Summary
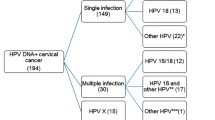
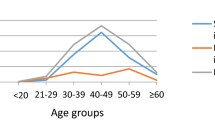
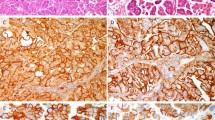
The p53 mutation has been found only in 0–6% of cervical carcinomas. In light of recent studies demonstrating that mutation of p53 gene was found in over 20% of the patients with vulvar carcinoma, a disease of elderly women and a known human papillomavirus (HPV)-related malignancy, we analysed mutation of the p53 gene in 46 women with cervical carcinomas at the age of 60 or more (mean; 71 years, range; 60–96 years). The presence of HPV and its type were analysed by polymerase chain reaction (PCR)-based assay using the consensus primers for L1 region. Mutation of the p53 gene was analysed by PCR-based single-strand conformation polymorphism and DNA sequencing technique. Point mutation of the p53 gene was detected in 5 out of 46 (11%) cervical carcinomas: 1 of 17 (6%) samples associated with high-risk HPVs (HPV 16 and HPV 18) and 4 of 27 samples (15%) with intermediate-risk HPVs (P = 0.36) whereas no mutation was found in 2 HPV negative cases. The mutated residues resided in the selective sequence known as a DNA-binding domain. The immunohistochemistry revealed the overexpression in cancer tissues positive for p53 mutation. All of the observed mutations of the p53 gene were transition type, suggesting that the mutation may be caused by endogenous mutagenesis. Although falling short of statistical significance reduces the strength of the conclusion, data presented here imply that p53 gene mutation, particularly along with intermediate-risk HPV types, may constitute one pathogenetic factor in cervical carcinoma affecting elderly women.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Cho, Y., Gorina, S., Jeffrey, P. D. & Pavletich, N. P. (1994). Crystal structure of a p53 tumor suppressor–DNA complex: understanding tumorigenic mutations. Science, 265: 346–355.
Choo, K. B. & Chong, K. Y. (1993). Absence of mutation in the p53 and retinoblastoma susceptibility genes in primary cervical carcinomas. Virology 193: 1042–1046.
Crook, T. & Vousden, K. H. (1992). Properties of p53 mutations detected in primary and secondary cervical cancers suggest mechanisms of metastasis and involvement of environmental carcinogens. EMBO J 11: 3935–3940.
Crook, T., Wrede, D. & Vousden, K. H. (1991). p53 point mutation in HPV-negative human cervical carcinoma cell lines. Oncogene, 6: 873–875.
Dittmer, D., Pati, S., Zambetti, G., Chu, S., Teresky, A. K., Moore, M., Finlay, C. & Levine, A. J. (1993). Gain of function mutation of p53. Nature Genet 4: 42–46.
Fujinaga, Y., Shimada, M., Okazawa, K., Fukushima, M., Kato, I. & Fujinaga, K. (1991). Simultaneous detection and typing of genital human papillomavirus DNA using the polymerase chain reaction. J Gen Virol 72: 1039–1044.
Fujita, M., Inoue, M., Tanizawa, O., Iwamoto, S. & Enomoto, T. (1992). Alterations of the p53 gene in human primary cervical carcinoma with and without human papillomavirus infection. Cancer Res 52: 5323–5328.
Harris, C. C. (1993). p53: At the crossroads of molecular carcinogenesis and risk assessment. Science, 262: 1980–1981.
Helland, A., Holm, R., Kristensen, G., Laern, J., Kaern, J., Karlsen, F., Trope, C., Nesland, J. M. & Borresen, A. L. (1993). Genetic alterations of the p53 gene, p53 protein expression and HPV infection in primary cervical carcinomas. J Path 171: 105–114.
Huibregtse, J. M., Scheffner, M. & Howley, P. M. (1993). Cloning and expression of the cDNA for E6-AP, a protein that mediates the interaction of the human papillomavirus E6 oncoprotein with p53. Mol Cell Biol 13: 775–784.
Kagie, M. J., Kenter, G. G., Tollenaar, RAEM, Hermans, J., Trimbos, J. B. & Fleuren, G. J. (1997). p53 protein overexpression is common and independent of human papillomavirus infection in squamous cell carcinoma of the vulva. Cancer 80: 1228–1233.
Kanda, T., Watanabe, S. & Yoshiike, K. (1988). Immortalization of primary rat cells by human papillomavirus type 16 subgenomic DNA fragment controlled by SV40 promoter. Virology 165: 321–325.
Kuerbitz, S. J., Plunkett, B. S., Walsh, W. V. & Kastan, M. B. (1992). Wild-type p53 is a cell cycle checkpoint determinant following irradiation. Proc Natl Acad Sci USA, 89: 7491–7495.
Lechner, M. S. & Laimins, L. A. (1994). Inhibition of p53 DNA binding by human papillomavirus E6 proteins. J Virol 68: 4262–4293.
Lee, Y. Y., Wilczynski, P., Chumakov, A., Chih, D. & Loeffler, H. P. (1994). Carcinoma of the vulva: HPV and p53 mutations. Oncogene 9: 1655–1659.
Lorincz, A. T., Reid, R., Jenson, A. B., Greenberg, M. D., Lancaster, W. & Kurman, R. J. (1992). Human papillomavirus infection of the cervix: relative risk associations of 15 common anogenital types. Obstet Gynecol, 79: 328–337.
Lowe, S. W., Schmitt, E. M., Smith, S. W., Osborne, B. A. & Jacks, T. (1993). p53 is required for radiation-induced apoptosis in mouse thymocytes. Nature 362: 847–849.
Milde-Langosch, K. M., Albrecht, K., Joram, S., Schlechte, H., Giessing, M. & Loning, T. (1995). Presence and persistence of HPV infection and p53 mutation in cancer of the cervix uteri and the vulva. Int J Cancer 63: 639–645.
Miwa, K., Miyamoto, S., Imamura, T., Nishida, M., Yoshikawa, Y., Nagata, Y. & Wake, N. (1995). The role of p53 inactivation in human cervical cell carcinoma development. Br J Cancer 71: 219–226.
Munger, K., Phelps, W. C., Bubb, V., Howley, P. M. & Schlegel, R. (1989a). The E6 and E7 genes of the human papillomavirus type 16 together are necessary and sufficient for transformation of primary human keratinocytes. J Virol, 63: 4417–4421.
Munger, K., Werness, B. A., Dyson, N., Phelps, W. C., Harlow, E. & Howley, P. M. (1989b). Complex formation of human papillomavirus E7 proteins with the retinoblastoma tumor suppressor gene product. EMBO J 8: 4099–4105.
Nagano, H., Yoshikawa, H., Kawana, T., Yokota, H., Taketani, Y., Igarashi, H., Yoshikura, H. & Iwamoto, A. (1996). Association of multiple human papillomavirus types with vulvar neoplasias. J Obstet Gynecol Res, 22: 1–8.
Naito, M., Satake, M., Sakai, E., Hirano, Y., Tsuchida, N., Kanzaki, H., Ito, Y. & Mori, T. (1992). Detection of p53 gene mutations in human ovarian and endometrial cancers by polymerase chain reaction-single strand conformation polymorphism analysis. Jpn J Cancer Res 83: 1030–1036.
Nakagawa, S., Watanabe, S., Yoshikawa, H., Taketani, Y., Yoshiike, K. & Kanda, T. (1995). Mutational analysis of human papillomavirus type 16 E6 protein: transforming function for human cells and degradation of p53 in vitro. Virology 212: 535–542.
Nakagawa, S., Yoshikawa, H., Onda, T., Kawana, T., Iwamoto, A. & Taketani, Y. (1996). Type of human papillomavirus is related to clinical features of cervical carcinoma. Cancer 78: 1935–1941.
Nevins, J. R. (1992). E2F: A link between the Rb tumor suppressor protein and viral oncoproteins. Science 258: 424–429.
Paquette, R. L., Lee, Y. Y., Wilczynski, S. P., Karmakar, A., Kizaki, M., Miller, C. W. & Koeffler, H. P. (1993). Mutation of p53 and human papillomavirus infection in cervical carcinoma. Cancer 72: 1272–1280.
Scheffner, M., Werness, B. A., Huibregtse, J. M., Levine, A. J. & Howley, P. M. (1990). The E6 oncoprotein encoded by human papillomavirus types 16 and 18 promotes the degradation of p53. Cell, 63: 1129–1136.
Snijders, P. J. F., Van den Brule, A. J. C., Schrijnemakers, H. F. J., Snow, G., Meijer, CJLM & Walboomers, J. M. M. (1990). The use of general primers in the polymerase chain reaction permits the detection of a broad spectrum of human papillomavirus genotypes. J Gen Virol 17: 173–181.
Storey, A., Pim, D., Murray, A., Osborn, K., Banks, L. & Crawford, L. (1988). Comparison of the in vitro transforming activities of human papillomavirus types. EMBO J, 7: 1815–1820.
van Ravenswaay Claasen, H. H., Kluin, P. M. & Fleuren, G. J. (1992). Tumor infiltrating cells in human cancer. On the possible role of CD16+ macrophages in anti-tumor cytotoxicity. Lab Invest, 67: 166–174.
Werness, B. A., Levine, A. J. & Howley, P. M. (1990). Association of human papillomavirus types 16 and 18 E6 proteins with p53. Science, 248: 76–79.
Yoshikawa, H., Kawana, T., Kitagawa, K., Mizuno, M., Yoshikura, H. & Iwamoto, A. (1991). Detection and typing of multiple genital human papillomaviruses by DNA amplification with consensus primers. Jpn J Cancer Res 82: 524–531.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Nakagawa, S., Yoshikawa, H., Jimbo, H. et al. Elderly Japanese women with cervical carcinoma show higher proportions of both intermediate-risk human papillomavirus types and p53 mutations. Br J Cancer 79, 1139–1144 (1999). https://doi.org/10.1038/sj.bjc.6690181
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690181