Abstract
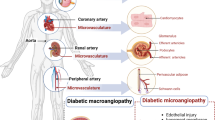
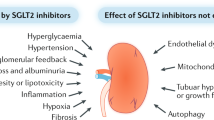
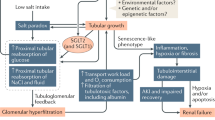
The mechanism of development of microangiopathy is incompletely understood, but relates to a number of ultrastructural, biochemical and haemostatic processes. These include capillary basement membrane thickening, non-enzymatic glycosylation, possibly increased free radical activity, increased flux through the polyol pathway and haemostatic abnormalities. The central feature appears to be hyperglycaemia, which is causally related to the above processes and culminates in tissue ischaemia. This article will briefly describe these processes and will discuss possible pathogenic interactions which may lead to the development of the pathological lesion.
Similar content being viewed by others
Article PDF
References
Spiro RG . Search for a biochemical basis of diabetic microangiopathy. Diabetologia 1976;12:1–14.
Beisswenger PJ, Spiro RG . Studies on the human glomer-ular basement membrane: composition, nature of the carbohydrate units and chemical changes in diabetes mellitus. Diabetes 1973;22:180–93.
Kefalides NA . Biochemical properties of human glomerular basement membrane in normal and diabetic kidneys. J Clin Invest 1974;53:403–7.
Kanwar YS, Hascall VC, Farquhar MG . Partial characterisation of newly synthesised proteoglycans isolated from the glomerular basement membrane. J Cell Biol 1981;90:527–32.
Uitto J, Perejda A, Grant GA, Rowold E, Kilo CA, Williamson JR . Glycosylation of human glomerular basement membrane collagen: increased content of hexose in ketoamine linkage and unaltered hydroxylysine-o-glycosides in patients with diabetes. Connect Tissue Res 1982;10:287–96.
Reynolds TM . Chemistry of non-enzymatic browning. Adv Food Res 1965;14:167–283.
Brownlee M, Pongor S, Cerami A . Covalent attachment of soluble proteins by non-enzymatically glycosylated collagen: role of in situ formation of immune complexes. J Exp Med 1983;158:1739–44.
Monnier VM, Vishwanath V, Frank KE, Elmets GA, Dau-chot P, Khon RR . Relation between complications of type 1 diabetes mellitus and collagen-linked fluorescence. N Engl J Med 1986;314:403–8.
Wickens DG, Norden AG, Lunec J, Dormandy TL . Fluorescence changes in human gamma-globulin induced by free radical activity. Biochim Biophys Acta 1983;742:607–16.
Wickens DG, Gaff TL, Lunec J, Dormandy TL . Free radical mediated aggregation of human gamma globulin. Agents Actions 1981;11:6–7.
Jennings PE, Barnett AH . New approaches to the pathogenesis and treatment of diabetic microangiopathy. Diabetic Med 1987;5:111–7.
Jones AF, Jennings PE, Wakefield A, Winkles JW, Lunec J, Barnett AH . Protein browning and diabetic complications. Lancet 1986;2:–459.
Nishigaki I, Hagihara M, Tsunekawa H, Maseki M, Yagi K . Lipid peroxide levels of serum lipoprotein fractions of diabetic patients. Biochem Med 1981;25:373–8.
Kaji H, Kurasaki M, Ito K, et al. Increased lipoperoxide value and glutathione peroxidase activity in blood plasma of type II (non-insulin dependent) diabetic women. Klin Wochenschr 1985;63:765–8.
Jennings PE, Jones AF, Florkowski CM, Lunec J, Barnett AH . Increased diene conjugates in diabetic subjects with microangiopathy. Diabetic Med 1987;4:452–6.
Hemler ME, Cook HW, Lands WEM . Prostaglandin biosynthesis can be triggered by lipid peroxides. Arch Biochem Biophys 1979;193:340–5.
Warso MA, Lands WEM . Lipid peroxidation in relation to prostacyclin and thromboxane physiology and pathophy-siology. Br Med Bull 1983;39:277–80.
Ham EG, Egan RW, Soderman DD, Gale PH, Kuehl FA . Peroxidase dependent deactivation of prostacyclin synthe-tase. J Biol Chem 1979;254:2191–4.
Chari SW, Nath N, Rathi AB . Glutathione and its redox system in diabetic polymorphonuclear leucocytes. Am J Med Sci 1984;287:14–5.
Stankova L, Riddle M, Larned J, et al. Plasma ascorbate concentrations and blood cell dehydroascorbate transport in patients with diabetes mellitus. Metabolism 1984;33:347–53.
Jennings PE, Chirico S, Jones AF, Lunec J, Barnett AH . Vitamin C metabolites and microangiopathy in diabetes mellitus. Diabetes Res 1987;6:151–4.
Sinclair AJ, Girling AJ, Gray L, Le Guen C, Lunec J, Barnett AH . Disturbed handling of ascorbic acid in diabetic patients with and without microangiopathy during high dose ascorbate supplementation. Diabetologia 1991;34:171–5.
Cogan DG . Aldose reductase and complications of diabetes. Ann Intern Med 1984;101:82–91.
Robinson WG, Kador PF, Kinoshita JH . Early retinal microangiopathy: prevention with aldose reductase inhibitors. Diabetic Med 1985;2:196–9.
Cohen M . Aldose reductase inhibition, glomerular metabolism and diabetic nephropathy. Diabetic Med 1985;2:203–6.
Jedziniak JA, Chylack LT, Cheng M-H, et al. The sorbitol pathway in the human lens: aldose reductase and polyol dehydrogenase. Invest Ophthalmol 1981;20:314–26.
Florkowski CM, Rowe BR, Nightingale S, Harvey TC, Barnett AH . Clinical and neurophysiological studies of aldose reductase inhibitor Ponalrestat in chronic symptomatic diabetic peripheral neuropathy. Diabetes 1991;40:129–33.
Gonzalez AM, Sochor M, McLean P . The effect of an aldose reductase inhibitor (Sorbinil) on the level of metabolites in the lenses of diabetic rats. Diabetes 1984;32:482–5.
Barnett PA, Gonzalez RG, Chylack LT, Chen H-M . The effect of oxidation on sorbitol pathway kinetics. Diabetes 1986;35:426–32.
Lee T-S, Saltsman A, Ohaski H, King GL . Activation of protein kinase C by elevation of glucose concentrations: proposal for a mechanism in the development of diabetic vascular complications. Proc Natl Acad Sci USA 1989;86:5141–5.
Porta M, Townsend C, Clover GM, et al. Evidence for functional endothelial cell damage in early diabetic retinopathy. Diabetologia 1981;20:597–601.
Carreras LO, Chamone DAF, Klerckx P, Vermylen J . Decreased vascular prostacyclin (PGI2) in diabetic rats: stimulation of PGI2 release in normal and diabetic rats by the antithrombotic compound BAYg 6575. Thromb Res 1980;19:663–70.
Hassen KF, Dahl-Jorgensen K, Lauritzen T, Feldt-Rasmus-sen B, Brinchmann-Hansen O, Deckert T . Diabetic control and microvascular complications: the near-normoglycaemic experience. Diabetologia 1986;29:677–84.
Ziboh VA, Maruta H, Lord J, Cagle WD, Lucky W . Increased biosynthesis of thromboxane A2 by diabetic platelets. Eur J Clin Invest 1979;9:223–8.
Butkus A, Shiney EK, Schumacher OP . Thromboxane biosynthesis in platelets of diabetic and coronary artery diseased patients. Artery 1982;11:238–51.
Chmielewski J, Farbiszewski R . Platelet factor 4 release during human platelet aggregation in diabetic patients. Thromb Haemost 1970;24:203–5.
Sacchni S, Curci G, Piccinin L, et al. Platelet alpha granule release in diabetes mellitus. Scand J Clin Lab Invest 1985;45:165–8.
Paulsen EP, McChung NM, Sabio H . Some characteristics of spontaneous platelet aggregation in young insulin dependent diabetic subjects. Horm Metab Res 1981;Suppl 11:15–21.
Dallinger KJC, Jennings PE, Toop MJ, Gyde OHB, Barnett AH . Platelet aggregation and coagulation factors in insulin dependent diabetics with and without microangiopathy. Diabetic Med 1987;4:44–8.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Barnett, A. Origin of the microangiopathic changes in diabetes. Eye 7, 218–222 (1993). https://doi.org/10.1038/eye.1993.52
Issue Date:
DOI: https://doi.org/10.1038/eye.1993.52
Keywords
This article is cited by
-
Vitreous vascular endothelial growth factor level during gestation
Archives of Gynecology and Obstetrics (2010)