« Prev Next »

Protein Function
The collection of proteins within a cell determines its health and function. Proteins are responsible for nearly every task of cellular life, including cell shape and inner organization, product manufacture and waste cleanup, and routine maintenance. Proteins also receive signals from outside the cell and mobilize intracellular response. They are the workhorse macromolecules of the cell and are as diverse as the functions they serve.
How Diverse Are Proteins?
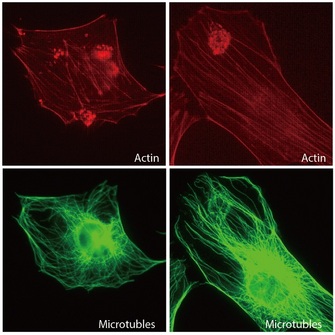
Not surprisingly, protein functions are as diverse as protein structures. For example, structural proteins maintain cell shape, akin to a skeleton, and they compose structural elements in connective tissues like cartilage and bone in vertebrates. Enzymes are another type of protein, and these molecules catalyze the biochemical reactions that occur in cells. Yet other proteins work as monitors, changing their shape and activity in response to metabolic signals or messages from outside the cell. Cells also secrete various proteins that become part of the extracellular matrix or are involved in intercellular communication.
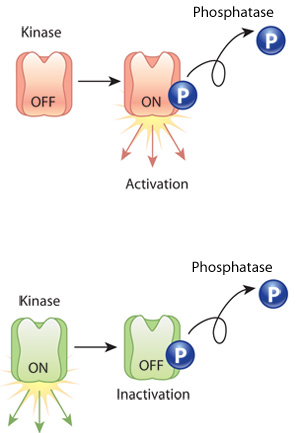
Proteins are sometimes altered after translation and folding are complete. In such cases, so-called transferase enzymes add small modifier groups, such as phosphates or carboxyl groups, to the protein. These modifications often shift protein conformation and act as molecular switches that turn the activity of a protein on or off. Many post-translational modifications are reversible, although different enzymes catalyze the reverse reactions. For example, enzymes called kinases add phosphate groups to proteins, but enzymes called phosphatases are required to remove these phosphate groups (Figure 1).
How Do Proteins Provide Structural Support for Cells?
How Do Proteins Aid the Biochemical Reactions of a Cell?
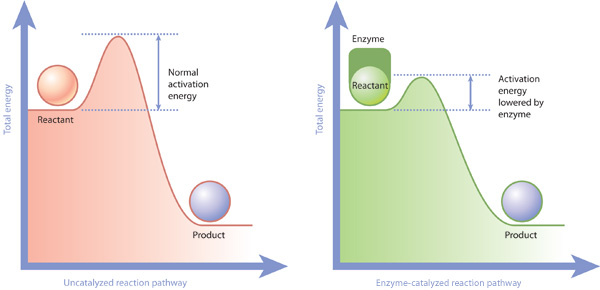
Cells rely on thousands of different enzymes to catalyze metabolic reactions. Enzymes are proteins, and they make a biochemical reaction more likely to proceed by lowering the activation energy of the reaction, thereby making these reactions proceed thousands or even millions of times faster than they would without a catalyst. Enzymes are highly specific to their substrates. They bind these substrates at complementary areas on their surfaces, providing a snug fit that many scientists compare to a lock and key. Enzymes work by binding one or more substrates, bringing them together so that a reaction can take place, and releasing them once the reaction is complete. In particular, when substrate binding occurs, enzymes undergo a conformational shift that orients or strains the substrates so that they are more reactive (Figure 3).
The name of an enzyme usually refers to the type of biochemical reaction it catalyzes. For example, proteases break down proteins, and dehydrogenases oxidize a substrate by removing hydrogen atoms. As a general rule, the "-ase" suffix identifies a protein as an enzyme, whereas the first part of an enzyme's name refers to the reaction that it catalyzes.
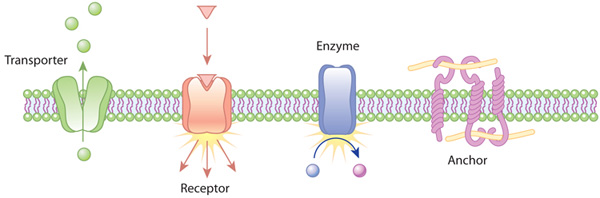
What Do Proteins Do in the Plasma Membrane?
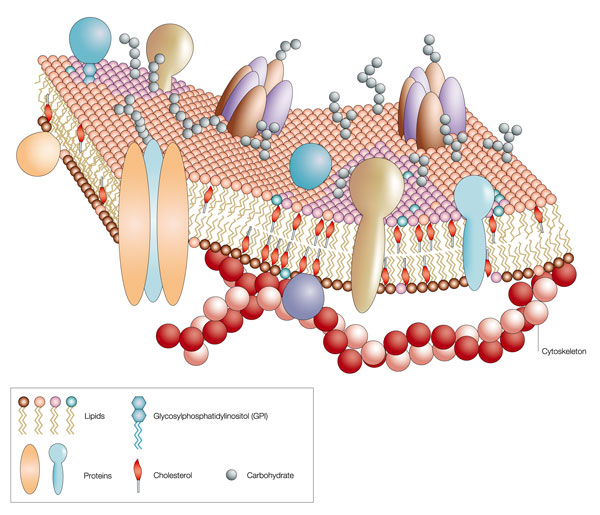
The proteins in the plasma membrane typically help the cell interact with its environment. For example, plasma membrane proteins carry out functions as diverse as ferrying nutrients across the plasma membrane, receiving chemical signals from outside the cell, translating chemical signals into intracellular action, and sometimes anchoring the cell in a particular location (Figure 4).
The overall surfaces of membrane proteins are mosaics, with patches of hydrophobic amino acids where the proteins contact lipids in the membrane bilayer and patches of hydrophilic amino acids on the surfaces that extend into the water-based cytoplasm. Many proteins can move within the plasma membrane through a process called membrane diffusion. This concept of membrane-bound proteins that can travel within the membrane is called the fluid-mosaic model of the cell membrane. The portions of membrane proteins that extend beyond the lipid bilayer into the extracellular environment are also hydrophilic and are frequently modified by the addition of sugar molecules. Other proteins are associated with the membrane but not inserted into it. They are sometimes anchored to lipids in the membrane or bound to other membrane proteins (Figure 5).
Conclusion
eBooks
This page appears in the following eBook