Abstract
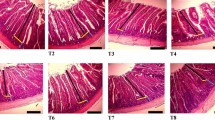
Effective iron fortification of foods is difficult, because water-soluble compounds that are well absorbed, such as ferrous sulphate (FeSO4), often cause unacceptable changes in the colour or taste of foods. Poorly water-soluble compounds, on the other hand, cause fewer sensory changes, but are not well absorbed. Here, we show that poorly water-soluble nanosized Fe and Fe/Zn compounds (specific surface area ∼190 m2 g−1) made by scalable flame aerosol technology have in vivo iron bioavailability in rats comparable to FeSO4 and cause less colour change in reactive food matrices than conventional iron fortificants. The addition of Zn to FePO4 and Mg to Fe/Zn oxide increases Fe absorption from the compounds, and doping with Mg also improves their colour. After feeding rats with nanostructured iron-containing compounds, no stainable Fe was detected in their gut wall, gut-associated lymphatics or other tissues, suggesting no adverse effects. Nanosizing of poorly water-soluble Fe compounds sharply increases their absorption and nutritional value.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Allen, L., de Benoist, B., Dary, O. & Hurrell, R. F. Guidelines on Food Fortification with Micronutrients (World Health Organization, 2006).
Zimmermann, M. B. & Hurrell, R. F. Nutritional iron deficiency. Lancet 370, 511–520 (2007).
International Zinc Nutrition Consultative Group. Assessment of the risk of zinc deficiency and options for its control. Food Nutr. Bull. 25, S91–S204 (2004).
Zimmermann, M. B., Chaouki, N. & Hurrell, R. F. Iron deficiency due to consumption of a habitual diet low in bioavailable iron: a longitudinal cohort study in Moroccan children. Am. J. Clin. Nutr. 81, 115–121 (2005).
Wieringa, F. T. et al. Combined iron and zinc supplementation in infants improved iron and zinc status, but interactions reduced efficacy in a multicountry trial in southeast asia. J. Nutr. 137, 466–471 (2007).
Davidsson, L., Almgren, A., Sandstrom, B. & Hurrell, R. F. Zinc-absorption in adult humans—the effect of iron fortification. Br. J. Nutr. 74, 417–425 (1995).
Hurrell, R. F. Fortification: overcoming technical and practical barriers. J. Nutr. 132, 806S–812S (2002).
Motzok, I., Pennell, M. D., Davies, M. I. & Ross, H. U. Effect of particle-size on biological availability of reduced iron. J. Assoc. Off. Anal. Chem. 58, 99–103 (1975).
Rohner, F. et al. Synthesis, characterization and bioavailability in rats of ferric phosphate nanoparticles. J. Nutr. 137, 614–619 (2007).
Mueller, R., Madler, L. & Pratsinis, S. E. Nanoparticle synthesis at high production rates by flame spray pyrolysis. Chem. Eng. Sci. 58, 1969–1976 (2003).
Hilty, F. M. et al. Development and optimization of iron- and zinc-containing nanostructured powders for nutritional applications. Nanotechnology 20, 475101 (2009).
Lynch, S. R. & Bothwel, T. A comparison of physical properties, screening procedures and a human efficacy trial for predicting the bioavailability of commercial elemental iron powders used for food fortification. Int. J. Vitam. Nutr. Res. 77, 107–124 (2007).
Swain, J. H., Newman, S. M. & Hunt, J. R. Bioavailability of elemental iron powders to rats is less than bakery-grade ferrous sulfate and predicted by iron solubility and particle surface area. J. Nutr. 133, 3546–3552 (2003).
Otten, J. J., Hellwig, J. P. & Mayers, L. D. Dietary Reference Intakes: The Essential Guide to Nutrient Requirements (Institute of Medicine of the National Academies, 2006).
Madler, L., Stark, W. J. & Pratsinis, S. E. Flame-made ceria nanoparticles. J. Mater. Res. 17, 1356–1362 (2002).
Jung, I. H., Decterov, S. A. & Pelton, A. D. Critical thermodynamic evaluation and optimization of the Fe–Mg–O system. J. Phys. Chem. Solids 65, 1683–1695 (2004).
Dheilly, R. M., Tudo, J. & Queneudec, M. Influence of climatic conditions on the carbonation of quicklime. J. Mater. Eng. Perform. 7, 789–795 (1998).
Jossen, R., Pratsinis, S. E., Stark, W. J. & Madler, L. Criteria for flame-spray synthesis of hollow, shell-like or inhomogeneous oxides. J. Am. Ceram. Soc. 88, 1388–1393 (2005).
Cunniff, P. & AOAC. in Official Methods of Analysis of AOAC International 62–63 (AOAC International, 1997).
Forbes, A. L. et al. Comparison of in vitro, animal and clinical determinations of iron bioavailability: International Nutritional Anemia Consultative Group Task Force report on iron bioavailability. Am. J. Clin. Nutr. 49, 225–238 (1989).
Fritz, J. C., Pla, G. W., Harrison, B. N. & Clark, G. A. Collaborative study of rat hemoglobin repletion test for bioavailability of iron. J. Assoc. Off. Anal. Chem. 57, 513–517 (1974).
Cornell, R. M. & Schwertmann, U. The Iron Oxides Structure, Properties, Reactions, Occurences and Uses. 2nd edn (Wiley-VCH, 2003).
Riedel, E. Anorganische Chemie. 4th edn (de Gruyter, 1999).
Yoder, C. H. & Flora, N. J. Geochemical applications of the simple salt approximation to the lattice energies of complex materials. Am. Miner. 90, 488–496 (2005).
Lide, D. R. CRC Handbook of Chemistry and Physics (Chapman & Hall/CRC, 2002).
Ifeacho, P., Wiggers, H. & Roth, P. SnO2/TiO2 mixed oxide particles synthesized in doped premixed H2/O2/Ar flames. Proc. Combust. Inst. 30, 2577–2584 (2005).
Tricoli, A., Righettoni, M. & Pratsinis, S. E. Minimal cross-sensitivity to humidity during ethanol detection by SnO2–TiO2 solid solutions. Nanotechnology 20, 315502 (2009).
Akurati, K. K. et al. One-step flame synthesis of SnO2/TiO2 composite nanoparticles for photocatalytic applications. Int. J. Photoenergy 7, 153–161 (2005).
Vemury, S. & Pratsinis, S. E. Dopants in flame synthesis of titania. J. Am. Ceram. Soc. 78, 2984–2992 (1995).
Jani, P., Halbert, G. W., Langridge, J. & Florence, A. T. Nanoparticle uptake by the rat gastrointestinal mucosa—quantitation and particle-size dependency. J. Pharm. Pharmacol. 42, 821–826 (1990).
Jani, P. U., McCarthy, D. E. & Florence, A. T. Titanium-dioxide (rutile) particle uptake from the rat GI-tract and translocation to systemic organs after oral-administration. Int. J. Pharm. 105, 157–168 (1994).
Wheby, M. S. Site of iron absorption in man. Scand. J. Haematol. 7, 56–62 (1970).
Fleming, M. D. et al. Microcytic anaemia mice have a mutation in nramp2, a candidate iron transporter gene. Nature Genet. 16, 383–386 (1997).
Garrick, M. D. & Garrick, L. M. Cellular iron transport. Biochim. Biophys. Acta—Gen. Subj. 1790, 309–325 (2009).
Wang, J. X. et al. Acute toxicity and biodistribution of different sized titanium dioxide particles in mice after oral administration. Toxicol. Lett. 168, 176–185 (2007).
Hussain, N., Jaitley, V. & Florence, A. T. Recent advances in the understanding of uptake of microparticulates across the gastrointestinal lymphatics. Adv. Drug Deliv. Rev. 50, 107–142 (2001).
Hillyer, J. F. & Albrecht, R. M. Gastrointestinal persorption and tissue distribution of differently sized colloidal gold nanoparticles. J. Pharm. Sci. 90, 1927–1936 (2001).
Karlsson, H. L., Cronholm, P., Gustafsson, J. & Moller, L. Copper oxide nanoparticles are highly toxic: a comparison between metal oxide nanoparticles and carbon nanotubes. Chem. Res. Toxicol. 21, 1726–1732 (2008).
Volkheim. G., Schulz, F. H., Lindenau, A. & Beitz, U. Persorption of metallic iron particles. Gut 10, 32–33 (1969).
Auffan, M. et al. Relation between the redox state of iron-based nanoparticles and their cytotoxicity toward Escherichia coli. Environ. Sci. Technol. 42, 6730–6735 (2008).
Hallberg, L., Brune, M. & Rossander, L. Low bioavailability of carbonyl iron in man—studies on iron fortification of wheat-flour. Am. J. Clin. Nutr. 43, 59–67 (1986).
Hurrell, R. How to ensure adequate iron absorption from iron-fortified food. Nutr. Rev. 60, S7–S15 (2002).
Fidler, M. C., Davidsson, L., Walczyk, T. & Hurrell, R. F. Iron absorption from fish sauce and soy sauce fortified with sodium iron EDTA. Am. J. Clin. Nutr. 78, 274–278 (2003).
Reeves, P. G., Nielsen, F. H. & Fahey, G. C. Ain-93 purified diets for laboratory rodents—final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the ain-76a rodent diet. J. Nutr. 123, 1939–1951 (1993).
Fluttert, M., Dalm, S. & Oitzl, M. S. A refined method for sequential blood sampling by tail incision in rats. Lab. Anim. 34, 372–378 (2000).
Amine, E. K., Hegsted, D. M. & Neff, R. Biological estimation of available iron using chicks or rats. J. Agric. Food Chem. 20, 246–251 (1972).
Finney, D. J. in Statistical Methods in Biological Assay 187–213 (Charles Griffin & Company, 1978).
Acknowledgements
The authors would like to thank L. Berger (ETH, Zurich) for her technical assistance, M. Haldimann (The Ministry of Health, Bern, Switzerland) for the ICP-MS measurements, F. Krumeich (Electron Microscopy Center ETH, Zurich) for the TEM images of the Fe powders, and L. Molinari (Children's Hospital, Zurich) for support in statistical analysis. We thank P. Lohmann GmbH (Emmerthal, Germany) for providing the FeSO4 for the study at no cost, and BHA Technologies (Muemliswil, Switzerland) for supplying the Teflon filters at no cost. The study was supported by the Swiss National Science Foundation, Bern, Switzerland; the Swiss Commission for Technology and Innovation (KTI Projekt 9765.1 PFLS-LS), Bern, Switzerland; IMP (Industrial Metal Powders (India) Pvt Ltd) and ETH Zurich, Switzerland.
Author information
Authors and Affiliations
Contributions
F.M.H., M.B.Z., R.F.H. and S.E.P. conceived the experiments. F.M.H., M.B.Z. and W.L. designed the experiments. F.M.H. produced the compounds tested in the rat study. J.T.N.K. produced Mg containing oxides without Zn. F.M.H. and M.A. performed the animal study. M.H. and F.E. performed the histological analysis. F.M.H., A.T. and M.H. analysed the data. F.M.H., A.T., R.F.H. S.E.P., W.L. and M.B.Z. co-wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 560 kb)
Rights and permissions
About this article
Cite this article
Hilty, F., Arnold, M., Hilbe, M. et al. Iron from nanocompounds containing iron and zinc is highly bioavailable in rats without tissue accumulation. Nature Nanotech 5, 374–380 (2010). https://doi.org/10.1038/nnano.2010.79
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2010.79
This article is cited by
-
Biological Selenium Nanoparticles in Quail Nutrition: Biosynthesis and its Impact on Performance, Carcass, Blood Chemistry, and Cecal Microbiota
Biological Trace Element Research (2023)
-
Iron from nanostructured ferric phosphate: absorption and biodistribution in mice and bioavailability in iron deficient anemic women
Scientific Reports (2022)
-
Iron nanoparticles as a promising compound for food fortification in iron deficiency anemia: a review
Journal of Food Science and Technology (2022)
-
Biocompatible FePO4 Nanoparticles: Drug Delivery, RNA Stabilization, and Functional Activity
Nanoscale Research Letters (2021)
-
Intestinal and hepatic effects of iron oxide nanoparticles
Archives of Toxicology (2021)