Abstract
Aim:
To quantitatively assess the effect of lowering external Ca2+ ([Ca2+]o) on both endothelium-dependent and -independent relaxations in rabbit aorta.
Methods:
Isometric contractions and relaxations of isolated aortae were recorded. When assessing the effect of reduced [Ca2+]o on relaxations, the normal [Ca2+]o solution was substituted with one of the reduced [Ca2+]o solutions for one aorta, while a paired aorta was replenished with normal [Ca2+]o solution.
Results:
The extent of acetylcholine (ACh)-induced relaxation, which is dependent on an intact endothelium, is time-dependent, and inversely related to [Ca2+]o in a range of 0.02–2 mmol/L. ACh-induced relaxations were not significantly altered by the magnitude of the precontraction induced by PGF2α. Nitroprusside-induced relaxations, which are independent of the endothelium, are also attenuated by reduced [Ca2+]o. Relaxant responses to ACh were significantly more susceptible to reduced [Ca2+]o than nitroprusside-induced relaxations. A maximally effective relaxing concentration of D600, an L-type Ca channel blocker methoxyverapamil, (10−5 mol/L) attenuated ACh-induced relaxations, whereas nitroprusside-induced relaxations were unaffected by D600.
Conclusion:
Thus, endothelium-dependent relaxation is more dependent on [Ca2+]o than endothelium-independent relaxation, and it seems likely that [Ca2+]o plays an important role not only in contractile processes, but also in relaxant processes as well.
Similar content being viewed by others
Introduction
Extracellular Ca2+ ([Ca2+]o) is required for full contractile responses of vascular smooth muscle1, 2, and this Ca2+ dependence varies depending upon specific stimulants and blood vessels3, 4, 5, 6. In contrast to a large number of investigations concerned with delineating the relationship between Ca2+ and the excitation/contraction coupling processes in vascular smooth muscle, it has not been clearly elucidated what general role [Ca2+]o might play in relaxant responses.
There is clear evidence that some vasodilators such as acetylcholine (ACh) cause relaxation by liberating an endothelium-derived relaxing factor (EDRF)7, nitric oxide (NO) which was identified later8, 9. On the other hand, other vasodilators such as nitroprusside, are generally regarded as endothelium-independent, and have direct inhibitory effects on vascular smooth muscle10, 11. It has been recently demonstrated that the presence of extracellular Ca2+ is required for release of EDRF11, 12, 13, 14. In these studies, Ca2+-free conditions abolished endothelium-dependent relaxation, whereas different. Ca2+ entry blockers had varied effects on similar endothelium-dependent relaxations. The use of Ca2+-free solutions frequently employed in these types of studies usually results in a noticeably diminished tension response which is generally required for quantitative analyses of relaxations, and this may have made it difficult to quantitatively assess the role of [Ca2+]o in relaxation processes. Thus, the present study was undertaken to quantitatively assess the effect of reducing [Ca2+]o in a concentration-dependent fashion on both endothelium-dependent and -independent relaxations in rabbit aorta.
Materials and methods
Isolated aortae
New Zealand white rabbits of either sex were anesthetized with 50 mg/kg (iv) of sodium pentobarbital, and then sacrificed by exsanguination from the common carotid arteries. Rabbits used in this study were 10–12 weeks old with a mean body weight of 1.86±0.03 kg (n=60). Thoracic aortae were quickly removed, isolated and cleaned, and cut into opened aortic rings 3 mm wide similar to the method of Carrier et al15. Tissues were fixed vertically between stainless steel clips in a tissue bath containing 20 mL nutrient solution, aerated with 95% O2+5% CO2, and maintained at 36–37 °C. The clip anchoring the upper end of the strip was connected to the lever of a force-displacement transducer (Grass FT 03). Using previously described procedures for passive force-active force relationships16, near optimum resting tension at which actively developed contractions were maximized in rabbit aortae was found to include a broad range (2.0–6.0 g) of passive forces (Authors, unpublished data). A resting force of 2.0 g was placed on each tissue to be consistent with previous studies17.
Procedures of aortic contraction/relaxation experiment in the bath
Constituents of the nutrient solution (normal Ca2+) were as follows (mmol/L): NaCl, 142; KCl, 5.4; CaCl2, 2.0; NaHCO3, 18.0; and dextrose, 11.0. The modified solutions of reduced [Ca2+]o contained the following concentrations of CaCl2 instead of 2.0 mmol/L: 1.0 mmol/L (1/2); 0.5 mmol/L (1/4); 0.2 mmol/L (1/10); 0.06 mmol/L (3/100); and 0.02 mmol/L (1/100 ) . The pH of continuously aerated solutions at 37 °C was 7.35–7.45. Osmotic adjustment was not made when KCl was increased up to 80 mmol/L. Before specific experimental protocols were initiated, preparations were allowed to equilibrate for 60-90 min in the bathing medium, during which time the solution was replaced every 10 to 15 min.
Isometric contractions and relaxations were recorded on an inkwriting oscillograph (Gould 2600S). Concentration-effect relationships were obtained by adding drugs directly to the bathing medium in a cumulative fashion. For studies on relaxant responses to ACh and nitroprusside, preparations were initially contracted with PGF2α (4×10−7–10−6 mol/L). At the end of each series of experiments, papaverine (10−4 mol/L) plus nitroprusside (10−4 mol/L) was added to obtain the maximum relaxation. In experiments conducted in reduced [Ca2+]o, 2 mmol/L CaCl2 was added with papaverine plus nitroprusside. Some aortae were dragged slowly for one min with intimal surface down7 over a sheet of sandpaper (3M, #320) wetted with nutrient solution to remove the endothelium. Abolition of relaxant responses to ACh was used to verify functional damage of endothelial cells in these intimal-rubbed preparations.
Substitution with reduced [Ca2+]o solutions
When assessing the effect of reduced [Ca2+]o on relaxations, the normal Ca2+ solution was substituted with one of the reduced [Ca2+]o solutions for one aorta, while a paired aorta was replenished with normal Ca2+ solution. Before substitution, modified solutions were maintained at 37 °C and continuously aerated. In every experiment, it was confirmed that substitution with normal Ca2+ solution did not appreciably alter resting tension. A substitution of normal Ca2+ solution with a low Ca was carefully made, since somewhat mechanical agitation of strip with change in the nutrient solution may alter aortic tension and/or endothelial activity as well as potential activation of some ion channels. However, no osmolarity adjustment was posed for Ca2+ within a small range of 2 mmol/L. KCl was added in a concentration of 80 mmol/L without osmorality adjustment, but this procedure and response was made at first in every aortic strip for checking the reactivity, suggesting a separate experiment from the low Ca study.
In experiments utilizing a 1/100 [Ca2+]o solution, responses to ACh were examined 30 min, 60 min, or 120 min after the substitution. PGF2α was added 15 min before the ACh-test relaxation to attain and maintain precontraction in 30 min studies and 15–30 min prior in 60 or 120 min studies. In all other studies using solutions with varied [Ca2+]o or added D600, aortae were exposed to either procedure for 120 min, including exposure to PGF2α for 15–30 min, prior to ACh- or nitroprusside-induced relaxations. Results are expressed as mean±SEM. Statistical analyses were made using Student's unpaired t-test.
Drugs
Drugs used were acetylcholine chloride (Sigma, St Louis, MO), sodium nitroprusside (Sigma), prostaglandin F2α-tromethamine (Upjohn, Kalamazoo, MI), methoxyverapamil (D600, Knoll AG, Ludwigshafen, West Germany) and histamine dihydrochloride (Sigma).
Results
Effect of reduced [Ca2+]o on ACh-induced relaxations
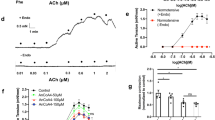
ACh elicited a relaxation of PGF2α -precontracted aortae that was readily abolished in endothelial rubbed preparations. The maximum relaxation due to 10−6 mol/L ACh was 68%±6% (n=8) of the papaverine plus nitroprusside-induced relaxation in aortae with intact endothelium, but -35±8% (n=8) (contraction) in aortae with damaged endothelium. Reducing [Ca2+]o to 0.02 mmol/L attenuated the ACh-induced relaxation in endothelium intact aortae. Significant attenuation occurred 30 min after the initial substitution with this reduced [Ca2+]o solution, reaching a maximum within 2 h (Figure 1). Thus in the following experiments concerned with defining the effects of various reductions in [Ca2+]o on relaxation, a 2 h exposure to the reduced [Ca2+]o solution was used. ACh-induced relaxation was attenuated by reducing [Ca2+]o from 2 to 0.2–0.02 mmol/L. The degree of attenuation was inversely related to [Ca2+]o, where reducing [Ca2+]o to 0.02 mmol/L essentially eliminated any ACh-induced relaxation (Figure 2). In a few experiments Ca2+ was added after the maximum ACh-induced relaxation without the concomitant addition of nitroprusside plus papaverine, and further relaxation ensued.
Inhibition of ACh-induced relaxation by reduced [Ca2+]o as a function of the duration of exposure to reduced [Ca2+]o. Preparations in normal Ca2+ or reduced [Ca2+]o (0.02 mmol/L) solutions were precontracted with PGF2α before adding ACh; contractions were 1825±233 mg (normal Ca2+) , 1284±386 mg (30 min), 1250±439 mg (60 min), and 1384±347 mg (120 min). Relaxant responses to ACh were expressed as a % of the papaverine plus nitroprusside-induced relaxation attained at the end of each experiment; relaxations were 2067±256 mg (control), 1368±400 mg (30 min), 1343±483 mg (60 min), and 1434±348 mg (120 min). bP<0.05, cP<0.01 vs control.
Effects of reduced [Ca2+]o on relaxant responses to ACh in preparations precontracted with PGF2α. Contractions were 1541±128 mg (normal Ca2+) , 1211±365 mg (1/4), 978±180 mg (1/10), 1341±361 mg (3/100), and 1249±178 mg (1/100). Papaverine plus nitroprusside-induced relaxation at the end of each experiment was taken as 100%; 1825±154 mg (normal Ca2+), 1400±356 mg (1/4), 1208±232 mg (1/10), 1582±347 mg (3/100), and 1331±188 mg (1/100).
When the level of precontraction was raised or lowered by increasing or decreasing the concentration of PGF2α rather than reducing [Ca2+]o, ACh-induced relaxation, expressed as a percent of the maximal papaverine plus nitroprusside-induced relaxation, did not differ (Figure 3). Also, if the level of tension in reduced [Ca2+]o was increased to approximate the same level as precontraction levels in normal Ca2+ by increasing the concentration of PGF2α accordingly, ACh-induced relaxatlon was still similarly depressed by reducing [Ca2+]o (data not shown).
Effects of varying the initial level of tone by altering the concentration of PGF2α on ACh-induced relaxations in a normal Ca2+ solution. Preparations were precontracted with 10−7 mol/L (I), 7×10−7 mol/L (II) or 10−5 mol/L (III) PGF2α; relative contractions shown at right in which contraction to concentration II was taken as 100%: 563±149 mg (I), 1792±194 mg (II), and 2258±155 mg (III).
When precontraction was induced by histamine (1×10−6– 3×10−6 mol/L) instead of PGF2α, reducing [Ca2+]o to 0.02 mmol/L also attenuated the maximum ACh-induced relaxation; 72%±4% of papaverine plus nitroprusside-induced relaxation in normal Ca2+ and 8%±8% in reduced [Ca2+]o (n=4).
Effects of reduced [Ca2+]o on nitroprusside-induced relaxations
Reducing the [Ca2+]o also attenuated relaxant responses to nitroprusside (Figure 4), which is not affected by endothelial damage (Figure 5, left). However, the attenuation was not apparent until [Ca2+]o was reduced to 0.06 mmol/L. Even in aortae without intact endothelium, reducing [Ca2+]o to 0.02 mmol/L depressed the nitroprusside-induced relaxation (Figure 5, right). Attenuation of the nitroprusside-induced relaxation by reducing [Ca2+]o to 0.02 mmol/L was also observed in aortae precontracted with histamine (1×10−6–3×10−6 mol/L); maximum response to nitroprusside was 95%±1% in normal Ca2+ and 4%±1% in reduced [Ca2+]o.
Effects of reduced [Ca2+]o on relaxant responses to nitroprusside in PGF2α-precontracted aortae. Precontractions were 1784±172 mg (normal Ca2+), 1842±392 mg (1/4), 1048±300 mg (1/10), 664±179 mg (3/100), and 1157±207 mg (1/100). Papaverine-induced relaxations at the end of each experiment were taken as 100%: 1979±198 mg (normal Ca2+), 2108±446 mg (1/4), 1214±318 mg (1/10), 794±175 mg (3/100), and 1271±243 mg (1/100).
Relaxant responses to nitroprusside in aortae with and without intact endothelium (left), and the effect of reducing [Ca2+]o to 0.02 mmol/L on relaxations in aortae without endothelium (right). Precontractions were 1547±285 mg and 2213±335 mg in aortae with (+) and without (−) endothelium (Endo), respectively, and 1833±223 mg (normal Ca2+) and 1230±317 mg (1/100 without endothelium) (right). Papaverine-induced relaxations after nitroprusside were taken as 100%: 1737±292 mg (Endo+), 2477±369 mg (Endo−), and 2104±241 mg (normal Ca2+), and 1332±331 mg (1/100) without endothelium (right). bP<0.05, cP<0.01 vs control.
Inhibitory effects of reduced [Ca2+]o on relaxant responses to nitroprusside were also compared to the inhibitory effects on ACh-induced responses (Figure 6). In terms of maximum responses, inhibition of nitroprusside-induced relaxations in a reduced [Ca2+]o solution was significantly less than that of ACh-induced relaxations. Furthermore, the relaxant response to 10−7 mol/L nitroprusside, which caused a similar degree of relaxation as the maximum effective concentration of ACh (71% vs 73%, respectively), was less attenuated by reduced [Ca2+]o than the ACh-induced relaxation.
Inhibitory effects of reduced [Ca2+]o on ACh-induced relaxations compared with similar effects on nitroprusside-induced relaxations. Comparisons were made based on data noted in Figure 2 and 4. The ordinate is expressed as inhibition (%) which represents the percent that maximum relaxant responses to ACh or nitroprusside in a reduced [Ca2+]o solution were to the maximum papaverine plus nitroprusside-induced relaxations divided by the same relationship in normal Ca2+ solution. Control maximum responses to ACh and nitroprusside were 73%±2% and 94%±1%, respectively, of papaverine plus nitroprusside-induced relaxations. Responses to 10−7 mol/L nitroprusside (closed triangles) were compared in normal and reduced [Ca2+]o solutions; relaxations in control were 71%±2% of the papaverine plus nitroprusside-induced relaxation. Significantly bP<0.05, cP<0.01 vs values of ACh (max).
Effects of Mg2+ and reduced [Ca2+]o on relaxant responses to ACh and nitroprusside
Mg2+ (0.6 mmol/L) did not significantly affect the ACh-induced relaxation in normal Ca2+; responses to ACh at concentrations of 10−7 and 10−6 mol/L were 63%±5% and 74%±5%; respectively, in the presence of Mg2+, and 63%±5% and 72%±4% in the absence (n=9). Mg2+ also did not prevent the inhibitory effects of reducing [Ca2+]o to 0.02 mmol/L on ACh-induced relaxations; maximum responses to ACh were 73%±4% in normal Ca2+ in the absence of Mg, and 14%±4% in reduced [Ca2+]o (0.02 mmol/L) plus Mg2+ (n=8) (P<0.001). Additionally, Mg2+ did not prevent the inhibitory effects of reduced [Ca2+]o on nitroprusside-induced relaxations; maximum responses to nitroprusside were 93%±2% in normal Ca2+ in the absence of Mg, and 41%±19% in the reduced [Ca2+]o (0.02 mmol/L) plus Mg2+ (n=5) (P<0.05).
Effects of reduced [Ca2+]o on contractile responses and resting tension
Reducing [Ca2+]o to 0.02 mmol/L for 2 h suppressed contractile responses to either PGF2α or KCl (Figure 7). Additionally, reducing [Ca2+]o to 0.02 mmol/L for 2 h resulted in a small, but appreciable and slow developing contraction of 255±74 mg, while tissue remaining in a normal Ca2+ solution for the same time period lost 63±28 mg (n=20) (P<0.001).
Effect of reducing [Ca2+]o to 0.02 mmol/L on contractile responses to PGF2α and KCl. Maximum contractions of control and Ca2+ deficient preparations in the presence of 2 mmol/L Ca2+ were taken as 100%. In the latter, Ca2+ was re-admitted at the end of each experiment. They were 2579±289 mg (control, left), 3093±453 mg (1/100, left), 3514±204 mg (control, right) and 3986±290 mg (1/100, right). cP<0.01 vs control.
Effects of D600 on ACh- and nitroprusside-induced relaxations
Addition of D600 to preparations precontracred with KCl (15 mmol/L) produced a concentration-dependent relaxation, reaching a maximum at l0−5 mol/L D600; which is 91%±2% of the papaverine plus nitroprusside-induced relaxation (n=7). Prior treatment with D600 (10−5 mol/L) for 120 min attenuated relaxant responses to ACh to a smaller extent than in normal [Ca2+]o (Figure 8). In contrast, nitroprusside-induced relaxations were not affected by prior D600.
Effect of D600 on relaxant responses to ACh and nitroprusside. Preparations with or without prior D600 (10−5 mol/L) were precontracted with PGF2α prior to relaxants; contractions were 1771±218 mg (control, left), 1191±291 mg (D600, left), 1583±219 mg (control, right), and 680±237 mg (D600, right). The papaverine plus nitroprusside-induced relaxation was taken as 100%. Relaxations were 1999±238 mg, 1409±321 mg, 1939±286 mg, and 1016±232 mg, respectively. bP<0.05, cP<0.01 vs control.
Discussion
The current study clearly illustrates in a quantitative manner, the concentration-dependent effects of lowering [Ca2+]o on ACh-induced relaxations in rabbit aorta, emphasizing the importance of extracellular Ca2+ in ACh-mediated release of EDRF and/or its subsequent action on the smooth muscle. This is an agreement with previous studies that noted a marked reduction in, or elimination of endothelium-dependent relaxation in both rabbit14 and rat aorta11, 12, 13 when these tissues were exposed to solutions from which Ca2+ had been omitted. All of these supports the original proposal by Furchgott and co-workers7 that Ca2+ plays a critical role in endothelium-dependent relaxations. In addition, this study has demonstrated the obvious importance of Ca2+ also in endothelium-independent relaxations, since nitroprusside-induced relaxations are depressed by reducing [Ca2+]o .
Since the level of precontraction generated by PGF2α was depressed by reduced [Ca2+]o, the lowered state of contraction could be responsible for depression of subsequent relaxation. However, ACh-induced relaxations were not different in a normal Ca2+ solution when the magnitude of the maintained contraction was varied by utilizing different concentrations of PGF2α (Figure 3). Inhibition by reduced [Ca2+]o of ACh-induced relaxation was still observed, when the depressed precontraction was similarly increased. Thus, a lower magnitude of contraction is unlikely to account for the depressed relaxant responses to these vasodilators noted when [Ca2+]o is reduced. Furthermore, both ACh- and nitroprusside-induced relaxations in aortae precontracted with histamine instead of PGF2α were also attenuated by a reduction in [Ca2+]o, illustrating that attenuation of relaxant responses in reduced [Ca2+]o is not unique for PGF2α-induced contractions.
Since membrane Ca2+ controls its own entry into smooth muscle18, an increased conductance to Ca2+ following a reduction in [Ca2+]o may effectively increase cytoplasmic Ca2+ levels, resulting in functional antagonism of relaxant responses. Furthermore, membrane potential may be altered by reduced [Ca2+]o19, and Casteels et al20 have demonstrated in rabbit mesenteric artery that a reduction in [Ca2+]o from 2.5 mmol/L to 0.16 mmol/L results in a progressive decrease in resting membrane potential. In the present study, in fact, a small but appreciable contraction was observed following 2 h exposure to a reduced. [Ca2+]o solution that may be attributed to this proposed membrane depolarization and increased permeability to Ca2+.
Webb and Bohr21 suggested that high concentrations of Ca2+ (>4 mmol/L) stimulate the Na+-K+ ATPase, resulting in relaxation in rat tail artery. Thus in opposition to raising [Ca2+]o, lowering [Ca2+]o might depress the Na+–K+ ATPase. Conditions that inhibit the Na+–K+ ATPase in smooth muscle, eg K+-deficient solutions or cardiac glycosides, cause contraction through direct excitatory actions on the smooth muscle membrane22, 23, 24, and can physiologically antagonize both ACh-25 and nitroprusside-induced relaxations10. Nitroprusside stimulates guanylate cyclase and increases cGMP, which has been suggested to result in activation of the Na+–K+ ATPase10. However, neither nitroprusside-induced increases in the levels of cGMP nor concurrent effects on Na+–K+ ATPase appear to be dependent on [Ca2+]o10, 26. In addition, lowering [Ca2+]o also depresses relaxant response to isoproterenol (Authors, unpublished observations), which is apparently mediated through an increase in cellular cAMP level27. Therefore, the depressant actions of reduced [Ca2+]o are not unique for relaxants that concomitantly increase the levels of cGMP (ACh and nitroprusside). On the other hand, since both increases in cGMP28 and cAMP29 may stimulate Ca2+ ATPase-mediated extrusion mechanisms in vascular smooth muscle, an effect of reduced [Ca2+]o on this system cannot be ruled out.
It has been recently shown that dichlorobenzamil, a proposed inhibitor of Ca influx via the Na+-Ca2+ exchange system depressed endothelium-dependent relaxations but did not affect nitroprusside-induced relaxations 11. Thus, the Na+-Ca2+ exchange system would not be contributing to the general attenuation of these two relaxant responses that occurs in reduced [Ca2+]o solutions.
Since there was an initial absence of Mg2+ in control and reduced [Ca2+]o solutions, the Mg2+ deficiency could contribute to increased tone. It has been shown that acute Mg2+ deficiency can contribute to increased tone and reactivity to vasoconstrictors in coronary arteries30. However, the lack of Mg2+ in the present study is not likely to contribute to the actions of reduced [Ca2+]o, since the presence of Mg2+ neither modified the ACh-induced relaxation in a normal Ca2+ solution nor prevented the attenuating effects of reduced [Ca2+]o.
Sex related differences in cardiovascular function31 are raised particularly for variation of existing hormonal status in female, while no specific comparison was made in this study. Greater endothelial responses to ACh in small femoral arteries of female mice than male mice are attributable to endothelium-derived hyperpolarizing factor (EDHF) in female32. It is of interest whether endothelial susceptibility to low Ca is linked to EDHF in female.
ACh at a high concentration of 10−5 mol/L elicited a contraction after the preceded relaxant responses. A contractile effect of ACh without relaxation effect occurs in newborn baboon cerebral artery33. The aorta may have excitatory muscarinic receptors that are different from M1 and M2 receptor binding sites as suggested in porcine coronary arteries34. As Vanhoutte and his colleagues proposed, it is conceivable that endothelium-derived contracting factor (EDCF) is attributable to the ACh-induced contraction, via endothelial cyclooxigenase-1, which stimulates thromboxane A2 on vascular smooth muscle35. It remains to clarify whether change in the contracting factor is involved in the low Ca2+ effect on the relaxing factor.
Quantitative analyses clearly reveal that reduced [Ca2+]o resulted in greater inhibition of ACh-induced relaxation than nitroprusside-induced (Figure 6). Likewise nitroprusside-induced relaxations were unaffected by D600, whereas relaxations to ACh were attenuated. Thus, it is apparent that endothelium-dependent relaxation is more dependent on [Ca2+]o. The greater inhibition of ACh-induced relaxations than nitroprusside-induced ones is probably directly related to the dual sites apparently affected by reduced [Ca2+]o: (1) obvious requirement of the endothelium for Ca2+ in production and/or release of EDRF resulting in less EDRF for a given stimulus and less relaxation, and (2) an apparent requirement of vascular smooth muscle for a critical amount of extracellular Ca2+for full expression of the direct action of EDRF, as well as other vasodilators on smooth muscle. Thus, endothelium-dependent relaxation is more dependent on [Ca2+]o than on endothelium-independent relaxatlon, and it seems likely that [Ca2+]o plays an important role in vascular smooth muscle responsiveness not only to vasoconstrictors, but also to vascular relaxants as well.
Author contribution
Shigehiro HAYASHI designed research, performed research, wrote the paper; R Kelly HESTER designed research, contributed reagents/tools, wrote the paper.
References
Bohr DF . Vascular smooth muscle updated. Circ Res 1973; 32: 665–72.
Johansson B, Somlyo AP . Electrophysiology and Excitation-contraction coupling. In: Bohr DF, Somlyo AP, and Sparks HV Jr, editors. Handbook of physiology, Sect 2, Vol II, Vascular Smooth Muscle, Bethesda, Maryland, USA: Am Physiol Soc; 1980. p 301–323.
Hayashi S, Toda N . Inhibition by Cd2+, verapamil and papaverine of Ca2+-induced contraction in isolated cerebral and peripheral arteries of the dog. Br J Pharmacol 1977; 60: 35–43.
Hester RK, Weiss GB, Fry WJ . Effects of norepinephrine, dopamine and potassium on tension and 45Ca fluxes in canine and rabbit renal arteries. J Pharmacol Exp Ther 1978; 207: 364–71.
Hester RK, Weiss GB . Comparison of the degree of dependence of canine renal arteries and veins on high and low affinity calcium for responses to norepinephrine and potassium. J Pharmacol Exp Ther 1981; 216: 239–46.
Ratz PH, Flaim SF . Species and blood vessel specificity in the use of calcium for contraction. In: Flaim SF and Zelis R, editors. Calcium Blockers. Baltimore, USA: Urban and Schwarzenberg; 1982. p 77–98.
Furchgott RF . Role of endothelium in responses of vascular smooth muscle. Circ Res 1983; 53: 557–73.
Ignarro LJ, Buga GM, Wood KS, Byrns RE, Chaudhuri G . Endothelium-derived relaxing factor produced and released from artery and vein is nitric oxide. Proc Natl Acad Sci USA 1987; 84: 9265–9.
Palmer RM, Ferrige AG, Moncada S . Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature 1987; 327: 524–6.
Rapoport RM, Schwartz K, Murad F . Effect of sodium-potassium pump inhibitors and membrane-depolarizing agents on sodium nitroprusside-induced relaxation and cyclic guanosine monophosphate accumulation in rat aorta. Circ Res 1985; 57: 164–70.
Winquist RJ, Bunting PB, Schofield TL . Blockade of endothelium-dependent relaxation by the amiloride analog dichlorobenzamil: Possible role of Na+/Ca2+ exchange in the release of endothelium-derived relaxant factor. J Pharmacol Exp Ther 1985; 235: 644–50.
Miller RC, Schoeffter P, Stoclet JC . Insensitivity of calcium-dependent endothelial stimulation in rat isolated aorta to the calcium entry blocker, flunarizine. Br J Pharmacol 1985; 85: 481–7.
Long CJ, Stone TW . The release of endothelium-derived relaxant factor is calcium dependent. Blood Vessels 1985; 22: 205–8.
Singer HA, Peach MJ . Calcium- and endothelial-mediated vascular smooth muscle relaxation in rabbit aorta. Hypertension 1982; 4: 19–25.
Carrier O Jr, Wedell EK, Barron KW . Specific α-adrenergic receptor desensitization in vascular smooth muscle. Blood Vessels 1978; 15: 247–58.
Hayashi S, Park MK, Kuehl TJ . Relaxant and contractile responses to prostaglandins in premature, newborn and adult baboon cerebral arteries. J Pharmacol Exp Ther 1985; 233: 628–35.
Hester RK . The effect of 2-nicotinamidoethyl nitrate on agonist-sensitive Ca2+ release and Ca2+ entry in rabbit aorta. J Pharmacol Exp Ther 1985; 233; 100–11.
Somlyo AP, Somlyo AV . Vascular smooth muscle. I. Normal structure, pathology, biochemistry, and biophysics. Pharmacol Rev 1968; 20: 197–272.
Shanes AM . Electrochemical aspects of physiological and pharmacological action in excitable cells. I. The resting cell and its alteration by extrinsic factors. Pharmacol Rev 1958; 10: 59–164.
Casteels R, Kitamura K, Kuriyama H, Suzuki H . The membrane properties of the smooth muscle cells of the rabbit main pulmonary artery. J Physiol 1977; 271: 41–61.
Webb RC, Bohr DF . Mechanism of membrane stabilization by calcium in vascular smooth muscle. Am J Physiol 1978; 235: C227–32.
Belardinelli L, Harder D, Sperelakis N, Rubio RM . Berne RM . Cardiac glycoside stimulation of inward Ca2+ current in vascular smooth muscle of canine coronary artery. J Pharmacol Exp Ther 1979; 209: 62–6.
Haddy FJ . Potassium effects on contraction in arterial smooth muscle mediated by Na+, K+-ATPase. Fed Proc 1983; 42: 239–45.
Hayashi S, Park MK . Neurogenic and myogenic contractile responses of dog mesenteric arteries to reduced K+ concentration and their interaction with ouabain. J Pharmacol Exp Ther 1984; 230: 527–33.
De Mey JG, Vanhoutte PM . Interaction between Na+, K+ exchanges and the direct inhibitory effect of acetylcholine on canine femoral arteries. Circ Res 1980; 46: 826–36.
Schultz KD, Schultz K, Schultz G . Sodium nitroprusside and other smooth muscle relaxants increase cyclic GMP levels in rat ductus deferens. Nature 1977; 265: 750–1.
Andersson R, Lundholm L, Mohme-Lundholm E, Nilsson K . Role of cyclic AMP and Ca2+ in metabolic and mechanical events in smooth muscle. Adv Cyclic Nucleotide Res 1972; 1: 213–29.
Popescu LM, Panoiu C, Hinescu M, Nutu O . The mechanism of cGMP-induced relaxation in vascular smooth muscle. Eur J Pharmacol 1985; 107: 393–4.
Bulbring E, den Hertog A . The action of isoprenaline on the smooth muscle of the guinea-pig taenia coli. J Physiol 1980; 304: 277–96.
Turlapaty, PDMV, Altura BM . Magnesium deficiency produces spasms of coronary arteries: Relationship to etiology of sudden death ischemic heart disease. Science 1980; 208: 198–200.
Kauser K, Rubanyi GM . Gender difference in endothelial dysfunction in the aorta of spontaneously hypertensive rats. Hypertension 1995; 25 (4 Pt 1): 517–23.
Luksha L, Poston L, Gustafsson JA, Hultenby K, Kublickiene K . The oestrogen receptor beta contributes to sex related differences in endothelial function of murine small arteries via EDHF. J Physiol 2006; 577(Pt-3): 945–5.
Hayashi S, Park MK and Kuehl TJ . Higher sensitivity of cerebral arteries isolated from premature and newborn baboons to adrenergic and cholinergic stimulation. Life Sci 1984; 35: 253–60.
Rinner I, Doods HN, van Charldorp KJ, Davidesko D, van Zwieten PA . Binding of muscarine receptor antagonists to pig coronary smooth muscle. Naunyn-Schmiedeberg's Arch Pharmacol 1988; 337: 146–50.
Gluais P, Lonchampt M, Morrow JD, Vanhoutte PM, Feletou M . Acetylcholine-induced endothelium-dependent contractions in the SHR aorta: the Janus face of prostacyclin. Br J Pharmacol 2005; 146: 834–45.
Acknowledgements
This work was supported by NHLBI grant NHL 26L2I. Methoxyverapamil (D600) was generously supplied by Knoll AG, Ludwigshafen, West Germany. The authors would like to thank Mrs Beth BECKER for her technical assistance and Mrs Patty SAMPSON for her kind assistance in preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hayashi, S., Hester, R. Reduction in extracellular Ca2+ attenuates endothelium-dependent relaxation more than nitroprusside-induced relaxation. Acta Pharmacol Sin 31, 19–26 (2010). https://doi.org/10.1038/aps.2009.164
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2009.164