Abstract
Little is known about the impact of multiple myeloma (MM) treatment on uninvolved immunoglobulins (Ig). We identified 448 patients who received high-dose dexamethasone (HD-DEX), lenalidomide and dexamethasone (RD), bortezomib and dexamethasone (VD), bortezomib, cyclophosphamide and dexamethasone (VCD) or bortezomib, lenalidomide and dexamethasone (VRD) for newly diagnosed MM at our institution between 2000 and 2013, and who had available data on absolute lymphocyte count (ALC) and quantitative uninvolved Ig at baseline and at the end of four cycles of therapy. Changes in ALC and uninvolved Ig were significantly different across treatments, with VCD and HD-DEX producing reductions in uninvolved Ig, and RD, VD and VRD leading to increases in uninvolved Ig. In addition, treatment with RD, VD and VRD was independently associated with higher odds of achieving a ⩾25% increase in or normalization of the primary uninvolved Ig on multivariate analysis. Although achievement of a humoral response in the primary uninvolved Ig was associated with a higher odds of achieving VGPR or better after four cycles of therapy, it was not associated with improved overall survival. These data highlight the different mechanisms of action of MM drugs and point toward a possible role for the use of VCD in treating antibody-mediated autoimmune disease.
Similar content being viewed by others
Introduction
Multiple myeloma (MM) is a clonal plasma cell proliferative disorder that is predicted to account for >15% of all hematologic malignancies diagnosed in the United States this year.1 The majority of patients present with an elevated monoclonal protein, and a reduction in one or more uninvolved, polyclonal immunoglobulin (Ig) is seen in >90% of patients, especially in individuals with IgA myeloma.2
Immunoparesis has been shown to have prognostic impact in MM, with inferior progression-free and overall survival (OS) seen in those with suppression of at least one uninvolved Ig compared with those in whom all uninvolved Ig are preserved;3 In addition, suppression of the uninvolved (polyclonal) Ig of the same isotype as the tumor is associated with a poorer prognosis.4 Similar findings have also been reported in MM-related disorders, including monoclonal gammopathy of undetermined significance,5 smoldering MM6 and light chain amyloidosis.7
There are, however, limited data on the changes that occur in uninvolved Ig during MM therapy. A pooled analysis of the phase 3 MM-009 and MM-010 lenalidomide trials showed that an increase in IgA occurred in >50% of patients with non-IgA MM and that these ‘humoral responders’ enjoyed longer progression-free and OS.8 A similar increase in progression-free survival among patients with an increase in uninvolved Ig after 6 months of treatment with lenalidomide has also been reported.9 However, these data are from patients with relapsed/refractory disease, which makes it difficult to discern the impact of the treatment regimen on immune function owing to prior exposure to other therapies, and there are also no data on agents such as bortezomib.10
We therefore sought to evaluate the changes in uninvolved Ig that occur during the first few months of therapy in a contemporary cohort of individuals with a new diagnosis of MM, and determine whether these changes are impacted by the treatment regimen, as well as the impact of such changes on treatment response and survival.
Subjects and methods
Study cohort
The study population was derived from an electronic database containing records of consecutive patients who were newly diagnosed with MM at Mayo Clinic between January 2000 and December 2013 (n=1155). Patients in whom Ig levels (IgA, IgG and IgM) were available both at the time of diagnosis as well at the end of approximately four cycles of first-line therapy were included; this was chosen as it typically represents the end of induction therapy in transplant-eligible patients.11 When Ig levels at the end of four cycles were unavailable, levels at the end of three (n=44), five (n=22) or six (n=15) cycles were used instead. Only those receiving the following first-line therapy were included: single-agent high-dose dexamethasone (HD-DEX), lenalidomide and dexamethasone (RD), bortezomib and dexamethasone (VD), bortezomib, cyclophosphamide and dexamethasone (VCD), and bortezomib, lenalidomide and dexamethasone (VRD). HD-DEX was included as it was used in the other treatments (RD, VD, VCD, VRD), and could therefore act as a useful comparator or control, whereas patient receiving thalidomide were excluded as it is not commonly used in contemporary induction regimes. Other exclusion criteria included biclonal or non-secretory MM, use of any plasma cell-directed therapy before the diagnosis of MM, and discontinuation or change of treatment prior to the end of cycle 4 of therapy.
Outcomes and definitions
After obtaining Institutional Review Board approval, baseline data on various parameters (Ig, difference in free light chains, white blood cell count, absolute lymphocyte count (ALC), percentage of bone marrow plasma cells), as well as the Ig and FLC levels at the end of four cycles of therapy were abstracted from the electronic medical record. Early transplant was defined as receipt of an autologous stem cell transplant after a maximum of six cycles of therapy. To assess change in uninvolved Ig, the mean of the percentage differences between baseline and four cycles for each uninvolved Ig (IgA and IgM for IgG myeloma, IgG and IgM for IgA myeloma, IgG and IgA for IgM and IgD myeloma, and IgG, IgM and IgA for light chain-only myeloma) was calculated. Change in the primary uninvolved Ig was determined by calculating the percentage change between baseline and four cycles in IgA (in cases of IgG myeloma) and IgG (for all other myeloma subtypes). Ig levels were categorized as ‘low’ or ‘normal’ based upon the reference ranges used in our laboratory (IgA: 61–356 mg/dl, IgM: 37–286 mg/dl, IgG: 767–1590 mg/dl). A humoral response in the primary uninvolved Ig constituted a conversion from a ‘low’ level to at least the lower limit of normal and/or a ⩾25% increase in levels between baseline and four cycles.8
Treatment response at the end of four cycles of therapy was determined according to International Myeloma Working Group criteria.12 Time to treatment failure (TTF) was defined as the duration from the start of initial therapy to the start of the next line of therapy (for disease progression) or death (if no additional treatment was received), or censored at the date of last follow-up. OS was calculated in a landmark fashion, as the duration between the date of follow-up after four cycles of therapy and the date of last follow-up or death, or censored at the date of last follow-up.
Statistical analysis
The Kruskal–Wallis and χ2-tests were used to detect differences in medians and proportions, respectively, and logistic regression used to test the association between variables and outcomes of interest. OS and TTF analyses were performed using the Kaplan––Meier method, and the log-rank test used to make comparisons between groups; a P-value of <0.05 was considered to be significant. Statistical analysis was performed using SPSS v.20 (IBM Corp., Armonk, NY, USA).
Results
A total of 448 patients were included in this analysis (Table 1). The median age of the cohort was 63 years (range 33–91), and the majority of patients (58%) were male. The monoclonal Ig subtype was IgG (59%), IgA (23%) or light chain-only (16%) in the majority of cases, and the most common first line of therapy was RD (n=221, 49%), followed by HD-DEX (n=82, 18%), VCD (n=77, 17%), VRD (n=46, 10%) and VD (n=22, 5%). Median OS was 7.8 years (95% confidence interval 7.0–8.5), and median TTF was 2.1 years (1.9−2.3); median follow-up was 6.4 years (5.7–7.0).
Changes in uninvolved Ig between baseline and four cycles of therapy
The levels of white blood cell count, ALC and difference in free light chains after four cycles, and the changes in white blood cell count, ALC and uninvolved Ig between baseline and four cycles are shown in Table 2. Treatment with VCD was associated with the greatest median decline in ALC, with the smallest reduction in ALC seen with the use of HD-DEX (HD-DEX: −4% (−71 to +376); RD: −11% (–84 to +719); VD: −21% (−70 to +386); VCD: −46% (−93 to +112); VRD: −21% (−76 to +141), P<0.001). Changes in uninvolved Ig were significantly different across treatments, with a median reduction in uninvolved Ig seen with the use of HD-DEX (−2% (−78 to +1094)) and VCD (−14% (−75 to +244)), and median increases observed with RD (+14% (−71 to +564)), VD (+29% (−63 to +329)) and VRD (+81% (−65 to +590), P<0.001), with VCD producing the largest decline in uninvolved Ig. Similar trends were seen when change in only the primary uninvolved Ig was considered, with HD-DEX, VD and VCD producing median reductions in the primary uninvolved Ig (HD-DEX: −27% (−80 to +630); VD: −2% (−87 to +320); VCD: −18% (−88 to +271)), and increases seen with RD and VRD (RD: +3% (−69 to +788); VRD: +9% (−81 to +683), P<0.001).
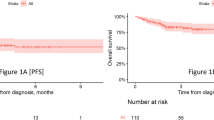
The same pattern of change in white blood cell count, ALC, uninvolved and primary Ig across treatment regimens was seen when only patients achieving VGPR or better after four cycles were considered (Supplementary Table 1). A dot-plot of the change in uninvolved Ig across different treatments and stratified by response after four cycles is shown in Figure 1.
Pattern of change in primary uninvolved Ig and humoral response
The pattern of change in the primary uninvolved Ig is shown in Table 3. Normalization of a low primary uninvolved Ig occurred in 43 patients overall (10%), and was more frequently seen with the RD (14%) and VRD (13%) compared with HD-DEX (5%), VD (5%) and VCD (1%, P=0.035). A similar pattern emerged when considering only the subgroup of patients in whom the primary uninvolved Ig remained low (n=314); within this cohort, an increase (but not to normal levels) was seen more frequently with the use of RD (49%), VD (44%) and VRD (53%) compared with HD-DEX (20%) and VCD (36%, P=0.001).
A humoral response in the primary uninvolved Ig occurred in 111 patients (25%), and was observed more frequently with RD (28%), VD (32%) and VRD (41%) in comparison with HD-DEX (11%) and VCD (18%, P=0.001). On multivariate analysis, adjusting for age, sex and monoclonal Ig subtype, treatment with RD (OR=3.06 (1.42−6.60), P=0.004), VD (OR=3.91 (1.23−12.46), P=0.021) and VRD (OR=6.34 (2.49−16.14)), P<0.001) was associated with significantly higher odds of producing a humoral response in the primary uninvolved Ig (Table 4).
Changes in uninvolved Ig, treatment response and survival
On multivariate analysis adjusting for age, sex, treatment regimen and monoclonal Ig subtype, a humoral response in the primary uninvolved Ig was associated with a significantly greater odds of being in VGPR or better after four cycles of therapy (OR=3.30 (1.91–5.69), P<0.001, Table 5). Median TTF was longer in patients in whom a humoral response was seen, compared with those without a humoral response (2.5 years vs 2.0 years, P=0.034), but no differences in OS were seen between the two groups (7.6 years vs 7.8 years, P=0.440, Figure 2). There were no significant differences in either TTF (1.9 years vs 2.2 years, P=0.513) or OS (7.8 years vs 8.2 years, P=0.748) between patients in whom the average percentage change in uninvolved Ig between baseline and four cycles was <0 and those in whom it was ⩾0 (Figure 3).
Discussion
MM is inextricably linked to immune dysfunction as it is a disorder of plasma cells, whose role is to produce antibodies during the adaptive immune response. Treatment of MM comprises of glucocorticoids, alkylating agents, thalidomide analogs and proteasome inhibitors, along with targeted therapies such as daratumumab13 and elotuzumab,14 which have recently been developed. It is apparent that studying the immune impact of MM treatment can generate information on the mechanism of action of these drugs, as well as providing evidence on the potential uses of these agents in immunologic disorders, where modulation of plasma cells and lymphocytes may be useful.
In this regard, our study has several noteworthy findings. First, our analysis has shown that MM treatments exert differential impacts on immune function in newly diagnosed patients, as measured by the change in ALC and uninvolved Ig during induction therapy. Treatment with VCD was associated with the greatest reductions in ALC and uninvolved Ig, whereas RD and VD tended to produce increases in uninvolved Ig, with the triplet VRD combination leading to the highest increases in uninvolved Ig. Similarly, RD, VD and VRD were independently associated with greater odds of leading to humoral responses in the primary uninvolved Ig. These results were generally replicated when only considering patients who achieved VGPR or better after four cycles of therapy, confirming that these immunologic effects are not response-specific. To our knowledge, these are the first data comparing changes in uninvolved Ig and ALC between myeloma treatments and provide insight into the mechanisms of action of these drugs.
The molecular basis underlying the reduction in uninvolved Ig in MM is thought to be mediated by interactions between B-cell maturation antigen (BCMA) and its ligands, BAFF (B-cell activating factor) and (APRIL) a proliferation inducing ligand, which stimulate B-cell differentiation and antibody production.15 Soluble BCMA has been identified in the serum of MM patients,16 and recent work suggests that this may bind to circulating BAFF, thereby interfering with its role in stimulating antibody production and leading to a reduction in polyclonal, uninvolved Ig.17 Pre-clinical data suggest that lenalidomide and bortezomib exert inhibitory effects on the BAFF/ARPIL/BCMA pathway,18 with xenograft studies showing that BCMA levels decrease after treatment with the latter.16 In addition, in vitro studies have shown that monoclonal antibodies directed against BCMA or APRIL augment the cytotoxicity of lenalidomide and dexamethasone against MM cells,19, 20 and such combinations are being investigated in Phase 1 trials.21 Taken together, these mechanisms could explain our findings that RD, VD and especially VRD produced the largest rises in uninvolved Ig and greatest humoral responses, as lenalidomide and bortezomib may inhibit pathways that lead to immunoparesis in MM.
In contrast to RD, VD and VRD, treatment with VCD led to significant reductions in uninvolved Ig, with significantly smaller humoral responses rates. The most straightforward explanation is that the VCD regimen includes a classic cytotoxic agent (cyclophosphamide), leading to greater immunosuppression (by means of lymphopenia) in comparison with RD and VRD, which contain an immunomodulatory drug (lenalidomide) and produce an enhanced immune response. In the case of VCD, this effect seems to override the more tumor-specific action of bortezomib. Nevertheless, despite the different effects on uninvolved Ig, the odds of achieving VGPR or better after four cycles of therapy were very similar with VCD and VRD, suggesting that general suppression of lymphocytes and B cells (with VCD) and a more tumor-specific action (with VRD) may be roughly comparable treatment strategies. Unfortunately, head-to-head studies of these regimens are not available.
We also found a greater suppression of uninvolved Ig with HD-DEX compared to RD, VD and VRD. This effect may be related to the dose intensity of dexamethasone; with HD-DEX, a total of 480 mg of dexamethasone is administered per month, whereas low-dose dexamethasone (120–160 mg per month) is the standard dose used in doublet and triplet regimens at our institution. Furthermore, the rates of achieving VGPR or better were lower with HD-DEX compared with combination therapy, and this may also limit recovery of uninvolved Igs.
In addition, we noted that the attainment of a humoral response in the primary uninvolved Ig was independently associated with higher odds of being in VGPR or better after four cycles of therapy, which translated into a marginal increase in TTF but had no impact on OS. Achieving better disease control likely enables recovery of normal plasma cells and restores the ability to mount a humoral immune response. The improvement in TTF in humoral responders is in keeping with previous studies showing improvement in progression-free survival among lenalidomide-treated patients in whom there were similar increases in uninvolved Ig;8, 9 nevertheless, the magnitude of the TTF benefit (~6 months) is smaller than previously reported, whereas there was no OS benefit for humoral responders, in contrast to prior data.8 Reasons for this include differences in patient cohort (we assessed newly diagnosed MM patients receiving induction therapy, whereas prior work was undertaken on either relapsed/refractory patients8 or in patients receiving maintenance or post-induction therapy9) as well as the wider range of treatments in our study in comparison to previous data that pertained only to lenalidomide. Our results also suggest that the importance placed on uninvolved Ig as a marker of immunodeficiency, and subsequent risk of grave infectious complications, may be overstated.
There are limitations of our analysis aside from those inherent to a retrospective, observational study. First, there may have been selection bias in the choice of initial therapy, and the patient population was heterogeneous, albeit limited to only newly diagnosed MM patients who had not received prior therapy. The number of patients who received VD was small (n=22), although the results of changes in ALC and uninvolved Ig seen with VRD seem to corroborate the findings with VD, given that the results with VRD were, to a degree, additive compared with RD and VD alone. Finally, we were unable to capture data on infectious complications in parallel with the changes in Ig. However, the large number of patients studied, long follow-up, and evaluation of several treatments, all myeloma subtypes, and all uninvolved Ig serve to ensure that these results are a valuable addition to the literature.
In summary, this study highlighted that the changes in uninvolved Ig and ALC during MM therapy differ according to treatment regimen. VCD and HD-DEX produce reductions in uninvolved Ig, whereas RD, VD and VRD are associated with increases in uninvolved Ig, suggesting differential mechanisms of action among these agents. Moreover, these data point towards a possible role for VCD in the treatment of autoimmune disease, where reduction of antibodies production by non-malignant, polyclonal plasma cells is a therapeutic goal. Studies should examine the use of VCD in the treatment of these disorders, since a short course of VCD may be more tolerable than chronic treatment with steroids and other immunosuppressive agents, which is the typical therapy for such patients.
References
Siegel RL, Miller KD, Jemal A . Cancer Statistics, 2017. CA Cancer J Clin 2017.
Kyle RA, Gertz MA, Witzig TE, Lust JA, Lacy MQ, Dispenzieri A et al. Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clin Proc 2003; 78: 21–33.
Kastritis E, Zagouri F, Symeonidis A, Roussou M, Sioni A, Pouli A et al. Preserved levels of uninvolved immunoglobulins are independently associated with favorable outcome in patients with symptomatic multiple myeloma. Leukemia 2014; 28: 2075–2079.
Bradwell A, Harding S, Fourrier N, Mathiot C, Attal M, Moreau P et al. Prognostic utility of intact immunoglobulin Ig'kappa/Ig'lambda ratios in multiple myeloma patients. Leukemia 2013; 27: 202–207.
Katzmann JA, Clark R, Kyle RA, Larson DR, Therneau TM, Melton LJ 3rd et al. Suppression of uninvolved immunoglobulins defined by heavy/light chain pair suppression is a risk factor for progression of MGUS. Leukemia 2013; 27: 208–212.
Perez-Persona E, Mateo G, Garcia-Sanz R, Mateos MV, de Las Heras N, de Coca AG et al. Risk of progression in smouldering myeloma and monoclonal gammopathies of unknown significance: comparative analysis of the evolution of monoclonal component and multiparameter flow cytometry of bone marrow plasma cells. Br J Haematol 2010; 148: 110–114.
Muchtar E, Dispenzieri A, Kumar SK, Buadi FK, Lacy MQ, Zeldenrust S et al. Immunoparesis in newly diagnosed AL amyloidosis is a marker for response and survival. Leukemia 2016; 31: 92–99.
Dimopoulos MA, Swern AS, Li JS, Hussein M, Weiss L, Nagarwala Y et al. Efficacy and safety of long-term treatment with lenalidomide and dexamethasone in patients with relapsed/refractory multiple myeloma. Blood Cancer J 2014; 4: e257.
Zamarin D, Devlin SM, Arcila ME, Landau H, Lesokhin A, Lendvai N et al. Polyclonal immune activation and marrow plasmacytosis in multiple myeloma patients receiving long-term lenalidomide therapy: incidence and prognostic significance. Leukemia 2013; 27: 2422–2424.
Moreau P, Richardson PG, Cavo M, Orlowski RZ, San Miguel JF, Palumbo A et al. Proteasome inhibitors in multiple myeloma: 10 years later. Blood 2012; 120: 947–959.
Rajkumar SV, Kumar S . Multiple myeloma. Diagnosis and Treatment. Mayo Clin Proc 2016; 91: 101–119.
Durie BG, Harousseau JL, Miguel JS, Blade J, Barlogie B, Anderson K et al. International uniform response criteria for multiple myeloma. Leukemia 2006; 20: 1467–1473.
Lokhorst HM, Plesner T, Laubach JP, Nahi H, Gimsing P, Hansson M et al. Targeting CD38 with Daratumumab Monotherapy in Multiple Myeloma. N Engl J Med 2015; 373: 1207–1219.
Lonial S, Dimopoulos M, Palumbo A, White D, Grosicki S, Spicka I et al. Elotuzumab Therapy for Relapsed or Refractory Multiple Myeloma. N Engl J Med 2015; 373: 621–631.
Moore PA, Belvedere O, Orr A, Pieri K, LaFleur DW, Feng P et al. BLyS: member of the tumor necrosis factor family and B lymphocyte stimulator. Science 1999; 285: 260–263.
Sanchez E, Li M, Kitto A, Li J, Wang CS, Kirk DT et al. Serum B-cell maturation antigen is elevated in multiple myeloma and correlates with disease status and survival. Br J Haematol 2012; 158: 727–738.
Sanchez E, Gillespie A, Tang G, Ferros M, Harutyunyan NM, Vardanyan S et al. Soluble B-cell maturation antigen mediates tumor-induced immune deficiency in multiple myeloma. Clin Cancer Res 2016; 22: 3383–3397.
Shen X, Guo Y, Qi J, Shi W, Wu X, Ni H et al. Study on the association between miRNA-202 expression and drug sensitivity in multiple myeloma cells. Pathol Oncol Res 2016; 22: 531–539.
Tai YT, Mayes PA, Acharya C, Zhong MY, Cea M, Cagnetta A et al. Novel anti-B-cell maturation antigen antibody-drug conjugate (GSK2857916) selectively induces killing of multiple myeloma. Blood 2014; 123: 3128–3138.
Tai YT, Acharya C, An G, Moschetta M, Zhong MY, Feng X et al. APRIL and BCMA promote human multiple myeloma growth and immunosuppression in the bone marrow microenvironment. Blood 2016; 127: 3225–3236.
Raje N, Faber E, Richardson PG, Schiller G, Hohl RJ, Cohen AD et al. Phase 1 study of tabalumab, a human anti-B-cell activating factor antibody, and bortezomib in patients with relapsed/refractory multiple myeloma. Clin Cancer Res 2016; 22: 5688–5695.
Acknowledgements
Supported in part by grants CA 107476, CA 168762 and CA186781 from the National Cancer Institute, Rockville, MD, USA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
SK has obtained research support for clinical trials from Celgene, Millennium, Novartis, Janssen and Sanofi. AD has received research support for clinical trials from Pfizer, Jannsen, Millennium, Alnylam and Celgene. MAG has received research support from ISIS and Prothena, and honoraria from Celgene, Millennium Pharmaceuticals and Novartis. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Blood Cancer Journal website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Ravi, P., Kumar, S., Gonsalves, W. et al. Changes in uninvolved immunoglobulins during induction therapy for newly diagnosed multiple myeloma. Blood Cancer J. 7, e569 (2017). https://doi.org/10.1038/bcj.2017.46
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bcj.2017.46
This article is cited by
-
International Myeloma Working Group risk stratification model for smoldering multiple myeloma (SMM)
Blood Cancer Journal (2020)