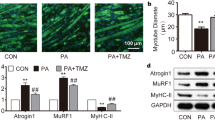
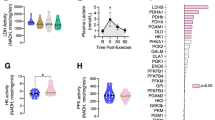
Abstract
Objective:
Skeletal muscle AMP-activated protein kinase (AMPK)α2 activity is impaired in obese, insulin-resistant individuals during exercise. We determined whether this defect contributes to the metabolic dysregulation and reduced exercise capacity observed in the obese state.
Design:
C57BL/6J wild-type (WT) mice and/or mice expressing a kinase dead AMPKα2 subunit in skeletal muscle (α2-KD) were fed chow or high-fat (HF) diets from 3 to 16 weeks of age. At 15 weeks, mice performed an exercise stress test to determine exercise capacity. In WT mice, muscle glucose uptake and skeletal muscle AMPKα2 activity was assessed in chronically catheterized mice (carotid artery/jugular vein) at 16 weeks. In a separate study, HF-fed WT and α2-KD mice performed 5 weeks of exercise training (from 15 to 20 weeks of age) to test whether AMPKα2 is necessary to restore work tolerance.
Results:
HF-fed WT mice had reduced exercise tolerance during an exercise stress test, and an attenuation in muscle glucose uptake and AMPKα2 activity during a single bout of exercise (P<0.05 versus chow). In chow-fed α2-KD mice, running speed and time were impaired ∼45 and ∼55%, respectively (P<0.05 versus WT chow); HF feeding further reduced running time ∼25% (P<0.05 versus α2-KD chow). In response to 5 weeks of exercise training, HF-fed WT and α2-KD mice increased maximum running speed ∼35% (P<0.05 versus pre-training) and maintained body weight at pre-training levels, whereas body weight increased in untrained HF WT and α2-KD mice. Exercise training restored running speed to levels seen in healthy, chow-fed mice.
Conclusion:
HF feeding impairs AMPKα2 activity in skeletal muscle during exercise in vivo. Although this defect directly contributes to reduced exercise capacity, findings in HF-fed α2-KD mice show that AMPKα2-independent mechanisms are also involved. Importantly, α2-KD mice on a HF-fed diet adapt to regular exercise by increasing exercise tolerance, demonstrating that this adaptation is independent of skeletal muscle AMPKα2 activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sigal RJ, Kenny GP, Wasserman DH, Castaneda-Sceppa C, White RD . Physical activity/exercise and Type 2 diabetes: a consensus statement from the american diabetes association. Diabetes Care 2006; 29: 1433–1438.
Steinberg GR, Kemp BE . AMPK in Health and Disease. Physiol Rev 2009; 89: 1025–1078.
Lee-Young RS, Griffee SR, Lynes SE, Bracy DP, Ayala JE, McGuinness OP et al. Skeletal Muscle AMP-Activated protein kinase is essential for the metabolic response to exercise In Vivo. J Biol Chem 2009; 284: 23925–23934.
Mu J, Barton ER, Birnbaum MJ . Selective suppression of AMP-activated protein kinase in skeletal muscle: update on ‘lazy mice’. Biochem Soc Trans 2003; 31: 236–241.
Sriwijitkamol A, Coletta DK, Wajcberg E, Balbontin GB, Reyna SM, Barrientes J et al. Effect of acute exercise on AMPK signaling in skeletal muscle of subjects with type 2 Diabetes: a time-course and dose-response study. Diabetes 2007; 56: 836–848.
Blaak EE, Saris WHM . Substrate oxidation, obesity and exercise training. Best Pract Res Clin Endocrinol Metab 2002; 16: 667–678.
Mu J, Brozinick Jr JT, Valladares O, Bucan M, Birnbaum MJ . A role for AMP-activated protein kinase in contraction- and hypoxia-regulated glucose transport in skeletal muscle. Mol Cell 2001; 7: 1085–1094.
Ayala JE, Bracy DP, McGuinness OP, Wasserman DH . Considerations in the design of hyperinsulinemic-euglycemic clamps in the conscious mouse. Diabetes 2006; 55: 390–397.
McGee SL, Howlett KF, Starkie RL, Cameron-Smith D, Kemp BE, Hargreaves M . Exercise increases nuclear AMPK α2 in human skeletal muscle. Diabetes 2003; 52: 926–928.
Wasserman K, Hansen JE, Sue DY, Casaburi R, Whipp BJ . Principles of exercise testing and interpretation, 3rd edn. Lippincott Williams and Wilkins: Baltimore, 1999.
Wasserman K, Sietsema KE . Assessing cardiac function by gas exchange. Cardiology 1988; 75: 307–310.
Fujii N, Seifert MM, Kane EM, Peter LE, Ho RC, Winstead S et al. Role of AMP-activated protein kinase in exercise capacity, whole body glucose homeostasis, and glucose transport in skeletal muscle—nsight from analysis of a transgenic mouse model. Diabetes Res Clin Pract 2007; 77: S92–S98.
Lee-Young RS, Ayala JE, Hunley CF, James FD, Bracy DP, Kang L et al. Endothelial nitric oxide synthase is central to skeletal muscle metabolic regulation and enzymatic signaling during exercise in vivo. Am J Physiol 2010; 298: R1399–R1408.
Sakamoto K, McCarthy A, Smith D, Green KA, Grahame Hardie D, Ashworth A et al. Deficiency of LKB1 in skeletal muscle prevents AMPK activation and glucose uptake during contraction. EMBO J 2005; 24: 1810–1820.
Beck Jørgensen S, O'Neill H, Hewitt K, Kemp B, Steinberg G . Reduced AMP-activated protein kinase activity in mouse skeletal muscle does not exacerbate the development of insulin resistance with obesity. Diabetologia 2009; 52: 2395–2404.
Anderson EJ, Lustig ME, Boyle KE, Woodlief TL, Kane DA, Lin C-T et al. Mitochondrial H2O2 emission and cellular redox state link excess fat intake to insulin resistance in both rodents and humans. J Clin Invest 2009; 119: 573–581.
Merry TL, Steinberg GR, Lynch GS, McConell GK . Skeletal muscle glucose uptake during contraction is regulated by nitric oxide and ROS independently of AMPK. Am J Physiol 2010; 298: E577–E585.
McGee SL, van Denderen BJW, Howlett KF, Mollica J, Schertzer JD, Kemp BE et al. AMP-activated protein kinase regulates GLUT4 transcription by phosphorylating histone deacetylase 5. Diabetes 2008; 57: 860–867.
De Filippis E, Alvarez G, Berria R, Cusi K, Everman S, Meyer C et al. Insulin-resistant muscle is exercise resistant: evidence for reduced response of nuclear-encoded mitochondrial genes to exercise. Am J Physiol 2008; 294: E607–E614.
Jorgensen SB, Treebak JT, Viollet B, Schjerling P, Vaulont S, Wojtaszewski JF et al. Role of AMPKα2 in basal, training-, and AICAR-induced GLUT4, hexokinase II, and mitochondrial protein expression in mouse muscle. Am J Physiol 2007; 292: E331–E339.
Maarbjerg SJ, Jorgensen SB, Rose AJ, Jeppesen J, Jensen TE, Treebak JT et al. Genetic impairment of α2-AMPK signaling does not reduce muscle glucose uptake during treadmill exercise in mice. Am J Physiol 2009; 297: E924–E934.
Kim F, Pham M, Maloney E, Rizzo NO, Morton GJ, Wisse BE et al. Vascular inflammation, insulin resistance, and reduced nitric oxide production precede the onset of peripheral insulin resistance. Arterioscler Thromb Vasc Biol 2008; 28: 1982–1988.
Acknowledgements
This work was supported by National Institutes of Health Grants U24 DK-59637 and R01 DK-54902 (to DHW). RS Lee-Young was supported by a Mentor-Based Fellowship from the American Diabetes Association.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Lee-Young, R., Ayala, J., Fueger, P. et al. Obesity impairs skeletal muscle AMPK signaling during exercise: role of AMPKα2 in the regulation of exercise capacity in vivo. Int J Obes 35, 982–989 (2011). https://doi.org/10.1038/ijo.2010.220
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2010.220
Keywords
This article is cited by
-
The exercise sex gap and the impact of the estrous cycle on exercise performance in mice
Scientific Reports (2018)
-
Type 2 diabetes mellitus and exercise impairment
Reviews in Endocrine and Metabolic Disorders (2013)