ABSTRACT
A monoclonal antibody, LC-1, recognizing lung cancer associated common antigens was obtained in authors laboratory. Its single chain Fv fragment (ScFv) named LC-1 ScFv was constructed based on recombinant phage displayed techniques. For expression on cell membrane, LC-1 ScFv was cloned into pDisplay vector, which directed the cloned gene to express as cell membrane bound protein. The resulting plasmid was sequenced and then introduced by the lipofectin method into a lung adenocarcinoma cell line SPC-A-1. G418 resistant cells were obtained by G418 selection. After transfection, LC-1 ScFv expression was observed by Western blot analysis and the expression of cognate antigens was down-regulated as shown in ELISA assay. SPC-A-1-pDisplay-ScFv cells grew in vitro at lower speed than the control intact cells and the cells transfected with vacant vector. Flow cytometry analysis detected a substantial increase in G1 phase and decrease in S phase in population of SPC-A-1-pDisplay-ScFv cells compared to SPC-A-1 and SPC-A-1-pDisplay cells. Semi-quantitative RT-PCR analysis showed that c-myc expression was down-regulated in SPC-A-1-pDisplay-ScFv cells. It seems that the antigens recognized by LC-1 may be in some way involved in a growth stimulating pathway and the antibody blocking of the function of the antigens shut down the pathway and thus down-regulate the expression of c-myc and growth of the cells.
Similar content being viewed by others
INTRODUCTION
Lung cancer remains the leading cause of cancer mortality in the world, accounting for more than one sixth of cancer deaths in the world1. Antibodies have been proved to be a powerful tool for the study of lung cancer. A monoclonal IgM antibody, LC-1, was obtained in our laboratory. It can react at a high rate with all four pathological types of lung cancers, including lung adenocarcinoma, lung squamous carcinoma, large cell lung cancer and small cell lung cancer, but not with normal and embryonic tissues2. In Western blot analysis, LC-1 detected three protein bands on cell surface of lung adenocarcinoma cell line, SPC-A-13. ABC staining revealed plenty of targeted antigens (LC-1Ags) on most lung cancer cells while few on normal tissues2. Further results indicated that LC-1 recognized the glyco-moiety of LC-1Ags(unpublished data). Experiments showed that when LC-1 bound SPC-A-1 cells, the antigen-antibody complexes were internalized via the receptor-mediated endocytic pathway, concentrated in the multivesicular bodies and finally transported to lysosomes for final proteolysis4. However, little is known about the nature of LC-1Ags except that they are cell surface glyco-proteins. Moreover, how they function in the lung cancer remains to be solved. All of these indicated that LC-1 may be a useful tool for the study of lung cancer.
Intracellular immunization, which was first introduced in 19885, is defined as the inhibition or inactivation of the function of a molecule by the ectopic intracellular expression of antisense RNA, ribozyme, dominant negative mutants and others alike which recognize the molecule. ScFvs were applied to intracellular immunization shortly after the first report of ScFv. Persic L. et al reported the creation of a set of vectors which permitted the localization of ScFvs to different intracellular compartments: the endoplasmic reticulum (scFvE-er), the nucleus (scFvE-nuclear), the mitochondria (scFvE-mit), the cytoplasm (scFvE-cyto), and as secreted proteins (scFvE-sec)6. Biocca S. and his colleagues reported the inactivation of p21ras in Xenopus laevis oocytes based on the intracellular expression of anti-p21ras ScFv genes. They found that cytosolic anti-p21ras ScFv not only markedly inhibited the H1 kinase activity induced by insulin but also blocked the ensuing meiotic maturation7.
Here we report the construction of ScFv fragment of LC-1(named LC-1 ScFv, Genbank accession number AF348616), blocking of the function of LC-1Ags with intracellularly expressed LC-1 ScFv, the ensuing inhibition cell growth of lung cancer cells and its possible mechanism.
MATERIALS AND METHODS
Cloning of V H and V L genes of LC-1
LC-1 Hybridoma was raised and kept in our lab. Total RNA was extracted from 107 cells with GIBCO-BRL TRIZOL Reagent (GIBCO Cat No. 15596, Rockville, USA) as directed by the manufacturers. cDNA was synthesized using GIBCO-BRL M-MLV (GIBCO Cat. No. 28025-013): 10 μml of 5× first strand buffer, DTT 0.01 M, 1 mM of each dNTPs, 5 μM of random hexamer primers and RNA 20 mg/L in a total volume of 48 μl. The mixture was denatured at 70 °C for 10 min then chilled on ice for 2 min. After adding 400 U of reverse Transcriptase M-MLV, the mixture was incubated in 37 °C water bath for 1 h and then inactivated at 95 ° C for 5 min. Primers specific for framework 1 and joining regions were used8, vhfor: TGA GGA GAC GGT GAC CGT GGT CCC TTG GCC CAG (3′ of VH) and vhbak: AG GTC CAA CTG CAG GAG TCA GG (5′ of VH), vlbak: GAC ATT GAG CTC ACC CAG TCT CCA (5′ of VL gene), vlfor: G TTT GAT CTC GAG CTT GGT CCC (3′ of VL gene). PCR was carried out as described8. Amplified fragments were cloned into pBluescript, and sequenced [ABI100 PRISMTM sequencer (PE co.)].
Construction of LC-1 ScFv
Linker primer: C TGG GGC CAA GGG ACC ACG GTC ACC GTC TCC TCA GGT GGA GGC GGT TCA GGC GGA GGT GGC TCT GGC GGT GGC GGA TCG GAC ATT GAG CTC ACC CAG TCT C9 and primer vlfor were used to modify and amplify VL gene. 200 ng of VH and modified VL gene fragments were each used to do SOE ligation. The resultant ScFv gene, named LC-1 ScFv, was amplified with vhbak and vlfor. Amplified LC-1 ScFv was cloned into pBluescript and sequenced.
Affinity maturation of LC-1 ScFv
Experiments were conducted according to the protocol given in users' manual (Cat No. 27-9401-01 Amersham Pharmacia Biotech. Co.). Briefly: LC-1 ScFv was modified and amplified with vhbaksfi: C ATG CCA TGA CTC GCG GCC CAG CCG CCG GCC ATG GCC CAG GTC CAA CTG CAG GAG TCA GG and vlfornot: GAG TCA TTC TGC GGC CGC CCG TTT GAT CTC GAG CTT GGT CCC. The modified and amplified LC-1 ScFv was digested with Sfi I (Cat No. R6391, Promega Co.) and Not I (Cat No. R6431, Promega co.), and subcloned into phagemid pCantab5E (Cat No. 27-9401-01, Amersham Pharmacia Biotech Co.). The resultant phagemid was transformed into F' E. coli TG1, rescued by KO7 helper phages. Recombinant phages were collected by PEG/NaCl (20% PEG, 2.5 M NaCl). 107 SPC-A-1 cells were coated onto a plate, blocked with 3% NCS (new born calf serum)/PBS and 1012 recombinant phages were applied to the plate for panning, and washed 20 times with PBS and another 20 times with 1% NCS/0.1% Tween 20/PBS. TG1 of OD600 0.3 was added to rescue the bound phages and reinfected for 2 h at 37 °C, the rescued phagemids were further subjected to another round of panning and three clones were sequenced.
Introduction of plasmid into SPC-A-1 cell line
Recombinant plasmid was introduced into lung adenocarcinoma cell line SPC-A-1 by lipofectin method as reported10 with some modification. Briefly: Mature LC-1 ScFv gene was amplified and modified with vlforsacII (5′-ATG CAT CCG CGG CCG TTT GAT CTC CAA CTT GGT CCC-3′) and vhbaksfi. The resultant LC-1 ScFv was digested with SfiI and SacII and cloned into identically digested pDisplay (Cat N0. V660-20, Invitrogen Co)(shown in Fig 1). The resulted recombinant plasmid was named pDisplay-ScFv. 15 μg pDisplay-ScFv was dissolved into 400 μl serum-free RPMI1640 medium and 15 μl lipofectin (Cat No. 18292-011, Gibco Co.) was dissolved into 400 μl serum-free RPMI1640 medium. These two components were mixed together and incubated at 37°C for 1 h. The mixture was used to incubate 5× 105 overnight pre-incubated SPC-A-1 cells for 6 h. The treated cells were incubated in RPMI1640/10%NCS for 24 h and then kept in 10% NCS/G418/RPMI1640 for three weeks. Cell colonies were seen. Vacant pDisplay vector and sterile water were also used to transform the cell as controls. SPC-A-1 transfected with vacant pDisplay vector was labeled as SPC-A-1-pDisplay, SPC-A-1 transfected with pDisplay-ScFv as SPC-A-1-pDisplay-ScFv.
Representative map of pDisplay plasmid Vacant pDisplay plasmid is digested with SfiI and SacII, and identically digested LC-1 ScFv fragment was ligated into it. SPC-A-1 was transformed with pDisplay, and pDisplay-ScFv. SPC-A-1-pDisplay-ScFv is the experimental series while SPCA-1 and SPC-A-1-pDisplay serve as control series. pDisplay vector ensures that the cloned gene is expressed at high level as membrane-bound protein.
ELISA assay of the expression of lung cancer associated antigens
5× 105 cells/well were coated onto each well of 96-well plate. The plate was blocked with 6% NCS/PBS for 1 h at 37° C, washed with 0.1% Tween-20/1% NCS/PBS three times. 0.03 μg/ml LC-1 antibody was used to bind cells for an h at 37 °C Horseradish peroxidase labeled anti-IgM second antibody (Cat No. A8786 m chain specific, Sigma Co.) was applied to bind LC-1 antibody. The plate was developed in OPD (O-phenylenediamine) substrate (50 mM Na2HPO4 0.04% OPD, 0.03% H2O2, 24 mM citrate). A490 was read by automatic reader.
ECL western blot analysis of the expression of c-myc tagged LC-1 ScFv
To assess the expression of LC-1 ScFv in the transfected SPC-A-1, ECL Western blot analysis was conducted as reported11 with some modification. Briefly: 5×106 cells were lysed in 500ml loading buffer (50 mM Tris-HCl pH 6.8, 100 mM DTT, 2% SDS, 10% glycerol). 20 ml aliquot was subjected to 12% SDS-PAGE eletrophoresis. The separated proteins were transferred to nitrocellulose membrane in transfer buffer (20 mM Tris-HCl, 190 mM Glycine, 20% methanol, pH 8.3) using Mini Trans-blot transfer system (Bio-Rad) at 100V for 1 h. The membrane was blocked with 5% nonfat dried milk in TBST (Tris-buffered saline containing 0.05% Tween-20) and then incubated with mouse anti-human C-MYC first antibody (SC002051, BBST Co). After three washes in TBST, the membrane was incubated with peroxidase-conjugated goat anti-mouse second antibody (SC002050, BBST Co). The blot was incubated with ECL substrate (RPN2109, Amersham Pharmacia Biotech) to reveal the antigen bands and the result was developed on x-ray film. 28-KD C-MYC tagged GUK domain of CASK protein (Genbank accession number AF035582) was used as positive control molecular marker.
Determination of cell growth rate
5×105 SPC-A-1-pDisplay-ScFv cells were seeded in each flask (5 flasks a series). The number of cells was counted in hematocytometer per 24 h. SPC-A-1 cells and SPC-A-1-pDisplay cells were used as control cells. The curve of cell growth rate of different series was drawn.
Flow cytometry analysis of cell cycle
Propidium iodide (PI) (Sigma, St. Louis, MO) stained cells were prepared and analyzed by flow cytometry (conducted by FACS laboratory of Shanghai Institute of Cell Biology, CAS). Briefly: 5×105 cells were seeded in each flask and kept at 37 ° C and 5% CO2 for two days. Cells were then collected, fixed in citrate buffer at room temperature for 1 h, centrifuged at 1000 g and resuspended in PBS at a final concentration of 6×106 cells/ml. Cells were stained using 1% PI and 250 U RNase cocktail (RNase A, RNase T1, Ambion Corp., Austin, TX) and incubated in the dark at room temperature for 30 min. Nuclear DNA content was measured based on PI fluorescence using a 488 nm argon laser and a Becton-Dickinson FACS Calibur flow cytometer (Becton-Dickinson Inc., San Jose, CA). Fluorescence data were collected in linear mode (8000 events) and the results were quantified using CELLQuest v1.01 (Becton-Dickinson Inc.). The percentages of G1 (1 N), S (1-2 N) and G2/M (2 N) cells were calculated based on defined gates for each population.
Semi-quantitative RT-PCR analysis of c-myc oncogene
Total RNA was extracted using GIBCO-BRL TRIZOL Reagent (Cat No. 15596 Gibco Life Tech) as directed by manufacturers. cDNA was synthesized using GIBCO-BRL M-MLV (Cat No. 28025-013, Gibco Life Tech). Semi-quantitative PCR was based on Chelly's protocol12 with some modifications. PCR amplification was performed on CEL-BIO Temperature Cycling System. PCR system: 12μl of 10× reaction buffer, 0.25 mM of each dNTPs, 0.75 pmol of each sense and antisense primers (C-myc sense (5′-GTT TTC CAA CTC CGG GAT CTG-3′) and c-myc antisense (5′-AAG ACT CCA GCG CCT TCT CTC-3′)(synthesized by Sangon Co.) for amplification of c-myc oncogene fragment and Gapdhfor and gapdhbak (kindly provided by Dr Changling LI, Institute of Cell Biology, CAS) for amplification of GAPDH fragment), and 6 ml of cDNA. Adjust total volume to 117 μl with double-distilled H2O. The mixture was denatured at 95 ° C for 5 min, then 3 μl of Taq DNA polymerase (3 U per m l, Cat No. MM0962, Sino-American Biotechnology Company, China) was added as a “hot start” method13 to eliminate non-specific annealing. GAPDH was used as an internal constant standard. PCR was performed in the following condition: denaturation for 45s at 94 °C annealing for 45s at 59 ° C, and extension for 45s at 72 ° C Initially, c-myc gene was amplified for 7-13 cycles and then the primers for internal standard, GAPDH, were added by the “primer-dropping”method14. After another 14-17 cycles, 17 μl of template of PCR reaction was collected from the following different cycles and templates were then electrophoresed through 1 % agarose gels, and stained with ethidium bromide and photographed. The density of target bands was measured by FURI Smartview-2000 (Shanghai FURI Science and Technology Co, Ltd,). The optimal numbers of PCR cycles were determined.
RESULTS
Sequences of VH and VL and affinity maturation of LC-1 ScFv
In electrophoresis, the isolated total RNA sample gave out three sharp bands, demonstrating the integrity of RNA extracted from hybridoma LC-1 (data not shown). PCR products of VH gene (Genbank accession number AF348614) and VL gene Fig 2 (Genbank accession number AF348615) can be amplified from reverse transcripted cDNAs. Electrophoresis showed that they were of expected molecular size. Sequences were aligned on NCBI BLAST (http://www.ncbi.nlm.nih.gov/blast/blast.cgi). Results showed that cloned VH and VL gene matched antibody sequence well. Sequence of LC-1 ScFv (Genbank accession number AF348616) showed that VH and VL gene were successfully spliced.
Electrophoresis of clone VL, VH and ScFv Lane 1 100bp marker; Lane 2 VL gene fragment; Lane 3 VH gene fragment; Lane 4 ScFv gene fragment. Results showed that VH and VL, and LC-1 ScFv gene were of expected molecular size. Sequences of these genes were registered in Genbank (accession number: 348614, 348615, 348616).
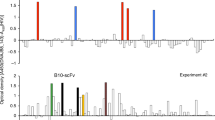
After affinity maturation using phage-display technique, three clones screened out were sequenced, and they gave out same sequences. No mutation was detected. (as shown in Fig 3)
Introduction of plasmid into SPC-A-1
Using lipofectin, foreign plasmid could be easily introduced into SPC-A-1 cell. Hundreds of clones were formed per 105 cells. SPC-A-1 cells treated with sterile water instead of plasmid in transformation died out under the selecting pressure of G418 after 4 changes of media, while hundreds of colonies were formed in SPC-A-1 treated with pDisplay and pDisplay-scfv.
Expression of lung cancer associated surface antigens on different cells detected by ELISA
SPC-A-1 cell and SPC-A-1 cells transfected with vacant pDisplay vector (SPC-A-1-pDisplay) expressed high level of LC-1Ags, while cells transformed with pDisplay-ScFv expressed few LC-1Ags detected by ELISA. (as shown in Fig 4)
LC-1 ScFv expression in transformed cells by ECL Western blot assay
To confirm that the lowered level of the expression of LC-1Ags was caused by the expression of LC-1 ScFv, we assayed the LC-1 ScFv expression by Western blot with anti-c-myc antibody, for pDisplay vector could endow a c-myc tag on the c-terminal of inframe protein as indicated in the representative map of the plasmid. Western blot results (Fig 5) showed that SPC-A-1-pDisplay-scfv gave a strong signal of c-myc tag but SPC-A-1 cells and SPC-A-1-pDisplay cells lacked of the 30 KD band in ECL Western blot assay.
Comparison of cell growth rate
An interesting phenomenon was that among these different kinds of cells, the SPC-A-1-pDisplay-ScFv cells grew much slower than SPC-A-1 cells or SPC-A-1-pDisplay cells (Fig 6). It showed that when SPC-A-1 or SPC-A-1-pDisplay cells reached over-confluence, the cell growth of SPC-A-1-pDisplay-ScFv only reached 40-50%.
Flow cytometry analysis of cell cycle
From Tab 1, Results revealed a substantial increase of cell percentage in G1 phase and decrease in S phase in population of SPC-A-1-pDisplay-ScFv cells compared to SPC-A-1 and SPC-A-1-pDisplay cells. This indicated that the proliferating SPC-A-1-pDisplay-ScFv cells were fewer than that of SPC-A-1 and SPC-A-1-pDisplay cells.
Analysis of c-myc oncogene expressed in these three different kinds of cells by semi-quantitative PCR
In Fig 7 it was indicated by semi-quantitative PCR analysis that c-myc expression was lower in SPC-A-1-pDisplay-scfv than that in SPC-A-1 or SPC-A-1-pDisplay cells. The results showed that c-myc gene expression was down-regulated in SPC-A-1-pDisplay-scfv cell. It means that the lower growth rate of SPC-A-1-pDisplay-ScFv may be correlated with down-regulation of c-myc gene in these cells.
Semi-quantitative PCR sample of different cell series Lane 1, RT-PCR sample of SPC-A-1-pDisplay-ScFv, c-myc fragment below the GAPDH fragment; Lane 2, RT-PCR sample of SPC-A-1-pDisplay, c-myc fragment below GAPDH fragment; Lane 3, RT-PCR sample of SPC-A-1, c-myc fragment below GAPDH fragment; Lane 4, 100 bp ladder molecule marker. Results showed expression of c-myc was down-regulated in SPC-A-1-pDisplay-ScFv cells while relatively high in SPC-A-1 cell and in SPC-A-1-pDisplay cells.
DISUCSSION
LC-1Ags are shared by different lung cancers, as reported earlier by our Lab2. In SPC-A-1 cells, the antigens appear to be 51, 70, or 91 KD3. Data show there are 7.2×104 antigen sites per SPC-A-1 cell 15.
Functionally blocking a specific protein has been succeeded with antagonists16, antisense oligo nucleotide17, and antibody18. Since Biocca et. al. introduced the idea of intracellular immunization with ScFv, many cellular proteins have been functionally blocked with ScFv7.
VH and VL gene of LC-1 have been cloned and their sequences were confirmed by twice fluorescent sequencing. LC-1 ScFv was constructed. After affinity maturation, no mutations occurred in sequence of VH and VL gene. It could be due to high binding activity of LC-1 antibody. It can effectively compete with other phage-display antibodies for binding to the immobilized SPC-A-1 cells and thus all the picked clones were identical with original sequence. We cloned the LC-1 ScFv gene into pDisplay to functionally block the cognate surface glycoproteins. The pDisplay vector features three elements: human cytomegalovirus (CMV) immediate-early promoter/enhancer which permits efficient, high level expression of the recombinant protein, the murine Ig k-chain leader sequence at the N-terminus and platelet derived growth factor receptor transmembrane domain (PDGFR-TM) at the C-terminus, which anchors the fusion protein to the plasma membrane and myc epitope which allows detection of the fusion protein with anti-myc antibody. In western blot analysis, high expression of LC-1 ScFv was detected in SPC-A-1-pDisplay-ScFv cells. In ELISA assay, no remarkable difference in LC-1Ags was detected between A490 of SPC-A-1-pDisplay-ScFv and that of PBS control, while A490 of SPC-A-1 and SPC-A-1-pDisplay cells was remarkably high, indicating that numerous epitopes were bound with LC-1 ScFvs.
Most probably, mechanism implicated in the low detection of LC-1Ags is that the immunocomplexes of LC-1 ScFv and LC-1Ags were internalized via the receptor-mediated endocytic pathway, concentrated in the multivesicular bodies and transported to lysosomes for final proteolysis, similar to the report by Xie W. et al4. But we are also aware of another possibility that the immunocomplexes remained on the cell membrane. But LC-1Ags were blocked by LC-1 ScFv, similar to the report by Biocca S. et al7.
In our experiments, SPC-A-1-pDisplay-ScFv cells were found to grow slower than SPC-A-1 and SPC-A-1-pDisplay cells. Result of flow cytometry analysis elucidated that there were fewer proliferating SPC-A-1-pDisplay-ScFv cells than that of SPC-A-1 and SPC-A-1-pDisplay cells. In our study, expression of c-myc was found to be down-regulated in SPC-A-1-pDisplay-ScFv cells, while remained high in SPC-A-1 and SPC-A-1-pDisplay cells as quantified by semi-quantitative RT-PCR analysis.
How is the blocking of LC-1Ags by LC-1 ScFv connected with the slowing-down of cell growth and c-myc down-regulation? Studies have shown that many growth factors or regulatory peptides and their receptors are expressed by cancer or adjacent normal cells in the lung, thus providing a series of autocrine or paracrine growth stimulatory loops19. Further studies revealed that formation of soft agar clones in vitro and the in vivo growth of xenografts of lung cancer cells in nude mouse were inhibited by neutralizing monoclonal antibody that blocked these stimulatory loops20, 21. Three best characterized autocrine systems in the lung cancer involve GRP/BN and their receptors20, Neu differentiation factors (NDF) and their ERBB family receptors22, and EGF/TGFa and their receptors23. The fact that lung adenocarcinoma cell line SPC-A-1 grows more slowly when intracellularly expressed LC-1 ScFvs block surface expression of LC-1Ags and that soft agar colony formation of SPC-A-1-pDisplay-ScFv is inhibited (data not published) are highly similar to the inhibition of soft agar colony formation by antibody disturbance as reported20, 21. This suggests that LC-1Ags of SPC-A-1 are possibly involved in an autocrine growth stimulatory loop which stimulates the growth of the cells. When LC-1Ags are blocked by LC-1 ScFv, thus leading to the shutdown of this autocrine loop, cells grow at lower speed. Most growth factor receptors are in some way involved in the signal transduction process, such as GRP/BN receptors belong to the G- protein coupled receptor superfamily24.
Myc proteins are transcription factors of helix-loop-helix/leucine zipper family that activate transcription as obligate heterodimers with a partner, protein Max25. A list of candidate genes activated at transcriptional level by myc was compiled, which contained a number of genes required for cell proliferation26. Myc activity is controlled at multiple levels by signal transduction cascades: one such level is c-myc transcription that is strongly induced by growth factors. A second level of control is through proteins that interact with the N-terminal of myc27. c-myc, among the so called class II cancer genes, is not altered at DNA level, but affects the phenotype of the host cells by changes in expression28. c-myc oncogene, a key oncoprotein in regulation of cell proliferation and apoptosis29, was reported to be up-regulated in lung cancer cells30. MYC protein promotes normal cell growth and proliferation through direct activation of genes involved in DNA synthesis, RNA metabolism and cell-cycle progression24.
We hypothesize that in SPC-A-1, LC-1Ags seem to pick up extracellular signals and then up- regulate the c-myc expression through their signal transduction pathway and thus promote cell growth. When the antigens are blocked, the signal transduction pathway is shut down, leading to downregulation of the c-myc expression and ensuing slowing down of cell growth.
Our experiments, together with our previous studies31, 32 showed LC-1Ags on SPC-A-1 cells could be a good target for lung cancer diagnosis and immunotherapy.
References
Hoffman PC, Mauer AM, Vokes EE . Lung cancer. Lancet 2000; 355(9202):479–85.
Ge XR, Wang J, Lin SJ, Yu NX, Che YF, Gu Y . McAb LC-1 against human lung cancer. Acta Biologiae Experimentalis Sinica 1989; 22(3):359–65.
Lin SJ, Ge XR, Wang J, Gu Y . Studies on antigens of human lung adenocarcinoma with McAb LC-1. Acta Biologiae Experimentalis Sinica 1990; 23(2):233–8.
Xie W, Zhang RX, Ge XR . Internalization of tumor associated antigen on human lung adenocarcinoma cell line SPC-A-1 by McAb LC-1. Acta Biologiae Experimentalis Sinica, 1996; 29(3):207–19.
Staeheli P . Intracellular immunization: A new strategy for producing disease-resistant transgenic livestock? Trends Biotechnol 1991; 9(3):71–2.
Persic L, Righi M, Roberts A, Hoogenboom HR, Cattaneo A, Bradbury A . Targeting vectors for intracellular immunisation. Gene 1997; 187(1):1–8.
Biocca S, Pierandrei-Amaldi P, Campioni N, Cattaneo A . Intracellular immunization with cytosolic recombinant antibodies. Biotechnology (N Y) 1994; 12(4):396–9.
Orlandi R, Gussow DH, Jones PT, Winter G . Cloning immunoglobulin variable domains for expression by the polymerase chain reaction. Proc Natl Acad Sci 1989; 86(10):3833–7.
Peter K, Graeber J, Kipriyanov S et al. Construction and functional evaluation of a single-chain antibody fusion protein with fibrin targeting and thrombin inhibition after activation by factor Xa. Circulation 2000; 101(10):1158–64.
Qin WX, DF Wan, FY SUN et al. Cloning and characterization of a novel gene (C17orf25) from the deletion region on chromosome 17p13. 3 in hepatocellular carcinoma. Cell Research 2001; 11(3):209–16.
Kong WH, Z Cu, JN Lu, JK Tso . Temperature dependent expression of cdc2 and cyclin B1 in spermatogenesis. Cell Research 2000; 10(4):289–302.
Chelly J, Kaplan JC, Maire P, Gautron S, Kahn A . Transcription of the dystrophin gene in human muscle and non muscle tissues. Nature 1988; 333:858–60.
Ailenberg M, Silverman M . Controlled hot start and improved specificity in carrying out PCR utilizing touch-up and loop incorporated primers (TULIPS). Biotechniques 2000; 29(5):1018–20, 1022–4.
Vehaskari VM, Hempe JM, Manning J, Aviles DH, Carmichael MC . Developmental regulation of ENaC subunit mRNA levels in rat kidney. Am J Physiol 1998; 274(6 Pt 1):C1661–6.
Li M, Lian BJ, Ge XR . Study on binding characteristics of 125I-labeled McAb LC-1 to lung adenocarcinoma cells in vitro and in vivo. Acta Biologiae Experimentalis Sinica 1992; 25(1):31–8.
Tramontana M, Lecci A, Meini S, Montserrat X, Pascual J, Giuliani S, Quartara L, Maggi CA . Differences between peptide and nonpeptide B(2) bradykinin receptor antagonists in blocking bronchoconstriction and hypotension induced by bradykinin in anesthetized Guinea pigs. J Pharmacol Exp Ther 2001; 296(3):1051–7.
Rajagopal S, Moskal TL, Wang H, Chakrabarty S . Efficacy and specificity of antisense laminin chain-specific expression vectors in blocking laminin induction by TGFbeta1: Effect of laminin blockade on TGFbeta1-mediated cellular responses. J Cell Physiol 1999; 178(3):296–303.
Yamada T, Okafuji T, Ohta K, Handwerker C, Drescher U, Tanaka H . Analysis of ephrin-A2 in the chick retinotectal projection using a function-blocking monoclonal antibody. J Neurobiol 2001; 47(4):245–54.
Viallet J, Sausville EA . Involvement of signal transduction pathways in lung cancer biology. J Cell Biochem Suppl 1996; 24:228–36.
Cuttitta C, Carney DN, Mulshine J, Moody TW, Fedorko J, Fischler A, Minna JD . Bombesin-like peptides can function as autocrine growth factors in human small-cell lung cancer. Nature 1985; 316(6031):823–6.
Halmos G, Schally AV . Reduction in receptor for bombesin and epidermal growth factor in Xenografts of human small cell lung cancer after treatment with bombesin antagonist RC-3095. PNAS 1997; 94(3):956–60.
Alroy I, Yarden Y . The ErbB signaling network in embryogenesis and oncogenesis: signal diversification through combinatorial ligand-receptor interactions. FEBS Lett 1997; 410(1):83–6.
Damstrup L, Rygaard K, Spang-Thomen M, Poulsen HS . Expression of the epidermal growth factor receptor in human small cell lung cancer cell lines. Cancer Res 1992; 52(11):3089–93.
Fathi Z, Way JW, Corjay MH, Viallet J, Sausville EA, Battey JF . Bombesin receptor structure and expression in human lung carcinama cell lines. J Cell Biochem (suppl) 1996; 24:237–46.
Henriksson M, Luscher B . Proteins of the Myc network: essential regulators of cell growth and differentiation. Adv Cancer Res 1996; 68:109–82.
Grandori C, Eisenman RN . Myc target genes. Trends Biochem Sci 1997; 22(5):177–81.
Bouchard C, Staller P, Eilers M . Control of cell proliferation by Myc. Trends Cell Biol 1998; 8(5):202–6.
Sager R . Expression genetics in cancer: Shifting the focus from DNA to RNA, PNAS 1997; 94:952–25.
Sekido Y, Fong KM, Minna JD . Progress in understanding the molecular pathogenesis of human lung cancer. Biochim Biophys Acta 1998; 1378(1):F21–59.
Pelengaris S, Rudopph B, Littlewood T . Action of Myc in vivo-proliferation and apoptosis. Current opinion in Genetics and Development 2000; 10:100–5.
Lin SJ, Wang J, Lin XK, Fan ZJ, Ge XR . Study on immunoradiological location of human lung cancers with McAb LC-1 IgM fragment. Shanghai J Immunol 1998; 18(4):201–3, 214.
Lin SJ, Zeng L, Zhong GR, Ge XR . Studies on treatment of nude mice bearing human lung cancer with 90Y-LC-1. J Shanghai Med Univer 1999; 26(3):206.
Acknowledgements
This work was supperted by Sciense and Technology Promoting Foundation of Shanghai No. 984419071.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
CHEN, L., LI, G., TANG, L. et al. The inhibition of lung cancer cell growth by intracellular immunization with LC-1 ScFv. Cell Res 12, 47–54 (2002). https://doi.org/10.1038/sj.cr.7290109
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/sj.cr.7290109