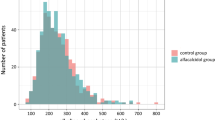
Abstract
Patients with end-stage renal disease often have derangements in calcium and phosphorus homeostasis and resultant secondary hyperparathyroidism (sHPT), which may contribute to the high prevalence of arterial stiffness and hypertension. We conducted a secondary analysis of the Evaluation of Cinacalcet Hydrochloride Therapy to Lower Cardiovascular Events (EVOLVE) trial, in which patients receiving hemodialysis with sHPT were randomly assigned to receive cinacalcet or placebo. We sought to examine whether the effect of cinacalcet on death and major cardiovascular events was modified by baseline pulse pressure as a marker of arterial stiffness, and whether cinacalcet yielded any effects on blood pressure. As reported previously, an unadjusted intention-to-treat analysis failed to conclude that randomization to cinacalcet reduces the risk of the primary composite end point (all-cause mortality or non-fatal myocardial infarction, heart failure, hospitalization for unstable angina or peripheral vascular event). However, after prespecified adjustment for baseline characteristics, patients randomized to cinacalcet experienced a nominally significant 13% lower adjusted risk (95% confidence limit 4–20%) of the primary composite end point. The effect of cinacalcet was not modified by baseline pulse pressure (Pinteraction=0.44). In adjusted models, at 20 weeks cinacalcet resulted in a 2.2 mm Hg larger average decrease in systolic blood pressure (P=0.002) and a 1.3 mm Hg larger average decrease in diastolic blood pressure (P=0.002) compared with placebo. In summary, in the EVOLVE trial, the effect of cinacalcet on death and major cardiovascular events was independent of baseline pulse pressure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Guerin AP, London GM, Marchais SJ, Metivier F . Arterial stiffening and vascular calcifications in end-stage renal disease. Nephrol Dial Transplant 2000; 15 (7): 1014–1021.
Pannier B, Guerin AP, Marchais SJ, Safar ME, London GM . Stiffness of capacitive and conduit arteries: Prognostic significance for end-stage renal disease patients. Hypertension 2005; 45 (4): 592–596.
Blacher J, Safar ME, Guerin AP, Pannier B, Marchais SJ, London GM . Aortic pulse wave velocity index and mortality in end-stage renal disease. Kidney Int 2003; 63 (5): 1852–1860.
Coen G, Calabria S, Bellinghieri G, Pecchini F, Conte F, Chiappini MG et al. Parathyroidectomy in chronic renal failure: Short- and long-term results on parathyroid function, blood pressure and anemia. Nephron 2001; 88 (2): 149–155.
Goldsmith DJA, Covic AA, Venning MC, Ackrill P . Blood pressure reduction after parathyroidectomy for secondary hyperparathyroidism: Further evidence implicating calcium homeostasis in blood pressure regulation. Am J Kidney Dis 1996; 27 (6): 819–825.
United States Renal Data System. USRDS 2013 Annual Data Report: Atlas of Chronic Kidney Disease and End-Stage Renal Disease in the United States 2013. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases: Bethesda, MD, USA.
Torres PA, De Broe M . Calcium-sensing receptor, calcimimetics, and cardiovascular calcifications in chronic kidney disease. Kidney Int 2012; 82 (1): 19–25.
Rybczynska A, Boblewski K, Lehmann A, Orlewska C, Foks H, Drewnowska K et al. Calcimimetic NPS R-568 induces hypotensive effect in spontaneously hypertensive rats. Am J Hypertens 2005; 18 (3): 364–371.
Odenwald T, Nakagawa K, Hadtstein C, Roesch F, Gohlke P, Ritz E et al. Acute blood pressure effects and chronic hypotensive action of calcimimetics in uremic rats. J Am Soc Nephrol 2006; 17 (3): 655–662.
Bonet J, Bayes B, Fernandez-Crespo P, Casals M, Lopez-Ayerbe J, Romero R . Cinacalcet may reduce arterial stiffness in patients with chronic renal disease and secondary hyperparathyroidism—results of a small-scale, prospective, observational study. Clin Nephrol 2011; 75 (3): 181–187.
Raggi P, Chertow GM, Torres PU, Csiky B, Naso A, Nossuli K et al. The ADVANCE study: a randomized study to evaluate the effects of cinacalcet plus low-dose vitamin D on vascular calcification in patients on hemodialysis. Nephrol Dial Transplant 2011; 26 (4): 1327–1339.
Suzuki H, Inoue T, Watanabe Y, Kikuta T, Sato T, Tsuda M et al. Does cinacalcet HCL, an oral calcimimetic agent for the treatment of secondary hyperparathyroidism, improve arterial stiffness in patients on continuous ambulatory peritoneal dialysis? Adv Perit Dial 2011; 27: 134–139.
Chertow GM, Block GA, Correa-Rotter R, Drueke TB, Floege J, Goodman WG et al. Effect of cinacalcet on cardiovascular disease in patients undergoing dialysis. N Engl J Med 2012; 367 (26): 2482–2494.
Chertow GM, Pupim LB, Block GA, Correa-Rotter R, Drueke TB, Floege J et al. Evaluation of cinacalcet therapy to lower cardiovascular events (EVOLVE): Rationale and design overview. Clin J Am Soc Nephrol 2007; 2 (5): 898–905.
Little RJ, RUBIN DB . Statistical analysis with missing data 2002. John Wiley & Sons, Inc..
Lewington S, Clarke R, Qizilbash N, Peto R, Collins S . Prospective Studies Collaboration Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 2002; 360 (9349): 1903–1913.
Chow KM, Szeto CC, Kum LC, Kwan BC, Fung TM, Wong TY et al. Improved health-related quality of life and left ventricular hypertrophy among dialysis patients treated with parathyroidectomy. J Nephrol 2003; 16 (6): 878–885.
Drüeke T, Fleury J, Toure Y, de Vernejoul P, Fauchet M, Lesourd P et al. Effect of parathyroidectomy on left-ventricular function in hæmodialysis patients. The Lancet 1980; 315 (8160): 112–114.
Bleyer AJ, Burkart J, Piazza M, Russell G, Rohr M, Carr JJ . Changes in cardiovascular calcification after parathyroidectomy in patients with esrd. Am J Kidney Dis 2005; 46 (3): 464–469.
Ifudu O, Matthew JJ, Macey LJ, Hong JS, Sumrani N, Sommer BG et al. Parathyroidectomy does not correct hypertension in patients on maintenance hemodialysis. Am J Nephrol 1998; 18 (1): 28–34.
Almirall J, Lopez T, Comerma I, Garcia E, Marques G . Effect of parathyroidectomy on blood pressure in dialysis patients. Nephron 2002; 92 (2): 495–496.
Zitt E, Woess E, Mayer G, Lhotta K . Effect of cinacalcet on renal electrolyte handling and systemic arterial blood pressure in kidney transplant patients with persistent hyperparathyroidism. Transplantation 2011; 92 (8): 883–889.
Henaut L, Boudot C, Massy ZA, Lopez-Fernandez I, Dupont S, Mary A et al. Calcimimetics increase CaSR expression and reduce mineralization in vascular smooth muscle cells: mechanisms of action. Cardiovasc Res 2014; 101 (2): 256–265.
Martinez-Miguel P, Medrano-Andres D, Lopes-Martin V, Arribas-Gomez I, Rodriguez-Puyol M, Rodriguez-Puyol D et al. Regulation of endothelin-converting enzyme-1 (ECE-1) by the calcimimetic R-568. Pharmacol Res 2013; 76: 106–118.
Zager PG, Nikolic J, Brown RH, Campbell MA, Hunt WC, Peterson D et al. "U" curve association of blood pressure and mortality in hemodialysis patients. Medical directors of dialysis clinic, inc. Kidney Int 1998; 54 (2): 561–569.
Li Z, Lacson E Jr., Lowrie EG, Ofsthun NJ, Kuhlmann MK, Lazarus JM et al. The epidemiology o f systolic blood pressure and death risk in hemodialysis patients. Am J Kidney Dis 2006; 48 (4): 606–615.
National Kidney Foundation. Kdoqi clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis 2005; 45: S1–S154.
Agarwal R . Blood pressure and mortality among hemodialysis patients. Hypertension 2010; 55 (3): 762–768.
Acknowledgements
This study was funded by Amgen, Inc. and led by an Executive Committee composed of academic members, two Sponsor members (nonvoting), and statisticians. The Executive Committee oversaw the design, conduct, and all analyses. Data were collected by the Sponsor and shared with the Executive Committee throughout the study and after unblinding. The analysis was performed by an independent biostatistician (SA) at Stanford University School of Medicine. The Sponsor provided the active medication and matching placebo. The lead author wrote the first draft of the manuscript, and the Executive Committee was responsible for data interpretation and manuscript completion. The Sponsor reviewed the manuscript, but decisions about the final manuscript were made by the lead author and academic members of the Executive Committee only. All authors vouch for the integrity of the data. The lead author (TC), Executive Committee Co-Chair (GC), with the statistician (SA) independently performed all prespecified and exploratory analyses; these analyses were confirmed by Amgen statisticians. The results of the independent analyses are those reported in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
All authors have completed and submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Grant, consulting fee/honorarium, speaking fees, stock options or travel support relevant to current manuscript: Abbott/Abbvie: JF, CH, RC-R, DW; Affymax: CH; Amgen: SA, GB, GC, TD, JF, CH, GL, SM, PP, RC-R, DW (note: no honoraria were received for writing or reviewing the manuscript); Astra Zeneca: RC-R; Chugai Pharmaceutical Co: JF; ClearView Healthcare Partners: CH; CorMedix: CH; Danone: RC-R; Davita: GB; Eli Lilly: SM; Fibrogen: CH; Fresenius Medical Care: TD, JF, RC-R, DW; Genzyme/Sanofi: GB, TD, JF, SM, RC-R, DW; Johnson & Johnson: CH, RC-R; KAI Pharmaceuticals: GB, SM; Keryx: GB, GC, CH; Medtronic: CH; Merck: DW, CH; Novartis: SM; Otsuka: DW; Pfizer: RC-R; Reata Pharmaceuticals: RC-R; Roche: RC-R; Satellite Healthcare: GC; Shire: JF, SM, DW; Vifor: JF, SM, DW; ZS Pharma: CH, DW. Employees and stock holders of Amgen: BD, WG. KM’s financial disclosures before 1 August 2013, can be viewed at https://www.dcri.org/about-us/conflict-of-interest/Mahaffey-COI_2011-2013.pdf; disclosures after 1 August 2013, can be viewed at http://med.stanford.edu/profiles/kenneth_mahaffey.
Rights and permissions
About this article
Cite this article
Chang, T., Abdalla, S., London, G. et al. The effects of cinacalcet on blood pressure, mortality and cardiovascular endpoints in the EVOLVE trial. J Hum Hypertens 30, 204–209 (2016). https://doi.org/10.1038/jhh.2015.56
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhh.2015.56
This article is cited by
-
Fibroblast growth factor 23—Klotho and hypertension: experimental and clinical mechanisms
Pediatric Nephrology (2021)