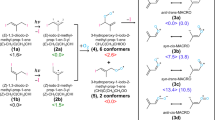
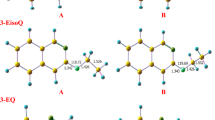
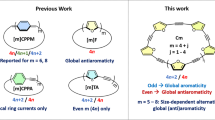
Abstract
RECENT work by Reppe1 has proved that Willstätter's hydrocarbon C8H8 has indeed the cyclo-octatetraene structure which was originally assigned to it. Its reactions enabled him to conclude that “the double bonds of cyclo-octatetraene are open to all the reactions of ethylenic chemistry”. This conclusion might at first sight be taken to indicate the absence of resonance in this molecule. However, this would be in contradiction to the physical data on the heat of combustion and the absorption of light, which can be taken as indicative of a resonating molecule. The heat of combustion leads to a value of 28 kcal. for the resonance energy, compared with 41 kcal. for benzene. The substance is described as a golden yellow liquid, which places the long wavelength ultra-violet system corresponding to the 2600 A. band of benzene at about 4000 A. If it contained fixed double bonds, then the absorption would be expected to lie below 2000 A. Thus the evidence from the heat of combustion and the absorption spectrum indicates a resonating molecule.
Similar content being viewed by others
Article PDF
References
Report on Cyclopolyolefins, W. Reppe (Chem. Soc. Library, 1944).
Proc Roy. Soc., A, 146, 223 (1934).
J. Chem. Phys., 3, 230 (1935).
Z. Elect., 45, 548 (1939).
Acta Physicochimica U.R.S.S., 19, 385 (1944). See also Pauling and Sherman, J. Chem. Phys., 1, 679 (1933), in which the value 30 kcal. is obtained.
Evans, Trans. Faraday Soc., 35, 824 (1939).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MACCOLL, A. Cyclo-octatetraene. Nature 157, 695 (1946). https://doi.org/10.1038/157695a0
Issue Date:
DOI: https://doi.org/10.1038/157695a0
This article is cited by
-
Structure of cyclo-Octatetraene
Nature (1950)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.