Abstract
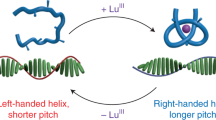
The helicity of biological macromolecules such as DNA and proteins is largely governed by the homochirality of their components (D-sugars and L-amino acids). In polymer and supramolecular chemistry, control of helicity is an attractive goal because of possible applications in materials science, chemical sensing and enantioselective catalysis1,2,3,4,5,6,7,8,9,10,11,12,13. We reported recently that macromolecular helicity can be induced in a polymer by an optically active amine14. Here we show that this helicity can be ‘memorized’ when the amine is replaced by various achiral amines. Although the maintenance of helicity in the polymer is not perfect, it can ‘repair’ itself over time. Small structural changes in the achiral amines influence the efficiency of helicity retention markedly.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lehn, J. M. Supramolecular Chemistry(VCH, Weinheim, (1995).
Constable, E. C. Oligopyridines as helicating ligands. Tetrahedron 46, 10013–10059 (1992).
Amabilino, D. B. & Stoddart, J. F. Interlocked and intertwined structures and superstructures. Chem. Rev. 95, 2725–2828 (1995).
Piguet, C., Bernardinelli, G. & Hopfgartner, G. Helicates as versatile supramolecular complexes. Chem. Rev. 97, 2005–2062 (1997).
Okamoto, Y. & Nakano, T. Asymmetric polymerization. Chem. Rev. 94, 349–372 (1994).
Nolte, R. J. M. Helical poly(isocyanides). Chem. Soc. Rev. 23, 11–19 (1994).
Green, M. M.et al. Ahelical polymer with a cooperative response to chiral information. Science 268, 1860–1866 (1995).
Rowan, A. E. & Nolte, R. J. M. Helical molecular programing. Angew. Chem. Int. Edn. 37, 703–706 (1998).
Schlitzer, D. S. & Novak, B. M. Trapped kinetic states, chiral amplification and molecular chaperoning in synthetic polymers: chiral induction in polyguanidines through ion pair interactions. J. Am. Chem. Soc. 120, 2196–2197 (1998).
Ruokolainen, J.et al. Switching supramolecular polymeric materials with multiple length scales. Science 280, 557–560 (1998).
Tsukruk, V. V. Assembly of supramolecular polymers in ultrathin films. Prog. Polym. Sci. 22, 247–311 (1997).
Pu, L. The study of chiral conjugated polymers. Acta Polym. 48, 116–141 (1997).
Okamoto, Y. & Yashima, E. Polysaccharide derivatives for chromatographic separation of enantiomers. Angew. Chem. Int. Edn. 37, 1020–1043 (1998).
Yashima, E., Matsushima, T. & Okamoto, Y. Chirality assignment of amines and amino alcohols based on circular dichroism induced by helix formation of a stereoregular poly(4-carboxyphenylacetylene) through acid-base complexation. J. Am. Chem. Soc. 119, 6345–6359 (1997).
Lindsey, J. S. Self-assembly in synthetic routes to molecular devices. Biological principles and chemical perspectives: a review. New J. Chem. 15, 153–180 (1991).
Harada, N. & Nakanishi, K. Circular Dichroic Spectroscopy—Exciton Coupling in Organic Stereochemistry(University Science Books, Mill Valley, CA, (1983).
Manabe, K., Okamura, K., Date, T. & Koga, K. Effects of ion-pair structure on relative basicity in chloroform: acid-base equilibria controlled by steric repulsion, π-stacking interactions, and hydrogen bonding within an ion pair. J. Am. Chem. Soc. 115, 5324–5325 (1993).
Acknowledgements
We thank Y. Maeda for his preliminary experimental support. We thank T. Sato, K.Akiyoshi and T. Aida for discussions. This work was supported in part by Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports, and Culture, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yashima, E., Maeda, K. & Okamoto, Y. Memory of macromolecular helicity assisted by interaction with achiral small molecules. Nature 399, 449–451 (1999). https://doi.org/10.1038/20900
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/20900
This article is cited by
-
Stapling strategy for slowing helicity interconversion of α-helical peptides and isolating chiral auxiliary-free one-handed forms
Nature Communications (2023)
-
Memorable full-color circularly polarized luminescence from chiral co-assembled polymer films enabled by multipath transfer
Science China Chemistry (2023)
-
Multi-color Tunable and White Circularly Polarized Luminescent Composite Nanofibers Electrospun from Chiral Helical Polymer
Advanced Fiber Materials (2022)
-
The folding propensity of α/sulfono-γ-AA peptidic foldamers with both left- and right-handedness
Communications Chemistry (2021)
-
Hierarchical communication of chirality for aromatic oligoamide sequences
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.