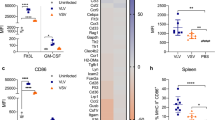
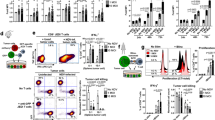
Abstract
Adenoviral (Adv) vectors are widely used in both experimental and clinical trials for vaccination and gene therapy. Recombinant Adv can evoke potent innate immune responses and adaptive immune responses to encoded antigens. However, how Adv infection affects the response to subsequently encountered antigens is poorly understood. We show that intravenously administered replication defective (E1 and E3 deleted) Adv educes functional changes in dendritic cells (DC) resulting in impaired priming of cytotoxic T lymphocytes (CTL) more than 7 days after Adv treatment. Generalized DC activation was indicated by transient upregulation of CD86 and reduced endocytosis of fluorescent beads. It is known that CD8+ DC are predominantly responsible for uptake and presentation (cross-presentation) of exogenous antigens to CD8+ CTL. Hence, impaired endocytosis in CD8+, but not CD8−, DC at 7 days after Adv administration provided an explanation for the impaired CTL response to antigen at this time. Shutdown of cross-presentation was confirmed using cytochrome c (cytc), an agent that selectively depletes cross-presenting DC. Adv-infection rendered CD8+ DC resistant to depletion by cytc. As the cross-presentation pathway underlies CD8 T-cell responses to many cancers and to vaccines or viruses that do not directly infect DC, systemic Adv administration may impair these responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Horwitz MS . Function of adenovirus E3 proteins and their interactions with immunoregulatory cell proteins. J Gene Med 2004; 6 (Suppl 1): S172–S183.
Rhee EG, Blattman JN, Kasturi SP, Kelley RP, Kaufman DR, Lynch DM et al. Multiple innate immune pathways contribute to the immunogenicity of recombinant adenovirus vaccine vectors. J Virol 2011; 85: 315–323.
Tripathy SK, Black HB, Goldwasser E, Leiden JM . Immune responses to transgene-encoded proteins limit the stability of gene expression after injection of replication-defective adenovirus vectors. Nat Med 1996; 2: 545–550.
Raper SE, Chirmule N, Lee FS, Wivel NA, Bagg A, Gao GP et al. Fatal systemic inflammatory response syndrome in a ornithine transcarbamylase deficient patient following adenoviral gene transfer. Mol Genet Metab 2003; 80: 148–158.
Zhang Y, Chirmule N, Gao GP, Qian R, Croyle M, Joshi B et al. Acute cytokine response to systemic adenoviral vectors in mice is mediated by dendritic cells and macrophages. Mol Ther 2001; 3: 697–707.
Korst RJ, Mahtabifard A, Yamada R, Crystal RG . Effect of adenovirus gene transfer vectors on the immunologic functions of mouse dendritic cells. Mol Ther 2002; 5: 307–315.
Zhu J, Huang X, Yang Y . Innate immune response to adenoviral vectors is mediated by both toll-like receptor-dependent and -independent pathways. J Virol 2007; 81: 3170–3180.
Yamaguchi T, Kawabata K, Koizumi N, Sakurai F, Nakashima K, Sakurai H et al. Role of MyD88 and TLR9 in the innate immune response elicited by serotype 5 adenoviral vectors. Hum Gene Ther 2007; 18: 753–762.
Garrett WS, Chen LM, Kroschewski R, Ebersold M, Turley S, Trombetta S et al. Developmental control of endocytosis in dendritic cells by Cdc42. Cell 2000; 102: 325–334.
West MA, Wallin RP, Matthews SP, Svensson HG, Zaru R, Ljunggren HG et al. Enhanced dendritic cell antigen capture via toll-like receptor-induced actin remodeling. Science 2004; 305: 1153–1157.
Wilson NS, Behrens GM, Lundie RJ, Smith CM, Waithman J, Young L et al. Systemic activation of dendritic cells by toll-like receptor ligands or malaria infection impairs cross-presentation and antiviral immunity. Nat Immunol 2006; 7: 165–172.
Gil-Torregrosa BC, Lennon-Dumenil AM, Kessler B, Guermonprez P, Ploegh HL, Fruci D et al. Control of cross-presentation during dendritic cell maturation. Eur J Immunol 2004; 34: 398–407.
Bedoui S, Prato S, Mintern J, Gebhardt T, Zhan Y, Lew AM et al. Characterization of an immediate splenic precursor of CD8+ dendritic cells capable of inducing antiviral T cell responses. J Immunol 2009; 182: 4200–4207.
den Haan JM, Bevan MJ . Constitutive versus activation-dependent cross-presentation of immune complexes by CD8(+) and CD8(−) dendritic cells in vivo. J Exp Med 2002; 196: 817–827.
den Haan JM, Lehar SM, Bevan MJ . CD8(+) but not CD8(−) dendritic cells cross-prime cytotoxic T cells in vivo. J Exp Med 2000; 192: 1685–1696.
Lin ML, Zhan Y, Villadangos JA, Lew AM . The cell biology of cross-presentation and the role of dendritic cell subsets. Immunol Cell Biol 2008; 86: 353–362.
Heath WR, Carbone FR . Cross-presentation in viral immunity and self-tolerance. Nat Rev Immunol 2001; 1: 126–134.
Brossart P, Goldrath AW, Butz EA, Martin S, Bevan MJ . Virus-mediated delivery of antigenic epitopes into dendritic cells as a means to induce CTL. J Immunol 1997; 158: 3270–3276.
Harui A, Roth MD, Kiertscher SM, Mitani K, Basak SK . Vaccination with helper-dependent adenovirus enhances the generation of transgene-specific CTL. Gene Therapy 2004; 11: 1617–1626.
Lin J, Somanathan S, Roy S, Calcedo R, Wilson JM . Lung homing CTLs and their proliferation ability are important correlates of vaccine protection against influenza. Vaccine 2010; 28: 5669–5675.
Malkevitch N, Patterson LJ, Aldrich K, Richardson E, Alvord WG, Robert-Guroff M . A replication competent adenovirus 5 host range mutant-simian immunodeficiency virus (SIV) recombinant priming/subunit protein boosting vaccine regimen induces broad, persistent SIV-specific cellular immunity to dominant and subdominant epitopes in Mamu-A*01 rhesus macaques. J Immunol 2003; 170: 4281–4289.
Varnavski AN, Calcedo R, Bove M, Gao G, Wilson JM . Evaluation of toxicity from high-dose systemic administration of recombinant adenovirus vector in vector-naive and pre-immunized mice. Gene Therapy 2005; 12: 427–436.
Lin ML, Zhan Y, Proietto AI, Prato S, Wu L, Heath WR et al. Selective suicide of cross-presenting CD8+ dendritic cells by cytochrome c injection shows functional heterogeneity within this subset. Proc Natl Acad Sci USA 2008; 105: 3029–3034.
Schnell MA, Zhang Y, Tazelaar J, Gao GP, Yu QC, Qian R et al. Activation of innate immunity in nonhuman primates following intraportal administration of adenoviral vectors. Mol Ther 2001; 3: 708–722.
Morelli AE, Larregina AT, Ganster RW, Zahorchak AF, Plowey JM, Takayama T et al. Recombinant adenovirus induces maturation of dendritic cells via an NF-kappaB-dependent pathway. J Virol 2000; 74: 9617–9628.
Perreau M, Mennechet F, Serratrice N, Glasgow JN, Curiel DT, Wodrich H et al. Contrasting effects of human, canine, and hybrid adenovirus vectors on the phenotypical and functional maturation of human dendritic cells: implications for clinical efficacy. J Virol 2007; 81: 3272–3284.
Hartman ZC, Kiang A, Everett RS, Serra D, Yang XY, Clay TM et al. Adenovirus infection triggers a rapid, MyD88-regulated transcriptome response critical to acute-phase and adaptive immune responses in vivo. J Virol 2007; 81: 1796–1812.
Lindsay RW, Darrah PA, Quinn KM, Wille-Reece U, Mattei LM, Iwasaki A et al. CD8+ T cell responses following replication-defective adenovirus serotype 5 immunization are dependent on CD11c+ dendritic cells but show redundancy in their requirement of TLR and nucleotide-binding oligomerization domain-like receptor signaling. J Immunol 2010; 185: 1513–1521.
Kamath AT, Henri S, Battye F, Tough DF, Shortman K . Developmental kinetics and lifespan of dendritic cells in mouse lymphoid organs. Blood 2002; 100: 1734–1741.
Idoyaga J, Suda N, Suda K, Park CG, Steinman RM . Antibody to langerin/CD207 localizes large numbers of CD8alpha+ dendritic cells to the marginal zone of mouse spleen. Proc Natl Acad Sci USA 2009; 106: 1524–1529.
Platt CD, Ma JK, Chalouni C, Ebersold M, Bou-Reslan H, Carano RA et al. Mature dendritic cells use endocytic receptors to capture and present antigens. Proc Natl Acad Sci USA 2010; 107: 4287–4292.
Shayakhmetov DM, Di Paolo NC, Mossman KL . Recognition of virus infection and innate host responses to viral gene therapy vectors. Mol Ther 2010; 18: 1422–1429.
Holst PJ, Orskov C, Thomsen AR, Christensen JP . Quality of the transgene-specific CD8+ T cell response induced by adenoviral vector immunization is critically influenced by virus dose and route of vaccination. J Immunol 2010; 184: 4431–4439.
Wakim LM, Waithman J, van Rooijen N, Heath WR, Carbone FR . Dendritic cell-induced memory T cell activation in nonlymphoid tissues. Science 2008; 319: 198–202.
Buchbinder SP, Mehrotra DV, Duerr A, Fitzgerald DW, Mogg R, Li D et al. Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the Step Study): a double-blind, randomised, placebo-controlled, test-of-concept trial. Lancet 2008; 372: 1881–1893.
Zheng C, Nikolov NP, Alevizos I, Cotrim AP, Liu S, McCullagh L et al. Transient detection of E1-containing adenovirus in saliva after the delivery of a first-generation adenoviral vector to human parotid gland. J Gene Med 2010; 12: 3–10.
Londrigan SL, Sutherland RM, Brady JL, Carrington EM, Cowan PJ, d'Apice AJ et al. In situ protection against islet allograft rejection by CTLA4Ig transduction. Transplantation 2010; 90: 951–957.
Acknowledgements
This work was supported by the National Health and Medical Research Council of Australia, Juvenile Diabetes Research Foundation and Diabetes Australia Research Trust. This work was made possible through Victorian State Government Operational Infrastructure Support and Australian Government NHMRC IRIIS. We are grateful to Nicole Ashman for technical assistance with mice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Sutherland, R., Londrigan, S., Brady, J. et al. Shutdown of immunological priming and presentation after in vivo administration of adenovirus. Gene Ther 19, 1095–1100 (2012). https://doi.org/10.1038/gt.2011.187
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2011.187