Abstract
We investigated whether the subservient relationship of peripheral to central hemodynamic parameters, such as the augmentation index (AI) and the second systolic (SBP2) and pulse pressures, were preserved with the progression of atherosclerosis in the Kurosawa and Kusanagi-hypercholesterolemic (KHC) rabbit, an animal model for hypercholesterolemia and atherosclerosis. Male KHC rabbits, aged 12 and 24 months, were anesthetized with pentobarbital sodium. Two catheter-tip transducers were introduced to the central (ascending aorta) and peripheral (distal region of the right brachial artery) arteries through the left common carotid and the right radial arteries, respectively. Pressure waves were simultaneously recorded under regular atrial pacing to investigate changes in response to the intravenous infusion of angiotensin II (Ang II) (30–40 ng kg−1 min−1) and sodium nitroprusside (NTP) (20–30 μg kg−1 min−1). Central systolic blood pressure (cSBP) and diastolic blood pressure (DBP), peripheral systolic blood pressure (pSBP) and DBP, and peripheral second systolic blood pressure (pSBP2) showed no significant difference between the 12- and 24-month-old groups before the administration of vasoactive drugs. There was no significant difference in central AI (cAI) between the two age groups before the drug infusion, even though atherosclerosis progressed with aging. Peripheral AI (pAI) changed in parallel with cAI in response to vasopressor and depressor actions due to the infusion of Ang II and NTP, respectively. We conclude that the subservience of pSBP2 to cSBP and pAI to cAI, in addition to the regression relationship of these parameters between peripheral and central arteries, were well preserved, irrespective of the progression of atherosclerosis.
Similar content being viewed by others
Introduction
The central systolic blood pressure (cSBP) is essentially different from the peripheral systolic blood pressure (pSBP), as measured using a brachial cuff sphygmomanometer in daily clinical settings. The former is mainly composed of forward waves, due to cardiac contraction, and reflected waves from the peripheral arterial sites of the whole body; the latter consists of forward waves that come from the central aorta and backward waves that are reflected only from the upper limb. Recent studies have revealed that cSBP was a better predictor of cardiovascular and hypertensive organ failure than the brachial blood pressure in hypertensive patients.1, 2, 3
The magnitude of systolic peak augmentation due to reflected waves is quantified as the augmentation index (AI).4 cSBP and central AI (cAI) could act as an invaluable reference source for central hemodynamics, including arterial distensibility. The ASCOT-CAFE study elucidated that vasodilating drugs produced preferable depressor effects on cSBP when compared with pSBP, which was measured using a brachial cuff sphygmomanometer.5 Miyashita et al.6 investigated the central pressure lowering effects of different classes of antihypertensive drugs cross-sectionally in Japanese hypertensive patients (Antihypertensives and Blood pressure of Central artery study in Japan). They elucidated that the vasodilators lowered cSBP significantly without class-specific differences, whereas non-vasodilators exerted no preferable effect on cSBP. Takase et al.7 recently showed the distribution of cSBP estimated from second pSBP (pSBP2) using an automated tonometry device in the Japanese general population, which could contribute to a more appropriate diagnosis and management of hypertension.
There is increasing evidence that non-invasively determined peripheral AI (pAI) correlated significantly with cAI that was obtained from the central pressure waves, which was derived using a generalized transfer function in healthy subjects8 and patients with cardiovascular or diabetic disease.9 In addition, there is evidence that pAI correlated with cAI obtained from directly measured central pressure waves with a catheter-tip micromanometer in patients who underwent cardiac catheterization or angiography.10, 11
Takazawa et al.11 recently investigated the relationship between invasively measured cSBP, using a high-fidelity micromanometer, with cSBP, estimated from non-invasively measured pSBP2 with an applanation tonometer (HEM-9000AI; Omron Healthcare Co. Ltd., Kyoto, Japan) without using generalized transfer function. They demonstrated that pSBP2 measured by the non-invasive method closely correlated with cSBP measured by the invasive method and that the relationship was preserved well after the administration of vasodilating drugs in patients with hypertension, diabetes mellitus, angina pectoris and myocardial infarction. Tomiyama et al.12 also recently reported that pSBP2 was a more potent predictor of the development of hypertension than the evaluation of stiffness of large- to middle-sized arteries.
Atherosclerosis could alter the pulse wave velocity, arrival time and magnitude of reflected waves to the aortic root.13 The contour of the pressure wave in the central artery is considerably deformed because of the progression of the atherosclerotic lesion with aging, which may affect the peripheral pressure waveform. cSBP and cAI have been shown to be higher in patients with hypercholesterolemia than in healthy subjects.14 It is of significance to investigate whether the relationship of pAI to cAI is preserved during the progression of atherosclerosis in an animal model.
In the present study, we investigated changes in the subservient relationships of pSBP2 to cSBP and pAI to cAI with aging in the Kurosawa and Kusanagi-hypercholesterolemic (KHC) rabbit, a useful animal model for familial hypercholesterolemia, atherosclerosis and mild hypertension,15, 16 when arterial pressure was altered due to infusion of a vasoconstrictor and a vasodilator.
Materials and methods
Animals
Nineteen KHC rabbits aged 12 (n=10) and 24 (n=9) months were used in the present study. They were bred in an individual stainless wire cage in an air-conditioned room at a temperature of 22–25 °C, a humidity level of 50–60% and a light/dark cycle of 12 light/12 dark in the Japan Laboratory Animals Inc., Tokyo, Japan. The animals were given a cholesterol-free commercial diet (RC-4; Oriental Yeast, Co., Ltd., Tokyo, Japan) at 100 g per animal per day and free access to water. The present study was approved by the Experimental Animal Committee of Fukushima Medical University and was performed according to the Guidelines for Animal Care and Handling of the Institute of Health (NIH).
Surgical procedure
The surgical procedure was almost similar to that described previously.16, 17 The rabbits were anesthetized by an intravenous administration of pentobarbital sodium (Nembutal; Abott Laboratories, Inc., North Chicago, IL, USA), fixed in a supine position and intubated through tracheotomy. A catheter-tip transducer (Mikro-Tip SPS-320, 2Fr; Millar Instruments, Inc., Dallas, TX, USA) was placed at the ascending aorta through the right common carotid artery. Another catheter-tip transducer (2Fr) was advanced to the distal end of the right brachial artery through the right radial artery. A median thoracotomy was performed without pneumothorax to maintain voluntary breathing. Electrodes for cardiac pacing were attached to the right atrial appendage. Pentobarbital sodium was added every 15–20 min at 5 mg per whole body. Procaine chloride was applied topically to the incised areas to reduce pain.
Pressure wave recording
Angiotensin II (Ang II) and sodium nitroprusside (NTP) were infused into the left ear vein using a syringe pump at respective doses of 30–40 ng kg−1 min−1 and 20–30 μg kg−1 min−1 until the mean arterial pressure (MAP) level at the ascending aorta reached ∼140 or 80 mm Hg, respectively. Changes in pressure waves at the ascending aorta and brachial artery were simultaneously recorded with a personal computer (PowerBook G4 M9691J/A; Apple Inc., Cupertino, CA, USA) through an analogue-to-digital converter (PowerLab System/16s; AD Instruments, Inc., Sydney, NSW, Australia) every 0.1 ms under regular cardiac pacing at 270 to 300 beats per min to investigate the effects of the vasoactive drugs on the vascular properties selectively, because changes in the heart rate through the baroreceptor reflex could affect AI.
Data processing
Twenty successive pressure waves at the ascending aorta and brachial artery were analyzed before and after the administration of vasoactive drugs. Figure 1 illustrates methods for the determination of AI in the central and peripheral pressure waves. The time at which the fourth derivative of the original wave crossed the baseline, from above to below, for the second time within each cardiac cycle was defined as the first systolic peak/shoulder when the pressure waves showed Type A and B patterns classified by Murgo et al.18 In a Type C pattern, the time at which the fourth derivative crossed the baseline, from below to above, for the third time within each cardiac cycle was defined as the second systolic peak (pSBP2), as reported by Takazawa et al.19 Type B was an intermediate pattern between Type A and Type C (not illustrated). cAI and pAI were calculated as S2/S1, where S1 and S2 were the first and second systolic peak pressures minus the diastolic pressure.19, 20
Methods for the determination of the augmentation index (AI) in different types of pressure waves. Central and peripheral arteries indicate the ascending aorta and brachial artery. Type of pressure waveform was classified by Murgo et al.17 S1 and S2 were the first and second systolic peak pressures minus the diastolic pressure.
Determination of total percent lesioned area
The total percent lesioned area of the aorta was calculated with almost the same methods as reported previously.17, 21 The aorta was excised from its origin to the bifurcation of the common iliac arteries and was cut open longitudinally. The intimal surface of the aorta was photocopied, and an outline of the atheromatous plaques and aorta were carefully traced. The traced outline was scanned, and the percent lesioned area was determined using free software (Scion Image; Scion Corporation, Houston, TX, USA).
Statistical procedure
The data were analyzed using a non-parametric two-way analysis of variance (Friedman test). If significant differences were observed, cSBP, pSBP, pSBP2 and AI were compared before and after drug administration, and between 12- and 24-month-old KHC rabbit groups, using a non-parametric multi-comparison test (Wilcoxon signed-rank test). The correlation coefficient (r) was also calculated using a non-parametric method. The significance of Spearman’s rank correlation coefficient was tested using the t-distribution. The difference between two overlapping correlation coefficients, such as between r(cSBP vs. pSBP) and r(cSBP vs. pSBP2), was tested using the Meng–Rosenthal–Rubin method.22 Two regression lines were compared using analysis of variance. A value of P<0.05 was considered significant.
Results
Pressor and depressor responses to Ang II and sodium NTP
Figure 2 shows the changes in the central and peripheral pressure waveforms in response to Ang II and NTP in the KHC rabbits that were aged 24 months. The change in the second peak pressure of the peripheral pressure wave was dominant compared with the change in the first peak pressure when vasoactive drugs were infused. The peripheral pressure waves shifted from Type C to Type A or Type B in most cases because of the infusion of Ang II, whereas all central pressure waves in KHC rabbits were Type A, irrespective of the infusion of vasoactive drugs, as illustrated in the previous report.17 Peaking of the central pressure wave appeared in the presence of Ang II. The peak pressure of the central pressure wave almost approximated the second peak pressure of the peripheral pressure wave, except in the presence of NTP. Similar change patterns in waveform were also observed in the 12-month-old KHC rabbit.
Table 1 shows the changes in cSBP, pSBP, pSBP2, central diastolic blood pressure (DBP) and peripheral DBP due to the infusion of vasoactive drugs. cSBP, pSBP and pSBP2 were elevated (P<0.001, for each parameter) and declined (P<0.001, for each parameter) significantly by the infusion of Ang II and NTP, respectively. There were no significant differences in cSBP, pSBP and pSBP2 between the 12- and 24-month-old KHC rabbit groups before and after the administration of vasoactive drugs. pSBP2 approximated cSBP before (control) and after the infusion of Ang II. The difference between pSBP2 and cSBP tended to increase after the infusion of NTP in the two age groups, although it did not reach statistical significance. Central DBP and peripheral DBP also increased and decreased significantly in response to the infusion of Ang II and NTP, respectively, in the two age groups, without a significant difference between the central and peripheral DBP values. Figure 3 illustrates the correlation diagrams between the central pulse pressure (cPP) and peripheral PP (pPP) (Figure 3a), between the cPP and peripheral second pulse pressure (pPP2) (Figure 3b), between the changes in central (ΔcPP) and peripheral (ΔpPP) pulse pressures (Figure 3c), and between the ΔcPP and changes in pPP2 (ΔpPP2) (Figure 3d) due to the infusion of the vasoactive drugs in KHC rabbits aged 12 and 24 months. The correlation coefficient between cPP and pPP2 (r=0.854, P<0.001 for 12 months and r=0.969, P<0.001 for 24 months) was significantly greater than between cPP and pPP (r=0.581, P<0.01 for 12 months and r=0.708, P<0.01 for 24 months) in the two age groups (P<0.05 for 12 months and P<0.001 for 24 months). There was no significant correlation between ΔpPP and ΔcPP in the two age groups (r=0.222, P>0.05 for 12 months and r=0.422, P>0.05 for 24 months), whereas ΔcPP correlated significantly with ΔpPP2 in the two age groups (r=0.769, P<0.001 for 12 months and r=0.949, P<0.001 for 24 months). In summary, a change in cPP due to the infusion of the vasoactive drugs was well reflected in pPP2 but not in pPP. Figure 4 depicts the correlation between the peripheral amplification of pressure waves and MAP. Peripheral amplification was determined as pPP/cPP × 100 (%). The amplification of peripheral pressure waves was greater when the MAP was in a relatively low range. There was a significant negative correlation between peripheral pressure amplification and MAP (r=−0.677, P<0.001 for 12 months and r=−0.836, P<0.001 for 24 months).
Correlation diagrams between central (cPP) and peripheral (pPP) pulse pressures (a), between cPP and amplitude of peripheral second pressure (pPP2) (b), between change in cPP due to infusion of vasoactive drugs (ΔcPP) and in pPP (ΔpPP) (c), and between (ΔcPP) and pPP2 (ΔpPP2) (d) in KHC rabbits aged 12 and 24 months.
Changes in cAI and pAI in response to vasoactive drugs
Changes in the amplitudes of central early and late (cPP) systolic peaks and of peripheral first (pPP) and second (pPP2) systolic peaks in response to vasoactive drugs are illustrated in Figure 5. The amplitude of the central early systolic peak and pPP did not show significant changes due to the infusion of vasoactive drugs in the two rabbit groups, whereas cPP and pPP2 decreased significantly due to the infusion of NTP in the two age groups, except for cPP at 12 months. Increases in cPP and pPP2 due to the infusion of Ang II were moderate in the 12-month-old group and were not observed in the 24-month-old group. Figure 6a depicts changes in cAI and pAI in response to the infusion of the vasoactive drugs. There were no significant inter-age group differences in cAI or pAI before, and even after, the infusion of the vasoactive drugs. cAI increased (P<0.001 for 12 months and P<0.01 for 24 months) and decreased (P<0.001 for each age group) significantly due to the infusion of Ang II and NTP in the two age groups, respectively. pAI also showed a significant increase (P<0.01 for 12 months and P<0.05 for 24 months) and a decrease (P<0.001 for each age group) in the two age groups. The change in pAI was almost in parallel with that seen in cAI in response to a change in the MAP level that was induced by vasoactive drugs in the two age groups. The slopes of the regression lines for cAI and for pAI were not significantly different between the two age groups. There was a strong positive correlation between pAI and cAI (r=0.887, P<0.001 for 12 months and r=0.925, P<0.001 for 24 months) (Figure 6b), with virtually identical regression lines for the two age groups. The difference in cAI before and after the infusion of the vasoactive drugs (ΔcAI) significantly correlated with the difference in pAI before and after the infusion of the vasoactive drugs (ΔpAI) in the two age groups (r=0.941, P<0.001 for 12 months and r=0.950, P<0.001 for 24 months) (Figure 6c). The slope of the regression line between ΔcAI and ΔpAI in the 12-month-old age group was almost the same as the slope in the 24-month-old age group.
Change in cAI and pAI respond to the intravenous infusion of sodium NTP and Ang II (a), relationship between cAI and pAI (b), and correlation diagram between the differences in cAI (ΔcAI) and pAI (ΔpAI) before and after the infusion of the vasoactive drugs (c) in KHC rabbits aged 12 and 24 months. The data are the mean±s.d. (a). **P<0.01; ***P<0.001; a, Control vs. Ang II; b, Control vs. NTP.
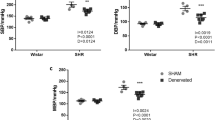
Macroscopic and histological findings of the atherosclerotic lesions with regard to age were almost similar to those reported previously.23, 24 Atheromatous plaques were present mainly in the ascending aorta and around the orifices of the main branch arteries at 12 months of age, and they progressed toward peripheral aortic regions at 24 months of age. The total percent lesioned area of the intimal surface of the aorta was 35.3±9.9 and 61.2±22.8 (%) (mean±s.d.) at 12 and 24 months of age, respectively, which was significant (P<0.01) between the two age groups. This result is similar to the results found in previous reports.17, 23 There were few atheromatous plaques in the axillary, brachial and radial arteries, except for a few smaller plaques localized around bifurcations of the branch arteries, even at 24 months old. The intima thickened with abundant foam cells at 12 months of age. Fibrous proliferation progressed among form cells with age. The medial layer was relatively preserved, except for the inner portion that is just beneath the intimal layer, even at an older age.
Discussion
The present findings in the rabbit model are consistent with those results observed in healthy human subjects25 and patients with cardiovascular disease.9, 10, 11 We observed that pSBP2 nearly approximated cSBP before and after the infusion of Ang II in the two age groups, whereas pSBP2 showed a trend toward underestimating cSBP in the presence of NTP. Recently, cSBP has been estimated from the late SBP of the peripheral pressure waves without generalized transfer function. Pauca et al.26, 27 found that the late SBP represented cSBP in aged hypertensive patients. Takazawa et al.10 reported that the changes in pSBP2 in the radial artery might enable a more accurate evaluation of changes in maximal SBP in the aorta. They also demonstrated that cSBP, approximated from pSBP2, was in good accordance with cSBP measured directly with a high-fidelity pressure sensor in patients with cardiovascular disease undergoing cardiac catheterization.11 Thus, pSBP2 has a key role in estimating cSBP and in determining pAI. However, Hickson et al.25 observed a discrepancy between radial pSBP2 and cSBP, mainly in normotensive younger subjects. This result was also consistent with our findings of cSBP and pSBP2 in response to the intravenous infusion of NTP, and it may suggest the possibility to extrapolate the rabbit data to humans. pSBP2 is considered to be valid for evaluating the depressor effect of vasodilators on cSBP in hypertensive patients, except for younger healthy subjects, who are rarely treated with a vasodilator. Hence, the underestimation issue is rarely problematic in the clinical setting.
Nichols et al.28 provided a conceptual explanation about the equivalence of pSBP2 to cSBP. The augmentation peak of the aortic pressure wave consists of low frequency components, which are reflected mainly from the lower body. The augmentation peak travels along the upper limb artery without significant amplification and attenuation, and it forms the second systolic peak in the radial artery. This process is a major reason for the approximate accordance of pPP2 with cPP and of pSBP2 with cSBP, because pSBP2 consists of pPP2 and an offset diastolic pressure, which is almost unchanged throughout the conduit arteries from the central aorta to the periphery, as shown in Table 1 and reported by Pauca et al.26, 27 in humans. The underlying mechanism for the equivalence of the radial second systolic peak and the aortic augmentation peak should be elucidated in future studies.
The changes in cAI that were induced by the infusion of vasoactive drugs and ΔcAI were well followed by changes in pAI and ΔpAI in the two age groups, respectively. These effects were due, in large part, to the fact that the change in cPP induced by the infusion of the vasoactive drugs was well reflected in pPP2, irrespective of the extent and severity of the atherosclerosis. There were no marked age-related characteristics in the regression relationship between ΔpAI and ΔcAI.
pAI correlated significantly with cAI, irrespective of the determination methods, in healthy subjects8 and in patients with cardiovascular disease.9, 10, 11 However, age, sex, height and type of cardiovascular or cardiovascular-related disease varied among subjects. The specific effect of extent or severity of atherosclerosis has not been fully understood.
There were no significant differences in cAI and pAI between the two age groups before and after the infusion of vasoactive drugs. This result could be due, in part, to the fact that the extent of differences in the heights of the central early and late systolic peaks and of the peripheral first and second systolic peaks were not significant between the two age groups, although an obvious inter-age group difference was observed in the height of each peak shown in Figure 5. We reported previously that the aortic pulse wave velocity and incremental elastic modulus of the ascending aortic wall were significantly greater in the 22- to 24-month-old KHC rabbit group compared with the 10- to 12-month-old KHC rabbit group.23, 24 The decreased distensibility of the aortic wall accounts for the increased early systolic peak. It also causes an increased early return of reflected waves to the aortic root. The observed increase in the height of the late systolic peak, which is proportional to the increased early peak, suggests that the effect of the earlier return of reflection may be cancelled by a possible reduction in peripheral impedance mismatch due to large artery stiffening.
Few atherosclerotic lesions were observed in the axillary and brachial arteries irrespective of age, whereas sclerotic lesions extended markedly toward the peripheral aortic regions. These findings suggest that the pressure wave transmission in the axillary and brachial arteries is rarely influenced by the progression of atherosclerosis, which may contribute to the preserved relationship between cAI and pAI, irrespective of age.
Limitations
pSBP2 was a useful estimate for cSBP before and after the infusion of Ang II in KHC rabbits, irrespective of age and the progression of atherosclerosis; however, the discrepancy between pSBP2 and cSBP was observed at a lower blood pressure level after the infusion of NTP. In the clinical setting, underestimation of cSBP could occur in younger hypotensive subjects. Furthermore, the linearity of the relationship between cAI and pAI covered a full range of MAP levels (Figure 6b), without the influence of the underestimation of cSBP by pSBP2.
In KHC rabbits, atherosclerotic lesions developed in the ascending aorta and around bifurcations of the branch arteries, such as the intercostal, mesenteric and renal arteries, and progressed toward the peripheral arterial sites. In contrast, atherosclerosis was prominent in the proximal abdominal aorta in humans,29 although histological findings of atheromatous plaques in rabbits closely resemble histological findings observed in humans. Vascular responses induced due to transient intravenous infusion under regular cardiac pacing could stimulate baroreceptors, which could partly modulate the effect of the vasoactive agents. Vasoactive drugs are usually evaluated due to chronic oral administration in clinical studies. The characteristics of pressure wave transmission along the upper limb arteries should be elucidated in the rabbit model and humans, and should be compared under conditions of intravenous and oral administration.
Conclusions
Changes in cSBP and cPP induced by vasoactive drugs are followed by changes in pSBP2 and pPP2 instead of pSBP and pPP, irrespective of the arterial pressure level and extent of atherosclerotic lesions. The good correlation between cAI and pAI is also preserved during atherosclerosis progression. The present study supports the validity of the estimation of cAI as well as cSBP based on the second systolic peak of the peripheral pressure wave in patients with atherosclerosis and/or hypertension.
References
Safar ME, Blacher J, Pannier B, Guerin AP, Marchais SJ, Guyonvarc’h PM, London GM . Central pulse pressure and mortality in end-stage renal disease. Hypertension 2002; 39: 735–738.
Roman MJ, Devereux RB, Kizer JR, Lee ET, Galloway JM, Ali T, Umans JG, Howard BV . Central pressure more strongly relates to vascular disease and outcome than does brachial pressure: the Strong Heart Study. Hypertension 2007; 50: 197–203.
Agabiti-Rosei E, Mancia G, O’Rourke MF, Roman MJ, Safar ME, Smulyan H, Wang JG, Wilkinson IB, Williams B, Vlachopoulos C . Central blood pressure measurements and antihypertensive therapy: a consensus document. Hypertension 2007; 50: 154–160.
Kelly R, Hayward C, Avolio A, O'Rourke M . Noninvasive determination of age-related changes in the human arterial pulse. Circulation 1989; 80: 1652–1659.
Williams B, Lacy PS, Thom SM, Cruickshank K, Stanton A, Collier D, Hughes AD, Thurston H, O'Rourke M . Differential impact of blood pressure−lowering drugs on central aortic pressure and clinical outcomes. Principal results of the Conduit Artery Function Evaluation (CAFE) Study. Circulation 2006; 113: 1213–1225.
Miyashita H, Aizawa A, Hashimoto J, Hirooka Y, Imai Y, Kawano Y, Kohara K, Sunagawa K, Suzuki H, Tabara Y, Takazawa K, Takenaka T, Yasuda H, Shimada K . Cross-sectional characterization of all classes of antihypertensives in terms of central blood pressure in Japanese hypertensive patients. Am J Hypertens 2010; 23: 260–268.
Takase H, Dohi Y, Kimura G . Distribution of central blood pressure values estimated by Omron HEM-9000AI in the Japanese general population. Hypertens Res 2013; 36: 50–57.
Kohara K, Tabara Y, Oshiumi A, Miyawaki Y, Kobayashi T, Miki T . Radial augmentation index: A useful and easily obtainable parameter for vascular aging. Am J Hypertens 2005; 18: 11–14.
Munir S, Guilcher A, Kamalesh T, Clapp B, Redwood S, Marber M, Chowienczyk P . Peripheral augmentation index defines the relationship between central and peripheral pulse pressure. Hypertension 2008; 51: 112–118.
Takazawa K, Kobayashi H, Shindo N, Tanaka N, Yamashina A . Relationship between radial and central arterial pulse wave and evaluation of central aortic pressure using the radial arterial pulse wave. Hypertens Res 2007; 30: 219–228.
Takazawa K, Kobayashi H, Kojima I, Aizawa A, Kinoh M, Sugo Y, Shimizu M, Miyawaki Y, Tanaka N, Yamashina A, Avolio A . Estimation of central aortic systolic pressure using late systolic inflection of radial artery pulse and its application to vasodilator therapy. J Hypertens 2012; 30: 908–916.
Tomiyama H, Yoshida M, Hashimoto H, Matsumoto C, Nagata M, Odaira M, O'rourke MF, Shiina K, Yamashina A . Central blood pressure: a powerful predictor of the development of hypertension. Hypertens Res 2013; 36: 19–24.
O’Rourke MF, Hashimoto J, O’Rourke MF, Hashimoto J . Mechanical factors in arterial aging. A clinical perspective. J Am Coll Cardiol 2007; 50: 1–13.
Wilkinson IB, Prasad K, Hall IR, Thomas A, MacCallum H, Webb DJ, Frenneaux MP, Cockcroft JR . Increased central pulse pressure and augmentation index in subjects with hypercholesterolemia. J Am Coll Cardiol 2002; 39: 1005–1011.
Kurosawa T, Kusanagi M, Yamasaki Y, Senga Y, Yamamoto T . New mutant rabbit straina with hypercholesterolemia and atherosclerotic lesions produced by serial inbreeding. Lab Anim Sci 1995; 45: 385–392.
Katsuda S, Miyashita H, Takazawa K, Machida N, Kusanagi M, Miyake M, Hazama A . Mild hypertension in young Kurosawa and Kusanagi-hypercholesterolemic (KHC) rabbits. Physiol Meas 2006; 27: 1361–1371.
Katsuda S, Miyake M, Kobayashi D, Hazama H, Kusanagi M, Takazawa K . Does the augmentation index of pulse waves truly increase with progression of atherosclerosis? An experimental study with hypercholesterolemic rabbits. Am J Hypertens 2013; 26: 311–317.
Murgo JP, Westerhof N, Giolma JP, Altobelli SA . Aortic input impedance in normal man: relationship to pressure wave forms. Circulation 1980; 62: 105–116.
Takazawa K, Tanaka N, Takeda K, Kurosu F, Ibukiyama C . Underestimation of vasodilator effects of nitroglycerin by upper limb blood pressure. Hypertension 1995; 26: 520–523.
Obara S, Hayashi S, Hazama A, Murakawa M, Katsuda S . Correlation between augmentation index and pulse wave velocity in rabbits. J Hypertens 2009; 27: 332–340.
Katsuda S, Suzuki K, Koyama N, Takahashi M, Miyake M, Hazama A, Takazawa K . Safflower seed polyphenols (N-(p-coumaroyl)serotonin and N-feruloylserotonin) ameliorate atherosclerosis and distensibility of the aortic wall in Kurosawa and Kusanagi-hypercholesterolemic (KHC) rabbits. Hypertens Res 2009; 32: 944–949.
Meng X-L, Rosenthal R, Rubin DB . Comparing correlated correlation coefficients. Psychol Bull 1992; 111: 172–175.
Katsuda S, Hasegawa M, Kusanagi M, Shimizu T . Comparison of pulse-wave velocity in different aortic regions in relation to the extent and severity of atherosclerosis between young and older Kurosawa and Kusanagi-hypercholesterolemic (KHC) rabbits. Clin Sci (Lond) 2000; 99: 393–404.
Katsuda S, Machida N, Hasegawa M, Miyashita H, Kusanagi M, Tsubone H, Hazama A . Change in the static rheological properties of the aorta in Kurosawa and Kusanagi-Hypercholesterolemic (KHC) rabbits with progress of atherosclerosis. Physiol Meas 2004; 25: 505–522.
Hickson SS, Butlin M, Mir FA, Graggaber J, Cheriyan J, Khan F, Grace AA, Cockcroft JR, Wilkinson IB, McEniery CM, on behalf of the Anglo-Cardiff Collaboration Trial Investigators. The accuracy of central SBP determined from the second systolic peak of the peripheral pressure waveform. J Hypertens 2009; 27: 1784–1788.
Pauca AL, Wallenhaupt SL, Kon ND, Tucker WY . Does radial artery pressure accurately reflect aortic pressure? Chest 1992; 102: 1193–1198.
Pauca AL, Kon ND, O’Rourke MF . The second peak of the radial artery pressure wave represents aortic systolic pressure in hypertensive and elderly patients. Br J Anaesth 2004; 92: 651–657.
Nichols WW, O'Rourke MF, Vlachopoulos C . Central arterial pressure. In:. McDonald’s Blood Flow in Arteries, 6th edn. Hodders Arnold: London. 2011 pp 569–578.
Stehbens WE . Atherosclerosis. In Thomas CC, Stehbens WE, (eds). Hemodynamics and the Blood Vessel Wall. Charles C Thomas: Illinois. 1979 pp 316–374.
Acknowledgements
This work was supported by JSPS KAKENHI Grant Number 23500522. We are grateful to Mr Haruyuki Wago and Mr Toshiyasu Okouchi for their technical assistance. The present study was partly supported by Omron Healthcare Co., Ltd.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Katsuda, Si., Miyashita, H., Shimada, K. et al. Subservient relationship of the peripheral second systolic pressure peak to the central hemodynamic parameters is preserved, irrespective of atherosclerosis progression in hypercholesterolemic rabbits. Hypertens Res 37, 19–25 (2014). https://doi.org/10.1038/hr.2013.104
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2013.104
Keywords
This article is cited by
-
Brachial and central blood pressure in HIV-infected subjects
Hypertension Research (2015)