Abstract
Heterozygosity for mutations in ribosomal protein genes frequently leads to a dominant phenotype of retarded growth and small adult bristles in Drosophila (the Minute phenotype). Cells with Minute genotypes are subject to cell competition, characterized by their selective apoptosis and removal in mosaic tissues that contain wild-type cells. Competitive apoptosis was found to depend on the pro-apoptotic reaper, grim and head involution defective genes but was independent of p53. Rp/+ cells are protected by anti-apoptotic baculovirus p35 expression but lacked the usual hallmarks of ‘undead’ cells. They lacked Dronc activity, and neither expression of dominant-negative Dronc nor dronc knockdown by dsRNA prevented competitive apoptosis, which also continued in dronc null mutant cells or in the absence of the initiator caspases dredd and dream/strica. Only simultaneous knockdown of dronc and dream/strica by dsRNA was sufficient to protect Rp/+ cells from competition. By contrast, Rp/Rp cells were also protected by baculovirus p35, but Rp/Rp death was dronc-dependent, and undead Rp/Rp cells exhibited typical dronc-dependent expression of Wingless. Independence of p53 and unusual dependence on Dream/Strica distinguish competitive cell death from noncompetitive apoptosis of Rp/Rp cells and from many other examples of cell death.
Similar content being viewed by others
Main
In Drosophila, heterozygous mutation of many ribosomal protein gene loci leads to the dominant ‘Minute’ phenotype, named for its small thin bristles.1, 2 Minute animals show a dominant developmental delay. In addition, Minute (that is, Rp/+) cells tend to be lost from mosaics that contain wild-type cells, making it difficult for clones of Rp/+ genotypes to survive and contribute to the adult.3, 4, 5, 6, 7 Such conditional cell viability that depends on a heterotypic cellular environment is termed ‘cell competition’.4
Competition of Rp/+ clones is suppressed by equalizing growth rates through starvation8 or nonmosaic mutation of a second Rp locus.4 Hyperplastic clones that express higher levels of myc9, 10 or lower levels of the Salvador-Hippo-Warts pathway tumor suppressors out-compete nearby wild-type cells, that is, they are ‘super-competitors’.7, 11 Competition based on c-myc also occurs in mouse embryogenesis.12 Differential growth is not always sufficient to cause cell competition, as cells growing rapidly due to elevated CyclinD/Cdk4 activity or higher activity of the insulin/IGF pathway are not super-competitive.9 Differences in Jak/Stat signaling, Wg signaling and cell adhesion are also reported to generate cell competition.13, 14, 15 These findings suggest that cell competition arises from specific interactions between cells, rather than as a general consequence of differential growth.
Apoptotic cell death is a fundamental part of cell competition. Elimination of Rp/+ clones is delayed by expression of the caspase inhibitor baculovirus p35.5 Apoptosis of Rp/+ cells also occurs when clones of wild-type cells arise in Rp/+ backgrounds, predominantly among Rp/+ cells nearby wild-type cells.6, 16 As expected, such apoptosis is prevented by expression of baculovirus p35 or DIAP1.6, 16, 17
Cell competition has been hypothesized to contribute to human cancer, because most tumors have an altered genotype, and because many genes implicated in cell competition are homologs of oncogenes and tumor suppressors.18, 19, 20, 21 Cell competition may contribute to homeostasis of organ growth4, 9 and to antitumor surveillance.22, 23, 24, 25, 26
Cell competition may be a means to eliminate certain categories of aneuploid cells.27, 28 Seventy-nine ribosomal protein genes, sixty-six of which are haploinsufficient Minute loci, are distributed throughout the Drosophila genome.2 Copy number changes to parts of the genome are likely to perturb relative dose of Rp/+ genes, and those that reduce Rp gene dose could be subject to cell competition. This suggests cell competition can eliminate some aneuploid cells even after DNA damage responses have ceased.27, 28, 29
In humans, heterozygosity for multiple different Rp mutations causes Diamond Blackfan Anemia.30 Accumulation of ribosomal assembly intermediates or of unassembled ribosomal proteins in these genotypes activates p53, for example through the binding of the p53 ubiquitin ligase Mdm2 by RpL11 or RpL5.31 The p53 pathway leads to cell cycle arrest and/or apoptosis,32 and loss of hematopoietic stem cells causes anemia. Diamond Blackfan Anemia is a condition of nonmosaic individuals, so its relationship to cell competition is unclear.
The uncertain nature of the cell interactions that trigger competition might be illuminated if the initiation of competitive apoptosis was understood. The Drosophila genome encodes three potential initiator caspases that might be activated through long prodomains, and four effector caspase zymogens lacking prodomains that are activated by initiator caspases and by one another.33 Here, the p53 and initiator caspase requirements for competitive cell death of Rp/+ cells were determined. Whereas Dronc is the initiator caspase for most apoptosis in Drosophila,34, 35, 36, 37 we found that competitive cell death could occur without dronc or p53. Experiments that eliminated multiple initiator caspases simultaneously demonstrated that competitive apoptosis of Rp/+ cells required Dronc and Dream/Strica redundantly, a difference from most other apoptotic genotypes in Drosophila, for example, Rp/Rp cells generated in these experiments died in a Dronc-dependent manner.
Results
Cell competition depended on Reaper, Hid and Grim
Cells dying during cell competition are positive in TUNEL, and immunoreactive to anti-active caspase antibodies. Elimination of RpL19/+ or RpS3/+ clones is delayed by p35 expression,5, 6 and elimination of RpS17/+ clones is delayed by p35 expression or DIAP1 expression.17 These findings establish that cell competition removes cells by caspase-dependent programmed cell death.
The pro-apoptotic proteins Hid, Grim and Reaper antagonize DIAP1 so that their expression releases caspase activity from negative regulation.38, 39 The role of rpr, grim and hid in competitive cell death was evaluated using a deficiency, Df(3L)H99, which removes all three genes.33, 40 When clones of RpL36/+ cells are generated in a wild-type background, making use of an RpL36+ transgene linked to FRT80B, these cells are eliminated by cell competition7(Figures 1a and b). By contrast, clones of RpL36/+; Df(3L)H99/Df(3L)H99 cells survived in the imaginal discs and adult eye (Figures 1c and e). These findings indicated that cell competition required one or more of Reaper, Hid and Grim.
Cell competition depends on Reaper, Hid and Grim. (a) Genotype: y w p[eyFLP ry+]; FRT80B/FRT80B p[w+]. Recombinant y w p[eyFLP ry+]; FRT80B/FRT80B cells are unpigmented (white); recombinant y w p[eyFLP ry+]; FRT80B p[w+]/FRT80B p[w+] cells are pigmented (red). (b) Genotype: w RpL36/y w p[eyFLP ry+]; p[RpL36+ w+] FRT80B/FRT80B. Cell competition causes elimination of unpigmented Rp/+ eye cells of the recombinant genotype w RpL36/y w p[eyFLP ry+]; FRT80B/FRT80B. (c) Genotype: w RpL36/y w p[eyFLP ry+]; p[RpL36+ w+] FRT80B/Df(3L)H99 FRT80B. Rp/+ cells of genotype w RpL36/y w p[eyFLP ry+]; Df(3L)H99 FRT80B survive in the adult eye. This indicates that genes deleted by Df(3L)H99 must be present for competitive elimination of Rp/+ cells. (d) Genotype: w RpL36/y w p[eyFLP ry+]; p[RpL36+ w+] p[arm-LacZ w+] FRT80B/FRT80B. Few RpL36/+ cells (black, unlabeled for β-galactosidase) remain in the eye imaginal disc, because of competition with neighboring cells that carry the RpL36+ transgene linked to p[arm-LacZ w+]. (e) Genotype: w RpL36/y w p[eyFLP ry+]; p[RpL36+ w+] p[arm-LacZ w+] FRT80B/Df(3L)H99 FRT80B. Many RpL36/+; Df(3L)H99 clones (black, unlabeled for β-galactosidase) occur in a mosaic eye imaginal disc, showing that genes deleted by Df(3L)H99 must be present for competitive elimination of Rp/+ cells. (f) Genotype: w RpL36/y w p[eyFLP ry+]; p[RpL36+ w+] p[arm-LacZ w+] FRT80B/Df(3L)IRER FRT80B. Like the control shown in d, few RpL36/+ cells survive in the eye disc (black, unlabeled for β-galactosidase). Unlike Df(3L)H99, the regulatory region deletion Df(3L)IRER did not rescue RpL36/+ cells from competition. (g) Genotype: hsFlp; FRT42 ubi-GFP M(2)56f/FRT42; hid20-10-LacZ/+. GFP (green: g′), β-galactosidase (blue: g′′), and anti-Dcp1 (red: g′′′). Hid-LacZ expression is elevated in all the M(2)56f/+ wing disc cells. It is sometimes higher still in those undergoing apoptosis, but not consistently. Clones homozygous for the FRT42 ubi-GFP M(2)56f chromosome should be generated in these experiments, but none were seen, reflecting cell lethality of the M(2)56f mutation. (h) Genotype: hsFlp; rprXRE-LacZ/+; FRT82 ubi-GFP M(3)95A/FRT82 labeled for GFP (green: h′), β-galactosidase (blue: h′′), and anti-Dcp1 (red: h′′′). Rpr-LacZ expression is elevated in all the M(3)95A/+ wing disc cells, and is not higher still in those undergoing apoptosis. Clones homozygous for the FRT82 ubi-GFP M(3)95A chromosome should be generated in these experiments, but none were seen, reflecting cell lethality of the M(3)95A mutation
To examine expression levels, β-galactosidase reporters for hid and rpr were examined. When clones of wild-type cells were induced in RpS3/+ animals, hid-LacZ expression was elevated in the RpS3/+ cells (Figure 1g). This applied to all RpS3/+ cells, however, not only cells dying through cell competition (Figure 1g). When clones of wild-type cells were induced in RpS18/+ animals, rpr-LacZ expression was elevated in all the RpS18/+ cells (Figure 1h).
Competitive cell death occurred independently of p53
A cis-regulatory element mediates stress-induced Reaper and Hid expression and apoptosis during embryogenesis, and in response to gamma-irradiation via p53.41 To examine cis-regulation of rpr and hid transcription in cell competition, we made use of the IRER (irradiation-responsive enhancer region) deficiency.41 In contrast to deletion of rpr, grim and hid (Figures 1d and e) loss of the IRER cis-regulatory region did not permit the survival of RpL36/+ (Figure 1f). Therefore, competitive cell death did not depend on transcriptional induction through the IRER cis-regulatory region.
To evaluate the contribution of p53 itself to cell competition, clones of wild-type cells were induced in RpS18/+ animals that also lacked p53. Apoptosis of RpS18/+ cells was not lowered significantly by p53 mutation (Figure 2). p53 null alleles also failed to rescue clones of RpL36/+ cells from competitive elimination during development (Figures 2d–f).
Competitive apoptosis in the absence of p53. (a and b) Wing imaginal discs labeled for the RpS18 chromosome in magenta (anti-GFP, a′, b′, h′, i′) and apoptotic cells in green (anti-active Dcp1, a″, b″, h″, i″). Clones of wild-type cells (lacking magenta labeling) have been induced by heat-shock-induced mitotic recombination. In (a) apoptotic cells RpS18/+ occur at interfaces between wild-type and RpS18/+ cells. Genotype: y w hsF; FRT42D/FRT42D p[Ubi-GFP] M(2)56F. (b) In the absence of p53, competitive apoptosis continues at the interfaces between wild-type and RpS18/+ cells. Genotype: y w hsF; FRT42D/FRT42D p[Ubi-GFP] M(2)56F; p535A-1-4/p5311-1B-1. RpS18/RpS18 cells should be generated as reciprocal recombinants in these experiments, but such clones were never seen, indicating that this genotype was cell-lethal in the presence or absence of p53. (c) Quantification of RpS18/+ cell death rates at interfaces with wild-type cells, normalized for the length of boundaries. (d) A mosaic eye from genotype y w p[eyFLP ry+]; FRT82/FRT82 p[w+]. The adult eye predominantly contains two recombinant genotypes. The y w p[eyFLP ry+]; FRT82/FRT82 cells are unpigmented (white). The y w p[eyFLP ry+]; FRT82 p[w+]/FRT82 p[w+] cells are pigmented (red). (e) A mosaic eye from genotype w RpL36/y w p[eyFLP ry+]; p[RpL36+ w+] FRT82/FRT82 Cell competition causes elimination of unpigmented Rp/+ eye cells of the recombinant genotype w RpL36/y w p[eyFLP ry+]; FRT82/FRT82. (f) A mosaic eye from genotype w RpL36/y w p[eyFLP ry+]; p[RpL36+ w+] FRT82/FRT82 p535A-1. Cell competition causes elimination of unpigmented recombinant Rp/+ eye cells, even though they are homozygous for the p53 null allele. (g) Properties of p535A-1 homozygous clones induced in hsFlp; FRT82 M95A armLacZ/FRT82 p535A-1 wing imaginal discs, induced at 92±4 h AEL at 25 °C, normalized to data from control clones induced in parallel in hsFlp; FRT82 M95A armLacZ/FRT82 wing discs. Clones lacking p53 did not significantly affect size of the wing discs, clone size, frequency of cell death among M95A/+ cells or frequency of competitive M95A/+ cell death along clone boundaries. Data represent the mean and S.D. from three separate experiments (significant differences were not seen in any of the three individual experiments). Measurements of cell death rate within the wild-type and p53 homozygous clones themselves exhibit high S.E.s because of the small absolute numbers of non-Minute cells that die, often zero or one per wing disc, a rate more than 20 times lower than the death of M95A/+ cells. (h) Wing imaginal disc labeled for the M95A/+ genotype (anti-β-galactosidase: magenta) and for apoptotic cells (anti-Dcp1: green). Most dying M95A/+ cells are at borders between wild-type clones and M95A/+ cells. (i) Wing imaginal disc labeled for the M95A/+ genotype (anti-β-galactosidase: magenta) and for apoptotic cells (anti-Dcp1: green). Most dying M95A/+ cells are at borders between p535A-1 homozygous clones and M95A/p535A-1 cells
Because p53 has a role in the growth stimulation of super-competitor cells overexpressing Myc,42 we examined the effect of p53 homozygosity in wild-type cells competing with RpS3/+ cells. RpS3/+ cells in contact with wild-type or with p53 mutant cells died with equal frequency, and p53 mutations did not significantly affect growth of wild-type cells in this context (Figures 2g–i). There was a trend to lower levels of apoptosis within p53 mutant cells themselves compared with wild-type cells, however, which could indicate that the occasional death of wild-type cells was p53-dependent (Figure 2g).
Baculovirus p35 allows Rp/Rp cells to survive and divide
The caspase inhibitor baculovirus p35 blocks apoptosis, but does not inhibit Dronc, the main initiator caspase for developmental cell death.43, 44, 45 Dronc substrate cleavage in cells protected by p35 is detected by a polyclonal antiserum raised against mouse Caspase-3 called ‘CM1’.46, 47 Rescued cells with persistent Dronc activity are termed ‘undead’,48, 49 and map locations of developmental cell death signaling.46, 50
To map Dronc-dependent apoptosis in cell competition, p35 was expressed in the posterior compartment of Rp/+ wing discs, and wild-type clones induced. Such p35 expression blocked competitive cell death completely, as measured by TUNEL (Figure 3a).6 In the anterior compartments (lacking p35 expression) CM1 labeled single Rp/+ cells at boundaries with wild-type clones, as described previously.6 TUNEL labeling confirmed that most of these cells were apoptotic (Figures 3a and b). By contrast to the anterior compartments, CM1 did not label single posterior cells but clusters of cells, typically numbering about 10 cells and near to +/+ clones (Figure 3a). Most clusters were not of the Rp/+ genotype. Rather, β-galactosidase labeling indicated the undead cell clusters were the β-gal Rp/β-gal Rp genotype arising as the reciprocal recombinant to +/+ cells (Figures 3a and c). Rescue by p35 indicated that the death of Rp/Rp cells was caspase-dependent, at least for mutations in RpS3.
Protection of Rp mutant cells by Baculovirus p35. (a) Wing imaginal disc labeled for the M(3)95A chromosome in blue (a′, anti-β-galactosidase labeling), apoptosis (a′′, TUNEL in green) and caspase cleavage (a′′′, CM1 antibody in red). Only occasionally were cells observed labeled by CM1 but not TUNEL or vice versa. In anterior compartments, most dying cells are coincidentally labeled by TUNEL and CM1, and represent Rp/+ cells with wild-type neighbors. In posterior compartments, baculovirus p35 completely suppressed cell death as detected by TUNEL. Caspase activation occurred in clusters of undead cells. Nearly all such undead cell clusters corresponded to the Rp/Rp genotype (magenta arrows); clusters of Rp/+ undead cells were less common (orange arrow), and also seen in Rp/+ discs without recombinant clones (data not shown). Three rectangles indicated regions enlarged in b–d. (b) Enlargement from a, indicating the general coincidence between TUNEL (b′′) and CM1 (b′) labels, and the location of most dying cells to the boundaries of the Rp/+ territory (b′′′) with wild-type cells. Note that apoptotic cells can often be recognized from the appearance of their β-galactosidase immunoreactivity, presumably a consequence of the concentration of cellular contents that is a defining morphological feature of apoptosis. A very few cells with such an appearance lacked either TUNEL or anti-caspase labeling, suggesting that some apoptotic cells may not be labeled by either method (data not shown). (c) Enlargement from a, showing a cluster of undead Rp/Rp cells labeled for caspase activation (CM1, c′). c′′, c′′′ definitions as for panel b. (d) Enlargement from a, showing a cluster of undead Rp/+ cells labeled for caspase activation (CM1). Genotype: y w hsF; en-GAL4 UAS-GFP/UAS-p35; FRT82B/FRT82B p[arm-LacZ] M(3)95A
CM1 labeling of Rp/+ cells expressing p35 was rare (Figure 3a). No undead Rp/+ cells were seen at boundaries between wild-type clones and Rp/+ cells. CM1 labeling was sometimes observed, however, in p35-expressing Rp/+ cells away from boundaries with +/+ clones (Figure 3d). These clusters were much less common than Rp/Rp clusters. This suggests that Rp/+ cells protected from competitive cell death by p35 expressed little Dronc activity, less than undead Rp/Rp cells.
Dronc activity promotes Wg expression in undead Rp/Rp cells but not in competed Rp/+ cells
Ectopic expression of Wingless proteins is a Dronc function that occurs in the presence of p35.48, 51, 52 The p35-expressing Rp/+ cells did not express ectopic Wg at boundaries with +/+ clones, although normal, developmentally localized Wg expression was readily detected (Figure 4a). In contrast, undead Rp/Rp cells expressed Wg, which was abolished by co-expressing Dronc-DN along with p35 (Figures 4a and b).
Characterization of undead Rp/Rp cells. (a) Wing imaginal disc labeled for genotype (magenta, a′) and Wingless protein (Green, a″). Arrows point to examples of undead Rp/Rp clones in the p35-expressing posterior compartment. Many express ectopic Wg protein. Ectopic Wg was not seen in cells of other genotypes. (b) Co-expression of baculovirus p35 and Dronc-DN, labeled for genotype (magenta, b′) and Wingless protein (Green, b″). Arrows indicate undead M/M clones. Ectopic Wg expression is suppressed in the presence of Dronc-DN. Genotype: y w hsF; en-GAL4 UAS-GFP/UAS-p35 UAS-DroncCARD; FRT82B/FRT82B p[arm-LacZ] M(3)95A. (c) Wing imaginal disc containing wild-type and M/M clones induced after 10-min heat shock. Arrows indicate examples of undead M/M clones in the posterior compartment. (d) Wing imaginal disc containing wild-type and M/M clones induced after 60-min heat shock. Arrows indicate examples of undead M/M clones in the posterior compartment. Compared with c, more clones are induced by the extended heat shock. (e) Overlay of wings without heat shock (red) and subject to 60-min heat shock (green), illustrating increased posterior compartment size following heat shock. Genotype: y w hsF; en-GAL4 UAS-GFP/UAS-p35; FRT82B/FRT82B p[arm-LacZ] M(3)95A, except for b. (f) Quantification of results shown in e. Ten-minute heat shock induction of clones did not significantly change posterior compartment size in comparison with the anterior, whereas 60-min heat shock did
Ectopic Wg expression from undead cells promotes growth.48, 51, 52 We and others have reported that posterior compartments are not larger when p35 is expressed during cell competition.6, 9 To re-examine this, we varied the number of Rp/Rp twin spots. A 10-min heat shock induced 10 recombination events per compartment, on average, with no detectable effect on wing size (Figure 4c). By contrast, 60-min heat shock resulted in 30 undead Rp/Rp clones in the posterior compartment, on average (Figure 4d), and a significant increase in the size of posterior compartments, detectable in the adult wings (Figures 4e and f). As no Wg protein was detected in Rp/+ cells, we attribute such ectopic growth to the undead Rp/Rp cells.
Dronc is not required for death of competed Rp/+ cells
As Dronc activity was not detectable in Rp/+ cells, its role was tested genetically. Clones of +/+ cells were induced in Rp/+ wing discs, where Dronc-DN expression was targeted to posterior compartments. Cell death was similar in the posterior compartments expressing Dronc-DN to the anterior compartments (Figure 5a), suggesting that Dronc was not essential for competitive cell death. A second approach used dronc null mutations.9 Homozygous mutant clones grew like their wild-type twins in non-Minute backgrounds (Figures 5b–d). When mitotic recombination was used to generate clones of RpL36/+ cells in a wild-type background, such clones were outcompeted and not recovered (Figure 5e). RpL36/+ clones were still eliminated when homozygous for dronc null mutations, indicating that dronc was not crucial for their competition (Figures 5f and g).
Requirements for dronc in Minute apoptosis. (a) Dronc-DN expression in posterior compartments did not suppress competitive death of Rp/+ cells. Rp/Rp cells were seen in the posterior compartments expressing Dronc-DN (arrows). However, some Rp/Rp clones were labeled by CM1 and appeared to contain apoptotic cells (arrows). (b) Clones of FRT80B control cells grew comparably to their twin spots. (c) Clones of droncl29 homozygous cells grew comparably to their twin spots. (d) Clones of droncl24 homozygous cells grew comparably to their twin spots. (e) Clones heterozygous for RpL36 were not recovered after mitotic recombination. (f) No RpL36/+ clones homozygous for droncl24 were recovered. (g) No RpL36/+ clones homozygous for droncl94 were recovered. (h) When baculovirus p35 and Dronc-DN were co-expressed in posterior compartments, Rp/Rp clones lacked the CM1 labeling typical of ‘undead’ cells. Genotypes: (a) y w hsF; en-GAL4 UAS-GFP/UAS-DroncCARD; FRT82B/FRT82B p[arm-LacZ] M(3)95A. (b) y w hsF; FRT80B/p{arm-LacZ] FRT80B. (c) y w hsF; droncI29 FRT80B/p{arm-LacZ] FRT80B. (d) y w hsF; droncI24 FRT80B/p{arm-LacZ] FRT80B. (e) y w hsF/w M(1)Bld; FRT80B/p[arm-LacZ] p[w+ RpL36+] FRT80B. (f) y w hsF/w M(1)Bld; droncI24 FRT80B/p[arm-LacZ] p[w+ RpL36+] FRT80B. (g) y w hsF/w M(1)Bld; droncI29 FRT80B/p[arm-LacZ] p[w+ RpL36+] FRT80B. (h) y w hsF; en-GAL4 UAS-GFP/UAS-p35 UAS-DroncCARD; FRT82B/FRT82B p[arm-LacZ] M(3)95A
Dronc participates in cell death of Rp/Rp cells
We also assessed dronc in undead Rp/Rp cells (Figure 3). Dronc-DN abolished CM1 labeling of Rp/Rp cells expressing p35, indicating Dronc activity in undead Rp/Rp cells (Figure 5h). These results also confirmed that Dronc-DN expression reduced Dronc activity in vivo, in the same preparations where Rp/+ cells were studied. When Dronc-DN was expressed without p35, Rp/Rp cells were still observed (Figure 5a). The protection afforded by DroncDN was incomplete in two respects: clones of Rp/Rp cells expressing Dronc-DN were smaller and less abundant than clusters of Rp/Rp cells expressing p35 (Figures 5a and 3a); some Rp/Rp cells expressing Dronc-DN were labeled by CM1 and showed the condensed morphology typical of apoptotic cells (Figure 5a, inset arrow). Taken together, the findings suggest that Dronc contributed to Rp/Rp cell death, but that some apoptosis continued in the presence of Dronc-DN.
Cell death of competed Rp/+ cells is independent of Dredd and Dream/strica
The other potential apical caspases Dream/strica and Dredd might be responsible for Dronc-independent competitive cell death. Both are inhibited by p35, consistent with the fact that p35 prevents both death and CM1 labeling of Rp/+ cells.
The role of Dream/strica was examined using a deletion in this gene, by inducing +/+ clones in Rp/+ animals that were dream4 homozygous. Competitive cell death occurred at rates indistinguishable between dream4 homozygotes and controls (Figures 6a, b, e and f). Therefore, Dream/strica was dispensable for competitive cell death. As Rp/Rp clones were not seen, Dream/strica may be dispensable for death of these cells also (Figure 6).
Dream/Strica and Dredd are dispensable for competitive death of Rp/+ cells. (a) Clones of wild-type cells (unlabeled, black) in RpS3/+ background (magenta). Cell death labeled by activated caspase-3 labeling (green). Genotype: y w hsF; FRT82B/FRT82B p[arm-LacZ] M(3)95A. (b) Clones of non-Minute cells (unlabeled, black) in RpS3/+ background (magenta) induced in animals homozygous for a dream null allele. Cell death labeled by activated caspase-3 labeling (green). Genotype: y w hsF; dream4/dream4; FRT82B/FRT82B p[arm-LacZ] M(3)95A. (c) Clones of non-Minute cells (unlabeled, black) in RpS18/+ background (magenta). Cell death labeled by activated Dcp-1 labeling (green). Genotype: y w hsF; FRT42D/FRT42D p[Ubi-GFP] M(2)56F. (d) Clones of wild type cells in RpS18/+ background in animals homozygous for a dredd null allele. Cell death labeled by activated Dcp-1 labeling (green). Genotype: dreddB118 hsflp; FRT42D/FRT42D p[Ubi-GFP] M(2)56F. (e–h) Quantification of Rp/+ cell death at clone boundaries for genotypes shown in a–d. For the y axis, cell death rates quantified per micron of boundary between genotypes
The role of Dredd was examined using a null allele, by inducing +/+ clones in Rp/+ animals that were dreddB118 homozygous. Competitive cell death occurred at rates indistinguishable between dreddB118 homozygotes and controls (Figures 6c, d, g and h). No Rp/Rp clones were seen. Therefore, dredd also appeared dispensable for competitive cell death and for death of Rp/Rp cells.
Competitive cell death depends redundantly on Dream/strica and Dronc
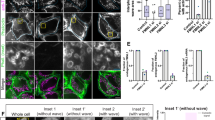
Although not individually required, initiator caspases might act redundantly in competitive apoptosis of Rp/+ cells. Although the dronc, dream and dredd triple mutant genotype has been constructed previously,53 severe viability and fertility effects were encountered when combining caspase mutations with Minute backgrounds. An alternative inducible RNAi approach was undertaken (Figure 7). When dsRNA for dronc was expressed in Rp/+ posterior compartments, competitive cell death occurred at boundaries with +/+ clones (Figure 7a), like the results with Dronc-DN (Figure 5a). The ~25% reduction compared with the control anterior compartments was not significant statistically (Figure 7d). Interestingly, dsRNA for dronc reduced apoptosis of Rp/+ cells that were not adjacent to wild-type clones. This was obvious in wing discs that lacked any wild-type clones, where dronc knockdown reduced apoptosis in posterior compartments (supplementary Figure 1). This finding suggested that some Rp/+ cells undergo dronc-dependent apoptosis independently of cell competition, and also that the dsRNA was effective. When dsRNA for dream was expressed in Rp/+ posterior compartments, cell death continued at boundaries with +/+ clones (Figure 7b), at rates not significantly different from controls (Figures 7b and e), similar to the results obtained using dream mutants (Figures 6a, b, e and f). By contrast, expressing dsRNAs for both dronc and dream simultaneously was sufficient to eliminate cell death almost completely in Rp/+ posterior compartments containing +/+ clones (Figures 7c and f).
Competitive death of Rp/+ cells depends on both Dronc and Dream/Strica. Third instar wing imaginal discs where +/+ cell clones are unlabelled (black), Rp/+ cells are labeled for β-galactosidase expression (magenta), cell death labeled in green and the compartmental boundary is marked in white line. Co-expression of dronc and dream specific dsRNA in the posterior compartment blocks cell death of Rp/+ cells due to competition but dsRNA against dronc or dream alone could not block it. (a) Clones of-wild type cells (unlabeled, black) in RpS3/+ background (magenta). Cell death labeled by activated caspase-3 labeling (green). Posterior compartment expression of dsRNA for dronc made little difference. Genotype: y w hsF/UAS-Dicer-2; en-GAL4 UAS-GFP/UAS-DroncRNAi; FRT82B/FRT82B p[arm-LacZ] M(3)95A. (b) Clones of wild type cells (unlabeled, black) in RpS3/+ background (magenta). Cell death labeled by activated caspase-3 labeling (green). Posterior compartment expression of dsRNA for dream/strica made no difference. Genotype: y w hsF/UAS-Dicer-2; en-GAL4 UAS-GFP/UAS-DreamRNAi; FRT82B/FRT82B p[arm-LacZ] M(3)95A. (c) Clones of wild type cells (unlabeled, black) in RpS3/+ background (magenta). Cell death labeled by activated caspase-3 labeling (green). Posterior compartment co-expression of dsRNAs for dronc and dream/strica abolished competitive apoptosis. Genotype: y w hsF/UAS-Dicer-2; en-GAL4 UAS-GFP/UAS-DroncRNAi -UAS-DreamRNAi; FRT82B/FRT82B p[arm-LacZ] M(3)95A. (d–f) Quantified data corresponding to a–c. Differences were not significant in d and e (P>0.05), but highly significant for f (P<0.01)
Caspase knockdown could also protect Rp/Rp clones (Supplementary Figure 2). When dsRNA for dronc was expressed, Rp/Rp clones survived (Supplementary Figure 2A), like when Dronc-DN was expressed (Figure 5a). No Rp/Rp clones survived when dsRNA for dream was expressed (Supplementary Figure 2B), like the results with dream mutants (Figure 6b). Rp/Rp clones did survive when both dronc and dream were knocked down (Supplementary Figure 2C),
Taken together, the findings indicate that competitive cell death of Rp/+ cells depended redundantly on Dronc and Dream initiator caspases, whereas death of Rp/Rp cells depended mainly on Dronc.
Discussion
Rp/+ cells in competition with wild-type cells undergo programmed cell death. Regardless of whether Rp/+ clones are in a wild-type background, or wild-type clones are in a Rp/+ background, cell death occurs that can be prevented by the caspase inhibitors baculovirus p35 and DIAP1, or by removing the pro-apoptotic genes grim, reaper and hid5, 6, 16, 54 (Figure 1). We show that competitive cell death involves less Dronc activity than other forms of apoptosis, and can be initiated by Dream/Strica.
Apoptosis of Rp/Rp cells
Unexpectedly, we found that Rp/Rp cells die through apoptosis, and therefore can survive and divide in the presence of baculovirus p35. In our experiments, cells homozygous for M(3)95A, a mutation thought to correspond to RpS3,2 were protected by p35, Dronc-DN or dsRNA for dronc (Figures 3a, c, 4b, c and 5a and Supplementary Figure 2). The findings are not specific to this mutation: p35 protected clones homozygous for the M(3)i55 mutation corresponding to RpS17 (see Figures 2f and 4d of Martin et al17).
It is doubtful that cells divide in the complete absence of essential ribosomal proteins. Even if these alleles are null, presumably each clone of Rp/Rp cells begins as a recombinant cell that inherits ribosomes from the Rp/+ mother cell. Our results suggest that as ribosome numbers diminish with time and growth, apoptosis is triggered before growth becomes impossible, and Rp/Rp cells can undergo a few more divisions if they are protected from apoptosis. Apoptosis of Rp/Rp cells provides a useful contrast with competitive death of Rp/+ cells within the same preparations.
Apoptotic Rp/+ cells lack dronc activity
When p35 protects cells from effector caspases, persistent Dronc activity marks the ‘undead’ cells.46, 51, 52 Undead Rp/Rp cells were labeled by CM1 and expressed Wg. They lost these undead markers when Dronc-DN was expressed along with p35, confirming Dronc activity (Figures 3, 4 and 5h). By contrast, Rp/+ cells protected by p35 were unlabeled by CM1 antibodies or for Wg (Figures 3 and 4). Therefore, competitive death of Rp/+ cells seems to involve little Dronc activity. Competitive death is initiated either not by Dronc, or by Dronc activity at low levels. The Dronc independence of competitive apoptosis precluded using undead cells to map competitive cell death signals, which has been possible for developmental apoptosis.46
Different Dronc requirements in competitive and noncompetitive apoptosis
The different levels of Dronc activity in Rp/+ and Rp/Rp cells mirrored different dronc requirements. Expression of Dronc-DN protected Rp/Rp cells, albeit less effectively than baculovirus p35 (Figure 5a), as did dsRNA for dronc (Supplementary Figure 2). Dronc-DN may be less effective than p35 because Dronc-DN does not inhibit Dronc activity completely. Alternatively, if Rp/Rp cells are subject to competition with Rp/+ cells in addition to cell-autonomous cell death, Rp/Rp cells that were protected from apoptosis by Dronc-DN would still be subject to Dronc-independent competition with neighboring Rp/+ cells, and for this reason would survive less well than Rp/Rp cells protected by p35.
By contrast, Rp/+ clones were eliminated in the absence of dronc (Figures 5e–g) or in the presence of Dronc-DN or dsRNA for dronc (Figures 5 and 7). Dronc-DN and dsRNA for dronc may retain some activity, and clones of cells homozygous for dronc null alleles may inherit Dronc protein or RNA, but as these reagents effectively blocked dronc-dependent processes in Rp/Rp cells (Figures 3, 4, 5 and Supplementary Figure 2), competitive death of Rp/+ cells must require less Dronc activity than is normal in other forms of apoptosis, such as that of Rp/Rp cells.
Competitive apoptosis depends redundantly on Dronc and Dream/Strica
The remaining initiator caspases Dredd and Dream/strica were also dispensable for competitive cell death (Figure 6). Effector caspases, which are clearly both activated and required, must either be activated by multiple initiator caspases redundantly, or by novel mechanisms. It proved possible to express dsRNA for both dronc and dream in competing cells, and this blocked competitive cell death almost completely, indicating that Dronc and Dream/Strica acted redundantly to initiate competitive cell death (Figures 7c and f). It was notable that dsRNA for dronc alone led to some reduction in cell death, although it was not significant statistically in our study (Figures 7a and d). This may indicate that dream does not substitute completely for dronc. However, reduced cell death of Rp/+ cells even in Rp/+ imaginal discs that lack wild-type clones indicates that Rp/+ cells were subject to a background of dronc-dependent cell death not due to cell competition (Supplementary Figure 1). Alone, dsRNA for dream/strica had no effect (Figures 7b and e). Overall, these data indicate that dronc and dream could each initiate competitive cell death, rendering competitive cell death less dependent on dronc than most apoptosis in Drosophila.
Another group concluded that competitive death of RpS17/+ cells is dronc-dependent, and that p35 expression during cell competition leads to undead Rp/+ cells. Their study, however, did not differentiate the apoptosis of Rp/Rp cells from Rp/+ cells, or note the presence of undead Rp/Rp in the presence of p35.17 In addition, the M(3)i55 chromosome that is mutant for RpS17 may exhibit a higher background of dronc-dependent, noncompetitive Rp/+ cell death than the M(3)95A chromosome used here (our unpublished observations, and compare Figure 3a of this work with Figure 4d from Martin et al17). It is important to distinguish these dronc-dependent death mechanisms from cell competition.
Role of pro-apoptotic genes in cell competition
Dronc is commonly activated in the apoptosome by expression of the pro-apoptotic genes rpr, grim or hid, which lead to the degradation of anti-apoptotic DIAP1 protein.55 Indeed, rpr and hid transcription was elevated in Rp/+ cells, and deleting rpr, grim and hid prevented cell competition (Figure 1). These data fall short of proof that elevated rpr, grim or hid expression initiate competitive cell death. Transcriptional reporters for hid and rpr were elevated in all Rp/+ cells, most of which were not apoptotic (Figure 1). As Rp/+ cells do not transcribe higher levels of DIAP1,56 elevated rpr or hid may sensitize Rp/+ cells to cell death without being the specific trigger. Elevating DIAP1 after rpr, grim and hid deletion might de-sensitize Rp/+ cells, rather than block a specific pathway. An interesting possibility is that Rp/+ cells elevate a nonautonomous survival factor. Such a factor would be present at lower levels near to wild-type cells, potentially providing a trigger for competitive cell death at clone boundaries. Such a scenario remains hypothetical until the factor is identified.
Competitive apoptosis is independent of p53
Transcription of rpr and hid in response to stress or gamma-irradiation depends on the IRER enhancer.41 Unlike some other examples of apoptosis, competitive cell death did not depend on this enhancer, ruling out at least one mechanism by which changes in pro-apoptotic gene expression levels could lead to cell competition. The IRER enhancer, as well as the transcription of hid and rpr, are transcriptional targets of p53, and p53 is activated by Rp mutations in vertebrates.31 Cell competition continued in the absence of p53, however (Figure 2). In addition, Rp/+ cells did not express ectopic Wg in the presence of p35 (Figure 4a), which would be expected if p53 was activated.57
It is significant that competitive cell death in Drosophila continued in the absence of p53 (Figure 2) for two reasons. First, it indicated that cell competition was not mediated by the vertebrate nucleolar stress pathway, whereby certain ribosomal proteins stabilize p53 and trigger death of hematopoietic stem cells.31 Accordingly, the MDM2 ubiquitin ligase that is targeted by vertebrate ribosomal proteins is not encoded in the Drosophila genome. It remains possible that other mechanisms of nucleolar stress occur in Drosophila. Secondly, it has been suggested that cell competition could be a mechanism to remove aneuploid cells.27, 28 As there is p53-independent removal of aneuploid cells,27, 28 our findings are consistent with cell competition as the mechanism.
Dream/Strica in competitive cell death
It could be proposed that expression levels determine the contributions of Dronc and Dream/strica to cell death. In this view, Dream/strica levels are inadequate to initiate developmental cell death, but higher levels of Dream/strica proteins in Rp/+ cells become sufficient to initiate competitive apoptosis in the absence of Dronc. There is evidence against this simple model, however, because the lack of undead Rp/+ cells in the presence of baculovirus p35 indicated that Dronc activity was unusually low in competitive apoptosis. In addition, apoptosis of Rp/Rp cells, as well as noncompetitive apoptosis of Rp/+ cells, were both dronc-dependent but independent of dream/strica. The fact that Rp/+ cells are capable of dronc-dependent and dronc/dream-dependent cell death processes is difficult to explain unless competitive cell death specifically activates Dream/strica in addition to Dronc.
Little is yet known concerning the molecular mechanisms of Dream/strica activation. Dream/strica differs from other initiator caspases in that its extensive prodomain is serine/threonine-rich and lacks a caspase recruitment domain or death effector domain, suggesting that it could be activated by a distinct mechanism.58 The protein interacts with both DIAP1 and DIAP2,58 and some studies suggest that Dream/strica accelerates aspects of dronc-dependent apoptosis. Dream/strica and Dronc are also required redundantly for nurse cell death during oogenesis and the programmed elimination of certain peptidergic neurons from the CNS during pupariation.53, 59 Further characterization of mechanisms of Dream/Strica activation may be revealing concerning the induction of competitive apoptosis by interactions between wild-type and Rp/+ cells.
Materials and Methods
Fly strains
Genotypes of each experiment are described in figure legends. The following genetic strains were used: Df(3L)H99;3 FRT80B;60 M(1)Bld; p{w+, rpL36+};7 DF(3L)IRER;41 p535A-1-4; and p5311-1B-1;61 en-Gal4 UAS-GFP;62 UAS-p35;43 arm-LacZ;63 ubi-GFP;64 UAS-DroncDN;45 droncI24; droncI29;37 dream4 and dreddB118;53 hid20-10-LacZ and rprXRE-LacZ;65 UAS-dsRNAdronc (VDRC100424) and UAS-dsRNAdream (VDRC22594) UAS-dcr2 (VDRC60007);66 for M(2)56F (a mutation of RpS18), M(3)i55 (a mutation of RpS17 ) and M(3)95A (a mutation of RpS3) see Marygold et al.2
Mosaics
Mosaic clones were generated by the FRT-FLP technique.67 Flies were maintained at 25°C on standard media unless otherwise noted. Unless otherwise specified, for Minute genotypes 37°C heat shock was administered for 60 min at 72±12 or 84±12 h after egg laying and imaginal discs were dissected 72 h later; other genotypes were heat-shocked 60±12 h after egg laying and imaginal discs dissected 60 h later.
Immunohistochemistry
Antibody and TUNEL labelings were performed as described.68 Confocal images were recorded using Biorad Radiance (Hercules, CA, USA) 2000 and Leica (Wetzlar, Germany) SP2 systems and processed using ImageJ (rsb.info.nih.gov) and Photoshop (Adobe Systems Inc., San Jose, CA, USA). Primary antibodies are as follows: mouse anti-β-galactosidase (mAb40-1a) from DSHB; rabbit anti-activated caspase 3 (CM1 from Idun Pharmaceuticals, San Diego, CA, USA);69 rabbit anti-activated-caspase 3 (Cell Signaling Technology, Danvers, MA, USA); rabbit anti-activated DCP1 (Cell Signaling Technology); rat anti-GFP (Nacalai Tesque Inc., Kyoto, Japan); rabbit anti-GFP (Invitrogen, ThermoFischer Scientific, Waltham, MA, USA). Labeling for anti-active Dcp1 is similar to CM1, but does not persist in the presence of p35 (our unpublished observations).
Secondary antibodies were multilabeling antibodies from Jackson ImmunoResearch Laboratories (West Grove, PA, USA).
Abbreviations
- Rp:
-
ribosomal protein
- TUNEL:
-
terminal deoxynucleotide transferase dUTP nick end labeling
References
Lambertsson A . The minute genes in Drosophila and their molecular functions. Adv Genet 1998; 38: 69–134.
Marygold SJ, Roote J, Reuter G, Lambertsson A, Ashburner M, Millburn GH et al. The ribosomal protein genes and Minute loci of Drosophila melanogaster. Genome Biol 2007; 8: R216.
Morata G, Ripoll P . Minutes: mutants of Drosophila autonomously affecting cell division rate. Dev Biol 1975; 42: 211–221.
Simpson P, Morata G . Differential mitotic rates and patterns of growth in compartments in the Drosophila wing. Dev Biol 1981; 85: 299–308.
Moreno E, Basler K, Morata G . Cells compete for decapentaplegic survival factor to prevent apoptosis in Drosophila wing development. Nature 2002; 416: 755–759.
Li W, Baker NE . Engulfment is required for cell competition. Cell 2007; 129: 1215–1225.
Tyler DM, Li W, Zhuo N, Pellock B, Baker NE . Genes affecting cell competition in Drosophila. Genetics 2007; 175: 643–657.
Simpson P . Parameters of cell competition in the compartments of the wing disc of Drosophila. Dev Biol 1979; 69: 182–193.
de la Cova C, Abril M, Bellosta P, Gallant P, Johnston LA . Drosophila myc regulates organ size by inducing cell competition. Cell 2004; 117: 107–116.
Moreno E, Basler K . dMyc transforms cells into super-competitors. Cell 2004; 117: 117–129.
Neto-Silva RM, de Beco S, Johnston LA . Evidence for a growth-stabilizing regulatory feedback mechanism between Myc and Yorkie, the Drosophila homolog of Yap. Dev Cell 2010; 19: 507–520.
Claveria C, Giovinazzo G, Sierra R, Torres M . Myc-driven endogenous cell competition in the early mammalian embryo. Nature 2013; 500: 39–44.
Vincent JP, Kolahgar G, Gagliardi M, Piddini E . Steep differences in wingless signaling trigger Myc-independent competitive cell interactions. Dev Cell 2011; 21: 366–374.
Hafezi Y, Bosch JA, Hariharan IK . Differences in levels of the transmembrane protein Crumbs can influence cell survival at clonal boundaries. Dev Biol 2012; 368: 358–369.
Rodrigues AB, Zoranovic T, Ayala-Camargo A, Grewal S, Reyes-Robles T, Krasny M et al. Activated STAT regulates growth and induces competitive interactions independently of Myc, Yorkie, Wingless and ribosome biogenesis. Development 2012; 139: 4051–4061.
Li W, Kale A, Baker NE . Oriented cell division as a response to cell death and cell competition. Curr Biol 2009; 19: 1821–1826.
Martin FA, Herrera SC, Morata G . Cell competition, growth and size control in the Drosophila wing imaginal disc. Development 2009; 136: 3747–3756.
Baker NE, Li W . Cell competition and its possible relation to cancer. Cancer Res 2008; 68: 5505–5507.
Moreno E . Is cell competition relevant to cancer? Nat Rev Cancer 2008; 8: 141–147.
Johnston LA . Competitive interactions between cells: death, growth, and geography. Science 2009; 324: 1679–1682.
Tamori Y, Deng WM . Cell competition and its implications for development and cancer. J Genet Genomics 2011; 38: 483–495.
Gateff E . Malignant neoplasms of genetic origin in Drosophila melanogaster. Science 1978; 200: 1448–1459.
Woods DF, Bryant PJ . The discs-large tumor suppressor gene of Drosophila encodes a guanylate kinase homolog localized at septate junctions. Cell 1991; 66: 451–464.
Agrawal N, Kango M, Mishra A, Sinha P . Neoplastic transformation and aberrant cell-cell interactions in genetic mosaics of lethal(2)giant larvae (lgl), a tumor suppressor gene of Drosophila. Dev Biol 1995; 172: 218–229.
Brumby AM, Richardson HE . scribble mutants cooperate with oncogenic Ras or Notch to cause neoplastic overgrowth in Drosophila. EMBO J 2003; 22: 5769–5779.
Ohsawa S, Sugimura K, Takino K, Xu T, Miyawaki A, Igaki T . Elimination of oncogenic neighbors by JNK-mediated engulfment in Drosophila. Dev Cell 2011; 20: 315–328.
Titen SW, Golic KG . Telomere loss provokes multiple pathways to apoptosis and produces genomic instability in Drosophila melanogaster. Genetics 2008; 180: 1821–1832.
McNamee LM, Brodsky MH . p53-independent apoptosis limits DNA damage-induced aneuploidy. Genetics 2009; 182: 423–435.
Baker NE . Cell competition. Curr Biol 2011; 21: R11–R15.
Vlachos A, Dahl N, Dianzani I, Lipton JM . Clinical utility gene card for: Diamond-Blackfan anemia—update 2013. Eur J Hum Genet 2013; 21; doi:doi:10.1038/ejhg.2013.34.
Fumagalli S, Di Cara A, Neb-Gulati A, Natt F, Schwemberger S, Hall J et al. Absence of nucleolar disruption after impairment of 40S ribosome biogenesis reveals an rpL11-translation-dependent mechanism of p53 induction. Nat Cell Biol 2009; 11: 501–508.
Moll UM, Wolff S, Speidel D, Deppert W . Transcription-independent pro-apoptotic functions of p53. Curr Opin Cell Biol 2005; 17: 631–636.
Hay BA, Guo M . Caspase-dependent cell death in Drosophila. Annu Rev Cell Dev Biol 2006; 22: 623–650.
Chew SK, Akdemir F, Chen P, Lu WJ, Mills K, Daish T et al. The apical caspase dronc governs programmed and unprogrammed cell death in Drosophila. Dev Cell 2004; 7: 897–907.
Daish TJ, Mills K, Kumar S . Drosophila caspase DRONC is required for specific developmental cell death pathways and stress-induced apoptosis. Dev Cell 2004; 7: 909–915.
Waldhuber M, Emoto K, Petritsch C . The Drosophila caspase DRONC is required for metamorphosis and cell death in response to irradiation and developmental signals. Mech Dev 2005; 122: 914–927.
Xu D, Li Y, Arcaro M, Lackey M, Bergmann A . The CARD-carrying caspase Dronc is essential for most, but not all, developmental cell death in Drosophila. Development 2005; 132: 2125–2134.
Bergmann A, Yang AY, Srivastava M . Regulators of IAP function: coming to grips with the grim reaper. Curr Opin Cell Biol 2003; 15: 717–724.
Hay BA, Huh JR, Guo M . The genetics of cell death: approaches, insights and opportunities in Drosophila. Nat Rev Genet 2004; 5: 911–922.
White K, Grether ME, Abrams JM, Young L, Farrell K, Steller H . Genetic control of programmed cell death in Drosophila. Science 1994; 264: 677–683.
Zhang Y, Lin N, Carroll PM, Chan G, Guan B, Xiao H et al. Epigenetic blocking of an enhancer region controls irradiation-induced proapoptotic gene expression in Drosophila embryos. Dev Cell 2008; 14: 481–493.
de la Cova C, Senoo-Matsuda N, Ziosi M, Wu DC, Bellosta P, Quinzii CM et al. Supercompetitor status of Drosophila Myc cells requires p53 as a fitness sensor to reprogram metabolism and promote viability. Cell Metab 2014; 19: 470–483.
Hay BA, Wolff T, Rubin GM . Expression of baculovirus P35 prevents cell death in Drosophila. Development 1994; 120: 2121–2129.
Hawkins CJ, Yoo SJ, Peterson EP, Wang SL, Vernooy SY, Hay BA . The Drosophila caspase DRONC cleaves following glutamate or aspartate and is regulated by DIAP1, HID, and GRIM. J Biol Chem 2000; 275: 27084–27093.
Meier P, Silke J, Leevers SJ, Evan GI . The Drosophila caspase DRONC is regulated by DIAP1. EMBO J 2000; 19: 598–611.
Yu SY, Yoo SJ, Yang L, Zapata C, Srinivasan A, Hay BA et al. A pathway of signals regulating effector and initiator caspases in the developing Drosophila eye. Development 2002; 129: 3269–3278.
Fan Y, Bergmann A . The cleaved-Caspase-3 antibody is a marker of Caspase-9-like DRONC activity in Drosophila. Cell Death Differ 2010; 17: 534–539.
Perez-Garijo A, Martin FA, Morata G . Caspase inhibition during apoptosis causes abnormal signalling and developmental aberrations in Drosophila. Development 2004; 131: 5591–5598.
Kondo S, Senoo-Matsuda N, Hiromi Y, Miura M . DRONC coordinates cell death and compensatory proliferation. Mol Cell Biol 2006; 26: 7258–7268.
Baker NE, Yu SY . The EGF receptor defines domains of cell cycle progression and survival to regulate cell number in the developing Drosophila eye. Cell 2001; 104: 699–708.
Huh JR, Guo M, Hay BA . Compensatory proliferation induced by cell death in the Drosophila wing disc requires activity of the apical cell death caspase Dronc in a nonapoptotic role. Curr Biol 2004; 14: 1262–1266.
Ryoo HD, Gorenc T, Steller H . Apoptotic cells can induce compensatory cell proliferation through the JNK and the Wingless signaling pathways. Dev Cell 2004; 7: 491–501.
Baum JS, Arama E, Steller H, McCall K . The Drosophila caspases Strica and Dronc function redundantly in programmed cell death during oogenesis. Cell Death Differ 2007; 14: 1508–1517.
Martin FA, Perez-Garijo A, Morata G . Apoptosis in Drosophila: compensatory proliferation and undead cells. Int J Dev Biol 2009; 53: 1341–1347.
Shapiro PJ, Hsu HH, Jung H, Robbins ES, Ryoo HD . Regulation of the Drosophila apoptosome through feedback inhibition. Nat Cell Biol 2008; 10: 1440–1446.
Wang L-H, Baker NE . Salvador-Warts-Hippo pathway in a developmental checkpoint monitoring Helix-Loop-Helix proteins. Dev Cell 2015 (in press).
Dichtel-Danjoy ML, Ma D, Dourlen P, Chatelain G, Napoletano F, Robin M et al. Drosophila p53 isoforms differentially regulate apoptosis and apoptosis-induced proliferation. Cell Death Differ 2013; 20: 108–116.
Doumanis J, Quinn L, Richardson H, Kumar S . STRICA, a novel Drosophila melanogaster caspase with an unusual serine/threonine-rich prodomain, interacts with DIAP1 and DIAP2. Cell Death Differ 2001; 8: 387–394.
Lee G, Wang Z, Sehgal R, Chen CH, Kikuno K, Hay B et al. Drosophila caspases involved in developmentally regulated programmed cell death of peptidergic neurons during early metamorphosis. J Comp Neurol 2011; 519: 34–48.
Cullen K, McCall K . Role of programmed cell death in patterning the Drosophila antennal arista. Dev Biol 2004; 275: 82–92.
Rong YS, Titen SW, Xie HB, Golic MM, Bastiani M, Bandyopadhyay P et al. Targeted mutagenesis by homologous recombination in D. melanogaster. Genes Dev 2002; 16: 1568–1581.
Neufeld TP, de la Cruz AF, Johnston LA, Edgar BA . Coordination of growth and cell division in the Drosophila wing. Cell 1998; 93: 1183–1193.
Vincent JP, Girdham CH, O'Farrell PH . A cell-autonomous, ubiquitous marker for the analysis of Drosophila genetic mosaics. Dev Biol 1994; 164: 328–331.
Janody F, Lee JD, Jahren N, Hazelett DJ, Benlali A, Miura GI et al. A mosaic genetic screen reveals distinct roles for trithorax and polycomb group genes in Drosophila eye development. Genetics 2004; 166: 187–200.
Fan Y, Lee TV, Xu D, Chen Z, Lamblin AF, Steller H et al. Dual roles of Drosophila p53 in cell death and cell differentiation. Cell Death Differ 2010; 17: 912–921.
Dietzl G, Chen D, Schnorrer F, Su KC, Barinova Y, Fellner M et al. A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila. Nature 2007; 448: 151–156.
Xu T, Rubin GM . Analysis of genetic mosaics in developing and adult Drosophila tissues. Development 1993; 117: 1223–1237.
Firth LC, Li W, Zhang H, Baker NE . Analyses of RAS regulation of eye development in Drosophila melanogaster. Methods Enzymol 2006; 407: 711–721.
Srinivasan A, Roth KA, Sayers RO, Shindler KS, Wong AM, Fritz LC et al. In situ immunodetection of activated caspase-3 in apoptotic neurons in the developing nervous system. Cell Death Differ 1998; 5: 1004–1016.
Acknowledgements
We thank P Meier, A Bergmann, B Lemaitre, L Zhou, K McCall, K White and J Treisman for Drosophila strains, and Idun Pharmaceuticals for the CM1 antibody. We thank L Zhou and K McCall for sharing unpublished information. Stocks obtained from the Bloomington Drosophila Stock Center (NIH P40OD018537) were used in this study. We thank L Johnston, J Secombe and L Zhou for comments on the manuscript, and G Rimesso for help with experiments. Confocal Imaging was performed at the Analytical Imaging Facility, Albert Einstein College of Medicine. This study was supported by grants from the NIH (GM061230 and GM104213) and by an unrestricted grant from Research to Prevent Blindness to the Department of Ophthalmology and Visual Sciences. Some data in this paper are from theses submitted in partial fulfillment of the requirements for the Degree of Doctor of Philosophy in the Graduate Division of Medical Sciences, Albert Einstein College of Medicine, Yeshiva University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by S Kumar
Supplementary Information accompanies this paper on Cell Death and Differentiation website
Rights and permissions
About this article
Cite this article
Kale, A., Li, W., Lee, CH. et al. Apoptotic mechanisms during competition of ribosomal protein mutant cells: roles of the initiator caspases Dronc and Dream/Strica. Cell Death Differ 22, 1300–1312 (2015). https://doi.org/10.1038/cdd.2014.218
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cdd.2014.218
This article is cited by
-
Emerging mechanisms of cell competition
Nature Reviews Genetics (2020)
-
Xrp1 is a transcription factor required for cell competition-driven elimination of loser cells
Scientific Reports (2018)
-
Chronic activation of JNK JAK/STAT and oxidative stress signalling causes the loser cell status
Nature Communications (2017)