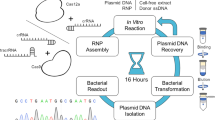
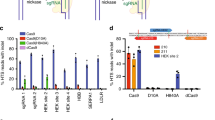
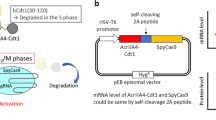
Abstract
Methods to introduce targeted double-strand breaks (DSBs) into DNA enable precise genome editing by increasing the rate at which externally supplied DNA fragments are incorporated into the genome through homologous recombination. The efficiency of these methods is limited by nonhomologous end joining (NHEJ), an alternative DNA repair pathway that competes with homology-directed repair (HDR). To promote HDR at the expense of NHEJ, we targeted DNA ligase IV, a key enzyme in the NHEJ pathway, using the inhibitor Scr7. Scr7 treatment increased the efficiency of HDR-mediated genome editing, using Cas9 in mammalian cell lines and in mice for all four genes examined, up to 19-fold. This approach should be applicable to other customizable endonucleases, such as zinc finger nucleases and transcription activator–like effector nucleases, and to nonmammalian cells with sufficiently conserved mechanisms of NHEJ and HDR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Change history
20 April 2015
In the version of this article initially published online, the received date of the paper was given as 17 October 2014; the correct date is 11 July 2014. On p.4, left column, second paragraph, 1 μM Scr7 should have been 1 mM Scr7; in Online Methods, second paragraph, Cas9 mRNA, sgRNA and template oligos were erroneously given in ng/ml rather than ng/μl. A technique was misidentified as ChIP; in all cases it should be “chip.” The possibility of using Scr7 as a means of enhancing homology-directed repair was recently also mentioned in a review article that was published while our manuscript was under review. A reference to this article has been added to the manuscript. Finally, the P values in Table 1 for Kell and Lgkc were reversed and should be *P<0.05 (Igkc), ***P<0.005 (Kell) and not *P<0.05 (Kell), ***P<0.005 (Igkc). The errors have been corrected for the print, PDF and HTML versions of this article.
References
Wiedenheft, B., Sternberg, S.H. & Doudna, J.A. RNA-guided genetic silencing systems in bacteria and archaea. Nature 482, 331–338 (2012).
Sander, J.D. & Joung, J.K. CRISPR-Cas systems for editing, regulating and targeting genomes. Nat. Biotechnol. 32, 347–355 (2014).
Jiang, W., Bikard, D., Cox, D., Zhang, F. & Marraffini, L.A. RNA-guided editing of bacterial genomes using CRISPR-Cas systems. Nat. Biotechnol. 31, 233–239 (2013).
Wang, T., Wei, J.J., Sabatini, D.M. & Lander, E.S. Genetic screens in human cells using the CRISPR-Cas9 system. Science 343, 80–84 (2014).
Yang, H. et al. One-step generation of mice carrying reporter and conditional alleles by CRISPR/Cas-mediated genome engineering. Cell 154, 1370–1379 (2013).
Wang, H. et al. One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell 153, 910–918 (2013).
Wu, Y. et al. Correction of a genetic disease in mouse via use of CRISPR-Cas9. Cell Stem Cell 13, 659–662 (2013).
Yin, H. et al. Genome editing with Cas9 in adult mice corrects a disease mutation and phenotype. Nat. Biotechnol. 32, 551–553 (2014).
Mali, P. et al. RNA-guided human genome engineering via Cas9. Science 339, 823–826 (2013).
Panier, S. & Boulton, S.J. Double-strand break repair: 53BP1 comes into focus. Nat. Rev. Mol. Cell Biol. 15, 7–18 (2014).
Pierce, A.J., Hu, P., Han, M., Ellis, N. & Jasin, M. Ku DNA end-binding protein modulates homologous repair of double-strand breaks in mammalian cells. Genes Dev. 15, 3237–3242 (2001).
Boboila, C. et al. Alternative end-joining catalyzes class switch recombination in the absence of both Ku70 and DNA ligase 4. J. Exp. Med. 207, 417–427 (2010).
Frit, P., Barboule, N., Yuan, Y., Gomez, D. & Calsou, P. Alternative end-joining pathway(s): bricolage at DNA breaks. DNA Repair (Amst.) 17, 81–97 (2014).
Beumer, K.J., Trautman, J.K., Mukherjee, K. & Carroll, D. Donor DNA Utilization during gene targeting with zinc-finger nucleases. G3 3, 657–664 (2013).
Adachi, N., Ishino, T., Ishii, Y., Takeda, S. & Koyama, H. DNA ligase IV-deficient cells are more resistant to ionizing radiation in the absence of Ku70: Implications for DNA double-strand break repair. Proc. Natl. Acad. Sci. USA 98, 12109–12113 (2001).
Frank, K.M. et al. Late embryonic lethality and impaired V(D)J recombination in mice lacking DNA ligase IV. Nature 396, 173–177 (1998).
Srivastava, M. et al. An inhibitor of nonhomologous end-joining abrogates double-strand break repair and impedes cancer progression. Cell 151, 1474–1487 (2012).
Sanyal, S. et al. Type I interferon imposes a TSG101/ISG15 checkpoint at the Golgi for glycoprotein trafficking during influenza virus infection. Cell Host Microbe 14, 510–521 (2013).
Van Kaer, L., Ashton-Rickardt, P.G., Ploegh, H.L. & Tonegawa, S. TAP1 mutant mice are deficient in antigen presentation, surface class I molecules, and CD4–8+ T cells. Cell 71, 1205–1214 (1992).
Christianson, J.C., Shaler, T.A., Tyler, R.E. & Kopito, R.R. OS-9 and GRP94 deliver mutant alpha1-antitrypsin to the Hrd1–SEL1L ubiquitin ligase complex for ERAD. Nat. Cell Biol. 10, 272–282 (2008).
Lee, C.C., Freinkman, E., Sabatini, D.M. & Ploegh, H.L. The protein synthesis inhibitor blasticidin S enters mammalian cells via leucine-rich repeat-containing protein 8D. J. Biol. Chem. 289, 17124–17131 (2014).
Rinkevich, Y. et al. Identification and prospective isolation of a mesothelial precursor lineage giving rise to smooth muscle cells and fibroblasts for mammalian internal organs, and their vasculature. Nat. Cell Biol. 14, 1251–1260 (2012).
Hovenden, M. et al. IgG subclass and heavy chain domains contribute to binding and protection by mAbs to the poly gamma-D-glutamic acid capsular antigen of Bacillus anthracis. PLoS Pathog. 9, e1003306 (2013).
Shi, J. et al. Engineered red blood cells as carriers for systemic delivery of a wide array of functional probes. Proc. Natl. Acad. Sci. USA 111, 10131–10136 (2014).
So, S., Adachi, N., Lieber, M.R. & Koyama, H. Genetic interactions between BLM and DNA ligase IV in human cells. J. Biol. Chem. 279, 55433–55442 (2004).
Simsek, D. & Jasin, M. Alternative end-joining is suppressed by the canonical NHEJ component Xrcc4-ligase IV during chromosomal translocation formation. Nat. Struct. Mol. Biol. 17, 410–416 (2010).
Song, L., Florea, L. & Langmead, B. Lighter: fast and memory-efficient sequencing error correction without counting. Genome Biol. 15, 509 (2014).
Robinson, J.T. et al. Integrative genomics viewer. Nat. Biotechnol. 29, 24–26 (2011).
Kamentsky, L. et al. Improved structure, function and compatibility for CellProfiler: modular high-throughput image analysis software. Bioinformatics 27, 1179–1180 (2011).
Popp, M.W., Antos, J.M. & Ploegh, H.L. Site-specific protein labeling via sortase-mediated transpeptidation. Curr. Protoc. Protein Sci. Chapter 15 Unit 15 13 (2009).
Hailemariam, T.K. et al. Sphingomyelin synthase 2 deficiency attenuates NFkappaB activation. Arterioscler. Thromb. Vasc. Biol. 28, 1519–1526 (2008).
Singh, P., Schimenti, J.C. & Bolcun-Filas, E. A mouse geneticists's practical guide to CRISPR applications. Genetics 199, 1–15 (2015).
Acknowledgements
We thank members of the Ploegh laboratory, especially N. Pishesha and F. Tafesse, for critical reading of the manuscript; P.A. Koenig, L.K. Swee, C.S. Shivalila, H. Wang, H. Yang and R. Jaenisch for discussions; T. Wang and D.M. Sabatini for pCW-Cas9; P. Thiru and G. Bell of BARC (WIBR) for assistance with statistical analysis; and F. Zhang (Addgene) for pX330. This work was supported by the National Institutes of Health (RO1 grant AI087879-01 to H.L.P.), Japan Society for the Promotion of Science (to T.M.), Japan Heart Foundation (to T.M.), AACR-Pancreatic Cancer Action Network (to S.K.D. and H.L.P.) and an SNSF Early Postdoc Mobility fellowship (to M.C.T.).
Author information
Authors and Affiliations
Contributions
T.M. and H.L.P. conceived of and designed experiments; T.M., S.K.D. and A.M.B. performed experiments; T.M. and M.T. analyzed data; T.M., J.R.I. and H.L.P. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9, Supplementary Tables 1–7 (PDF 16545 kb)
Supplementary Protocol
CellProfiler pipeline protocol (TXT 9 kb)
Rights and permissions
About this article
Cite this article
Maruyama, T., Dougan, S., Truttmann, M. et al. Increasing the efficiency of precise genome editing with CRISPR-Cas9 by inhibition of nonhomologous end joining. Nat Biotechnol 33, 538–542 (2015). https://doi.org/10.1038/nbt.3190
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.3190
This article is cited by
-
A potential paradigm in CRISPR/Cas systems delivery: at the crossroad of microalgal gene editing and algal-mediated nanoparticles
Journal of Nanobiotechnology (2023)
-
CATI: an efficient gene integration method for rodent and primate embryos by MMEJ suppression
Genome Biology (2023)
-
The applications of CRISPR/Cas-mediated genome editing in genetic hearing loss
Cell & Bioscience (2023)
-
Versatile generation of precise gene edits in bovines using SEGCPN
BMC Biology (2023)
-
SERCA2 phosphorylation at serine 663 is a key regulator of Ca2+ homeostasis in heart diseases
Nature Communications (2023)