Featured
-
-
Article
| Open Access Cryo-EM structures of lipidic fibrils of amyloid-β (1-40)
Cryo-EM structures of lipidic fibrils of amyloid-β (1-40)
Alzheimer’s plaques contain a high amount of Aβ fibrils and a high concentration of lipids. The authors determined structures of Aβ40 fibrils grown in the presence of lipids, revealing high-resolution details of potentially disease-relevant fibril-lipid interactions.
- Benedikt Frieg
- , Mookyoung Han
- & Gunnar F. Schröder
-
Article
| Open Access Protein thermal sensing regulates physiological amyloid aggregation
Protein thermal sensing regulates physiological amyloid aggregation
Cells form non-pathological amyloids to survive stressful conditions. Marijan et al. show that heat shock-induced aggregation is self-regulated by protein stability, with high-ordered motifs acting as thermo-switches that control amyloidogenesis.
- Dane Marijan
- , Evgenia A. Momchilova
- & Timothy E. Audas
-
Article
| Open Access Excess PrPC inhibits muscle cell differentiation via miRNA-enhanced liquid–liquid phase separation implicated in myopathy
Excess PrPC inhibits muscle cell differentiation via miRNA-enhanced liquid–liquid phase separation implicated in myopathy
The prion protein PrPC is known to play a role in skeletal muscle development and physiology, including in myopathy. Here, the authors report that excess PrPC binds microRNAs that enhance its aggregation, which inhibits autophagy in muscle cells.
- Jing Tao
- , Yanping Zeng
- & Yi Liang
-
Article
| Open Access Cryo-EM structure and polymorphic maturation of a viral transduction enhancing amyloid fibril
Cryo-EM structure and polymorphic maturation of a viral transduction enhancing amyloid fibril
In this work, the authors report the Cryo-EM structure of PNF-18, a biotechnologically engineered peptide fibril that enhances retroviral infectivity. The peptide fibrils mature into polymorphic amyloid structures in a time-dependent manner. The structure provides insights into the molecular basis of peptide nanofibrils as retroviral transduction enhancers.
- Thomas Heerde
- , Desiree Schütz
- & Marcus Fändrich
-
Article
| Open Access FTD-tau S320F mutation stabilizes local structure and allosterically promotes amyloid motif-dependent aggregation
FTD-tau S320F mutation stabilizes local structure and allosterically promotes amyloid motif-dependent aggregation
The authors used multi-disciplinary approaches to understand the structural mechanism underlying spontaneous aggregation of tau encoding an S320F FTD-tau mutant. Understanding the mechanisms of tau aggregation will help identify novel methods to regulate its misfolding.
- Dailu Chen
- , Sofia Bali
- & Lukasz A. Joachimiak
-
Article
| Open Access Network of hotspot interactions cluster tau amyloid folds
Network of hotspot interactions cluster tau amyloid folds
The authors developed a computational approach to probe the stability of amyloid fibrils and discover networks of hotspot interactions. Understanding the mechanisms of amyloid folding will help identify novel methods to treat protein (mis)folding diseases.
- Vishruth Mullapudi
- , Jaime Vaquer-Alicea
- & Lukasz A. Joachimiak
-
Article
| Open Access Disease-associated mutations within the yeast DNAJB6 homolog Sis1 slow conformer-specific substrate processing and can be corrected by the modulation of nucleotide exchange factors
Disease-associated mutations within the yeast DNAJB6 homolog Sis1 slow conformer-specific substrate processing and can be corrected by the modulation of nucleotide exchange factors
Here the authors describe mechanisms through which analogous LGMDD1 (Limb-Girdle Muscular Dystrophy Type D1) mutations affect Sis1 (a yeast functional homolog of human DNAJB6) chaperone activity and poison the function of wild-type protein; potentially uncovering a new therapeutic route to explore.
- Ankan K. Bhadra
- , Michael J. Rau
- & Heather L. True
-
Comment
| Open Access The shape of things to come: structural insights into how prion proteins encipher heritable information
The shape of things to come: structural insights into how prion proteins encipher heritable information
The prion hypothesis embodies the radical concept that prion proteins contain the necessary information for infectious replication within their shape, thus obviating the requirement for genomic material. Two elegant papers by Hoyt et al. and Manka et al. describing high-resolution structures of infectious prions bring us closer to answering the long-standing question of how different prion conformations produce heritably distinct diseases.
- Glenn C. Telling
-
Article
| Open Access 2.7 Å cryo-EM structure of ex vivo RML prion fibrils
2.7 Å cryo-EM structure of ex vivo RML prion fibrils
High-resolution structures of mammalian prions have remained elusive. Here, Manka et al. report the cryo-EM structure of infectious RML prion fibrils from mice. Structural similarity with recently reported infectious 263K prion fibrils from hamsters now suggests a common prion architecture.
- Szymon W. Manka
- , Wenjuan Zhang
- & Jonathan D. F. Wadsworth
-
Article
| Open Access Cryo-EM demonstrates the in vitro proliferation of an ex vivo amyloid fibril morphology by seeding
Cryo-EM demonstrates the in vitro proliferation of an ex vivo amyloid fibril morphology by seeding
Here, the authors present the cryo-EM structure of in vitro amyloid fibrils from recombinant SAA1.1 protein that were formed by seeding with fibrils purified from systemic AA amyloidosis tissue. This in vitro fibril structure resembles the structure of the ex vivo fibrils but differs from unseeded in vitro fibrils. These findings show that fibril morphologies can be propagated in vitro by seeding.
- Thomas Heerde
- , Matthies Rennegarbe
- & Marcus Fändrich
-
Article
| Open Access Aβ receptors specifically recognize molecular features displayed by fibril ends and neurotoxic oligomers
Aβ receptors specifically recognize molecular features displayed by fibril ends and neurotoxic oligomers
PrPC, a receptor for Aβ oligomers, blocks polarized elongation of Aβ fibrils by binding to the rapidly growing end of each fibril. PrPC and other receptors may trap Aβ oligomers and protofibrils on the neuronal surface by binding to a common molecular determinant, initiating a neurotoxic signal.
- Ladan Amin
- & David A. Harris
-
Article
| Open Access NSs amyloid formation is associated with the virulence of Rift Valley fever virus in mice
NSs amyloid formation is associated with the virulence of Rift Valley fever virus in mice
Rift Valley fever virus (RVFV) can cause severe diseases in humans, including encephalitis. Here the authors show that NSs, the major virulence factor of RVFV, is an amyloidogenic protein forming fibrils in infected mouse brains and causing increased mortality in mice.
- Psylvia Léger
- , Eliana Nachman
- & Pierre-Yves Lozach
-
Article
| Open Access Single-molecule detection on a portable 3D-printed microscope
Single-molecule detection on a portable 3D-printed microscope
Single-molecule in vitro assays require dedicated confocal microscopes equipped with fluorescence correlation spectroscopy (FCS) modules. Here the authors present a compact, cheap and open-source 3D-printed confocal microscope for single photon counting and FCS measurements, and use it to detect α-synuclein aggregation.
- James W. P. Brown
- , Arnaud Bauer
- & Yann Gambin
-
Article
| Open Access Cryo-EM structure of a transthyretin-derived amyloid fibril from a patient with hereditary ATTR amyloidosis
Cryo-EM structure of a transthyretin-derived amyloid fibril from a patient with hereditary ATTR amyloidosis
Systemic amyloidosis of the ATTR is one of the most abundant forms of systemic amyloidosis and caused by misfolding of the circulating blood protein transthyretin (TTR). Here the authors present the cryo-EM structure of patient-derived Val30Met ATTR amyloid fibrils which reveals that the protofilament consists of an N-terminal and a C-terminal TTR fragment and discuss implications for the mechanism of misfolding.
- Matthias Schmidt
- , Sebastian Wiese
- & Marcus Fändrich
-
Article
| Open Access The prion-like protein kinase Sky1 is required for efficient stress granule disassembly
The prion-like protein kinase Sky1 is required for efficient stress granule disassembly
The factors regulating stress granule dissolution are not fully understood. Here, the authors identify Sky1 as a stress granule component in yeast, and show that Sky1 kinase activity is required for timely stress granule disassembly during stress recovery.
- Jenifer E. Shattuck
- , Kacy R. Paul
- & Eric D. Ross
-
Article
| Open Access Production of seedable Amyloid-β peptides in model of prion diseases upon PrPSc-induced PDK1 overactivation
Production of seedable Amyloid-β peptides in model of prion diseases upon PrPSc-induced PDK1 overactivation
Aβ plaques have been detected in brains of patients with prion diseases. Here, using mice, the authors show that prion infection enhances Aβ production via a PDK1-TACE mechanism and that brain deposition of Aβ induced by Aβ seeds co-transmitted with PrPSc contributes to mortality in prion disease.
- Juliette Ezpeleta
- , Vincent Baudouin
- & Benoit Schneider
-
Article
| Open Access Tau local structure shields an amyloid-forming motif and controls aggregation propensity
Tau local structure shields an amyloid-forming motif and controls aggregation propensity
The biophysical mechanisms of how disease-associated tau mutations drive amyloid formation are not well understood. Here the authors use biophysical approaches, cell models and MD simulations and find that the intrinsically disordered repeat domain of tau encodes a metastable local structure and perturbations through mutations and proline isomerization cause an aggregation phenotype in vitro and in cells.
- Dailu Chen
- , Kenneth W. Drombosky
- & Lukasz A. Joachimiak
-
Article
| Open Access Cryo-EM fibril structures from systemic AA amyloidosis reveal the species complementarity of pathological amyloids
Cryo-EM fibril structures from systemic AA amyloidosis reveal the species complementarity of pathological amyloids
Systemic AA amyloidosis is caused by misfolding of the acute phase protein serum amyloid A1. Here the authors present the cryo-EM structures of murine and human AA amyloid fibrils that were isolated from tissue samples and describe how the fibrils differ in their fundamental structural properties.
- Falk Liberta
- , Sarah Loerch
- & Matthias Schmidt
-
Article
| Open Access A viral expression factor behaves as a prion
A viral expression factor behaves as a prion
Prion-forming proteins have been found in animals, plants, fungi, and bacteria. Here, Nan et al. report that a baculovirus-encoded protein behaves as prion in a yeast system and forms aggregates at high multiplicity of infection in insect cells that affect baculovirus replication.
- Hao Nan
- , Hongying Chen
- & Xiaodong Xu
-
Article
| Open Access Artificial strain of human prions created in vitro
Artificial strain of human prions created in vitro
Synthetic prions have previously been generated from recombinant rodent PrP. Here the authors generate synthetic human prions, by seeding human PrP with CJD prions, and characterize its infectivity in mice.
- Chae Kim
- , Xiangzhu Xiao
- & Jiri G. Safar
-
Article
| Open Access Experimental transfusion of variant CJD-infected blood reveals previously uncharacterised prion disorder in mice and macaque
Experimental transfusion of variant CJD-infected blood reveals previously uncharacterised prion disorder in mice and macaque
It is hypothesised that exposure to bovine spongiform encephalopathy through contaminated food could have resulted in a large proportion of latent variant Creutzfeldt-Jakob disease cases in humans. Here the authors demonstrate that inoculation with blood from non-symptomatic, vCJD infected humans, results in a unique prion-like disorder in mice and macaques.
- Emmanuel E. Comoy
- , Jacqueline Mikol
- & Jean-Philippe Deslys
-
Article
| Open Access Species-dependent structural polymorphism of Y145Stop prion protein amyloid revealed by solid-state NMR spectroscopy
Species-dependent structural polymorphism of Y145Stop prion protein amyloid revealed by solid-state NMR spectroscopy
Prion diseases can be transmitted across species. Here the authors use solid-state NMR to study prion protein (PrP) amyloids from human, mouse and Syrian hamster and show that their structural differences are mainly governed by two residues, which helps to understand interspecies PrP propagation on a molecular level.
- Theint Theint
- , Philippe S. Nadaud
- & Christopher P. Jaroniec
-
Article
| Open Access Divergent prion strain evolution driven by PrPC expression level in transgenic mice
Divergent prion strain evolution driven by PrPC expression level in transgenic mice
PrPC protein plays a key role in prion transmission across species. Here, the authors compare transmission of a representative scrapie isolate to transgenic mice expressing variable levels of the same Prnp allele as the donor sheep, and find divergent strain propagation regulated by PrPCgene dosage.
- Annick Le Dur
- , Thanh Lan Laï
- & Hubert Laude
-
Article |
 Evidence for zoonotic potential of ovine scrapie prions
Evidence for zoonotic potential of ovine scrapie prions
Scrapie, a form of prion disease that affects sheep and goats, is believed not to be transmissible to humans. Using transgenic mice expressing human prion protein as a model of cross-species prion transmission, the authors show that ovine scrapie may possess potential to be passed on to humans.
- Hervé Cassard
- , Juan-Maria Torres
- & Olivier Andréoletti
-
Article |
 Amyloid-associated activity contributes to the severity and toxicity of a prion phenotype
Amyloid-associated activity contributes to the severity and toxicity of a prion phenotype
The yeast prion Sup35/[PSI+] confers a translation termination defect in its amyloid form. Pezza et al.reveal that aggregated Sup35 retains translation termination activity, and find that fluctuations in the size and composition of aggregates play an important role in determining their activity and toxicity.
- John A. Pezza
- , Janice Villali
- & Tricia R. Serio
-
Article |
 Loss of amino-terminal acetylation suppresses a prion phenotype by modulating global protein folding
Loss of amino-terminal acetylation suppresses a prion phenotype by modulating global protein folding
While N-terminal acetylation has been shown to regulate the function or stability of a limited number of specific proteins, Holmes et al.report that global loss of this modification results in widespread protein misfolding, and show that the resulting stress response contributes to the suppression of a yeast prion phenotype.
- William M. Holmes
- , Brian K. Mannakee
- & Tricia R. Serio
-
Article
| Open Access Prion neuropathology follows the accumulation of alternate prion protein isoforms after infective titre has peaked
Prion neuropathology follows the accumulation of alternate prion protein isoforms after infective titre has peaked
Prions (PrP) are infectious agents that cause lethal neurodegenerative diseases. Here the authors study the kinetics of prion propagation in mice and show that the onset of neuropathology occurs during the late phase of disease and is hypothesized to be due to increases in a toxic isoform of PrP that is different from the infectious species.
- Malin K. Sandberg
- , Huda Al-Doujaily
- & John Collinge
-
Article
| Open Access mGlu5 receptors and cellular prion protein mediate amyloid-β-facilitated synaptic long-term depression in vivo
mGlu5 receptors and cellular prion protein mediate amyloid-β-facilitated synaptic long-term depression in vivo
In Alzheimer's disease, the soluble amyloid beta peptide is known to modulate synaptic function by forming a complex with prion proteins and metabotropic glutamate receptors. Here, Hu et al.show that amyloid beta signalling via this complex facilitates the induction of long-term depression at synapses.
- Neng-Wei Hu
- , Andrew J. Nicoll
- & Michael J. Rowan
-
Article
| Open Access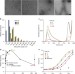 Amyloid-β nanotubes are associated with prion protein-dependent synaptotoxicity
Amyloid-β nanotubes are associated with prion protein-dependent synaptotoxicity
Prion protein has been suggested to bind toxic amyloid-ß oligomers. Nicollet al.demonstrate that binding to prion protein and prion protein-dependent synaptotoxicity correlate with the presence of a tubular form of amyloid-ß with a defined triple helical structure.
- Andrew J. Nicoll
- , Silvia Panico
- & John Collinge
-
Article
| Open Access Prion protein facilitates uptake of zinc into neuronal cells
Prion protein facilitates uptake of zinc into neuronal cells
Prion proteins are implicated in a range of neurodegenerative diseases, which are, in part, due to a disruption of metal homeostasis. Wattet al.use selective antagonists to show that prion proteins mediate zinc uptake by interacting with GluA2-lacking, GluA1-containing AMPA receptors.
- Nicole T. Watt
- , David R. Taylor
- & Nigel M. Hooper
-
Article
| Open Access Interaction between prion protein and toxic amyloid β assemblies can be therapeutically targeted at multiple sites
Interaction between prion protein and toxic amyloid β assemblies can be therapeutically targeted at multiple sites
The ability of synthetic amyloid β-protein to bind to prion proteins and alter synaptic plasticity has been previously reported. Here the relevance of this binding is investigated in brains of Alzheimer's disease patients and the interaction is shown to be blocked by antibodies to two distinct regions of prion proteins.
- Darragh B. Freir
- , Andrew J. Nicoll
- & John Collinge
-
Article
| Open Access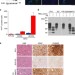 Rapid cell-surface prion protein conversion revealed using a novel cell system
Rapid cell-surface prion protein conversion revealed using a novel cell system
The study of prion diseases has been hampered as there is no method to distinguish newly formed abnormal prion protein conformers. Here, the authors describe a method to study newly formed abnormal prion protein and demonstrate that it is produced within 1 minute of cell exposure to prions.
- R. Goold
- , S. Rabbanian
- & S.J. Tabrizi

 A Protein Misfolding Shaking Amplification-based method for the spontaneous generation of hundreds of bona fide prions
A Protein Misfolding Shaking Amplification-based method for the spontaneous generation of hundreds of bona fide prions