Key Points
-
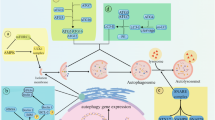
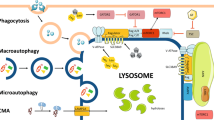
Lysosomes contain catabolic hydrolases that participate in the digestion of autophagic material (after fusion between lysosomes and autophagosomes), in acute cell death (after lysosomal-membrane permeabilization, LMP) or in tissue invasion (after their release into the extracellular space).
-
LMP can be induced by classic apoptotic stimuli, intracellular second messengers such as reactive oxygen species and sphingosine, as well as by lysosomotropic toxins. Often, LMP causes mitochondrial outer-membrane permeabilization and caspase activation, which are two of the hallmarks of apoptosis.
-
Lysosomal alterations are common in cancer cells in which increased expression of lysosomal enzymes participates in tissue invasion and tumour growth, yet overexpressed heat-shock protein 70 (HSP70) locally prevents LMP. Targeting of lysosomes by lysosomotropic toxins, inhibitors of the vacuolar H+-ATPase or downmodulation/inhibition of HSP70 might have cancer-selective cytotoxic effects.
-
The autophagic sequestration of parts of the cytosol and/or cytoplasmic organelles is involved in the cellular adaptation to nutrient deprivation and sub-lethal damage. Therefore, autophagy might prevent cell death under some conditions. However, in some instances, particularly when apoptosis is inhibited, high levels of autophagy can function as a cell death effector mechanism.
-
Defective autophagy might participate in carcinogenesis, possibly owing to a reduced removal of defective organelles and/or damaged cells. Some chemotherapeutic agents can induce morphological changes, which indicate that autophagic cell death is occurring in the treated cells, but there is no formal proof that this type of cell death is involved in endogenous or therapeutic tumour suppression.
-
Specific therapeutic strategies might exploit the structural and functional abnormalities that affect the lysosomal and autophagic compartments in cancer cells.
Abstract
Lysosomal hydrolases participate in the digestion of endocytosed and autophagocytosed material inside the lysosomal/autolysosomal compartment in acute cell death when released into the cytosol and in cancer progression following their release into the extracellular space. Lysosomal alterations are common in cancer cells. The increased expression and altered trafficking of lysosomal enzymes participates in tissue invasion, angiogenesis and sensitization to the lysosomal death pathway. But lysosomal heat-shock protein 70 locally prevents lysosomal-membrane permeabilization. Similarly, alterations in the autophagic compartment are linked to carcinogenesis and resistance to chemotherapy. Targeting these pathways might constitute a novel approach to cancer therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Green, D. R. & Kroemer, G. The pathophysiology of mitochondrial cell death. Science 305, 626–629 (2004).
Leist, M. & Jäättelä, M. Four deaths and a funeral: from caspases to alternative mechanisms. Nature Rev. Mol. Cell Biol. 2, 589–598 (2001). A thorough review describing different types of cell death.
Jäättelä, M. Multiple cell death pathways as regulators of tumour initiation and progression. Oncogene 23, 2746–2756 (2004).
Guicciardi, M. A., Leist, M. & Gores, G. J. Lysosomes in cell death. Oncogene 23, 2881–2890 (2004).
Levine, B. & Klionsky, D. J. Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev. Cell 6, 463–477 (2004).
Shintani, T. & Klionsky, D. J. Autophagy in health and disease: a double-edged sword. Science 306, 990–995 (2004). These two reviews provide a state-of-the-art introduction to certainties and uncertainties within the field of autophagy.
Savill, J. & Fadok, V. Corpse clearance defines the meaning of cell death. Nature 407, 784–788 (2000).
de Duve, C. Lysosomes revisited. Eur. J. Biochem. 137, 391–397 (1983).
Schotte, P., Declercq, W., Van Huffel, S., Vandenabeele, P. & Beyaert, R. Non-specific effects of methyl ketone peptide inhibitors of caspases. FEBS Lett. 442, 117–121 (1999).
Foghsgaard, L. et al. Cathepsin B acts as a dominant execution protease in tumor cell apoptosis induced by tumor necrosis factor. J. Cell Biol. 153, 999–1009 (2001). The first identification of cathepsin B as a specific mediator of tumour cell death.
Brunk, U. T., Neuzil, J. & Eaton, J. W. Lysosomal involvement in apoptosis. Redox Rep. 6, 91–97 (2001).
Brunk, U. T., Dalen, H., Roberg, K. & Hellquist, H. B. Photo-oxidative disruption of lysosomal membranes causes apoptosis of cultured human fibroblasts. Free Radic. Biol. Med. 23, 616–626 (1997).
Kågedal, K., Zhao, M., Svensson, I. & Brunk, U. T. Sphingosine-induced apoptosis is dependent on lysosomal proteases. Biochem. J. 359, 335–343 (2001).
Cirman, T. et al. Selective disruption of lysosomes in HeLa cells triggers apoptosis, mediated by cleavage of Bid by multiple papain-like lysosomal cathepsins. J. Biol. Chem. 279, 3578–3587 (2004).
Boya, P. et al. Lysosomal membrane permeabilization induces cell death in a mitochondrion-dependent fashion. J. Exp. Med. 197, 1323–1334 (2003). The first report that lysosomal membrane permeabilization might induce cell death through a caspase-independent, mitochondrion-dependent pathway.
Brunk, U. T. & Svensson, I. Oxidative stress, growth factor starvation and Fas activation may all cause apoptosis through lysosomal leak. Redox Rep. 4, 3–11 (1999).
Nakayama, M. et al. Multiple pathways of TWEAK-induced cell death. J. Immunol. 168, 734–743 (2002).
Yuan, X. M. et al. Lysosomal destabilization in p53-induced apoptosis. Proc. Natl Acad. Sci. USA 99, 6286–6291 (2002).
Broker, L. E. et al. Cathepsin B mediates caspase-independent cell death induced by microtubule stabilizing agents in non-small cell lung cancer cells. Cancer Res. 64, 27–30 (2004).
Ostenfeld, M. S. et al. Effective tumor cell death by sigma-2 receptor ligand siramesine invovles lysosomal leakage and oxidative stress. Cancer Res. (in the press).
Bidere, N. et al. Cathepsin D triggers Bax activation, resulting in selective AIF relocation in T lymphocytes entering the early commitment phase to apoptosis. J. Biol. Chem. 278, 31401–31411 (2003).
Nylandsted, J. et al. Heat shock protein 70 promotes cell survival by inhibiting lysosomal membrane permeabilization. J. Exp. Med. 200, 425–435 (2004). The first report of a cancer-associated protein that stabilizes lysosomal membranes.
Roberg, K., Kagedal, K. & Ollinger, K. Microinjection of cathepsin d induces caspase-dependent apoptosis in fibroblasts. Am. J. Pathol. 161, 89–96 (2002). The first report showing that cytosolic cathepsin D can trigger mitochondrial outer-membrane permeabilization.
Guicciardi, M. E. et al. Cathepsin B contributes to TNF-α-mediated hepatocyte apoptosis by promoting mitochondrial release of cytochrome c. J. Clin. Invest. 106, 1127–1137 (2000). The first report that cathepsin B can mediate death receptor-induced apoptosis.
Fehrenbacher, N. et al. Sensitization to the lysosomal cell death pathway upon immortalization and transformation. Cancer Res. 64, 5301–5310 (2004). The first indication of a switch in death pathways on immortalization and transformation.
Zhao, M., Brunk, U. T. & Eaton, J. W. Delayed oxidant-induced cell death involves activation of phospholipase A2. FEBS Lett. 509, 399–404 (2001).
Foghsgaard, L., Lademann, U., Wissing, D., Poulsen, B. & Jäättelä, M. Cathepsin B mediates tumor necrosis factor-induced arachidonic acid release in tumor cells. J. Biol. Chem. 277, 39499–39506 (2002).
Heinrich, M. et al. Cathepsin D links TNF-induced acid sphingomyelinase to Bid-mediated caspase-9 and -3 activation. Cell Death Differ. 11, 550–563 (2004).
Schneider-Brachert, W. et al. Compartmentalization of TNF receptor 1 signaling: internalized TNF receptosomes as death signaling vesicles. Immunity 21, 415–428 (2004).
Heinrich, M. et al. Cathepsin D targeted by acid sphingomyelinase-derived ceramide. EMBO J. 18, 5252–5263 (1999).
Deiss, L. P., Galinka, H., Berissi, H., Cohen, O. & Kimchi, A. Cathepsin D protease mediates programmed cell death induced by interferon-γ, Fas/APO-1 and TNF-α. EMBO J. 15, 3861–3870 (1996). The first indication of lysosomal proteases as mediators of death receptor-induced apoptosis.
Wu, G. S., Saftig, P., Peters, C. & El-Deiry, W. S. Potential role for cathepsin D in p53-dependent tumor suppression and chemosensitivity. Oncogene 16, 2177–2183 (1998).
Liu, N. et al. NF-κB protects from the lysosomal pathway of cell death. EMBO J. 22, 5313–5322 (2003).
Michallet, M. C., Saltel, F., Flacher, M., Revillard, J. P. & Genestier, L. Cathepsin-dependent apoptosis triggered by supraoptimal activation of T lymphocytes: a possible mechanism of high dose tolerance. J. Immunol. 172, 5405–5414 (2004).
Vancompernolle, K. et al. Atractyloside-induced release of cathepsin B, a protease with caspase-processing activity. FEBS Lett. 438, 150–158 (1998).
Jäättelä, M. & Tschopp, J. Caspase-independent cell death in T lymphocytes. Nature Immunol. 4, 416–423 (2003).
Khwaja, A. & Tatton, L. Resistance to the cytotoxic effects of tumor necrosis factor α can be overcome by inhibition of a FADD/caspase-dependent signaling pathway. J. Biol. Chem. 274, 36817–36823 (1999).
Werneburg, N., Guicciardi, M. E., Yin, X. M. & Gores, G. J. TNF-α-mediated lysosomal permeabilization is FAN and caspase 8/Bid dependent. Am. J. Physiol. Gastrointest. Liver Physiol. 287, G436–G443 (2004).
Wiegmann, K., Schutze, S., Machleidt, T., Witte, D. & Kronke, M. Functional dichotomy of neutral and acidic sphingomyelinases in tumor necrosis factor signaling. Cell 78, 1005–1015 (1994).
Segui, B. et al. Involvement of FAN in TNF-induced apoptosis. J. Clin. Invest. 108, 143–151 (2001).
Werneburg, N. W., Guicciardi, M. E., Bronk, S. F. & Gores, G. J. Tumor necrosis factor-α-associated lysosomal permeabilization is cathepsin B dependent. Am. J. Physiol. Gastrointest. Liver Physiol. 283, G947–G956 (2002).
Holler, N. et al. Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nature Immunol. 1, 489–495 (2000).
Rochefort, H. et al. Estrogen regulated proteases and antiproteases in ovarian and breast cancer cells. J. Steroid Biochem. Mol. Biol. 76, 119–124 (2001).
Joyce, J. A. & Hanahan, D. Multiple roles for cysteine cathepsins in cancer. Cell Cycle 3, 1516–1519 (2004).
Roshy, S., Sloane, B. F. & Moin, K. Pericellular cathepsin B and malignant progression. Cancer Metastasis Rev. 22, 271–286 (2003).
Rao, J. S. Molecular mechanisms of glioma invasiveness: the role of proteases. Nature Rev. Cancer 3, 489–501 (2003).
Joyce, J. A. et al. Cathepsin cysteine proteases are effectors of invasive growth and angiogenesis during multistage tumorigenesis. Cancer Cell 5, 443–453 (2004). An elegant study showing that cathepsins have an important role in cancer progression in vivo.
Mohanam, S. et al. Down-regulation of cathepsin B expression impairs the invasive and tumorigenic potential of human glioblastoma cells. Oncogene 20, 3665–3673 (2001).
Nishimura, Y., Itoh, K., Yoshioka, K., Ikeda, K. & Himeno, M. A role for small GTPase RhoA in regulating intracellular membrane traffic of lysosomes in invasive rat hepatoma cells. Histochem. J. 34, 189–213 (2002).
Nishimura, Y., Itoh, K., Yoshioka, K., Tokuda, K. & Himeno, M. Overexpression of ROCK in human breast cancer cells: evidence that ROCK activity mediates intracellular membrane traffic of lysosomes. Pathol. Oncol. Res. 9, 83–95 (2003).
Nishimura, Y., Yoshioka, K., Bernard, O., Himeno, M. & Itoh, K. LIM kinase 1: evidence for a role in the regulation of intracellular vesicle trafficking of lysosomes and endosomes in human breast cancer cells. Eur. J. Cell Biol. 83, 369–380 (2004).
Brown, W. J., DeWald, D. B., Emr, S. D., Plutner, H. & Balch, W. E. Role for phosphatidylinositol 3-kinase in the sorting and transport of newly synthesized lysosomal enzymes in mammalian cells. J. Cell Biol. 130, 781–796 (1995).
Cheng, K. W. et al. The RAB25 small GTPase determines aggressiveness of ovarian and breast cancers. Nature Med. 10, 1251–1256 (2004).
Lackner, M. R. et al. Chemical genetics identifies Rab geranylgeranyl transferase as an apoptotic target of farnesyl transferase inhibitors. Cancer Cell 7, 325–336 (2005). Identification of RABGGT and its effectors as regulators of cell death by advanced genetic screens in C. elegans.
Nylandsted, J. et al. Selective depletion of heat shock protein 70 (hsp 70) activates a tumor-specific death program that is independent of caspases and bypasses Bcl-2. Proc. Natl Acad. Sci. USA 97, 7871–7876 (2000).
Nylandsted, J. et al. Eradication of glioblastoma, and breast and colon carcinoma xenografts by Hsp70 depletion. Cancer Res. 62, 7139–7142 (2002).
Jäättelä, M. Heat shock proteins as cellular lifeguards. Ann. Med. 31, 261–271 (1999).
Hantschel, M. et al. Hsp70 plasma membrane expression on primary tumor biopsy material and bone marrow of leukemic patients. Cell Stress Chaperones 5, 438–442 (2000).
Madge, L. A., Li, J. H., Choi, J. & Pober, J. S. Inhibition of phosphatidylinositol 3-kinase sensitizes vascular endothelial cells to cytokine-initiated cathepsin-dependent apoptosis. J. Biol. Chem. 278, 21295–21306 (2003).
Kos, J., Werle, B., Lah, T. & Brunner, N. Cysteine proteinases and their inhibitors in extracellular fluids: markers for diagnosis and prognosis in cancer. Int. J. Biol. Markers 15, 84–89 (2000).
Silverman, G. A. et al. Human clade B serpins (ov-serpins) belong to a cohort of evolutionarily dispersed intracellular proteinase inhibitor clades that protect cells from promiscuous proteolysis. Cell. Mol. Life Sci. 61, 301–325 (2004).
Pennacchio, L. A. et al. Progressive ataxia, myoclonic epilepsy and cerebellar apoptosis in cystatin B-deficient mice. Nature Genet. 20, 251–258 (1998).
Jones, B., Roberts, P. J., Faubion, W. A., Kominami, E. & Gores, G. J. Cystatin A expression reduces bile salt-induced apoptosis in a rat hepatoma cell line. Am. J. Physiol. 275, G723–G730 (1998).
Kuopio, T. et al. Cysteine proteinase inhibitor cystatin A in breast cancer. Cancer Res. 58, 432–436 (1998).
Suminami, Y. et al. Inhibition of apoptosis in human tumour cells by the tumour-associated serpin, SCC antigen-1. Br. J. Cancer 82, 981–989 (2000).
Klionsky, D. J. The molecular machinery of autophagy: unanswered questions. J. Cell Sci. 118, 7–18 (2005).
Edinger, A. L. & Thompson, C. B. Death by design: apoptosis, necrosis and autophagy. Curr. Opin. Cell Biol. 16, 663–669 (2004).
Mizushima, N., Yamamoto, A., Matsui, M., Yoshimori, T. & Ohsumi, Y. In vivo analysis of autophagy in response to nutrient starvation using transgenic mice expressing a fluorescent autophagosome marker. Mol. Biol. Cell. 15, 1101–1111 (2004). The first sytemic study of autophagy in mammals, based on an elegant transgenic approach that visualizes autophagy.
Kuma, A. et al. The role of autophagy during the early neonatal starvation period. Nature 432, 1032–1036 (2004). The first report indicating that autophagy might be required for catabolic reactions furnishing essential metabolites at the whole-organism level.
Lum, J. J. et al. Growth factor regulation of autophagy and cell survival in the absence of apoptosis. Cell 120, 237–248 (2005).
Boya, P. et al. Inhibition of macroautophagy triggers apoptosis. Mol. Biol. Cell 25, 1025–1040 (2005).
Gonzalez-Polo, R. A. et al. The apoptosis/autophagy paradox. Accumulation of autophagic vacuoles triggers apoptosis. J. Cell Sci. 118, 3091–3102 (2005).
Kabeya, Y. et al. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J. 19, 5720–5728 (2000).
Martin, D. N. & Baehrecke, E. H. Caspases function in autophagic programmed cell death in Drosophila. Development 131, 275–284 (2004). A tentalizing report suggesting that caspase activation is essential for salivary gland cell death, which has been thought to occur through autophagy.
Daish, T. J., Mills, K. & Kumar, S. Drosophila caspase DRONC is required for specific developmental cell death pathways and stress-induced apoptosis. Dev. Cell. 7, 909–915 (2004).
Xue, L., Fletcher, G. C. & Tolkovsky, A. M. Mitochondria are selectively eliminated from eukaryotic cells after blockade of caspases during apoptosis. Curr. Biol. 6, 361–365 (2001).
Mills, K. R., Reginato, M., Debnath, J., Queenan, B. & Brugge, J. S. Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) is required for induction of autophagy during lumen formation in vitro. Proc. Natl Acad. Sci. USA 101, 3438–3443 (2004).
Yu, L. et al. Regulation of an ATG7–beclin 1 program of autophagic cell death by caspase-8. Science 304, 1500–1502 (2004).
Shimizu, S. et al. A role of Bcl-2 family of proteins in non-apoptotic programmed cell death dependent on autophagy genes. Nature Cell Biol. 6, 1221–1228 (2004). References 79 and 80 are the first two reports suggesting that inhibition of apoptotic machinery might unravel an alternative cell death pathway that involves autophagy.
Høyer-Hansen, M., Bastholm, L., Mathiasen, I., Elling, F. & Jäättelä, M. Vitamin D analogue EB1089 triggers dramatic lysosomal changes and Beclin 1-mediated autophagic cell death. Cell Death Differ. 12, 1297–1309 (2005).
Liang, X. H. et al. Induction of autophagy and inhibition of tumorigenesis by beclin 1. Nature 402, 672–676 (1999). The description of BECN1 as an essential autophagy gene endowed with an additional tumour-suppressing function.
Qu, X. et al. Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene. J. Clin. Invest. 112, 1809–1820 (2003).
Yue, Z., Jin, S., Yang, C., Levine, A. J. & Heintz, N. Beclin 1, an autophagy gene essential for early embryonic development, is a haploinsufficient tumor suppressor. Proc. Natl Acad. Sci. USA 100, 15077–15082 (2003).
Aita, V. M. et al. Cloning and genomic organization of beclin 1, a candidate tumor suppressor gene on chromosome 17q21. Genomics 59, 59–65 (1999).
Ravikumar, B. & Rubinsztein, D. C. Can autophagy protect against neurodegeneration caused by aggregate-prone proteins? Neuroreport 15, 2443–2445 (2004).
Elmore, S. P., Qian, T., Grissom, S. F. & Lemasters, J. J. The mitochondrial permeability transition initiates autophagy in rat hepatocytes. FASEB J. 15, 2286–2287 (2002).
Gu, Y., Wang, C. & Cohen, A. Effect of IGF-1 on the balance between autophagy of dysfunctional mitochondria and apoptosis. FEBS Lett. 577, 357–360 (2004).
Kissova, I., Deffieu, M., Manon, S. & Camougrand, N. Uth1p is involved in the autophagic degradation of mitochondria. J. Biol. Chem. 279, 39068–39074 (2004).
Saeki, K. et al. Bcl-2 down-regulation causes autophagy in a caspase-independent manner in human leukemic HL60 cells. Cell Death Differ. 7, 1263–1269 (2000).
Huynh, C., Roth, D., Ward, D. M., Kaplan, J. & Andrews, N. W. Defective lysosomal exocytosis and plasma membrane repair in Chediak–Higashi/beige cells. Proc. Natl Acad. Sci. USA 101, 16795–16800 (2004).
Ono, K., Kim, S. O. & Han, J. Susceptibility of lysosomes to rupture is a determinant for plasma membrane disruption in tumor necrosis factor α-induced cell death. Mol. Cell. Biol. 23, 665–676 (2003).
Dell'Angelica, E. C., Mullins, C., Caplan, S. & Bonifacino, J. S. Lysosome-related organelles. FASEB J. 14, 1265–1278 (2000).
Edinger, A. L., Cinalli, R. M. & Thompson, C. B. Rab7 prevents growth factor-independent survival by inhibiting cell-autonomous nutrient transporter expression. Dev. Cell 5, 571–582 (2003).
Schmitt, E. et al. Chemosensitization by a non-apoptogenic heat shock protein 70-binding apoptosis-inducing factor mutant. Cancer Res. 63, 8233–8240 (2003).
Rohde, M. et al. Members of the heat-shock protein 70 family promote cancer cell growth by distinct mechanisms. Genes Dev. 19, 570–582 (2005).
Evdonin, A. L., Guzhova, I. V., Margulis, B. A. & Medvedeva, N. D. Phospholipse c inhibitor, u73122, stimulates release of hsp-70 stress protein from A431 human carcinoma cells. Cancer Cell Int. 4, 2 (2004).
Ulbrich, K. & Subr, V. Polymeric anticancer drugs with pH-controlled activation. Adv. Drug Deliv. Rev. 56, 1023–1050 (2004).
Luciani, F. et al. Effect of proton pump inhibitor pretreatment on resistance of solid tumors to cytotoxic drugs. J. Natl Cancer Inst. 96, 1702–1713 (2004).
Kanzawa, T., Kondo, Y., Ito, H., Kondo, S. & Germano, I. Induction of autophagic cell death in malignant glioma cells by arsenic trioxide. Cancer Res. 63, 2103–2108 (2003).
Kanzawa, T. et al. Role of autophagy in temozolomide-induced cytotoxicity for malignant glioma cells. Cell Death Differ. 11, 4484–4457 (2004).
Paglin, S. et al. A novel response of cancer cells to radiation involves autophagy and formation of acidic vesicles. Cancer Res. 61, 439–444 (2001).
Nakashima, S. et al. Vacuolar H+-ATPase inhibitor induces apoptosis via lysosomal dysfunction in the human gastric cancer cell line MKN-1. J. Biochem. (Tokyo) 134, 359–364 (2003).
Scaringi, L. et al. Omeprazole induces apoptosis in jurkat cells. Int. J. Immunopathol. Pharmacol. 17, 331–342 (2004).
Dutcher, J. P. Mammalian target of rapamycin inhibition. Clin. Cancer Res. 10, 6382S–6387S (2004).
Beuvink, I. et al. The mTOR inhibitor RAD001 sensitizes tumor cells to DNA-damaged induced apoptosis through inhibition of p21 translation. Cell 120, 747–759 (2005).
Xu, G., Zhang, W., Bertram, P., Zheng, X. F. & McLeod, H. Pharmacogenomic profiling of the PI3K–PTEN–AKT–mTOR pathway in common human tumors. Int. J. Oncol. 24, 893–900 (2004).
Majumder, P. K. et al. mTOR inhibition reverses Akt-dependent prostate intraepithelial neoplasia through regulation of apoptotic and HIF-1-dependent pathways. Nature Med. 10, 594–601 (2004).
Opipari, A. W. J. et al. Resveratrol-induced autophagocytosis in ovarian cancer cells. Cancer Res. 64, 696–703 (2004).
Scarlatti, F. et al. Ceramide-mediated macroautophagy involves inhibition of protein kinase B and up-regulation of beclin 1. J. Biol. Chem. 279, 18384–18391 (2004).
Daido, S. et al. Pivotal role of the cell death factor BNIP3 in ceramide-induced autophagic cell death in malignant glioma cells. Cancer Res. 64, 4286–4293 (2004).
Erdal, H. et al. Induction of lysosomal membrane permeabilization by compounds that activate p53-independent apoptosis. Proc. Natl Acad. Sci. USA 102, 192–197 (2005). The first reported small-molecule screen identifying compounds that induce lysosomal-membrane permeabilization.
Rawlings, N. D., Tolle, D. P. & Barrett, A. J. MEROPS: the peptidase database. Nucleic Acids Res. 32, D160–D164 (2004).
Turk, V., Turk, B. & Turk, D. Lysosomal cysteine proteases: facts and opportunities. EMBO J. 20, 4629–4633 (2001).
Eskelinen, E. L., Tanaka, Y. & Saftig, P. At the acidic edge: emerging functions for lysosomal membrane proteins. Trends Cell Biol. 13, 137–145 (2003).
Ghosh, P., Dahms, N. M. & Kornfeld, S. Mannose 6-phosphate receptors: new twists in the tale. Nature Rev. Mol. Cell Biol. 4, 202–212 (2003).
Andrews, N. W. Regulated secretion of conventional lysosomes. Trends Cell Biol. 10, 316–321 (2000).
Reddy, A., Caler, E. V. & Andrews, N. W. Plasma membrane repair is mediated by Ca2+-regulated exocytosis of lysosomes. Cell 106, 157–169 (2001).
Acknowledgements
G.K. is supported by the Ligue Nationale contre le Cancer and the European Union. M.J. is supported by the Danish Cancer Society, the Danish National Research Foundation, the Danish Medical Research Council and the Association for International Cancer Research.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Glossary
- CASPASES
-
A family of cysteine proteases that cleave after asparagine residues. Initiator caspases (for example, caspase 8) are typically activated in response to particular stimuli, whereas effector caspases (such as caspase 3) are important for the ordered dismantling of vital cellular structures.
- MITOCHONDRIAL OUTER-MEMBRANE PERMEABILIZATION
-
An apoptosis-associated process that results in the release of apoptosis-inducing proteins, which are normally retained in the mitochondrial intermembrane space, through the outer membrane and into the cytosol.
- CATHEPSINS
-
Proteases that locate mostly to lysosomes and lysosome-like organelles.
- AUTOPHAGY
-
In this review, autophagy is used synonymous to macro-autophagy, a process in which a portion of the cytoplasm is wrapped into a specific membrane and later digested by lysosomal enzymes.
- LYSOSOMAL-MEMBRANE PERMEABILIZATION
-
A perturbation of lysosomal membrane function that leads to the translocation of lysosomal hydrolases (including cathepsins) from the lysosomal lumen to the rest of the cell. LMP can be induced by endogenous signal transducers (such as reactive oxygen species and sphingosine) as well as by lysosomotropic drugs.
- PROGRAMMED CELL DEATH
-
An active cell death process that can be blocked by inhibition of a signal or activity within the target cell.
- DEATH RECEPTORS
-
A family of cell-surface receptors that are capable of mediating cell death on ligand-induced trimerization. The best-studied members include tumour-necrosis factor receptor 1 (TNFR1), FAS (also known as CD95), and the two receptors for TNF-related apoptosis-inducing ligand (TRAILR1 and TRAILR2).
- REACTIVE OXYGEN SPECIES
-
Collective term comprising classic oxygen radicals and peroxides that are formed inside the cell.
- Bcl2 FAMILY
-
Proteins that contain at least one BCL2 homology (BH) region. The family is divided into anti-apoptotic proteins (such as BCL2 and BCL-XL) that contain four BH domains (BH1, BH2, BH3 and BH4), pro-apoptotic proteins (for example, BAX and BAK) that contain BH1, BH2 and BH3, and the pro-apoptotic BH3-only proteins.
- CYTOCHROME C
-
A haem protein that is normally confined to the mitochondrial intermembrane space. On induction of apoptosis, cytochrome c is released from mitochondria and triggers the formation of the apoptosome, a caspase-activation complex.
- FENTON-TYPE CHEMISTRY
-
The Fenton reaction is the formation of OH−, OH− and Fe3+ from the non-enzymatic reaction of Fe2+ with H2O2; an important reaction in the oxidative stress of blood cells and various tissues.
- Rab GTPASES
-
Monomeric, small GTPases that, together with their effectors, mediate the first event during membrane fusion — the tethering of vesicles to each other or to other membranes.
- SIGMA-2 RECEPTOR
-
Sigma receptors (1 and 2) are orphan receptors that bind psychoactive substances and modulate neuronal function and cancer cell survival.
- AMPHISOME
-
A vesicle formed by fusion of an autophagosome with an endosome.
- AUTOLYSOSOME
-
Also called autophagolysosome. A vesicle formed by the fusion of an autophagosome (or an amphisome) with a lysosome.
- CHEDIAK–HIGASHI SYNDROME
-
A rare autosomal-recessive genetic disorder caused by mutations in CHS1 (which is involved in lysosomal fission and secretion).
- GRISCELLI SYNDROME TYPE 2
-
A rare autosomal-recessive genetic disorder caused by mutations in RAB27A (which is involved in the movement of secretory lysosomes).
- APOPTOSIS-INDUCING FACTOR
-
(AIF). A flavoprotein that is normally present in the mitochondrial intermembrane space. On induction of apoptosis, AIF translocates to the nucleus where it activates a molecular complex that causes large-scale DNA fragmentation, presumably in a caspase-independent fashion.
Rights and permissions
About this article
Cite this article
Kroemer, G., Jäättelä, M. Lysosomes and autophagy in cell death control. Nat Rev Cancer 5, 886–897 (2005). https://doi.org/10.1038/nrc1738
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc1738
This article is cited by
-
Bioorthogonal Chemistry in Cellular Organelles
Topics in Current Chemistry (2024)
-
Genetic assessment of pathogenic germline alterations in lysosomal genes among Asian patients with pancreatic ductal adenocarcinoma
Journal of Translational Medicine (2023)
-
Serine hydroxymethyltransferase 2 knockdown induces apoptosis in ccRCC by causing lysosomal membrane permeabilization via metabolic reprogramming
Cell Death & Disease (2023)
-
Targeting autophagy increases the efficacy of proteasome inhibitor treatment in multiple myeloma by induction of apoptosis and activation of JNK
BMC Cancer (2022)
-
Effect of morpholine and charge distribution of cyanine dyes on cell internalization and cytotoxicity
Scientific Reports (2022)