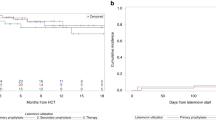
Summary:
Cytomegalovirus (CMV) disease remains an important cause of morbidity and mortality in patients undergoing hematopoietic stem cell transplantation (HSCT). We evaluated high-dose acyclovir (HDACV) and pre-emptive ganciclovir to prevent CMV disease in 76 children who underwent peripheral blood stem cell transplantation (PBSCT) and were at risk for CMV reactivation and disease (both recipient and donor seropositive) from May 1998 to April 2003. All received HDACV from day −9 to 6 months post transplant in conjunction with weekly CMV pp65 antigenemia monitoring. The incidence of antigenemia in this cohort was 19.7%, at a median of 22 days post-PBSCT. The frequencies were 26.4 and 4.4% in allogeneic and autologous groups, respectively (P=0.03). Patients with nonmalignant disease had higher CMV antigenemia than those with malignant disease (30.8 vs 8.1%, P=0.02). Age at PBSCT, sex, graft-versus-host disease (GVHD) prophylaxis regimen and presence of acute GVHD did not affect the risk of CMV antigenemia. None of the patients who had positive pp65 antigenemia developed CMV disease during the study period. We conclude that pp65 antigenemia-guided HDACV and pre-emptive ganciclovir may prevent CMV disease in children undergoing PBSCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Meyers JD, Flournoy N, Thomas E . Risk factors for cytomegalovirus infection after human bone marrow transplantation. J Infect Dis 1986; 153: 478–488.
Forman SJ, Zaia JA . Treatment and prevention of cytomegalovirus pneumonia after bone marrow transplantation: where do we stand? Blood 1994; 83: 2392–2398.
Wingard JR, Chen DYH, Burns WH et al. Cytomegalovirus infection after autologous bone marrow transplantation with comparison to infection after allogeneic bone marrow transplantation. Blood 1998; 71: 1432–1437.
Osarogiagbon RU, Defor TE, Weisdorf MA et al. CMV antigenemia following bone marrow transplantation: risk factors and outcomes. Biol Blood Marrow Transplant 2000; 6: 280–288.
Broers AE, van der Hold R, van Esser JWJ et al. Increased transplant-related morbidity and mortality in CMV-seropositive patients despite highly effective prevention of CMV disease after allogeneic T-cell-depleted stem cell transplantation. Blood 2000; 95: 2240–2245.
Kanda Y, Mineishi S, Saito T et al. Pre-emptive therapy against cytomegalovirus (CMV) disease guided by CMV antigenemia assay after allogeneic hematopoietic stem cell transplantation: a single-center experience in Japan. Bone Marrow Transplant 2001; 27: 437–444.
Reed EC, Bowden RA, Dandliker PS et al. Treatment of cytomegalovirus pneumonia with ganciclovir and intravenous cytomegalovirus immunoglobulin in patients with bone marrow transplants. Ann Intern Med 1988; 109: 783–788.
Winston DJ, Ho WG, Bartoni RN et al. Ganciclovir prophylaxis of cytomegalovirus infection and disease in allogeneic bone marrow transplant recipients. Results of a placebo-controlled, double blind trial. Ann Intern Med 1993; 118: 179–184.
Bueno J, Ramil C, Green M . Current management strategies for the prevention and treatment of cytomegalovirus infection in pediatric transplant recipients. Paediatr Drugs 2002; 4: 279–290.
Meyers JD, Reed EC, Shepp DH et al. Acyclovir for prevention of cytomegalovirus infection and disease after allogeneic marrow transplantation. N Engl J Med 1988; 318: 70–75.
Schmidt GM, Horak DA, Niland JC et al. A randomized controlled trial of prophylactic ganciclovir for cytomegalovirus pulmonary infection in recipients of allogeneic bone marrow transplants: The City of Hope-Stanford-Syntex CMV Study Group. N Engl J Med 1991; 324: 1005–1011.
Goodrich JM, Bowden R, Fisher L et al. Ganciclovir prophylaxis to prevent cytomegalovirus disease after allogeneic marrow transplant. Ann Intern Med 1993; 118: 173–178.
Thyms AS, Seamans EM, Naim HM . The in vitro activity of acyclovir and related compounds against cytomegalovirus infections. J Antimicrob Chemother 1991; 8: 65–72.
Prentice HG, Gluckman E, Powles R et al. For the European Acyclovir for CMV Prophylaxis Study Group. Impact of long-term acyclovir on cytomegalovirus infection and survival after allogeneic bone marrow transplantation. Lancet 1994; 343: 749–753.
Prentice HG, Gluckman E, Powles R et al. Long-term survival in allogeneic bone marrow transplant recipients following acyclovir prophylaxis for CMV infection. Bone Marrow Transplant 1997; 19: 129–133.
Nakamura R, Cortez K, Solomon S et al. High-dose acyclovir and pre-emptive ganciclovir to prevent cytomegalovirus disease in myloablative and non-myeloablative allogeneic stem cell transplantation. Bone Marrow Transplant 2002; 30: 235–242.
Boeckh M, Gooley TA, Bowden RA . Effect of high-dose acyclovir on survival in allogeneic marrow transplant recipients who received ganciclovir at engraftment or for cytomegalovirus pp65 antigenemia. J Infect Dis 1998; 178: 1153–1157.
Ottinger HD, Beelen DW, Schaefer UW, Grosse-Wilde H . Improved immune reconstitution after allotransplantation of peripheral blood stem cells instead of bone marrow. Blood 1996; 7: 2775–2779.
Trenschel R, Bernier M, Delforge A et al. Myeloid and lymphoid recovery following bone marrow transplantation: a comparative study between related, unrelated bone marrow and allogeneic stem cell transplantation. Leuk Lymphoma 1998; 30: 325–352.
Trenschel R, Ross S, Hüsing J et al. Reduced risk of persisting cytomegalovirus pp65 antigenemia and cytomegalovirus interstitial pneumonia following allogeneic PBSCT. Bone Marrow Transplant 2000; 25: 665–672.
Boeckh M, Gooley TA, Myerson D et al. Cytomegalovirus pp65 antigenemia-guided treatment with ganciclovir versus ganciclovir at engraftment after allogeneic marrow transplantation: a randomized double-blind study. Blood 1996; 88: 4063–4071.
Koehler M, George KS, Ehrlich GD et al. Prevention of CMV disease in allogeneic BMT recipients by cytomegalovirus antigenemia-guided preemptive ganciclovir therapy. J Pediatr Hematol Oncol 1997; 19: 43–47.
Boeckh M, Bowden RA, Gooley T et al. Successful modification of a pp65 antigenemia-based early treatment strategy for prevention of cytomegalovirus disease in allogeneic marrow transplant recipients. Blood 1999; 93: 1781–1782.
Shimokawa T, Morishima Y, Kitaori K et al. Early treatment of CMV antigenemia with ganciclovir for prevention of fatal CMV disease in patients receiving marrow from HLA-matched unrelated donors. Int J Hematol 1999; 70: 119–126.
Mori T, Okamoto S, Matsuoka S et al. Risk-adapted pre-emptive therapy for cytomegalovirus disease in patients undergoing allogeneic bone marrow transplantation. Bone Marrow Transplant 2000; 25: 765–769.
Verdonck LF, Dekker AW, Rozenberg-Arska M, van den Hoek MR . A risk-adapted approach with a short course of ganciclovir to prevent cytomegalovirus (CMV) pneumonia in CMV-seropositive recipients of allogeneic bone marrow transplantation. Clin Infect Dis 1997; 24: 901–907.
Manteiga R, Martino R, Sureda A et al. Cytomegalovirus pp65 antigenemia-guided pre-emptive treatment with ganciclovir after allogeneic stem cell transplantation: a single center experience. Bone Marrow Transplant 1998; 22: 899–904.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper was partly presented at EBMT 29th Meeting in Istanbul.
Rights and permissions
About this article
Cite this article
Hazar, V., Kansoy, S., Küpesiz, A. et al. High-dose acyclovir and pre-emptive ganciclovir in prevention of cytomegalovirus disease in pediatric patients following peripheral blood stem cell transplantation. Bone Marrow Transplant 33, 931–935 (2004). https://doi.org/10.1038/sj.bmt.1704463
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704463
Keywords
This article is cited by
-
High-dose aciclovir in CMV infection prophylaxis after allogeneic HSCT: a single-center long-term experience
Bone Marrow Transplantation (2023)