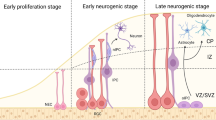
Abstract
Epigenetic mechanisms regulate cell differentiation during embryonic development and also serve as important interfaces between genes and the environment in adulthood. Neurogenesis in adults, which generates functional neural cell types from adult neural stem cells, is dynamically regulated by both intrinsic state-specific cell differentiation cues and extrinsic neural niche signals. Epigenetic regulation by DNA and histone modifiers, non-coding RNAs and other self-sustained mechanisms can lead to relatively long-lasting biological effects and maintain functional neurogenesis throughout life in discrete regions of the mammalian brain. Here, we review recent evidence that epigenetic mechanisms carry out diverse roles in regulating specific aspects of adult neurogenesis and highlight the implications of such epigenetic regulation for neural plasticity and disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Cameron, H.A. & McKay, R.D. Adult neurogenesis produces a large pool of new granule cells in the dentate gyrus. J. Comp. Neurol. 435, 406–417 (2001).
Altman, J. & Das, G.D. Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J. Comp. Neurol. 124, 319–335 (1965).
Gage, F.H. Mammalian neural stem cells. Science 287, 1433–1438 (2000).
Alvarez-Buylla, A. & Lim, D.A. For the long run: maintaining germinal niches in the adult brain. Neuron 41, 683–686 (2004).
Ming, G.L. & Song, H. Adult neurogenesis in the mammalian central nervous system. Annu. Rev. Neurosci. 28, 223–250 (2005).
Lledo, P.M., Alonso, M. & Grubb, M.S. Adult neurogenesis and functional plasticity in neuronal circuits. Nat. Rev. Neurosci. 7, 179–193 (2006).
Zhao, C., Deng, W. & Gage, F.H. Mechanisms and functional implications of adult neurogenesis. Cell 132, 645–660 (2008).
Ma, D.K., Bonaguidi, M.A., Ming, G.L. & Song, H. Adult neural stem cells in the mammalian central nervous system. Cell Res. 19, 672–682 (2009).
Duan, X., Kang, E., Liu, C.Y., Ming, G.L. & Song, H. Development of neural stem cell in the adult brain. Curr. Opin. Neurobiol. 18, 108–115 (2008).
Kempermann, G., Wiskott, L. & Gage, F.H. Functional significance of adult neurogenesis. Curr. Opin. Neurobiol. 14, 186–191 (2004).
Deng, W., Aimone, J.B. & Gage, F.H. New neurons and new memories: how does adult hippocampal neurogenesis affect learning and memory? Nat. Rev. Neurosci. 11, 339–350 (2010).
Kitabatake, Y., Sailor, K.A., Ming, G.L. & Song, H. Adult neurogenesis and hippocampal memory function: new cells, more plasticity, new memories? Neurosurg. Clin. N. Am. 18, 105–113 (2007).
Holliday, R. Mechanisms for the control of gene activity during development. Biol. Rev. Camb. Philos. Soc. 65, 431–471 (1990).
Goldberg, A.D., Allis, C.D. & Bernstein, E. Epigenetics: a landscape takes shape. Cell 128, 635–638 (2007).
Waddington, C.H. Principles of Development and Differentiation (Macmillan, New York, 1966).
Holliday, R. Epigenetics: a historical overview. Epigenetics 1, 76–80 (2006).
Jaenisch, R. & Bird, A. Epigenetic regulation of gene expression: how the genome integrates intrinsic and environmental signals. Nat. Genet. 33 Suppl., 245–254 (2003).
Zhang, T.Y. & Meaney, M.J. Epigenetics and the environmental regulation of the genome and its function. Annu. Rev. Psychol. 61, 439–466 (2010).
Covic, M., Karaca, E. & Lie, D.C. Epigenetic regulation of neurogenesis in the adult hippocampus. Heredity 105, 122–134 (2010).
Hsieh, J. & Eisch, A.J. Epigenetics, hippocampal neurogenesis and neuropsychiatric disorders: unraveling the genome to understand the mind. Neurobiol. Dis. 39, 73–84 (2010).
Ma, D.K., Kim, W.R., Ming, G.L. & Song, H. Activity-dependent extrinsic regulation of adult olfactory bulb and hippocampal neurogenesis. Ann. NY Acad. Sci. 1170, 664–673 (2009).
Cedar, H. & Bergman, Y. Linking DNA methylation and histone modification: patterns and paradigms. Nat. Rev. Genet. 10, 295–304 (2009).
Holliday, R. Epigenetic inheritance based on DNA methylation. EXS 64, 452–468 (1993).
Kriaucionis, S. & Heintz, N. The nuclear DNA base 5-hydroxymethylcytosine is present in Purkinje neurons and the brain. Science 324, 929–930 (2009).
Tahiliani, M. et al. Conversion of 5-methylcytosine to 5-hydroxymethylcytosine in mammalian DNA by MLL partner TET1. Science 324, 930–935 (2009).
Ito, S. et al. Role of Tet proteins in 5mC to 5hmC conversion, ES-cell self-renewal and inner cell mass specification. Nature 466, 1129–1133 (2010).
Jenuwein, T. & Allis, C.D. Translating the histone code. Science 293, 1074–1080 (2001).
Serio, T.R. & Lindquist, S.L. Protein-only inheritance in yeast: something to get [PSI+]-ched about. Trends Cell Biol. 10, 98–105 (2000).
Ptashne, M. On the use of the word 'epigenetic'. Curr. Biol. 17, R233–R236 (2007).
Capela, A. & Temple, S. LeX/ssea-1 is expressed by adult mouse CNS stem cells, identifying them as nonependymal. Neuron 35, 865–875 (2002).
Pastrana, E., Cheng, L.C. & Doetsch, F. Simultaneous prospective purification of adult subventricular zone neural stem cells and their progeny. Proc. Natl. Acad. Sci. USA 106, 6387–6392 (2009).
Kanki, H., Shimabukuro, M.K., Miyawaki, A. & Okano, H. “Color Timer” mice: visualization of neuronal differentiation with fluorescent proteins. Mol. Brain 2, 3 (2010).
Encinas, J.M. & Enikolopov, G. Identifying and quantitating neural stem and progenitor cells in the adult brain. Methods Cell Biol. 85, 243–272 (2008).
Heiman, M. et al. A translational profiling approach for the molecular characterization of CNS cell types. Cell 135, 738–748 (2008).
Goren, A. et al. Chromatin profiling by directly sequencing small quantities of immunoprecipitated DNA. Nat. Methods 7, 47–49 (2010).
O'Neill, L.P., VerMilyea, M.D. & Turner, B.M. Epigenetic characterization of the early embryo with a chromatin immunoprecipitation protocol applicable to small cell populations. Nat. Genet. 38, 835–841 (2006).
Ball, M.P. et al. Targeted and genome-scale strategies reveal gene-body methylation signatures in human cells. Nat. Biotechnol. 27, 361–368 (2009).
Lister, R. et al. Human DNA methylomes at base resolution show widespread epigenomic differences. Nature 462, 315–322 (2009).
Bernstein, B.E. et al. A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125, 315–326 (2006).
Roh, T.Y., Ngau, W.C., Cui, K., Landsman, D. & Zhao, K. High-resolution genome-wide mapping of histone modifications. Nat. Biotechnol. 22, 1013–1016 (2004).
Jin, S.G., Kadam, S. & Pfeifer, G.P. Examination of the specificity of DNA methylation profiling techniques towards 5-methylcytosine and 5-hydroxymethylcytosine. Nucleic Acids Res. 38, e125 (2010).
Molofsky, A.V. et al. Bmi-1 dependence distinguishes neural stem cell self-renewal from progenitor proliferation. Nature 425, 962–967 (2003).
Ringrose, L. & Paro, R. Polycomb/Trithorax response elements and epigenetic memory of cell identity. Development 134, 223–232 (2007).
Ng, R.K. & Gurdon, J.B. Epigenetic inheritance of cell differentiation status. Cell Cycle 7, 1173–1177 (2008).
Fasano, C.A. et al. Bmi-1 cooperates with Foxg1 to maintain neural stem cell self-renewal in the forebrain. Genes Dev. 23, 561–574 (2009).
Fasano, C.A. et al. shRNA knockdown of Bmi-1 reveals a critical role for p21-Rb pathway in NSC self-renewal during development. Cell Stem Cell 1, 87–99 (2007).
He, S. et al. Bmi-1 over-expression in neural stem/progenitor cells increases proliferation and neurogenesis in culture but has little effect on these functions in vivo. Dev. Biol. 328, 257–272 (2009).
Zhao, X. et al. Mice lacking methyl-CpG binding protein 1 have deficits in adult neurogenesis and hippocampal function. Proc. Natl. Acad. Sci. USA 100, 6777–6782 (2003).
Martín Caballero, I., Hansen, J., Leaford, D., Pollard, S. & Hendrich, B.D. The methyl-CpG binding proteins Mecp2, Mbd2 and Kaiso are dispensable for mouse embryogenesis, but play a redundant function in neural differentiation. PLoS ONE 4, e4315 (2009).
Li, X. et al. Epigenetic regulation of the stem cell mitogen Fgf-2 by Mbd1 in adult neural stem/progenitor cells. J. Biol. Chem. 283, 27644–27652 (2008).
Episkopou, V. SOX2 functions in adult neural stem cells. Trends Neurosci. 28, 219–221 (2005).
Suh, H. et al. In vivo fate analysis reveals the multipotent and self-renewal capacities of Sox2+ neural stem cells in the adult hippoampus. Cell Stem Cell 1, 515–528 (2007).
Boyer, L.A. et al. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell 122, 947–956 (2005).
Jaenisch, R. & Young, R. Stem cells, the molecular circuitry of pluripotency and nuclear reprogramming. Cell 132, 567–582 (2008).
Hu, Q. et al. The EGF receptor-sox2-EGF receptor feedback loop positively regulates the self-renewal of neural precursor cells. Stem Cells 28, 279–286 (2010).
Lim, D.A. et al. Chromatin remodeling factor Mll1 is essential for neurogenesis from postnatal neural stem cells. Nature 458, 529–533 (2009).
Jepsen, K. et al. SMRT-mediated repression of an H3K27 demethylase in progression from neural stem cell to neuron. Nature 450, 415–419 (2007).
Hsieh, J., Nakashima, K., Kuwabara, T., Mejia, E. & Gage, F.H. Histone deacetylase inhibition-mediated neuronal differentiation of multipotent adult neural progenitor cells. Proc. Natl. Acad. Sci. USA 101, 16659–16664 (2004).
Sun, G., Yu, R.T., Evans, R.M. & Shi, Y. Orphan nuclear receptor TLX recruits histone deacetylases to repress transcription and regulate neural stem cell proliferation. Proc. Natl. Acad. Sci. USA 104, 15282–15287 (2007).
Jawerka, M. et al. The specific role of histone deacetylase 2 in adult neurogenesis. Neuron Glia Biol. published online, doi:10.1017/S1740925X10000049 (14 April 2010).
Crepaldi, L. & Riccio, A. Chromatin learns to behave. Epigenetics 4, 23–26 (2009).
Zocchi, L. & Sassone-Corsi, P. Joining the dots: from chromatin remodeling to neuronal plasticity. Curr. Opin. Neurobiol. 20, 432–440 (2010).
Wang, Z. et al. Genome-wide mapping of HATs and HDACs reveals distinct functions in active and inactive genes. Cell 138, 1019–1031 (2009).
Morris, K.V. Non-coding RNAs, epigenetic memory and the passage of information to progeny. RNA Biol. 6, 242–247 (2009).
Hobert, O. Gene regulation by transcription factors and microRNAs. Science 319, 1785–1786 (2008).
Iorio, M.V., Piovan, C. & Croce, C.M. Interplay between microRNAs and the epigenetic machinery: an intricate network. Biochim. Biophys. Acta published online, doi:10.1016/j.bbagrm.2010.05.005 (20 May 2010).
Liu, C. et al. Epigenetic regulation of miR-184 by MBD1 governs neural stem cell proliferation and differentiation. Cell Stem Cell 6, 433–444 (2010).
Szulwach, K.E. et al. Cross talk between microRNA and epigenetic regulation in adult neurogenesis. J. Cell Biol. 189, 127–141 (2010).
Smrt, R.D. et al. MicroRNA miR-137 regulates neuronal maturation by targeting ubiquitin ligase mind bomb-1. Stem Cells 28, 1060–1070 (2010).
Cheng, L.C., Pastrana, E., Tavazoie, M. & Doetsch, F. miR-124 regulates adult neurogenesis in the subventricular zone stem cell niche. Nat. Neurosci. 12, 399–408 (2009).
Kuwabara, T., Hsieh, J., Nakashima, K., Taira, K. & Gage, F.H. A small modulatory dsRNA specifies the fate of adult neural stem cells. Cell 116, 779–793 (2004).
Juliandi, B., Abematsu, M. & Nakashima, K. Chromatin remodeling in neural stem cell differentiation. Curr. Opin. Neurobiol. 20, 408–415 (2010).
Yoo, A.S., Staahl, B.T., Chen, L. & Crabtree, G.R. MicroRNA-mediated switching of chromatin-remodelling complexes in neural development. Nature 460, 642–646 (2009).
Coufal, N.G. et al. L1 retrotransposition in human neural progenitor cells. Nature 460, 1127–1131 (2009).
Muotri, A.R. et al. Somatic mosaicism in neuronal precursor cells mediated by L1 retrotransposition. Nature 435, 903–910 (2005).
Muotri, A.R. & Gage, F.H. Generation of neuronal variability and complexity. Nature 441, 1087–1093 (2006).
Kuwabara, T. et al. Wnt-mediated activation of NeuroD1 and retro-elements during adult neurogenesis. Nat. Neurosci. 12, 1097–1105 (2009).
Muotri, A.R., Zhao, C., Marchetto, M.C. & Gage, F.H. Environmental influence on L1 retrotransposons in the adult hippocampus. Hippocampus 19, 1002–1007 (2009).
Ma, D.K., Ming, G.L. & Song, H. Glial influences on neural stem cell development: cellular niches for adult neurogenesis. Curr. Opin. Neurobiol. 15, 514–520 (2005).
Ma, D.K. et al. Neuronal activity-induced Gadd45b promotes epigenetic DNA demethylation and adult neurogenesis. Science 323, 1074–1077 (2009).
Ma, D.K., Guo, J.U., Ming, G.L. & Song, H. DNA excision repair proteins and Gadd45 as molecular players for active DNA demethylation. Cell Cycle 8, 1526–1531 (2009).
Martinowich, K. et al. DNA methylation-related chromatin remodeling in activity-dependent BDNF gene regulation. Science 302, 890–893 (2003).
Zhou, Z. et al. Brain-specific phosphorylation of MeCP2 regulates activity-dependent Bdnf transcription, dendritic growth and spine maturation. Neuron 52, 255–269 (2006).
He, D.Y., Neasta, J. & Ron, D. Epigenetic regulation of BDNF expression via the scaffolding protein rack1. J. Biol. Chem. 285, 19043–19050 (2010).
Smrt, R.D. et al. Mecp2 deficiency leads to delayed maturation and altered gene expression in hippocampal neurons. Neurobiol. Dis. 27, 77–89 (2007).
Feng, J. et al. Dnmt1 and Dnmt3a maintain DNA methylation and regulate synaptic function in adult forebrain neurons. Nat. Neurosci. 13, 423–430 (2010).
Qiu, Z. & Ghosh, A. A calcium-dependent switch in a CREST-BRG1 complex regulates activity-dependent gene expression. Neuron 60, 775–787 (2008).
Mu, Y., Lee, S.W. & Gage, F.H. Signaling in adult neurogenesis. Curr. Opin. Neurobiol. 20, 416–423 (2010).
Dirks, P. Bmi1 and cell of origin determinants of brain tumor phenotype. Cancer Cell 12, 295–297 (2007).
Nagarajan, R.P. & Costello, J.F. Epigenetic mechanisms in glioblastoma multiforme. Semin. Cancer Biol. 19, 188–197 (2009).
Banasr, M. & Duman, R.S. Regulation of neurogenesis and gliogenesis by stress and antidepressant treatment. CNS Neurol. Disord. Drug Targets 6, 311–320 (2007).
Sahay, A. & Hen, R. Adult hippocampal neurogenesis in depression. Nat. Neurosci. 10, 1110–1115 (2007).
Allan, A.M. et al. The loss of methyl-CpG binding protein 1 leads to autism-like behavioral deficits. Hum. Mol. Genet. 17, 2047–2057 (2008).
Li, Y. et al. TrkB regulates hippocampal neurogenesis and governs sensitivity to antidepressive treatment. Neuron 59, 399–412 (2008).
Roth, T.L. & Sweatt, J.D. Epigenetic marking of the BDNF gene by early-life adverse experiences. Horm. Behav. published online, doi:10.1016/j.yhbeh.2010.05.005 (17 May 2010).
Kuzumaki, N. et al. Hippocampal epigenetic modification at the brain-derived neurotrophic factor gene induced by an enriched environment. Hippocampus published online, doi:10.1002/hipo.20775 (15 March 2010).
Nelson, E.D., Kavalali, E.T. & Monteggia, L.M. Activity-dependent suppression of miniature neurotransmission through the regulation of DNA methylation. J. Neurosci. 28, 395–406 (2008).
Bath, K.G. et al. Variant brain-derived neurotrophic factor (Val66Met) alters adult olfactory bulb neurogenesis and spontaneous olfactory discrimination. J. Neurosci. 28, 2383–2393 (2008).
Chen, Z.Y. et al. Genetic variant BDNF (Val66Met) polymorphism alters anxiety-related behavior. Science 314, 140–143 (2006).
Christian, K., Song, H. & Ming, G.L. Adult neurogenesis as a cellular model to study schizophrenia. Cell Cycle 9, 636–637 (2010).
Acknowledgements
We thank M.L. Gage and K. Christian for comments on the manuscript and J. Simon for illustrations. This work was supported by the National Institutes of Health grants NS048271 and HD069184 (G.-l.M.), MH088485 and MH090258 (F.H.G.), and AG024984, NS047344 and MH087874 (H.S.), the Brain Science Institute at Johns Hopkins and National Alliance for Research on Schizophrenia and Depression (G.-l.M.), the Mathers Foundation, the Lookout Fund and the J.S. McDonnell Foundation (F.H.G.), a Helen Hay Whitney Foundation postdoctoral fellowship (D.K.M.), and the Dr. Richard and Mavis Fowler and the Foundation for Advanced Research in the Medical Sciences Fellowship (J.U.G.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Ma, D., Marchetto, M., Guo, J. et al. Epigenetic choreographers of neurogenesis in the adult mammalian brain. Nat Neurosci 13, 1338–1344 (2010). https://doi.org/10.1038/nn.2672
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2672
This article is cited by
-
Computational analysis of crosstalk between transcriptional regulators and RNA-binding proteins suggests mutual regulation of polycomb proteins and SRSF1 influencing adult hippocampal neurogenesis
Discover Mental Health (2023)
-
The role of m6A modification in the biological functions and diseases
Signal Transduction and Targeted Therapy (2021)
-
Epigenetic mechanisms in schizophrenia and other psychotic disorders: a systematic review of empirical human findings
Molecular Psychiatry (2020)
-
The Impact of Estradiol on Neurogenesis and Cognitive Functions in Alzheimer’s Disease
Cellular and Molecular Neurobiology (2020)
-
Primary microcephaly with an unstable genome
Genome Instability & Disease (2020)