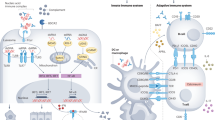
Abstract
Lupus nephritis is a complication of systemic lupus erythematosus, a heterogeneous autoimmune syndrome involving multiple pathways. Accumulating data from the fields of genetics, clinical science, transcriptomics and basic immunology indicate that antiviral immunity has relevance in the pathogenesis of lupus nephritis. This idea is based on the existence of genetic variants that promote the persistence of nuclear particles in the extracellular space or inside lysosomes. Such nuclear particles mimic viral particles and their RNA or DNA components activate viral nucleic acid recognition receptors in antigen-presenting cells. These autoadjuvant effects of endogenous nucleic acids promote an inappropriate immune interpretation of the nuclear particles during antigen presentation. This process fosters the expansion of autoreactive T cells and B cells, which promotes autoantibody production and immune complex glomerulonephritis. The release of interferon α sets off an antiviral immune response with a coordinated induction of hundreds of antiviral genes both inside and outside the kidney. In this article we summarize the available data indicating that innate immunity triggers antiviral immunity in systemic lupus erythematosus. We also discuss the related implications for innovative therapeutic strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Rahman, A. & Isenberg, D. A. Systemic lupus erythematosus. N. Engl. J. Med. 358, 929–939 (2008).
Goodnow, C. C. Multistep pathogenesis of autoimmune disease. Cell 130, 25–35 (2007).
Gaffney, P. M. et al. A genome-wide search for susceptibility genes in human systemic lupus erythematosus sib-pair families. Proc. Natl Acad. Sci. USA 95, 14875–14879 (1998).
Gregersen, P. K. & Olsson, L. M. Recent advances in the genetics of autoimmune disease. Annu. Rev. Immunol. 27, 363–391 (2009).
Kanta, H. & Mohan, C. Three checkpoints in lupus development: central tolerance in adaptive immunity, peripheral amplification by innate immunity and end-organ inflammation. Genes Immun. 10, 390–396 (2009).
Moser, K. L., Kelly, J. A., Lessard, C. J. & Harley, J. B. Recent insights into the genetic basis of systemic lupus erythematosus. Genes Immun. 10, 373–379 (2009).
Quintero-Del-Rio, A. I., Kelly, J. A., Kilpatrick, J., James, J. A. & Harley, J. B. The genetics of systemic lupus erythematosus stratified by renal disease: linkage at 10q22.3 (SLEN1), 2q34–35 (SLEN2), and 11p15.6 (SLEN3). Genes Immun. 3 (Suppl. 1), S57–S62 (2002).
Hom, G. et al. Association of systemic lupus erythematosus with C8orf13-BLK and ITGAM-ITGAX. N. Engl. J. Med. 358, 900–909 (2008).
Münz, C., Lünemann, J. D., Getts, M. T. & Miller, S. D. Antiviral immune responses: triggers of or triggered by autoimmunity? Nat. Rev. Immunol. 9, 246–258 (2009).
Kojima, M., Motoori, T., Asano, S. & Nakamura, S. Histological diversity of reactive and atypical proliferative lymph node lesions in systemic lupus erythematosus patients. Pathol. Res. Pract. 203, 423–431 (2007).
McClain, M. T. et al. Early events in lupus humoral autoimmunity suggest initiation through molecular mimicry. Nat. Med. 11, 85–89 (2005).
Barzilai, O. et al. Epstein-Barr virus and cytomegalovirus in autoimmune diseases: are they truly notorious? A preliminary report. Ann. NY Acad. Sci. 1108, 567–577 (2007).
Zandman-Goddard, G. et al. Exposure to Epstein-Barr virus infection is associated with mild systemic lupus erythematosus disease. Ann. NY Acad. Sci. 1173, 658–663 (2009).
Marshak-Rothstein, A. & Rifkin, I. R. Immunologically active autoantigens: the role of toll-like receptors in the development of chronic inflammatory disease. Annu. Rev. Immunol. 25, 419–441 (2007).
Anders, H. J., Krug, A. & Pawar, R. D. Molecular mimicry in innate immunity? The viral RNA recognition receptor TLR7 accelerates murine lupus. Eur. J. Immunol. 38, 1795–1799 (2008).
Rönnblom, L., Alm, G. V. & Eloranta, M. L. Type I interferon and lupus. Curr. Opin. Rheumatol. 21, 471–477 (2009).
Poole, B. D. et al. Aberrant Epstein-Barr viral infection in systemic lupus erythematosus. Autoimmun. Rev. 8, 337–342 (2009).
Rekvig, O. P., Bendiksen, S. & Moens, U. Immunity and autoimmunity induced by polyomaviruses: clinical, experimental and theoretical aspects. Adv. Exp. Med. Biol. 577, 117–147 (2006).
Feehally, J. et al. HLA has strongest association with IgA nephropathy in genome-wide analysis. J. Am. Soc. Nephrol. 21, 1791–1797 (2010).
Stanescu, H. C. et al. Risk HLA-DQA1 and PLA2R1 alleles in idiopathic membranous nephropathy. N. Engl. J. Med. 364, 616–626 (2011).
Smith, K. G. & Clatworthy, M. R. FcγRIIB in autoimmunity and infection: evolutionary and therapeutic implications. Nat. Rev. Immunol. 10, 328–343 (2010).
Hotchkiss, R. S., Strasser, A., McDunn, J. E. & Swanson, P. E. Cell death. N. Engl. J. Med. 361, 1570–1583 (2009).
Strasser, A., Jost, P. J. & Nagata, S. The many roles of FAS receptor signaling in the immune system. Immunity 30, 180–192 (2009).
Wu, J. et al. Fas ligand mutation in a patient with systemic lupus erythematosus and lymphoproliferative disease. J. Clin. Invest. 98, 1107–1113 (1996).
Bygrave, A. E. et al. Spontaneous autoimmunity in 129 and C57BL/6 mice-implications for autoimmunity described in gene-targeted mice. PLoS Biol. 2, E243 (2004).
Russell, A. I. et al. Polymorphism at the C-reactive protein locus influences gene expression and predisposes to systemic lupus erythematosus. Hum. Mol. Genet. 13, 137–147 (2004).
Truedsson, L., Bengtsson, A. A. & Sturfelt, G. Complement deficiencies and systemic lupus erythematosus. Autoimmunity 40, 560–566 (2007).
Muñoz, L. E. et al. Autoimmunity and chronic inflammation–two clearance-related steps in the etiopathogenesis of SLE. Autoimmun. Rev. 10, 38–42 (2010).
Hanayama, R. et al. Autoimmune disease and impaired uptake of apoptotic cells in MFG-E8-deficient mice. Science 304, 1147–1150 (2004).
Mortensen, E. S. & Rekvig, O. P. Nephritogenic potential of anti-DNA antibodies against necrotic nucleosomes. J. Am. Soc. Nephrol. 20, 696–704 (2009).
Yasutomo, K. et al. Mutation of DNASE1 in people with systemic lupus erythematosus. Nat. Genet. 28, 313–314 (2001).
Muñoz, L. E., Lauber, K., Schiller, M., Manfredi, A. A. & Herrmann, M. The role of defective clearance of apoptotic cells in systemic autoimmunity. Nat. Rev. Rheumatol. 6, 280–289 (2010).
Anders, H. J. Innate pathogen recognition in the kidney: toll-like receptors, NOD-like receptors, and RIG-like helicases. Kidney Int. 72, 1051–1056 (2007).
Takeuchi, O. & Akira, S. Pattern recognition receptors and inflammation. Cell 140, 805–820 (2010).
Hornung, V. et al. 5′-Triphosphate RNA is the ligand for RIG-I. Science 314, 994–997 (2006).
Pichlmair, A. et al. RIG-I-mediated antiviral responses to single-stranded RNA bearing 5′-phosphates. Science 314, 997–1001 (2006).
Stacey, K. J. et al. The molecular basis for the lack of immunostimulatory activity of vertebrate DNA. J. Immunol. 170, 3614–3620 (2003).
Karikó, K., Buckstein, M., Ni, H. & Weissman, D. Suppression of RNA recognition by Toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA. Immunity 23, 165–175 (2005).
Huck, S., Deveaud, E., Namane, A. & Zouali, M. Abnormal DNA methylation and deoxycytosine-deoxyguanine content in nucleosomes from lymphocytes undergoing apoptosis. FASEB J. 13, 1415–1422 (1999).
Hof, D., Raats, J. M. & Pruijn, G. J. Apoptotic modifications affect the autoreactivity of the U1 snRNP autoantigen. Autoimmun. Rev. 4, 380–388 (2005).
Richardson, B. et al. Evidence for impaired T cell DNA methylation in systemic lupus erythematosus and rheumatoid arthritis. Arthritis Rheum. 33, 1665–1673 (1990).
Cornacchia, E. et al. Hydralazine and procainamide inhibit T cell DNA methylation and induce autoreactivity. J. Immunol. 140, 2197–2200 (1988).
Takeuchi, O. & Akira, S. Innate immunity to virus infection. Immunol. Rev. 227, 75–86 (2009).
Anders, H. J. Pseudoviral immunity–a novel concept for lupus. Trends Mol. Med. 15, 553–561 (2009).
Savarese, E. et al. U1 small nuclear ribonucleoprotein immune complexes induce type I interferon in plasmacytoid dendritic cells through TLR7. Blood 107, 3229–3234 (2006).
Lau, C. M. et al. RNA-associated autoantigens activate B cells by combined B cell antigen receptor/Toll-like receptor 7 engagement. J. Exp. Med. 202, 1171–1177 (2005).
Barrat, F. J. et al. Nucleic acids of mammalian origin can act as endogenous ligands for Toll-like receptors and may promote systemic lupus erythematosus. J. Exp. Med. 202, 1131–1139 (2005).
Pisitkun, P. et al. Autoreactive B cell responses to RNA-related antigens due to TLR7 gene duplication. Science 312, 1669–1672 (2006).
Subramanian, S. et al. A Tlr7 translocation accelerates systemic autoimmunity in murine lupus. Proc. Natl Acad. Sci. USA 103, 9970–9975 (2006).
Christensen, S. R. et al. Toll-like receptor 7 and TLR9 dictate autoantibody specificity and have opposing inflammatory and regulatory roles in a murine model of lupus. Immunity 25, 417–428 (2006).
Savarese, E. et al. Requirement of Toll-like receptor 7 for pristane-induced production of autoantibodies and development of murine lupus nephritis. Arthritis Rheum. 58, 1107–1115 (2008).
Leadbetter, E. A. et al. Chromatin-IgG complexes activate B cells by dual engagement of IgM and Toll-like receptors. Nature 416, 603–607 (2002).
Means, T. K. et al. Human lupus autoantibody-DNA complexes activate DCs through cooperation of CD32 and TLR9. J. Clin. Invest. 115, 407–417 (2005).
Lartigue, A. et al. Role of TLR9 in anti-nucleosome and anti-DNA antibody production in lpr mutation-induced murine lupus. J. Immunol. 177, 1349–1354 (2006).
Wu, X. & Peng, S. L. Toll-like receptor 9 signaling protects against murine lupus. Arthritis Rheum. 54, 336–342 (2006).
Lenert, P. et al. DNA-like class R inhibitory oligonucleotides (INH-ODNs) preferentially block autoantigen-induced B-cell and dendritic cell activation in vitro and autoantibody production in lupus-prone MRL-Faslpr/lpr mice in vivo. Arthritis Res. Ther. 11, R79 (2009).
Patole, P. S. et al. G-rich DNA suppresses systemic lupus. J. Am. Soc. Nephrol. 16, 3273–3280 (2005).
Dong, L., Ito, S., Ishii, K. J. & Klinman, D. M. Suppressive oligodeoxynucleotides delay the onset of glomerulonephritis and prolong survival in lupus-prone NZB × NZW mice. Arthritis Rheum. 52, 651–658 (2005).
Wang, J. et al. The functional effects of physical interactions among Toll-like receptors 7, 8, and 9. J. Biol. Chem. 281, 37427–37434 (2006).
Lech, M. et al. Different roles of TiR8/Sigirr on toll-like receptor signaling in intrarenal antigen-presenting cells and tubular epithelial cells. Kidney Int. 72, 182–192 (2007).
Lech, M. et al. Interleukin-1 receptor-associated kinase-M suppresses systemic lupus erythematosus. Ann. Rheum. Dis. 70, 2207–2212 (2011).
Gilliet, M., Cao, W. & Liu, Y. J. Plasmacytoid dendritic cells: sensing nucleic acids in viral infection and autoimmune diseases. Nat. Rev. Immunol. 8, 594–606 (2008).
Allam, R., Sayyed, S. G., Kulkarni, O., Lichtnekert, J. & Anders, H. J. Mdm2 promotes systemic lupus erythematosus and lupus nephritis. J. Am. Soc. Nephrol. 22, 2016–2027 (2011).
Theofilopoulos, A. N., Baccala, R., Beutler, B. & Kono, D. H. Type I interferons (α/β) in immunity and autoimmunity. Annu. Rev. Immunol. 23, 307–336 (2005).
Han, G. M. et al. Analysis of gene expression profiles in human systemic lupus erythematosus using oligonucleotide microarray. Genes Immun. 4, 177–186 (2003).
Bekeredjian-Ding, I. B. et al. Plasmacytoid dendritic cells control TLR7 sensitivity of naive B cells via type I IFN. J. Immunol. 174, 4043–4050 (2005).
Nacionales, D. C. et al. Deficiency of the type I interferon receptor protects mice from experimental lupus. Arthritis Rheum. 56, 3770–3783 (2007).
Santiago-Raber, M. L. et al. Type-I interferon receptor deficiency reduces lupus-like disease in NZB mice. J. Exp. Med. 197, 777–788 (2003).
Braun, D., Geraldes, P. & Demengeot, J. Type I interferon controls the onset and severity of autoimmune manifestations in lpr mice. J. Autoimmun. 20, 15–25 (2003).
Espeli, M. et al. Local renal autoantibody production in lupus nephritis. J. Am. Soc. Nephrol. 22, 296–305 (2011).
Lech, M. et al. IRF4 deficiency abrogates lupus nephritis despite enhancing systemic cytokine production. J. Am. Soc. Nephrol. 22, 1443–1452 (2011).
Enocsson, H. et al. Interferon-α mediates suppression of C-reactive protein: explanation for muted C-reactive protein response in lupus flares? Arthritis Rheum. 60, 3755–3760 (2009).
Becker, G. J., Waldburger, M., Hughes, G. R. & Pepys, M. B. Value of serum C-reactive protein measurement in the investigation of fever in systemic lupus erythematosus. Ann. Rheum. Dis. 39, 50–52 (1980).
Gabay, C. et al. Absence of correlation between interleukin 6 and C-reactive protein blood levels in systemic lupus erythematosus compared with rheumatoid arthritis. J. Rheumatol. 20, 815–821 (1993).
Patole, P. S. et al. Expression and regulation of Toll-like receptors in lupus-like immune complex glomerulonephritis of MRL-Fas(lpr) mice. Nephrol. Dial. Transplant. 21, 3062–3073 (2006).
Patole, P. S. et al. Coactivation of Toll-like receptor-3 and -7 in immune complex glomerulonephritis. J. Autoimmun. 29, 52–59 (2007).
Pawar, R. D. et al. Ligands to nucleic acid-specific toll-like receptors and the onset of lupus nephritis. J. Am. Soc. Nephrol. 17, 3365–3373 (2006).
Patole, P. S. et al. Viral double-stranded RNA aggravates lupus nephritis through Toll-like receptor 3 on glomerular mesangial cells and antigen-presenting cells. J. Am. Soc. Nephrol. 16, 1326–1338 (2005).
Allam, R. & Anders, H. J. The role of innate immunity in autoimmune tissue injury. Curr. Opin. Rheumatol. 20, 538–544 (2008).
Fluer, K. et al. Viral RNA induces type I interferon-dependent cytokine release and cell death in mesangial cells via MDA5. Implications for viral infection-associated glomerulonephritis. Am. J. Pathol. 175, 2014–2022 (2009).
Allam, R. et al. Viral RNA and DNA sense common antiviral responses including type I interferons in mesangial cells. J. Am. Soc. Nephrol. 20, 1986–1996 (2009).
Hägele, H., Allam, R., Pawar, R. D. & Anders, H. J. Double-stranded RNA activates type I interferon secretion in glomerular endothelial cells via retinoic acid-inducible gene (RIG)-1. Nephrol. Dial. Transplant. 24, 3312–3318 (2009).
Hägele, H. et al. Double-stranded DNA activates glomerular endothelial cells and enhances albumin permeability via a toll-like receptor-independent cytosolic DNA recognition pathway. Am. J. Pathol. 175, 1896–1904 (2009).
Peterson, K. S. et al. Characterization of heterogeneity in the molecular pathogenesis of lupus nephritis from transcriptional profiles of laser-captured glomeruli. J. Clin. Invest. 113, 1722–1733 (2004).
Rozzo, S. J. et al. Evidence for an interferon-inducible gene, Ifi202, in the susceptibility to systemic lupus. Immunity 15, 435–443 (2001).
Grimley, P. M. et al. Tubuloreticular inclusions in peripheral blood mononuclear cells related to systemic therapy with alpha-interferon. Lab. Invest. 52, 638–649 (1985).
Strauss, J. et al. Renal disease in children with the acquired immunodeficiency syndrome. N. Engl. J. Med. 321, 625–630 (1989).
Rich, S. A. Human lupus inclusions and interferon. Science 213, 772–775 (1981).
Anders, H. J., Lichtnekert, J. & Allam, R. Interferon-α and -β in kidney inflammation. Kidney Int. 77, 848–854 (2010).
Yang, A. H. et al. The clinicopathological implications of endothelial tubuloreticular inclusions found in glomeruli having histopathology of idiopathic membranous nephropathy. Nephrol. Dial. Transplant. 24, 3419–3425 (2009).
Lafyatis, R., York, M. & Marshak-Rothstein, A. Antimalarial agents: closing the gate on Toll-like receptors? Arthritis Rheum. 54, 3068–3070 (2006).
Pons-Estel, G. J. et al. Protective effect of hydroxychloroquine on renal damage in patients with lupus nephritis: LXV, data from a multiethnic US cohort. Arthritis Rheum. 61, 830–839 (2009).
Guiducci, C. et al. TLR recognition of self nucleic acids hampers glucocorticoid activity in lupus. Nature 465, 937–941 (2010).
Pawar, R. D. et al. Inhibition of Toll-like receptor-7 (TLR-7) or TLR-7 plus TLR-9 attenuates glomerulonephritis and lung injury in experimental lupus. J. Am. Soc. Nephrol. 18, 1721–1731 (2007).
Barrat, F. J., Meeker, T., Chan, J. H., Guiducci, C. & Coffman, R. L. Treatment of lupus-prone mice with a dual inhibitor of TLR7 and TLR9 leads to reduction of autoantibody production and amelioration of disease symptoms. Eur. J. Immunol. 37, 3582–3586 (2007).
Yao, Y. et al. Neutralization of interferon-α/β-inducible genes and downstream effect in a phase I trial of an anti-interferon-α monoclonal antibody in systemic lupus erythematosus. Arthritis Rheum. 60, 1785–1796 (2009).
Acknowledgements
A. Migliorini is a graduate of the Deutsche Forschungsgemeinschaft Graduate College 1202. H.-J. Anders is supported by the Deutsche Forschungsgemeinschaft via AN372/9-2, 10-1, and 11-1.
Author information
Authors and Affiliations
Contributions
A. Migliorini and H.-J. Anders contributed equally to all aspects of this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Migliorini, A., Anders, HJ. A novel pathogenetic concept—antiviral immunity in lupus nephritis. Nat Rev Nephrol 8, 183–189 (2012). https://doi.org/10.1038/nrneph.2011.197
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2011.197
This article is cited by
-
Characterization of virus-mediated autoimmunity and the consequences for pathological process in patients with systemic lupus erythematosus
Clinical Rheumatology (2023)
-
Lupus nephritis
Nature Reviews Disease Primers (2020)
-
Toll-like receptors in lupus nephritis
Journal of Biomedical Science (2018)
-
The role of extracellular histone in organ injury
Cell Death & Disease (2017)
-
Links between coagulation, inflammation, regeneration, and fibrosis in kidney pathology
Laboratory Investigation (2016)