Abstract
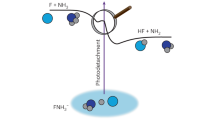
In neutral atoms and molecules, electrons are kept within their orbitals by attractive electrostatic interactions with positively charged nuclei, with relatively few neutral molecules being able to bind more than one extra electron. For multiply charged molecular anions, dynamic stability plays an important role: the superposition of long-range Coulomb repulsion and short-range electron binding gives rise to a repulsive Coulomb barrier (RCB) that traps the excess electrons1. The RCB has profound effects on the physical and chemical properties of multiply charged anions in the gas phase1,2,3,4,5. For example, it has recently been shown to prevent the detachment of electrons from a doubly charged anion, even when the excitation energies exceed the electron binding energy6,7. Here we report photodetachment experiments which demonstrate that the RCB can even trap electrons in molecular orbitals characterized by a negative binding energy. We show that the addition of sulphonate groups (–SO3−) to cyclic copper phthalocyanine (CuPc; ref. 8) systematically increases the energy of the corresponding molecular orbitals, culminating in the highest occupied molecular orbital of the tetra-anion, [CuPc(SO3)4]4−, being unstable by 0.9 eV. The increase in molecular orbital energy and the negative electron binding energy we observe are due to charge localization in the sulphonate groups and the resultant RCB. The unusually large height of the repulsive barrier also ensures that the anion remains metastable, and continues to store 0.9 eV excess electrostatic energy, throughout the 400 seconds we are able to observe it.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Scheller, M. K., Compton, R. N. & Cederbaum, L. S. Gas-phase multiply charged anions. Science 270, 1160–1166 (1995).
Jin, C. et al. Attachment of two electrons to C60F48: Coulomb barrier in doubly charged anions. Phys. Rev. Lett. 73, 2821–2824 (1994).
Compton, R. N., Tuinman, A. A., Klots, C. E., Peterson, MR. & Patton, D. C. Electron attachment to a negative ion: e + C84− → C842−. Phys. Rev. Lett. 78, 4367–4370 (1997).
Martin, R. L. & Ritchie, J. P. Coulomb and exchange interactions in C60n−. Phys. Rev. B 48, 4845–4849 (1993).
Yannouleas, C. & Landman, U. Stabilized-jellium description of neutral and multiply charged fullerenes C60x±. Chem. Phys. Lett. 217, 175–185 (1994).
Wang, X. B., Ding, C. F. & Wang, L. S. Photodetachment spectroscopy of a doubly charged anion: direct observation of the repulsive Coulomb barrier. Phys. Rev. Lett. 81, 3351–3354 (1998).
Wang, L. S., Ding, C. F., Wang, X. B. & Nicholas, J. B. Probing the potential barriers and intramolecular electrostatic interactions in free doubly charged anions. Phys. Rev. Lett. 81, 2667–2670 (1998).
Leznoff, C. C. & Lever, A. B. P. (eds) Phthalocyanines: Properties and Applications (VCH, New York, (1989).
Wang, L. S., Ding, C. F., Wang, X. B. & Barlow, S. E. Photodetachment photoelectron spectroscopy of multiply charged anions using electrospray ionization. Rev. Sci. Instrum. 70, 1957–1966 (1999).
Berkowitz, J. Photoelectron spectroscopy of phthalocyanine vapors. J. Chem. Phys. 70, 2819–2828 (1979).
Brown, C. J. Crystal structure of β-copper phthalocyanine. J. Chem. Soc. A 2488–2493 (1968).
Rosa, A. & Baerends, E. J. Metal-macrocycle interaction in phthalocyanines: density functional calculations of ground and excited states. Inorg. Chem. 33, 584–595 (1994).
PC Spartan Plus 5.1(Wavefunction, Inc., Von Carman Ave., Irvine, California 92612, USA).
Stewart, J. J. Optimization of parameters for semiempirical methods II: Applications. J. Comput. Chem. 10, 221–264 (1989); MOPAC: A semiempirical molecular orbital program. J. Comput. Aided Mol. Design 4, 1–105 (1990).
D'Haennens, I. J. in Encyclopedia of Physics (eds Lerner, R. G. & Trigg, G. L.) 1251–1253 (VCH, New York, (1991).
Hodgson, P. E., Gadioli, E. & Erba, E. G. Introductory Nuclear Physics (Clarendon, Oxford, (1997).
Scheller, M. K. & Cederbaum, L. S. Aconstruction principle for stable multiply charged molecular anions in the gas phase. Chem. Phys. Lett. 216, 141–146 (1993).
Weikert, H.-G. & Cederbaum, L. S. Free doubly negative tetrahalides. J. Chem. Phys. 99, 8877–8891 (1993).
Boldyrev, A. I., Gutowski, M. & Simons, J. Small multiply charged anions as building blocks in chemistry. Acc. Chem. Res. 29, 497–502 (1996).
Vekey, K. Multiply charged ions. Mass Spectrom. Rev. 14, 195–225 (1995).
Brechignac, C., Cahuzac, P., Kebaili, N. & Leygnier, J. Temperature effects in the Coulombic fission of strontium clusters. Phys. Rev. Lett. 81, 4612–4615 (1998).
Schroder, D., Harvey, J. N. & Schwarz, H. Long-lived, multiply charged diatomic TiFn+ ions (n = 1–3). J. Phys. Chem. A 102, 3639–3642 (1998).
Acknowledgements
We thank R. S. Disselkamp for discussions and K. Ferris for help in the theoretical calculations. This work was supported by the US Department of Energy, Office of Basic Energy Sciences, Chemical Sciences Division and is conducted at the Pacific Northwest National Laboratory, operated for the US Department of Energy by Battelle Memorial Institute. L.-S.W. is an Alfred P. Sloan research fellow.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, XB., Wang, LS. Observation of negative electron-binding energy in a molecule. Nature 400, 245–248 (1999). https://doi.org/10.1038/22286
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/22286
This article is cited by
-
Influence of heteroelement on dipole and quadrupole moments of a series of three-membered rings containing a second, third, fourth, or fifth-row atom: a theoretical investigation
Structural Chemistry (2019)
-
Accurate Electron Affinity of Iron and Fine Structures of Negative Iron ions
Scientific Reports (2016)
-
Structures and Chemical Bonding in NbS n 2−/−/0 (n = 3–5) Clusters: Effects of Sulfur Content and Charge States
Journal of Cluster Science (2016)
-
A systematic density functional and wavefunction-based study on dicarboxyl dianions −O2C–R–CO2 − with R = C2, C2X2, C2X4, and C6X4 (X = H, F)
Theoretical Chemistry Accounts (2010)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.