Key Points
-
Medulloblastoma is the most common malignant paediatric brain tumour and is a leading cause of cancer-related morbidity and mortality in children.
-
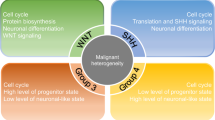
Integrative genomic studies have recently identified at least four distinct molecular subgroups of medulloblastoma — WNT, sonic hedgehog (SHH), Group 3 and Group 4 — which exhibit highly discriminate transcriptional, cytogenetic and mutational spectra in addition to divergent patient demographics and clinical behaviour.
-
Recent next-generation sequencing of medulloblastoma samples and their matched constitutional DNA has led to the identification of a multitude of previously unknown candidate genes that are somatically mutated in this cancer, including many that are mutated in a subgroup-specific manner.
-
Different mechanisms of structural variation, including somatic copy number aberrations, chromothripsis and tetraploidy, contribute to a considerable proportion of medulloblastomas and may be more prevalent than recurrent somatic mutations in Group 3 and Group 4 tumours.
-
Despite the extensive amount of copy number and sequence data that has become available for large cohorts of medulloblastoma, most Group 3 and Group 4 tumours cannot be attributed to a specific driver mutation or mutations, suggesting that additional mechanisms, such as epigenetic deregulation, may also have a prominent role.
-
Research on medulloblastoma during the next few years will be focused on the functional validation of candidate genes and molecular processes that were reported to be deregulated in the recent human genomic studies. The generation of faithful models that recapitulate these mutational events will become a priority, and such reagents will be a valuable resource for the development of rational, molecularly targeted therapies.
Abstract
The division of medulloblastoma into different subgroups by microarray expression profiling has dramatically changed our perspective of this malignant childhood brain tumour. Now, the availability of next-generation sequencing and complementary high-density genomic technologies has unmasked novel driver mutations in each medulloblastoma subgroup. The implications of these findings for the management of patients are readily apparent, pinpointing previously unappreciated diagnostic and therapeutic targets. In this Review, we summarize the 'explosion' of data emerging from the application of modern genomics to medulloblastoma, and in particular the recurrent targets of mutation in medulloblastoma subgroups. These data are currently making their way into clinical trials as we seek to integrate conventional and molecularly targeted therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Pui, C. H., Gajjar, A. J., Kane, J. R., Qaddoumi, I. A. & Pappo, A. S. Challenging issues in pediatric oncology. Nature Rev. Clin. Oncol. 8, 540–549 (2011).
Siegel, R., Naishadham, D. & Jemal, A. Cancer statistics, 2012. CA Cancer J. Clin. 62, 10–29 (2012).
Gajjar, A. et al. Risk-adapted craniospinal radiotherapy followed by high-dose chemotherapy and stem-cell rescue in children with newly diagnosed medulloblastoma (St. Jude Medulloblastoma-96): long-term results from a prospective, multicentre trial. Lancet Oncol. 7, 813–820 (2006).
Rutkowski, S. et al. Survival and prognostic factors of early childhood medulloblastoma: an international meta-analysis. J. Clin. Oncol. 28, 4961–4968 (2010).
Ellison, D. W. et al. β-Catenin status predicts a favorable outcome in childhood medulloblastoma: the United Kingdom Children's Cancer Study Group Brain Tumour Committee. J. Clin. Oncol. 23, 7951–7957 (2005). This study was the first to show that CTNNB1 -mutated medulloblastomas, which would subsequently define the WNT subgroup, have an excellent prognosis compared with non- CTNNB1 -mutated cases.
Packer, R. J. & Vezina, G. Management of and prognosis with medulloblastoma: therapy at a crossroads. Arch. Neurol. 65, 1419–1424 (2008).
Mabbott, D. J., Penkman, L., Witol, A., Strother, D. & Bouffet, E. Core neurocognitive functions in children treated for posterior fossa tumors. Neuropsychology 22, 159–168 (2008).
Mabbott, D. J. et al. Serial evaluation of academic and behavioral outcome after treatment with cranial radiation in childhood. J. Clin. Oncol. 23, 2256–2263 (2005).
Spiegler, B. J., Bouffet, E., Greenberg, M. L., Rutka, J. T. & Mabbott, D. J. Change in neurocognitive functioning after treatment with cranial radiation in childhood. J. Clin. Oncol. 22, 706–713 (2004).
Low, J. A. & de Sauvage, F. J. Clinical experience with Hedgehog pathway inhibitors. J. Clin. Oncol. 28, 5321–5326 (2010).
Ng, J. M. & Curran, T. The Hedgehog's tale: developing strategies for targeting cancer. Nature Rev. Cancer 11, 493–501 (2011).
Taylor, M. D., Mainprize, T. G. & Rutka, J. T. Molecular insight into medulloblastoma and central nervous system primitive neuroectodermal tumor biology from hereditary syndromes: a review. Neurosurgery 47, 888–901 (2000).
Hahn, H. et al. Mutations of the human homolog of Drosophila patched in the nevoid basal cell carcinoma syndrome. Cell 85, 841–851 (1996).
Johnson, R. L. et al. Human homolog of patched, a candidate gene for the basal cell nevus syndrome. Science 272, 1668–1671 (1996). References 13 and 14 implicated mutations in PTCH1 as the underlying cause of Gorlin syndrome.
Gailani, M. R. et al. Developmental defects in Gorlin syndrome related to a putative tumor suppressor gene on chromosome 9. Cell 69, 111–117 (1992).
Raffel, C. et al. Sporadic medulloblastomas contain PTCH mutations. Cancer Res. 57, 842–845 (1997).
Pietsch, T. et al. Medulloblastomas of the desmoplastic variant carry mutations of the human homologue of Drosophila patched. Cancer Res. 57, 2085–2088 (1997).
Wolter, M., Reifenberger, J., Sommer, C., Ruzicka, T. & Reifenberger, G. Mutations in the human homologue of the Drosophila segment polarity gene patched (PTCH) in sporadic basal cell carcinomas of the skin and primitive neuroectodermal tumors of the central nervous system. Cancer Res. 57, 2581–2585 (1997).
Kool, M. et al. Molecular subgroups of medulloblastoma: an international meta-analysis of transcriptome, genetic aberrations, and clinical data of WNT, SHH, Group 3, and Group 4 medulloblastomas. Acta Neuropathol. 123, 473–484 (2012). This study combined all published genomic data of medulloblastomas and effectivley summarized the main clinical and genomic features defining the medulloblastoma subgroups.
Hamilton, S. R. et al. The molecular basis of Turcot's syndrome. New Engl. J. Med. 332, 839–847 (1995).
Zurawel, R. H., Chiappa, S. A., Allen, C. & Raffel, C. Sporadic medulloblastomas contain oncogenic β-catenin mutations. Cancer Res. 58, 896–899 (1998).
Eberhart, C. G., Tihan, T. & Burger, P. C. Nuclear localization and mutation of β-catenin in medulloblastomas. J. Neuropathol. Exp. Neurol. 59, 333–337 (2000).
Pastorino, L. et al. Identification of a SUFU germline mutation in a family with Gorlin syndrome. Am. J. Med. Genet. A 149A, 1539–1543 (2009).
Taylor, M. D. et al. Mutations in SUFU predispose to medulloblastoma. Nature Genet. 31, 306–310 (2002). This study identified germline and somatic mutations of SUFU in patients with medulloblastoma, establishing it as an important TSG in the SHH pathway.
Dahmen, R. P. et al. Deletions of AXIN1, a component of the WNT/wingless pathway, in sporadic medulloblastomas. Cancer Res. 61, 7039–7043 (2001).
Baeza, N., Masuoka, J., Kleihues, P. & Ohgaki, H. AXIN1 mutations but not deletions in cerebellar medulloblastomas. Oncogene 22, 632–636 (2003).
Smyth, I. et al. Isolation and characterization of human patched 2 (PTCH2), a putative tumour suppressor gene inbasal cell carcinoma and medulloblastoma on chromosome 1p32. Hum. Mol. Genet. 8, 291–297 (1999).
Koch, A. et al. Mutations of the Wnt antagonist AXIN2 (Conductin) result in TCF-dependent transcription in medulloblastomas. Int. J. Cancer 121, 284–291 (2007).
Lam, C. W. et al. A frequent activated smoothened mutation in sporadic basal cell carcinomas. Oncogene 18, 833–836 (1999).
Meyerson, M., Gabriel, S. & Getz, G. Advances in understanding cancer genomes through second-generation sequencing. Nature Rev. Genet. 11, 685–696 (2010).
Stratton, M. R. Exploring the genomes of cancer cells: progress and promise. Science 331, 1553–1558 (2011).
Robinson, G. et al. Novel mutations target distinct subgroups of medulloblastoma. Nature 488, 43–48 (2012).
Jones, D. T. et al. Dissecting the genomic complexity underlying medulloblastoma. Nature 488, 100–105 (2012).
Pugh, T. J. et al. Medulloblastoma exome sequencing uncovers subtype-specific somatic mutations. Nature 488, 106–110 (2012). References 32 to 34 were the first studies to apply next-generation sequencing to large cohorts of medulloblastoma samples in order to catalogue the prevalent somatic mutations in the disease.
Rausch, T. et al. Genome sequencing of pediatric medulloblastoma links catastrophic DNA rearrangements with TP53 mutations. Cell 148, 59–71 (2012). This study used WGS to describe the genomic landscape of medulloblastomas arising in patients with LFS, uncovering chromothripsis as a signature event in this subset of patients.
Northcott, P. A. et al. The miR-17/92 polycistron is up-regulated in sonic hedgehog-driven medulloblastomas and induced by N-myc in sonic hedgehog-treated cerebellar neural precursors. Cancer Res. 69, 3249–3255 (2009).
Thompson, M. C. et al. Genomics identifies medulloblastoma subgroups that are enriched for specific genetic alterations. J. Clin. Oncol. 24, 1924–1931 (2006).
Kool, M. et al. Integrated genomics identifies five medulloblastoma subtypes with distinct genetic profiles, pathway signatures and clinicopathological features. PLoS ONE 3, e3088 (2008). References 37 and 38 were the first to report the existence of distinct molecular subgroups of medulloblastoma based on unsupervised analyses of transcriptional profiles.
Cho, Y. J. et al. Integrative genomic analysis of medulloblastoma identifies a molecular subgroup that drives poor clinical outcome. J. Clin. Oncol. 29, 1424–1430 (2011).
Northcott, P. A. et al. Medulloblastoma comprises four distinct molecular variants. J. Clin. Oncol. 29, 1408–1414 (2011). References 39 and 40 are highly complementary studies on the genomic and clinical characteristics of medulloblastoma subgroups and instigated the definition of the currently recognized consensus subgroups.
Remke, M. et al. FSTL5 is a marker of poor prognosis in non-WNT/non-SHH medulloblastoma. J. Clin. Oncol. 29, 3852–3861 (2011).
Taylor, M. D. et al. Molecular subgroups of medulloblastoma: the current consensus. Acta Neuropathol. 123, 465–472 (2012). This report is the consensus statement on the four subgroup structure of medulloblastoma agreed on by the medulloblastoma community.
Northcott, P. A., Korshunov, A., Pfister, S. M. & Taylor, M. D. The clinical implications of medulloblastoma subgroups. Nature Rev. Neurol. 8, 340–351 (2012).
Northcott, P. A., Dubuc, A. M., Pfister, S. & Taylor, M. D. Molecular subgroups of medulloblastoma. Expert Rev. Neurother. 12, 871–884 (2012).
Packer, R. J. Risk stratification of medulloblastoma: a paradigm for future childhood brain tumor management strategies. Curr. Neurol. Neurosci. Rep. 11, 124–126 (2011).
Leary, S. E. & Olson, J. M. The molecular classification of medulloblastoma: driving the next generation clinical trials. Curr. Opin. Pediatr. 24, 33–39 (2012).
Pomeroy, S. L. et al. Prediction of central nervous system embryonal tumour outcome based on gene expression. Nature 415, 436–442 (2002). This study used gene expression array profiling to discriminate different histologies of paediatric brain tumours and provided definitive molecular evidence of medulloblastoma as being distinct from other primitive neuroectodermal tumours.
Clifford, S. C. et al. Wnt/Wingless pathway activation and chromosome 6 loss characterize a distinct molecular sub-group of medulloblastomas associated with a favorable prognosis. Cell Cycle 5, 2666–2670 (2006).
Pfaff, E. et al. TP53 mutation is frequently associated with CTNNB1 mutation or MYCN amplification and is compatible with long-term survival in medulloblastoma. J. Clin. Oncol. 28, 5188–5196 (2010).
Lindsey, J. C. et al. TP53 mutations in favorable-risk Wnt/Wingless-subtype medulloblastomas. J. Clin. Oncol. 29, e344–e348 (2011).
Northcott, P. A. et al. Rapid, reliable, and reproducible molecular sub-grouping of clinical medulloblastoma samples. Acta Neuropathol. 123, 615–626 (2012).
Gibson, P. et al. Subtypes of medulloblastoma have distinct developmental origins. Nature 468, 1095–1099 (2010). This study described the first mouse model to faithfully recapitulate human WNT medulloblastoma and proposed LRPs as their putative cell-of-origin.
Northcott, P. A. et al. Pediatric and adult sonic hedgehog medulloblastomas are clinically and molecularly distinct. Acta Neuropathol. 122, 231–240 (2011).
Ellison, D. W. et al. Medulloblastoma: clinicopathological correlates of SHH, WNT, and non-SHH/WNT molecular subgroups. Acta Neuropathol. 121, 381–396 (2011).
Brugieres, L. et al. High frequency of germline SUFU mutations in children with desmoplastic/nodular medulloblastoma younger than 3 years of age. J. Clin. Oncol. 30, 2087–2093 (2012).
Brugieres, L. et al. Incomplete penetrance of the predisposition to medulloblastoma associated with germ-line SUFU mutations. J. Med. Genet. 47, 142–144 (2010).
Garre, M. L. et al. Medulloblastoma variants: age-dependent occurrence and relation to Gorlin syndrome-a new clinical perspective. Clin. Cancer Res. 15, 2463–2471 (2009).
Huse, J. T. & Holland, E. C. Genetically engineered mouse models of brain cancer and the promise of preclinical testing. Brain Pathol. 19, 132–143 (2009).
Wu, X., Northcott, P. A., Croul, S. & Taylor, M. D. Mouse models of medulloblastoma. Chin. J. Cancer 30, 442–449 (2011).
Goodrich, L. V., Milenkovic, L., Higgins, K. M. & Scott, M. P. Altered neural cell fates and medulloblastoma in mouse patched mutants. Science 277, 1109–1113 (1997). This article introduced the first sporadic mouse model of medulloblastoma generated through germline deletion of one allele of the Ptch1 TSG.
Wetmore, C., Eberhart, D. E. & Curran, T. The normal patched allele is expressed in medulloblastomas from mice with heterozygous germ-line mutation of patched. Cancer Res. 60, 2239–2246 (2000).
Hatton, B. A. et al. The Smo/Smo model: hedgehog-induced medulloblastoma with 90% incidence and leptomeningeal spread. Cancer Res. 68, 1768–1776 (2008).
Hallahan, A. R. et al. The SmoA1 mouse model reveals that notch signaling is critical for the growth and survival of sonic hedgehog-induced medulloblastomas. Cancer Res. 64, 7794–7800 (2004).
Oliver, T. G. et al. Loss of patched and disruption of granule cell development in a pre-neoplastic stage of medulloblastoma. Development 132, 2425–2439 (2005).
Yang, Z. J. et al. Medulloblastoma can be initiated by deletion of Patched in lineage-restricted progenitors or stem cells. Cancer Cell 14, 135–145 (2008).
Schuller, U. et al. Acquisition of granule neuron precursor identity is a critical determinant of progenitor cell competence to form Shh-induced medulloblastoma. Cancer Cell 14, 123–134 (2008).
Grammel, D. et al. Sonic hedgehog-associated medulloblastoma arising from the cochlear nuclei of the brainstem. Acta Neuropathol. 123, 601–614 (2012).
Rudin, C. M. et al. Treatment of medulloblastoma with hedgehog pathway inhibitor GDC-0449. New Engl. J. Med. 361, 1173–1178 (2009). This study demonstrated proof-of-principle for targeted therapy of medulloblastoma through administration of a pharmacological inhibitor of SMO to a single patient with metastatic medulloblastoma, resulting in a profound but transient response and subsequent therapeutic resistance.
Yauch, R. L. et al. Smoothened mutation confers resistance to a Hedgehog pathway inhibitor in medulloblastoma. Science 326, 572–574 (2009).
Buonamici, S. et al. Interfering with resistance to smoothened antagonists by inhibition of the PI3K pathway in medulloblastoma. Sci. Transl. Med. 2, 51ra70 (2010).
Dijkgraaf, G. J. et al. Small molecule inhibition of GDC-0449 refractory smoothened mutants and downstream mechanisms of drug resistance. Cancer Res. 71, 435–444 (2011).
Metcalfe, C. & de Sauvage, F. J. Hedgehog fights back: mechanisms of acquired resistance against Smoothened antagonists. Cancer Res. 71, 5057–5061 (2011).
Northcott, P. A. et al. Multiple recurrent genetic events converge on control of histone lysine methylation in medulloblastoma. Nature Genet. 41, 465–472 (2009). This study used SNP arrays to identify significant copy number aberrations in the medulloblastoma genome and was the first to implicate deregulation of chromatin modifiers as an important theme in medulloblastoma biology.
Kimura, H., Ng, J. M. & Curran, T. Transient inhibition of the Hedgehog pathway in young mice causes permanent defects in bone structure. Cancer Cell 13, 249–260 (2008).
Pei, Y. et al. An animal model of MYC-driven medulloblastoma. Cancer Cell 21, 155–167 (2012).
Kawauchi, D. et al. A mouse model of the most aggressive subgroup of human medulloblastoma. Cancer Cell 21, 168–180 (2012). References 75 and 76 describe the successful generation of in vivo models of MYC-driven Group 3 medulloblastoma.
Remke, M. et al. Adult medulloblastoma comprises three major molecular variants. J. Clin. Oncol. 29, 2717–2723 (2011).
Eberhart, C. G. Three down and one to go: modeling medulloblastoma subgroups. Cancer Cell 21, 137–138 (2012).
Swartling, F. J. et al. Distinct neural stem cell populations give rise to disparate brain tumors in response to N-MYC. Cancer Cell 21, 601–613 (2012).
Swartling, F. J. et al. Pleiotropic role for MYCN in medulloblastoma. Genes Dev. 24, 1059–1072 (2010). This article introduced an elegant mouse model of MYCN-driven medulloblastoma relying on a bidirectional transgenic promoter expressing both MYCN and luciferase in the developing mouse cerebellum that generated highly penetrant mouse medulloblastomas with similar features of the human disease.
Northcott, P. A., Rutka, J. T. & Taylor, M. D. Genomics of medulloblastoma: from Giemsa-banding to next-generation sequencing in 20 years. Neurosurg. Focus 28, e6 (2010).
Ciara, E. et al. Heterozygous germ-line mutations in the NBN gene predispose to medulloblastoma in pediatric patients. Acta Neuropathol. 119, 325–334 (2010).
Distel, L., Neubauer, S., Varon, R., Holter, W. & Grabenbauer, G. Fatal toxicity following radio- and chemotherapy of medulloblastoma in a child with unrecognized Nijmegen breakage syndrome. Med. Pediatr. Oncol. 41, 44–48 (2003).
Taylor, M. D. et al. Medulloblastoma in a child with Rubenstein-Taybi Syndrome: case report and review of the literature. Pediatr. Neurosurg. 35, 235–238 (2001).
Parsons, D. W. et al. The genetic landscape of the childhood cancer medulloblastoma. Science 331, 435–439 (2011). This study was the first unbiased exome-wide sequencing study of medulloblastoma and identified novel recurrent somatic mutations in the chromatin modifiers MLL2 and MLL3.
Parsons, D. W. et al. An integrated genomic analysis of human glioblastoma multiforme. Science 321, 1807–1812 (2008).
Hodis, E. et al. A landscape of driver mutations in melanoma. Cell 150, 251–263 (2012).
Banerji, S. et al. Sequence analysis of mutations and translocations across breast cancer subtypes. Nature 486, 405–409 (2012).
Cancer Genome Atlas Research Network. Integrated genomic analyses of ovarian carcinoma. Nature 474, 609–615 (2011).
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of human colon and rectal cancer. Nature 487, 330–337 (2012).
Barbieri, C. E. et al. Exome sequencing identifies recurrent SPOP, FOXA1 and MED12 mutations in prostate cancer. Nature Genet. 44, 685–689 (2012).
Pek, J. W. & Kai, T. DEAD-box RNA helicase Belle/DDX3 and the RNA interference pathway promote mitotic chromosome segregation. Proc. Natl Acad. Sci. USA 108, 12007–12012 (2011).
Lai, M. C., Chang, W. C., Shieh, S. Y. & Tarn, W. Y. DDX3 regulates cell growth through translational control of cyclin E1. Mol. Cell. Biol. 30, 5444–5453 (2010).
Schroder, M. Human DEAD-box protein 3 has multiple functions in gene regulation and cell cycle control and is a prime target for viral manipulation. Biochem. Pharmacol. 79, 297–306 (2010).
Lee, C. S. et al. Human DDX3 functions in translation and interacts with the translation initiation factor eIF3. Nucleic Acids Res. 36, 4708–4718 (2008).
Lai, M. C., Lee, Y. H. & Tarn, W. Y. The DEAD-box RNA helicase DDX3 associates with export messenger ribonucleoproteins as well as tip-associated protein and participates in translational control. Mol. Biol. Cell 19, 3847–3858 (2008).
Rosner, A. & Rinkevich, B. The DDX3 subfamily of the DEAD box helicases: divergent roles as unveiled by studying different organisms and in vitro assays. Curr. Med. Chem. 14, 2517–2525 (2007).
Hogbom, M. et al. Crystal structure of conserved domains 1 and 2 of the human DEAD-box helicase DDX3X in complex with the mononucleotide AMP. J. Mol. Biol. 372, 150–159 (2007).
Northcott, P. A. et al. Subgroup-specific structural variation across 1,000 medulloblastoma genomes. Nature 488, 49–56 (2012). This study described the SCNAs present in a series of >1,000 primary medulloblastomas and led to the discovery of several novel medulloblastoma subgroup-specific alterations, including PVT1–MYC fusion genes in Group 3 medulloblastoma and tandem duplication of SNCAIP in Group 4 medulloblastoma.
Sengoku, T. & Yokoyama, S. Structural basis for histone H3 Lys 27 demethylation by UTX/KDM6A. Genes Dev. 25, 2266–2277 (2011).
Kim, E. & Song, J. J. Diverse ways to be specific: a novel Zn-binding domain confers substrate specificity to UTX/KDM6A histone H3 Lys 27 demethylase. Genes Dev. 25, 2223–2226 (2011).
Kooistra, S. M. & Helin, K. Molecular mechanisms and potential functions of histone demethylases. Nature Rev. Mol. Cell Biol. 13, 297–311 (2012).
Lee, M. G., Wynder, C., Cooch, N. & Shiekhattar, R. An essential role for CoREST in nucleosomal histone 3 lysine 4 demethylation. Nature 437, 432–435 (2005).
Schnetz, M. P. et al. CHD7 targets active gene enhancer elements to modulate ES cell-specific gene expression. PLoS Genet. 6, e1001023 (2010).
Bajpai, R. et al. CHD7 cooperates with PBAF to control multipotent neural crest formation. Nature 463, 958–962 (2010).
Schnetz, M. P. et al. Genomic distribution of CHD7 on chromatin tracks H3K4 methylation patterns. Genome Res. 19, 590–601 (2009).
Greer, E. L. & Shi, Y. Histone methylation: a dynamic mark in health, disease and inheritance. Nature Rev. Genet. 13, 343–357 (2012).
Margueron, R. & Reinberg, D. The Polycomb complex PRC2 and its mark in life. Nature 469, 343–349 (2011).
Alimova, I. et al. Targeting the enhancer of zeste homologue 2 in medulloblastoma. International journal of cancer. J. Inter. Cancer 131, 1800–1809 (2012).
Oberoi, J. et al. Structural basis for the assembly of the SMRT/NCoR core transcriptional repression machinery. Nature Struct. Mol. Biol. 18, 177–184 (2011).
Chin, L., Hahn, W. C., Getz, G. & Meyerson, M. Making sense of cancer genomic data. Genes Dev. 25, 534–555 (2011).
Satow, R., Kurisaki, A., Chan, T. C., Hamazaki, T. S. & Asashima, M. Dullard promotes degradation and dephosphorylation of BMP receptors and is required for neural induction. Dev. Cell 11, 763–774 (2006).
Kim, Y. et al. A conserved phosphatase cascade that regulates nuclear membrane biogenesis. Proc. Natl Acad. Sci. USA 104, 6596–6601 (2007).
Han, S. et al. Nuclear envelope phosphatase 1-regulatory subunit 1 (formerly TMEM188) is the metazoan Spo7p ortholog and functions in the lipin activation pathway. J. Biol. Chem. 287, 3123–3137 (2012).
Cogen, P. H. et al. Involvement of multiple chromosome 17p loci in medulloblastoma tumorigenesis. Am. J. Hum. Genet. 50, 584–589 (1992).
Cogen, P. H., Daneshvar, L., Metzger, A. K. & Edwards, M. S. Deletion mapping of the medulloblastoma locus on chromosome 17p. Genomics 8, 279–285 (1990).
Biegel, J. A., Burk, C. D., Barr, F. G. & Emanuel, B. S. Evidence for a 17p tumor related locus distinct from p53 in pediatric primitive neuroectodermal tumors. Cancer Res. 52, 3391–3395 (1992).
Adamson, D. C. et al. OTX2 is critical for the maintenance and progression of Shh-independent medulloblastomas. Cancer Res. 70, 181–191 (2010).
Pfister, S. et al. Outcome prediction in pediatric medulloblastoma based on DNA copy-number aberrations of chromosomes 6q and 17q and the MYC and MYCN loci. J. Clin. Oncol. 27, 1627–1636 (2009).
Mendrzyk, F. et al. Genomic and protein expression profiling identifies CDK6 as novel independent prognostic marker in medulloblastoma. J. Clin. Oncol. 23, 8853–8862 (2005).
Li, M. et al. Multiple CDK/CYCLIND genes are amplified in medulloblastoma and supratentorial primitive neuroectodermal brain tumor. Cancer Genet. 205, 220–231 (2012).
Le Guezennec, X. & Bulavin, D. V. WIP1 phosphatase at the crossroads of cancer and aging. Trends Biochem. Sci. 35, 109–114 (2010).
Castellino, R. C. et al. Medulloblastomas overexpress the p53-inactivating oncogene WIP1/PPM1D. J. Neurooncol. 86, 245–256 (2008).
Doucette, T. A. et al. WIP1 enhances tumor formation in a sonic hedgehog-dependent model of medulloblastoma. Neurosurgery 70, 1003–1010 (2012).
Huppi, K. et al. The identification of microRNAs in a genomically unstable region of human chromosome 8q24. Mol. Cancer Res. 6, 212–221 (2008).
Beck-Engeser, G. B. et al. Pvt1-encoded microRNAs in oncogenesis. Retrovirology 5, 4 (2008).
Guan, Y. et al. Amplification of PVT1 contributes to the pathophysiology of ovarian and breast cancer. Clin. Cancer Res. 13, 5745–5755 (2007).
Engelender, S. et al. Synphilin-1 associates with α-synuclein and promotes the formation of cytosolic inclusions. Nature Genet. 22, 110–114 (1999).
Chung, K. K. et al. Parkin ubiquitinates the α-synuclein-interacting protein, synphilin-1: implications for Lewy-body formation in Parkinson disease. Nature Med. 7, 1144–1150 (2001).
Eyal, A. & Engelender, S. Synphilin isoforms and the search for a cellular model of lewy body formation in Parkinson's disease. Cell Cycle 5, 2082–2086 (2006).
Pfister, S. et al. BRAF gene duplication constitutes a mechanism of MAPK pathway activation in low-grade astrocytomas. J. Clin. Invest. 118, 1739–1749 (2008).
Jones, D. T. et al. Tandem duplication producing a novel oncogenic BRAF fusion gene defines the majority of pilocytic astrocytomas. Cancer Res. 68, 8673–8677 (2008).
Malkin, D. et al. Germ line p53 mutations in a familial syndrome of breast cancer, sarcomas, and other neoplasms. Science 250, 1233–1238 (1990).
Srivastava, S., Zou, Z. Q., Pirollo, K., Blattner, W. & Chang, E. H. Germ-line transmission of a mutated p53 gene in a cancer-prone family with Li-Fraumeni syndrome. Nature 348, 747–749 (1990). References 133 and 134 established germline TP53 mutations as the underlying cause of LFS.
Stephens, P. J. et al. Massive genomic rearrangement acquired in a single catastrophic event during cancer development. Cell 144, 27–40 (2011).
Boveri, T. Concerning the origin of malignant tumours by Theodor Boveri. Translated and annotated by Henry Harris. J. Cell Sci. 121, 1–84 (2008).
Gordon, D. J., Resio, B. & Pellman, D. Causes and consequences of aneuploidy in cancer. Nature Rev. Genet. 13, 189–203 (2012).
Storchova, Z. & Pellman, D. From polyploidy to aneuploidy, genome instability and cancer. Nature Rev. Mol. Cell Biol. 5, 45–54 (2004).
Fujii, Y., Hongo, T. & Hayashi, Y. Chromosome analysis of brain tumors in childhood. Genes Chromosomes Cancer 11, 205–215 (1994).
Tomita, T., Das, L. & Radkowski, M. A. Bone metastases of medulloblastoma in childhood; correlation with flow cytometric DNA analysis. J. Neurooncol. 8, 113–120 (1990).
Giangaspero, F. et al. “Desmoplastic” versus “classic” medulloblastoma: comparison of DNA content, histopathology and differentiation. Virchows Archiv. A Pathol. Anat. Histopathol. 418, 207–214 (1991).
Zerbini, C. et al. Prognostic factors in medulloblastoma, including DNA ploidy. J. Clin. Oncol. 11, 616–622 (1993).
Keles, G. E. et al. Establishment and characterization of four human medulloblastoma-derived cell lines. Oncol. Res. 7, 493–503 (1995).
Rajcan-Separovic, E. et al. Interphase fluorescence in situ hybridization and DNA flow cytometry analysis of medulloblastomas with a normal karyotype. Cancer Genet. Cytogenet. 133, 94–97 (2002).
Davoli, T. & de Lange, T. Telomere-driven tetraploidization occurs in human cells undergoing crisis and promotes transformation of mouse cells. Cancer Cell 21, 765–776 (2012).
Fujiwara, T. et al. Cytokinesis failure generating tetraploids promotes tumorigenesis in p53-null cells. Nature 437, 1043–1047 (2005).
Castedo, M. et al. Selective resistance of tetraploid cancer cells against DNA damage-induced apoptosis. Ann. NY Acad. Sci. 1090, 35–49 (2006).
Rello-Varona, S. et al. Preferential killing of tetraploid tumor cells by targeting the mitotic kinesin Eg5. Cell Cycle 8, 1030–1035 (2009).
Vitale, I. et al. Inhibition of Chk1 kills tetraploid tumor cells through a p53-dependent pathway. PLoS ONE 2, e1337 (2007).
Strahm, B. & Malkin, D. Hereditary cancer predisposition in children: genetic basis and clinical implications. Int. J. Cancer 119, 2001–2006 (2006).
Searles Nielsen, S. et al. Family cancer history and risk of brain tumors in children: results of the SEARCH international brain tumor study. Cancer Causes Control 19, 641–648 (2008).
Marth, G. T. et al. The functional spectrum of low-frequency coding variation. Genome Biol. 12, R84 (2011).
1000 Genomes Project Consortium. A map of human genome variation from population-scale sequencing. Nature 467, 1061–1073 (2010).
Dawson, M. A. & Kouzarides, T. Cancer epigenetics: from mechanism to therapy. Cell 150, 12–27 (2012).
Baylin, S. B. & Jones, P. A. A decade of exploring the cancer epigenome - biological and translational implications. Nature Rev. Cancer 11, 726–734 (2011).
Ozsolak, F. & Milos, P. M. RNA sequencing: advances, challenges and opportunities. Nature Rev. Genet. 12, 87–98 (2011).
Curtis, C. et al. The genomic and transcriptomic architecture of 2,000 breast tumours reveals novel subgroups. Nature 486, 346–352 (2012).
Parmigiani, G. et al. Design and analysis issues in genome-wide somatic mutation studies of cancer. Genomics 93, 17–21 (2009).
Stratton, M. R., Campbell, P. J. & Futreal, P. A. The cancer genome. Nature 458, 719–724 (2009).
Premsrirut, P. K. et al. A rapid and scalable system for studying gene function in mice using conditional RNA interference. Cell 145, 145–158 (2011).
Zuber, J. et al. Toolkit for evaluating genes required for proliferation and survival using tetracycline-regulated RNAi. Nature Biotechnol. 29, 79–83 (2011).
Copeland, N. G. & Jenkins, N. A. Harnessing transposons for cancer gene discovery. Nature Rev. Cancer 10, 696–706 (2010).
Wu, X. et al. Clonal selection drives genetic divergence of metastatic medulloblastoma. Nature 482, 529–533 (2012). This study used the SB transposon system to mutagenize the developing mouse cerebellum and generate highly penetrant metastatic mouse medulloblastomas that allowed for a direct comparison of the genes suspected to drive the primary tumours with those targeted in the matched metastases.
Ellison, D. W. et al. Definition of disease-risk stratification groups in childhood medulloblastoma using combined clinical, pathologic, and molecular variables. J. Clin. Oncol. 29, 1400–1407 (2011).
von Hoff, K. et al. Long-term outcome and clinical prognostic factors in children with medulloblastoma treated in the prospective randomised multicentre trial HIT'91. Eur. J. Cancer 45, 1209–1217 (2009).
Taylor, R. E. et al. Outcome for patients with metastatic (M2-3) medulloblastoma treated with SIOP/UKCCSG PNET-3 chemotherapy. Eur. J. Cancer 41, 727–734 (2005).
Packer, R. J. Childhood brain tumors: accomplishments and ongoing challenges. J. Child Neurol. 23, 1122–1127 (2008).
Gilbertson, R. J. Mapping cancer origins. Cell 145, 25–29 (2011).
Marino, S., Vooijs, M., van Der Gulden, H., Jonkers, J. & Berns, A. Induction of medulloblastomas in p53-null mutant mice by somatic inactivation of Rb in the external granular layer cells of the cerebellum. Genes Dev. 14, 994–1004 (2000).
Wechsler-Reya, R. J. & Scott, M. P. Control of neuronal precursor proliferation in the cerebellum by Sonic Hedgehog. Neuron 22, 103–114 (1999). This study identified the link between SHH signalling and cerebellar development.
Acknowledgements
This work was principally supported by the PedBrain Tumour Project contributing to the International Cancer Genome Consortium (ICGC), funded by German Cancer Aid (109252) and the German Federal Ministry of Education and Research (BMBF, NGFNplus #01GS0883). The authors also acknowledge the Pediatric Brain Tumour Foundation (PBTFUS), the US National Institutes of Health (R01CA148699 and R01CA159859) and the Dutch Cancer Foundations KWF (2010-4713) and KIKA. P.A.N. is a Roman Herzog PostDoctoral Fellow supported by the Hertie Foundation and the DKFZ. The authors are deeply indebted to C. Smith of Creative Science Studios for assistance with artwork. They also acknowledge C. Imbusch (DKFZ) for bioinformatic support pertaining to the meta-analysis of next-generation sequencing studies. Last, special acknowledgments are warranted for T. J. Pugh (Broad Institute), N. Jäger (DKFZ) and D. J. H. Shih (SickKids) who played lead bioinformatic roles in the respective medulloblastoma genomic studies highlighted in this Review.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Leptomeningeal dissemination
-
Metastasis to the leptomeninges (arachnoid mater and pia mater) that wrap the brain and spinal cord. Metastatic medulloblastoma cells typically disseminate to the leptomeninges via the cerebrospinal fluid.
- Gorlin syndrome
-
Also known as nevoid basal cell carcinoma syndrome (NBCCS). An autosomal dominant condition in which affected individuals harbour germline mutations in patched 1 and the cells of which display aberrant activation of the sonic hedgehog signalling pathway. Individuals exhibit a variety of developmental defects including jaw cysts, palmar pits and skeletal abnormalities and they are predisposed to the development of extensive basal cell carcinomas and medulloblastoma, among other cancers.
- Turcot syndrome
-
Typically an autosomal dominant condition characterized by multiple adenomatous colon polyps and predisposition to colorectal cancer and brain tumours. The genetic basis is linked to mutations in adenomatous polyposis coli, which predisposes to medulloblastoma, or to the mismatch repair genes MLH1 or PMS2, which predispose to glioblastoma.
- Next-generation sequencing
-
Sequencing technologies emerging since 2005 that have substantially increased the output of the nucleic acid sequencing process. They produce millions to hundreds of millions of typically short sequence reads (50–400 bp) from amplified DNA clones.
- Classic histology
-
The most common histological subtype of medulloblastoma, displaying prototypical sheets of repetitive small cells with a high nuclear/cytoplasmic ratio and round nuclei. Found in all medulloblastoma subgroups.
- External granule cell layer
-
(EGL). A germinal zone comprised of cerebellar granule neuron precursors that line the surface of the developing cerebellum.
- Lower rhombic lip
-
(LRL). A division of the rhombic lip, a specialized germinal matrix situated at the interface between the neural tube and the roofplate of the fourth ventricle of the developing cerebellum.
- Dorsal brainstem
-
The hindmost parts of the structure of the brainstem.
- Desmoplastic (or nodular) histology
-
Histological variant of medulloblastoma characterized by the presence of a varying number of nodules that consist of differentiated neurocytic cells and internodular desmoplasia (fibrous or connective tissue), best demonstrated by reticulin staining. Mostly restricted to the sonic hedgehog medulloblastoma subgroup.
- Large-cell or anaplastic
-
(LCA). An admixture of two usually co-occuring histological variants of medulloblastoma associated with a more aggressive clinical phenotype and the Group 3 subgroup. Collectively, LCA medulloblastomas appear as groups of large cells with round nuclei (large cell) and cells exhibiting marked cytological pleomorphisms (anaplasia), especially nuclear pleomorphism.
- Cerebellar granule neuron precursors
-
(CGNPs). As precursors to the most abundant neuron in the brain, CGNPs arise in the hindbrain during late embryonic development and migrate postnatally to the external granule cell layer of the cerebellum. They then undergo a period of massive proliferation before eventually migrating inwards to the internal granule layer and terminally differentiating.
- Cochlear nuclei
-
Structures in the brainstem that derive from the auditory lower rhombic lip, receiving inputs on sound from the cochlear nerve. Cerebellar granule neuron precursors residing in the cochlear nuclei have been implicated as a possible cell-of-origin for sonic hedgehog medulloblastoma.
- Isochromosome
-
An abnormal chromosome produced during mitosis or meiosis that is characterized by the presence of two genetically and morphologically identical chromosome arms fused at the centromere. Strictly, the common alteration of chromosome 17 in medulloblastoma is more often an isodicentric (17q), as the breakpoint is typically in the p-arm proximal to the centromere rather than at the centromere itself.
- Single nucleotide variants
-
(SNVs). A nucleotide substitution resulting in the exchange of one nucleotide for another. In cancer, SNVs refer to single nucleotide changes present in the tumour DNA that are not present in the patient's normal genome.
- Indels
-
Mutations involving the insertion or deletion of nucleotides, which often lead to a loss of function by causing a shift in the reading frame of a gene.
- Chromothripsis
-
A process of erroneous DNA repair following a single catastrophic breakage event that results in massive genomic rearrangement.
- Tetraploidy
-
A whole-genome duplication event such that each chromosome is present as four, rather than two, copies.
- Lewy bodies
-
Abnormal protein aggregates found in the brain of individuals with Parkinson's disease, Lewy body dementia and other related disorders.
- Tandem duplication
-
A structural variant caused by a duplication event that results in two genomic segments of at least 1 kb in size that share >90% sequence identity positioned in a contiguous manner within the genome.
- Double-minute chromosomes
-
Typically acentric, extra-chromosomally amplified chromatin, usually containing a particular chromosomal segment or gene. Double minutes are common in cancer and often result in oncogene amplification through replication and asymmetric distribution after cell division.
- Single nucleotide polymorphism
-
(SNP). A single nucleotide DNA sequence variant that differs between individuals or paired chromosomes from the same individual. In contrast to single nucleotide variants, these are present in the normal genome of an individual.
- Aneuploidy
-
An abnormal number of chromosomes.
Rights and permissions
About this article
Cite this article
Northcott, P., Jones, D., Kool, M. et al. Medulloblastomics: the end of the beginning. Nat Rev Cancer 12, 818–834 (2012). https://doi.org/10.1038/nrc3410
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3410
This article is cited by
-
Molecular and functional profiling of chemotolerant cells unveils nucleoside metabolism-dependent vulnerabilities in medulloblastoma
Acta Neuropathologica Communications (2023)
-
Neurodegeneration-associated protein VAPB regulates proliferation in medulloblastoma
Scientific Reports (2023)
-
Superenhancers as master gene regulators and novel therapeutic targets in brain tumors
Experimental & Molecular Medicine (2023)
-
Hedgehog signaling in tissue homeostasis, cancers, and targeted therapies
Signal Transduction and Targeted Therapy (2023)
-
Smurf1 and Smurf2 mediated polyubiquitination and degradation of RNF220 suppresses Shh-group medulloblastoma
Cell Death & Disease (2023)