Abstract
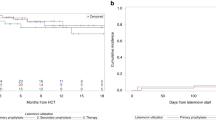
Despite significant advances in prevention and therapy, cytomegalovirus (CMV) infection continues to be an important cause of morbidity and mortality in the hematopoietic stem cell transplant (HSCT) recipient. The standard drug for pre-emptive therapy is intravenous ganciclovir (GCV). Valganciclovir (VGC), the oral pro-drug of GCV, has excellent bioavailability and is ideal for oral therapy. Since March 2002, VGC was adopted in our center for outpatient pre-emptive therapy in all patients undergoing allogeneic HSCT. Fifty-two allogeneic HSCT recipients were followed weekly via Digene hybrid capture assay®. Patients with a positive assay were treated with VGC 900 mg p.o. b.i.d. × 14 days followed by 900 mg p.o. QD until at least 7 days after a negative test. Eighteen patients (14 sib, four MUD) had 30 episodes of CMV DNA detection treated with oral VGC. Median duration of therapy was 21 days (range 10–21 days). The rate of response was 93% (28/30) as confirmed by a negative assay within 14 days. No significant toxicity was encountered. Two patients failed oral VGC. One case of CMV enteritis was diagnosed in a patient with acute GVHD. Pre-emptive therapy of CMV infection with oral VGC is safe and effective in allogeneic HSCT recipients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Meyers JD, Fluornoy N, Thomas ED . Nonbacterial pneumonia after allogeneic marrow transplantation. A review of ten years experience. Rev Infect Dis 1982; 4: 1119–1132.
Guidelines for Preventing Opportunistic Infections Among Hematopoietic Stem Cell Transplant Recipients. Recommendations of CDC, the Infectious Disease Society of America and the American Society of Blood and Marrow Transplantation. Biol blood Marrow Transplant 2000; 6: 659–713.
Boeckh M, Nichols WG, Papanicolaou G, Rubn R, Wingard JR, Zaia J . Cytomegalovirus in hematopoietic stem cell transplant recipients: current status, known challenges, and future strategies. Biol Blood Marrow Transplant 2003; 9: 543–558.
Ljungman P, Aschan J, Lewensohn-Fuchs I, Carlens S, Larsson K, Lonnqvist B et al. Results of different strategies for reducing cytomegalovirus-associated mortality in allogeneic stem cell transplantation recipients. Transplantation 1998; 66: 1330–1334.
Chakrabarti S, Mackinnon S, Chopra R, Kottaridis PD, Peggs K, O'Gorman P et al. High incidence of cytomegalovirus infection after nonmyeloablative stem cell transplantation: potential role of Campath-1H in delaying immune reconstitution. Blood 2002; 99: 4357–4363.
Junghanss C, Boeckh M, Carter RA, Sandmaier BM, Maris MB, Maloney DG et al. Incidence and outcome of cytomegalovirus infections following nonmyeloablative compared with myeloablative allogeneic stem cell transplantation, a matched control study. Blood 2002; 99: 1978–1985.
Martino R, Rovira M, Carreras E, Solano C, Jorge S, De La Rubia J et al. Severe infections after allogeneic peripheral blood stem cell transplantation: a matched-pair comparison of unmanipulated and CD34+ cell-selected transplantation. Haematologica 2001; 86: 1075–1086.
Kroger N, Zabelina T, Kruger W, Renges H, Stute N, Schrum J et al. Patient cytomegalovirus seropositivity with or without reactivation is the most important prognostic factor for survival and treatment-related mortality in stem cell transplantation from unrelated donors using pretransplant in vivo T-cell depletion with anti-thymocyte globulin. Br J Hematol 2001; 113: 1060–1071.
Bacigalupo A, Mordini N, Pitto A, Piaggio G, Podesta M, Benvenuto F et al. Transplantation of HLA-mismatched CD34+ selected cells in patients with advanced malignancies: severe immunodeficiency and related complications. Br J Haematol 1997; 98: 760–766.
Verdonck LF, Dekker AW, Rozenberg-Arska M, van den Hoek MR . A risk-adapted approach with a short course of ganciclovir to prevent cytomegalovirus (CMV) pneumonia in CMV-seropositive recipients of allogeneic bone marrow transplants. Clin Infect Dis 1997; 24: 901–907.
Zaia JA . Prevention and management of CMV-related problems after hematopoietic stem cell transplantation. Bone Marrow Transplant 2002; 29: 633–638.
Ljungman P . Prevention and treatment of viral infections in stem cell transplant recipients. Br J Haematol 2002; 118: 44–57.
Einsele H, Ehninger G, Hebart H, Wittkowski KM, Schuler U, Jahn G et al. Polymerase chain reaction monitoring reduces the incidence of cytomegalovirus disease and the duration and side effects of antiviral therapy after bone marrow transplantation. Blood 1995; 86: 2815–2820.
Goodrich JM, Bowden RA, Fisher L, Keller C, Schoch G, Meyers JD . Ganciclovir prophylaxis to prevent cytomegalovirus disease after allogeneic marrow transplant. Ann Internal Med 1993; 118: 173–178.
Winston DJ, Ho WG, Bartoni K, Du Mond C, Ebeling DF, Buhles WC et al. Ganciclovir prophylaxis of cytomegalovirus infection and disease in allogeneic bone marrow transplant recipients. Results of a placebo-controlled, double-blind trial. Ann Internal Med 1993; 118: 179–184.
Boeckh M, Gooley TA, Myerson D, Cunningham T, Schoch G, Bowden RA . Cytomegalovirus pp65 antigenemia-guided early treatment with ganciclovir versus ganciclovir at engraftment after allogeneic marrow transplantation: a randomized double-blind study. Blood 1996; 80: 4063–4071.
Schmidt GM, Horak DA, Niland JC, Duncan SR, Forman SJ, Zaia JA . A randomized controlled trial of prophylactic ganciclovir for cytomegalovirus pulmonary infection in recipients of allogeneic bone marrow transplants. N Engl J Med 1991; 324: 1005–1011.
Goodrich JM, Mori M, Gleaves CA, Du Mond C, Cays M, Ebeling DF et al. Early treatment with ganciclovir to prevent cytomegalovirus disease after allogeneic bone marrow transplantation. N Engl J Med 1991; 325: 1601–1607.
Cvetkovic RS, Wellington K . Valganciclovir: a review of its use in the management of CMV infection and disease in immunocompromised patients. Drugs 2005; 65: 859–878.
Martin DF, Sierra-Madero J, Walmsley S, Wolitz RA, Macey K, Georgiou P et al. A controlled trial of valganciclovir as induction therapy for cytomegalovirus retinitis. N Engl J Med 2002; 346: 1119–1126.
Crowder L, Ruge D, Johnson J. Use of the Digene Hybrid Capture CMV Signal Amplification Assay ver. 2.0 (HC) in a cancer hospital helps modify ganciclovir use for cytomegalovirus from a prophylactic to a preemptive approach. Poster Session #126/C, Abstract C-138, American Society for Microbiology General Meeting, Orlando, FL, May 2001.
Schirm J, Kooistra A, van Son WJ, van der Bij W, Verschuuren E, Sprenge HG et al. Comparison of the Murex Hybrid Capture CMV DNA (v2.0) assay and the pp65 antigenemia test for the detection and quantitation of CMV in blood samples from immunocompromised patients. J Clin Virol 1999; 14: 153–165.
Hebart H, Wuchter P, Loeffler J, Gscheidle B, Hamprecht K, Sinzger C et al. Evaluation of the Murex CMV DNA Hybrid Capture assay (version 2.0) for early diagnosis of cytomegalovirus infection in recipients of an allogeneic stem cell transplant. Bone Marrow Transplant 2001; 28: 213–218.
Weinberg A, Schissel D, Giller R . Molecular methods for cytomegalovirus surveillance in bone marrow transplant recipients. J Clin Microbiol 2002; 40: 4203–4206.
Ljungman P, Griffith P . Definitions of Cytomegalovirus infection and disease. In: Michelson S, Plotkin S (eds). Multidisciplinary Approach to Understanding Cytomegalovirus Disease. Elsevier Science Publishers BV: Amsterdam, 1993.
Leather HL, Meyer CL, Wingard JR . Oral (PO) valganciclovir (VGC) for preemptive therapy of cytomegalovirus (CMV) antigenemia (AG) in hematopoietic stem cell transplant (HSCT) recipients. Biol Blood Marrow Transplant 2003; 9: 238.
Einsele H . Preemptive therapy with IV ganciclovir vs valganciclovir: PK and preliminary safety and efficacy data. Oral presentation in CMV in Stem Cell Transplantation: New insights and options. Symposium presented at the Tandem Transplantation Meetings, Orlando, Florida, 2004.
Reusser P, Einsele H, Lee J, Volin L, Rovira M, Engelhard D et al. Randomized multicenter trial of foscarnet vs ganciclovir for preemptive therapy of cytomegalovirus infection after allogeneic stem cell transplantation. Blood 2002; 99: 1159–1164.
Acknowledgements
We thank all the clinicians, nurses, and pharmacists from our transplantation team who helped to take care of this group of patients.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ayala, E., Greene, J., Sandin, R. et al. Valganciclovir is safe and effective as pre-emptive therapy for CMV infection in allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 37, 851–856 (2006). https://doi.org/10.1038/sj.bmt.1705341
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705341
Keywords
This article is cited by
-
Empirical treatment against cytomegalovirus and tuberculosis in HIV-infected infants with severe pneumonia: study protocol for a multicenter, open-label randomized controlled clinical trial
Trials (2022)
-
Infectious diseases in allogeneic haematopoietic stem cell transplantation: prevention and prophylaxis strategy guidelines 2016
Annals of Hematology (2016)