Abstract
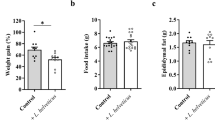
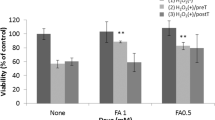
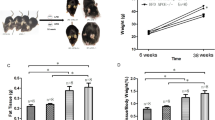
Many sight-threatening diseases have two critical phases, vessel loss followed by hypoxia-driven destructive neovascularization. These diseases include retinopathy of prematurity and diabetic retinopathy, leading causes of blindness in childhood and middle age affecting over 4 million people in the United States. We studied the influence of ω-3- and ω-6-polyunsaturated fatty acids (PUFAs) on vascular loss, vascular regrowth after injury, and hypoxia-induced pathological neovascularization in a mouse model of oxygen-induced retinopathy1. We show that increasing ω-3-PUFA tissue levels by dietary or genetic means decreased the avascular area of the retina by increasing vessel regrowth after injury, thereby reducing the hypoxic stimulus for neovascularization. The bioactive ω-3-PUFA-derived mediators neuroprotectinD1, resolvinD1 and resolvinE1 also potently protected against neovascularization. The protective effect of ω-3-PUFAs and their bioactive metabolites was mediated, in part, through suppression of tumor necrosis factor-α. This inflammatory cytokine was found in a subset of microglia that was closely associated with retinal vessels. These findings indicate that increasing the sources of ω-3-PUFA or their bioactive products reduces pathological angiogenesis. Western diets are often deficient in ω-3-PUFA, and premature infants lack the important transfer from the mother to the infant of ω-3-PUFA that normally occurs in the third trimester of pregnancy2. Supplementing ω-3-PUFA intake may be of benefit in preventing retinopathy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Smith, L.E. et al. Oxygen-induced retinopathy in the mouse. Invest. Ophthalmol. Vis. Sci. 35, 101–111 (1994).
Crawford, M.A. et al. Are deficits of arachidonic and docosahexaenoic acids responsible for the neural and vascular complications of preterm babies? Am. J. Clin. Nutr. 66, 1032S–1041S (1997).
Kermorvant-Duchemin, E. et al. Trans-arachidonic acids generated during nitrative stress induce a thrombospondin-1-dependent microvascular degeneration. Nat. Med. 11, 1339–1345 (2005).
Fierro, I.M., Kutok, J.L. & Serhan, C.N. Novel lipid mediator regulators of endothelial cell proliferation and migration: aspirin-triggered-15R-lipoxin A(4) and lipoxin A(4). J. Pharmacol. Exp. Ther. 300, 385–392 (2002).
Fliesler, S.J. & Anderson, R.E. Chemistry and metabolism of lipids in the vertebrate retina. Prog. Lipid Res. 22, 79–131 (1983).
SanGiovanni, J.P. & Chew, E.Y. The role of omega-3 long-chain polyunsaturated fatty acids in health and disease of the retina. Prog. Retin. Eye Res. 24, 87–138 (2005).
Calder, P.C. Polyunsaturated fatty acids and inflammation. Prostaglandins Leukot. Essent. Fatty Acids 75, 197–202 (2006).
Salem, N., Jr., Litman, B., Kim, H.Y. & Gawrisch, K. Mechanisms of action of docosahexaenoic acid in the nervous system. Lipids 36, 945–959 (2001).
Serhan, C.N. & Savill, J. Resolution of inflammation: the beginning programs the end. Nat. Immunol. 6, 1191–1197 (2005).
Kang, J.X., Wang, J., Wu, L. & Kang, Z.B. Transgenic mice: fat-1 mice convert n-6 to n-3 fatty acids. Nature 427, 504 (2004).
Moriguchi, T. et al. Effects of an n-3-deficient diet on brain, retina, and liver fatty acyl composition in artificially reared rats. J. Lipid Res. 45, 1437–1445 (2004).
Serhan, C.N., Arita, M., Hong, S. & Gotlinger, K. Resolvins, docosatrienes, and neuroprotectins, novel omega-3-derived mediators, and their endogenous aspirin-triggered epimers. Lipids 39, 1125–1132 (2004).
Hong, S., Gronert, K., Devchand, P.R., Moussignac, R.L. & Serhan, C.N. Novel docosatrienes and 17S-resolvins generated from docosahexaenoic acid in murine brain, human blood, and glial cells. Autacoids in anti-inflammation. J. Biol. Chem. 278, 14677–14687 (2003).
Tjonahen, E. et al. Resolvin E2: identification and anti-inflammatory actions: pivotal role of human 5-lipoxygenase in resolvin E series biosynthesis. Chem. Biol. 13, 1193–1202 (2006).
Arita, M. et al. Stereochemical assignment, antiinflammatory properties, and receptor for the omega-3 lipid mediator resolvin E1. J. Exp. Med. 201, 713–722 (2005).
Meder, W. et al. Characterization of human circulating TIG2 as a ligand for the orphan receptor ChemR23. FEBS Lett. 555, 495–499 (2003).
Goukassian, D.A. et al. Tumor necrosis factor-α receptor p75 is required in ischemia-induced neovascularization. Circulation 115, 752–762 (2007).
Kishore, R. et al. The cytoskeletal protein ezrin regulates EC proliferation and angiogenesis via TNF-α–induced transcriptional repression of cyclin A. J. Clin. Invest. 115, 1785–1796 (2005).
Gardiner, T.A. et al. Inhibition of tumor necrosis factor-α improves physiological angiogenesis and reduces pathological neovascularization in ischemic retinopathy. Am. J. Pathol. 166, 637–644 (2005).
Vassalli, P. The pathophysiology of tumor necrosis factors. Annu. Rev. Immunol. 10, 411–452 (1992).
Ritter, M.R. et al. Myeloid progenitors differentiate into microglia and promote vascular repair in a model of ischemic retinopathy. J. Clin. Invest. 116, 3266–3276 (2006).
Checchin, D., Sennlaub, F., Levavasseur, E., Leduc, M. & Chemtob, S. Potential role of microglia in retinal blood vessel formation. Invest. Ophthalmol. Vis. Sci. 47, 3595–3602 (2006).
Collart, M.A., Baeuerle, P. & Vassalli, P. Regulation of tumor necrosis factor alpha transcription in macrophages: involvement of four κB-like motifs and of constitutive and inducible forms of NF-κB. Mol. Cell. Biol. 10, 1498–1506 (1990).
Millet, I. et al. Inhibition of NF-κB activity and enhancement of apoptosis by the neuropeptide calcitonin gene-related peptide. J. Biol. Chem. 275, 15114–15121 (2000).
Duffield, J.S. et al. Resolvin D series and protectin D1 mitigate acute kidney injury. J. Immunol. 177, 5902–5911 (2006).
Mukherjee, P.K., Marcheselli, V.L., Serhan, C.N. & Bazan, N.G. Neuroprotectin D1: a docosahexaenoic acid-derived docosatriene protects human retinal pigment epithelial cells from oxidative stress. Proc. Natl. Acad. Sci. USA 101, 8491–8496 (2004).
Arita, M. et al. Resolvin E1, an endogenous lipid mediator derived from omega-3 eicosapentaenoic acid, protects against 2,4,6-trinitrobenzene sulfonic acid-induced colitis. Proc. Natl. Acad. Sci. USA 102, 7671–7676 (2005).
Aiello, L.P. et al. Suppression of retinal neovascularization in vivo by inhibition of vascular endothelial growth factor (VEGF) using soluble VEGF-receptor chimeric proteins. Proc. Natl. Acad. Sci. USA 92, 10457–10461 (1995).
Bannenberg, G.L. et al. Molecular circuits of resolution: formation and actions of resolvins and protectins. J. Immunol. 174, 4345–4355 (2005).
Grounds, M.D. et al. Silencing TNFα activity by using Remicade or Enbrel blocks inflammation in whole muscle grafts: an in vivo bioassay to assess the efficacy of anti-cytokine drugs in mice. Cell Tissue Res. 320, 509–515 (2005).
Acknowledgements
We thank C. DiMartino, N. Liu, J.-R. Mo and K. Percarpio for technical help and J.-Y. Tsai for discussions. We thank the US National Institutes of Health Office of Dietary Supplements. This research was generously supported by the V. Kann Rasmussen Foundation and the US National Institutes of Health (EY008670, EY017017, EY14811 (L.E.H.S.); 1F32 EY017789, 5 T32 EY07145 (K.M.C.); P50-DE016191, GM38765 (C.N.S.); and Children's Hospital Boston Mental Retardation and Developmental Disabilities Research Center, P01 HD18655). We thank the Juvenile Diabetes Research Foundation for fellowship support (J.C.). This work was also supported by the Research to Prevent Blindness Lew Wasserman Merit Award (L.E.H.S.). The sponsors had no role in the design or conduct of the study, in the collection, analysis and interpretation of data or in the preparation, review or approval of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
C.N.S. is an inventor on patents held by Brigham and Women's Hospital that relate to novel composition of matter: isolation, characterization, and use in treating diseases. These patents are the subject of licensing agreements for Brigham and Women's Hospital and consultantships related to clinical development for C.N.S.
Supplementary information
Supplementary Fig. 1
Normal vascular regrowth. (PDF 16 kb)
Supplementary Fig. 2
Intraocular injections of the TNF-α receptor fusion protein in ω-6 fed mice. (PDF 85 kb)
Supplementary Fig. 3
DHA and EPA suppress TNF-α mRNA induction in LPS-activated macrophages. (PDF 54 kb)
Supplementary Table 1
Omega-6 and omega-3 PUFA diet analysis. (PDF 24 kb)
Supplementary Table 2
Fat-1 mouse diet analysis. (PDF 13 kb)
Supplementary Table 3
Resolvin and neuroprotectin detection in mice on an ω-6 or ω-3 PUFA diet. (PDF 16 kb)
Supplementary Table 4
Feed lot analysis. (PDF 19 kb)
Rights and permissions
About this article
Cite this article
Connor, K., SanGiovanni, J., Lofqvist, C. et al. Increased dietary intake of ω-3-polyunsaturated fatty acids reduces pathological retinal angiogenesis. Nat Med 13, 868–873 (2007). https://doi.org/10.1038/nm1591
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1591
This article is cited by
-
ω-3PUFAs Inhibit Hypoxia-Induced Retinal Neovascularization via Regulating Microglial Pyroptosis through METTL14-Mediated m6A Modification of IFNB1 mRNA
Applied Biochemistry and Biotechnology (2024)
-
HIF1α-dependent hypoxia response in myeloid cells requires IRE1α
Journal of Neuroinflammation (2023)
-
Retinopathy of prematurity: from oxygen management to molecular manipulation
Molecular and Cellular Pediatrics (2023)
-
Study protocol of OmegaROP-2 prospective study: expression of placental fatty acid receptors in preterm newborns with retinopathy of prematurity
BMC Ophthalmology (2023)
-
Improvement of retinal function in Alzheimer disease-associated retinopathy by dietary lysophosphatidylcholine-EPA/DHA
Scientific Reports (2023)