Abstract
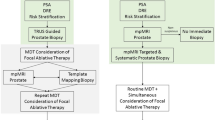
Treatment of prostate cancer using high-intensity focused ultrasound (HIFU) focal therapy will become a reliable treatment option only if several conditions are fulfilled. These conditions concern patient selection, assessment of the tumour location and aggressiveness, evaluation of target tissue destruction, and detection of local recurrence or appearance of new tumours. Regarding patient selection, standard transrectal biopsies are not accurate enough and, although perineal template biopsies can detect tumours, they are invasive, expensive procedures, and there is a risk of incidental detection of insignificant cancers. In turn, multiparametric MRI is accurate for detecting and localizing high-grade (Gleason score ≥7) cancers and may provide non-invasive assessment of tumour aggressiveness. Moreover, contrast-enhanced imaging—ultrasonography or MRI—can assess post-HIFU tissue destruction and provide accurate detection of tumour recurrence, which is a key element for follow up. This Perspectives article will assess whether our current methods for cancer diagnosis, tissue targeting, and treatment follow up are accurate enough to allow the design of robust HIFU focal therapy protocols.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lindner, U., Trachtenberg, J. & Lawrentschuk, N. Focal therapy in prostate cancer: modalities, findings and future considerations. Nat. Rev. Urol. 7, 562–571 (2010).
Lawrentschuk, N. & Klotz, L. Active surveillance for low-risk prostate cancer: an update. Nat. Rev. Urol. 8, 312–320 (2011).
Suardi, N. et al. Currently used criteria for active surveillance in men with low-risk prostate cancer: an analysis of pathologic features. Cancer 113, 2068–2072 (2008).
Ahmed, H. U. et al. Focal therapy for localised unifocal and multifocal prostate cancer: a prospective development study. Lancet Oncol. 13, 622–632 (2012).
Crouzet, S. et al. Multicentric oncologic outcomes of high-intensity focused ultrasound for localized prostate cancer in 803 patients. Eur. Urol. 58, 559–566 (2010).
Murat, F. J. et al. Mid-term results demonstrate salvage high-intensity focused ultrasound (HIFU) as an effective and acceptably morbid salvage treatment option for locally radiorecurrent prostate cancer. Eur. Urol. 55, 640–647 (2009).
Ahmed, H. et al. Focal salvage therapy for localized prostate cancer recurrence after external beam radiotherapy. Cancer http://dx.doi.org/10.1002/cncr.27394
Rouvière, O. et al. Transrectal high-intensity focused ultrasound ablation of prostate cancer: effective treatment requiring accurate imaging. Eur. J. Radiol. 63, 317–327 (2007).
Salomir, R. et al. Image-based control of the magnetic resonance imaging-guided focused ultrasound thermotherapy. Top. Magn. Reson. Imaging 17, 139–151 (2006).
Marberger, M. et al. Novel approaches to improve prostate cancer diagnosis and management in early-stage disease. BJU Int. 109 (Suppl. 2), 1–7 (2012).
Scattoni, V. et al. Is extended and saturation biopsy necessary? Int. J. Urol. 17, 432–447 (2010).
Shariat, S. F. & Roehrborn, C. G. Using biopsy to detect prostate cancer. Rev. Urol. 10, 262–280 (2008).
Igel, T. C. et al. Systematic transperineal ultrasound guided template biopsy of the prostate in patients at high risk. J. Urol. 165, 1575–1579 (2001).
Sartor, A. O. et al. Evaluating localized prostate cancer and identifying candidates for focal therapy. Urology 72, S12–S24 (2008).
Dominguez-Escrig, J. L., McCracken, S. R. & Greene, D. Beyond diagnosis: evolving prostate biopsy in the era of focal therapy. Prostate Cancer 2011, 386207 (2011).
Mian, B. M. et al. Predictors of cancer in repeat extended multisite prostate biopsy in men with previous negative extended multisite biopsy. Urology 60, 836–840 (2002).
Taneja, S. S. & Mason, M. Candidate selection for prostate cancer focal therapy. J. Endourol. 24, 835–841 (2010).
Scales, C. D. Jr et al. Predicting unilateral prostate cancer based on biopsy features: implications for focal ablative therapy--results from the SEARCH database. J. Urol. 178, 1249–1252 (2007).
Tsivian, M. et al. Predicting unilateral prostate cancer on routine diagnostic biopsy: sextant vs extended. BJU Int. 105, 1089–1092 (2010).
Tareen, B. et al. Can contemporary transrectal prostate biopsy accurately select candidates for hemi-ablative focal therapy of prostate cancer? BJU Int. 104, 195–199 (2009).
Walz, J. et al. High incidence of prostate cancer detected by saturation biopsy after previous negative biopsy series. Eur. Urol. 50, 498–505 (2006).
Keetch, D. W., Catalona, W. J. & Smith, D. S. Serial prostatic biopsies in men with persistently elevated serum prostate specific antigen values. J. Urol. 151, 1571–1574 (1994).
Tsivian, M., Hruza, M., Mouraviev, V., Rassweiler, J. & Polascik, T. J. Prostate biopsy in selecting candidates for hemiablative focal therapy. J. Endourol. 24, 849–853 (2010).
Ashley, R. A. et al. Reassessing the diagnostic yield of saturation biopsy of the prostate. Eur. Urol. 53, 976–981 (2008).
Kawakami, S. et al. Direct comparison between transrectal and transperineal extended prostate biopsy for the detection of cancer. Int. J. Urol. 14, 719–724 (2007).
Falzarano, S. M. et al. Can saturation biopsy predict prostate cancer localization in radical prostatectomy specimens: a correlative study and implications for focal therapy. Urology 76, 682–687 (2010).
Abdollah, F. et al. The role of transrectal saturation biopsy in tumour localization: pathological correlation after retropubic radical prostatectomy and implication for focal ablative therapy. BJU Int. 108, 366–371 (2011).
Merrick, G. S. et al. Prostate cancer distribution in patients diagnosed by transperineal template-guided saturation biopsy. Eur. Urol. 52, 715–723 (2007).
Taira, A. V. et al. Performance of transperineal template-guided mapping biopsy in detecting prostate cancer in the initial and repeat biopsy setting. Prostate Cancer Prostatic Dis. 13, 71–77 (2010).
Zhan, Y. et al. Targeted prostate biopsy using statistical image analysis. IEEE Trans. Med. Imaging 26, 779–788 (2007).
Narayanan, R. et al. Adaptation of a 3D prostate cancer atlas for transrectal ultrasound guided target-specific biopsy. Phys. Med. Biol. 53, 397–406 (2008).
Ou, Y. et al. Sampling the spatial patterns of cancer: optimized biopsy procedures for estimating prostate cancer volume and Gleason score. Med. Image Anal. 13, 609–620 (2009).
Megwalu, I. I. et al. Evaluation of a novel precision template-guided biopsy system for detecting prostate cancer. BJU Int. 102, 546–550 (2008).
Epstein, J. I., Sanderson, H., Carter, H. B. & Scharfstein, D. O. Utility of saturation biopsy to predict insignificant cancer at radical prostatectomy. Urology 66, 356–360 (2005).
Ahmed, H. U. et al. Characterizing clinically significant prostate cancer using template prostate mapping biopsy. J. Urol. 186, 458–464 (2011).
Smeenge, M. et al. Role of transrectal ultrasonography (TRUS) in focal therapy of prostate cancer: report from a Consensus Panel. BJU Int. http://dx.doi.org/10.1111/j.1464-410X.2012.11072.x.
Smeenge, M., Mischi, M., Laguna Pes, M. P., de la Rosette, J. J. & Wijkstra, H. Novel contrast-enhanced ultrasound imaging in prostate cancer. World J. Urol. 29, 581–587 (2011).
Walz, J. et al. Identification of the prostate cancer index lesion by real-time elastography: considerations for focal therapy of prostate cancer. World J. Urol. 29, 589–594 (2011).
Simmons, L. A. et al. Detection, localisation and characterisation of prostate cancer by Prostate HistoScanning™. BJU Int. 110, 28–35 (2012).
Smeenge, M. et al. Role of transrectal ultrasound in focal therapy of prostate cancer: report from a consensus panel. BJU Int. http://dx.doi.org/10.1111/j.1464-410X.2012.11072.x.
de la Rosette, J. et al. Focal therapy in prostate cancer-report from a consensus panel. J. Endourol. 24, 775–780 (2010).
Girouin, N. et al. Prostate dynamic contrast-enhanced MRI with simple visual diagnostic criteria: is it reasonable? Eur. Radiol. 17, 1498–1509 (2007).
Fütterer, J. J. et al. Prostate cancer localization with dynamic contrast-enhanced MR imaging and proton MR spectroscopic imaging. Radiology 241, 449–458 (2006).
Tan, C. H., Wang, J. & Kundra, V. Diffusion weighted imaging in prostate cancer. Eur. Radiol. 21, 593–603 (2011).
Katahira, K. et al. Ultra-high-b-value diffusion-weighted MR imaging for the detection of prostate cancer: evaluation in 201 cases with histopathological correlation. Eur. Radiol. 21, 188–196 (2011).
Reinsberg, S. A. et al. Combined use of diffusion-weighted MRI and 1H MR spectroscopy to increase accuracy in prostate cancer detection. AJR Am. J. Roentgenol. 188, 91–98 (2007).
Ahmed, H. U. et al. Is it time to consider a role for MRI before prostate biopsy? Nat. Rev. Clin. Oncol. 6, 197–206 (2009).
Sciarra, A. et al. Advances in magnetic resonance imaging: how they are changing the management of prostate cancer. Eur. Urol. 59, 962–977 (2011).
Akin, O. et al. Transition zone prostate cancers: features, detection, localization, and staging at endorectal MR imaging. Radiology 239, 784–792 (2006).
Oto, A. et al. Prostate cancer: differentiation of central gland cancer from benign prostatic hyperplasia by using diffusion-weighted and dynamic contrast-enhanced MR imaging. Radiology 257, 715–723 (2010).
Lemaitre, L. et al. Dynamic contrast-enhanced MRI of anterior prostate cancer: morphometric assessment and correlation with radical prostatectomy findings. Eur. Radiol. 19, 470–480 (2009).
Villers, A. et al. Dynamic contrast enhanced, pelvic phased array magnetic resonance imaging of localized prostate cancer for predicting tumor volume: correlation with radical prostatectomy findings. J. Urol. 176, 2432–2437 (2006).
Turkbey, B. et al. Prostate cancer: value of multiparametric MR imaging at 3 T for detection--histopathologic correlation. Radiology 255, 89–99 (2010).
Wang, L. et al. Assessment of biologic aggressiveness of prostate cancer: correlation of MR signal intensity with Gleason grade after radical prostatectomy. Radiology 246, 168–176 (2008).
Woodfield, C. A. et al. Diffusion-weighted MRI of peripheral zone prostate cancer: comparison of tumor apparent diffusion coefficient with Gleason score and percentage of tumor on core biopsy. AJR Am. J. Roentgenol. 194, W316–W322 (2010).
Verma, S. et al. Assessment of aggressiveness of prostate cancer: correlation of apparent diffusion coefficient with histologic grade after radical prostatectomy. AJR Am. J. Roentgenol. 196, 374–381 (2011).
Oto, A. et al. Diffusion-weighted and dynamic contrast-enhanced MRI of prostate cancer: correlation of quantitative MR parameters with Gleason score and tumor angiogenesis. AJR Am. J. Roentgenol. 197, 1382–1390 (2011).
Hambrock, T. et al. Relationship between apparent diffusion coefficients at 3.0-T MR imaging and Gleason grade in peripheral zone prostate cancer. Radiology 259, 453–461 (2011).
Giles, S. L. et al. Apparent diffusion coefficient as a predictive biomarker of prostate cancer progression: value of fast and slow diffusion components. AJR Am. J. Roentgenol. 196, 586–591 (2011).
Zakian, K. L. et al. Correlation of proton MR spectroscopic imaging with gleason score based on step-section pathologic analysis after radical prostatectomy. Radiology 234, 804–814 (2005).
Franiel, T., Hamm, B. & Hricak, H. Dynamic contrast-enhanced magnetic resonance imaging and pharmacokinetic models in prostate cancer. Eur. Radiol. 21, 616–626 (2011).
Rouvière, O. et al. Characterization of time-enhancement curves of benign and malignant prostate tissue at dynamic MR imaging. Eur. Radiol. 13, 931–942 (2003).
Hambrock, T. et al. Prospective assessment of prostate cancer aggressiveness using 3-T diffusion-weighted magnetic resonance imaging-guided biopsies versus a systematic 10-core transrectal ultrasound prostate biopsy cohort. Eur. Urol. 61, 177–184 (2012).
Rouvière, O. et al. Recurrent prostate cancer after external beam radiotherapy: value of contrast-enhanced dynamic MRI in localizing intraprostatic tumor--correlation with biopsy findings. Urology 63, 922–927 (2004).
Rouviere, O., Vitry, T. & Lyonnet, D. Imaging of prostate cancer local recurrences: why and how? Eur. Radiol. 20, 1254–1266 (2010).
Haider, M. A. et al. Dynamic contrast-enhanced magnetic resonance imaging for localization of recurrent prostate cancer after external beam radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 70, 425–430 (2008).
Rouvière, O. et al. Is it possible to model the risk of malignancy of focal abnormalities found at prostate multiparametric MRI? Eur. Radiol. 22, 1149–1157 (2012).
Heidenreich, A. Consensus criteria for the use of magnetic resonance imaging in the diagnosis and staging of prostate cancer: not ready for routine use. Eur. Urol. 59, 495–497 (2011).
Dickinson, L. et al. Magnetic resonance imaging for the detection, localisation, and characterisation of prostate cancer: recommendations from a European consensus meeting. Eur. Urol. 59, 477–494 (2011).
Barentsz, J. O. et al. ESUR prostate MR guidelines 2012. Eur. Radiol. 22, 746–757 (2012).
Niaf, E., Rouvière, O., Mege-Lechevallier, F., Bratan, F. & Lartizien, C. Computer-aided diagnosis of prostate cancer in the peripheral zone using multiparametric MRI. Phys. Med. Biol. 57, 3833–3851 (2012).
Artan, Y. et al. Prostate cancer localization with multispectral MRI using cost-sensitive support vector machines and conditional random fields. IEEE Trans. Image Process. 19, 2444–2455 (2010).
Puech, P. et al. Computer-assisted diagnosis of prostate cancer using DCE-MRI data: implementation and preliminary results. Int. J. Comput. Assist. Radiol. Surg. 4, 1–10 (2009).
Jager, G. J. et al. Local staging of prostate cancer with endorectal MR imaging: correlation with histopathology. AJR Am. J. Roentgenol. 166, 845–852 (1996).
Langer, D. L. et al. Intermixed normal tissue within prostate cancer: effect on MR imaging measurements of apparent diffusion coefficient and T2--sparse versus dense cancers. Radiology 249, 900–908 (2008).
Rosenkrantz, A. B., Mendrinos, S., Babb, J. S. & Taneja, S. S. Prostate cancer foci detected on multiparametric magnetic resonance imaging are histologically distinct from those not detected. J. Urol. 187, 2032–2038 (2012).
Onik, G., Miessau, M. & Bostwick, D. G. Three-dimensional prostate mapping biopsy has a potentially significant impact on prostate cancer management. J. Clin. Oncol. 27, 4321–4326 (2009).
Ukimura, O. et al. 3-Dimensional elastic registration system of prostate biopsy location by real-time 3-dimensional transrectal ultrasound guidance with magnetic resonance/transrectal ultrasound image fusion. J. Urol. 187, 1080–1086 (2012).
Pinto, P. A. et al. Magnetic resonance imaging/ultrasound fusion guided prostate biopsy improves cancer detection following transrectal ultrasound biopsy and correlates with multiparametric magnetic resonance imaging. J. Urol. 186, 1281–1285 (2011).
Rouvière, O. et al. MRI appearance of prostate following transrectal HIFU ablation of localized cancer. Eur. Urol. 40, 265–274 (2001).
Rouvière, O. et al. Prostate cancer ablation with transrectal high-intensity focused ultrasound: assessment of tissue destruction with contrast-enhanced US. Radiology 259, 583–591 (2011).
Rouvière, O. et al. Prostate cancer transrectal HIFU ablation: detection of local recurrences using T2-weighted and dynamic contrast-enhanced MRI. Eur. Radiol. 20, 48–55 (2010).
Riviere, J. et al. Salvage radiotherapy after high-intensity focussed ultrasound for recurrent localised prostate cancer. Eur. Urol. 58, 567–573 (2010).
Stolzenburg, J. U. et al. Salvage laparoscopic extraperitoneal radical prostatectomy after failed high-intensity focused ultrasound and radiotherapy for localized prostate cancer. Urology 70, 956–960 (2007).
Siddiqui, K. et al. MRI-guided transurethral ultrasound therapy of the prostate gland using real-time thermal mapping: initial studies. Urology 76, 1506–1511 (2010).
Acknowledgements
The authors thank Andrew Fowler for editing the manuscript.
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article and contributed to the discussion of the content. O. Rouvière contributed to the writing and editing the manuscript before submission. A. Gelet also contributed to the writing and J. Y. Chapelon contributed to editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
A. Gelet declares he is a consultant who also receives grant support from EDAP TMS. The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Rouvière, O., Gelet, A., Crouzet, S. et al. Prostate focused ultrasound focal therapy—imaging for the future. Nat Rev Clin Oncol 9, 721–727 (2012). https://doi.org/10.1038/nrclinonc.2012.136
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2012.136
This article is cited by
-
Multimodal imaging in focal therapy planning and assessment in primary prostate cancer
Clinical and Translational Imaging (2017)
-
Tumor boundary detection in ultrasound imagery using multi-scale generalized gradient vector flow
Journal of Medical Ultrasonics (2015)
-
Influence of imaging and histological factors on prostate cancer detection and localisation on multiparametric MRI: a prospective study
European Radiology (2013)