Abstract
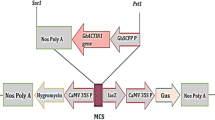
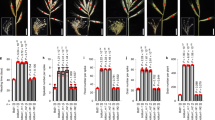
The capacity of conventional breeding to simultaneously improve the yield and quality of cotton fiber is limited1. The accumulation of the plant hormone indole-3-acetic acid (IAA) in cotton fiber initials prompted us to investigate the effects of genetically engineering increased IAA levels in the ovule epidermis. Targeted expression of the IAA biosynthetic gene iaaM, driven by the promoter of the petunia MADS box gene Floral Binding protein 7 (FBP7)2, increased IAA levels in the epidermis of cotton ovules at the fiber initiation stage. This substantially increased the number of lint fibers, an effect that was confirmed in a 4-year field trial. The lint percentage of the transgenic cotton, an important component of fiber yield, was consistently higher in our transgenic plants than in nontransgenic controls, resulting in a >15% increase in lint yield. Fiber fineness was also notably improved.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Meredith, W.R. Cotton yield progress: why has it reached a plateau? Better Crops 84, 6–9 (2000).
Colombo, L. et al. Downregulation of ovule-specific MADS box genes from petunia results in maternally controlled defects in seed development. Plant Cell 9, 703–715 (1997).
Chen, Z.J. et al. Toward sequencing cotton (Gossypium) genomes. Plant Physiol. 145, 1303–1310 (2007).
Meredith, W.R. Quantitative genetics. in Cotton (eds. Kohel, R.J. & Lewis, C.F.) 131–150 (American Society of Agronomy, Madison, Wisconsin, USA, 1984).
Gordon, S. Cotton fibre quality. in Cotton: Science and Technology (eds. Gordon, S. & Hsieh, Y.L.) 68–100 (CRC Press, Boca Raton, FL, USA, 2007).
Benedict, C.R., Kohel, J.R. & Lewis, H.L. Cotton fiber quality. in Cotton (eds. Smith, C.W. & Cothren, J.T.) 270 (John Wiley & Sons, New York, USA, 1999).
Zhang, J.F., Lu, Y., Adragna, H. & Hughs, E. Genetic improvement of New Mexico acala cotton germplasm and their genetic diversity. Crop Sci. 45, 2363–2373 (2005).
Stewart, J.M. Fiber initiation on the cotton ovule (Gossypium hirsutum). Am. J. Bot. 62, 723–730 (1975).
Basra, A.S. & Saha, S. Growth regulation of cotton fibers. in Cotton Fibers: Developmental Biology, Quality Improvement, and Textile Processing (ed. Basra, A.S.) 47–58 (The Haworth Press, New York, USA, 1999).
Beasley, C.A. Hormonal regulation of growth in unfertilized cotton ovules. Science 179, 1003–1005 (1973).
Beasley, C.A. & Ting, I.P. Effects of plant growth substances on in vitro fiber development from unfertilized cotton ovules. Am. J. Bot. 61, 188–194 (1974).
Gialvalis, S. & Seagull, R.W. Plant hormones alter fiber initiation in unfertilized, cultured ovules of Gossypium hirsutum. J. Cotton Sci. 5, 252–258 (2001).
Seagull, R.W. & Giavalis, S. Pre- and post-anthesis application of exogenous hormones alters fiber production in Gossypium hirsutum L. cultivar Maxxa GTO. J. Cotton Sci. 8, 105–111 (2004).
Guinn, G. & Brummett, D.L. Changes in abscisic acid and indoleacetic acid before and after anthesis relative to changes in abscission rates of cotton fruiting forms. Plant Physiol. 87, 629–631 (1988).
John, M.E. Genetic engineering strategies of cotton fiber modification. in Cotton Fibers: Developmental Biology, Quality Improvement and Textile Processing (ed. Basra, A.S.) 271–292 (The Haworth Press, New York, USA, 1999).
Chen, J.G., Du, X.M., Zhou, X. & Zhao, H.Y. Levels of cytokinins in the ovules of cotton mutants with altered fiber development. J. Plant Growth Regul. 16, 181–185 (1997).
Shi, Y. et al. Transcriptome profiling, molecular biological, and physiological studies reveal a major role for ethylene in cotton fiber cell elongation. Plant Cell 18, 651–664 (2006).
Li, Y. et al. Transgenics of plant hormones and their potential application in horticultural crops. in Genetically Modified Crops, their Development, Uses and Risks (eds. Liang, G.H. & Skinner, D.Z.) 101–117 (The Haworth Press, New York, USA, 2004).
Wilkins, T.A., Rajasekaran, K. & Anderson, D.M. Cotton biotechnology. Crit. Rev. Plant Sci. 19, 511–550 (2000).
Vanneste, S. & Friml, J. Auxin: a trigger for change in plant development. Cell 136, 1005–1016 (2009).
Hou, Z. & Huang, W. Immunohistochemical localization of IAA and ABP1 in strawberry shoot apexes during floral induction. Planta 222, 678–687 (2005).
Ji, S. et al. Isolation and analyses of genes preferentially expressed during early cotton fiber development by subtractive PCR and cDNA array. Nucleic Acids Res. 31, 2534–2543 (2003).
Tu, L. et al. Genes expression analyses of sea-island cotton (Gossypium barbadense L.) during fiber development. Plant Cell Rep. 26, 1309–1320 (2007).
Morgan, D.G. Influence of 1-naphthylphthalamic acid on the movement of indolyl-3-acetic acid in plants. Nature 201, 476–477 (1964).
Savidge, B., Rounsley, S.D. & Yanofsky, M.F. Temporal relationship between the transcription of two Arabidopsis MADS box genes and the floral organ identity genes. Plant Cell 7, 721–733 (1995).
Barg, R. et al. The tomato early fruit specific gene Lefsm1 defines a novel class of plant-specific SANT/MYB domain proteins. Planta 221, 197–211 (2005).
Hou, L. et al. SCFP, a novel fiber-specific promoter in cotton. Chin. Sci. Bull. 53, 2639–2645 (2008).
John, M.E. & Crow, L.J. Gene expression in cotton (Gossypium hirsutum L.) fiber: cloning of the mRNAs. Proc. Natl. Acad. Sci. USA 89, 5769–5773 (1992).
Phillips, A.L. Genetic and transgenic approaches to improving crop performance via hormones. in Plant Hormones: Biosynthesis, Signal Transduction, Action, edn 2 (ed. Davies, P.J.) 582–609 (Springer, Dordrecht, The Netherlands, 2007).
Luo, M. et al. GhDET2, a steroid 5α-reductase, plays an important role in cotton fiber cell initiation and elongation. Plant J. 51, 419–430 (2007).
Glass, N.L. & Kosuge, T. Cloning of the gene for indoleacetic acid-lysine synthetase from Pseudomonas syringae subsp. savastanoi. J. Bacteriol. 166, 598–603 (1986).
Ryser, U. Cotton fiber initiation and histodifferentiation. in Cotton Fibers: Developmental Biology, Quality Improvement, and Textile Processing (ed. Basra, A.S.) 1–34 (The Haworth Press, New York, USA, 1999).
Debeaujon, I. et al. Proanthocyanidin-accumulating cells in Arabidopsis testa: Regulation of differentiation and role in seed development. Plant Cell 15, 2514–2531 (2003).
Wan, C.Y. & Wilkins, T.A. A modified hot borate method significantly enhances the yield of high-quality RNA from cotton (Gossypium hirsutum L.). Anal. Biochem. 223, 7–12 (1994).
Gou, J. et al. Gibberellins regulate lateral root formation in Populus through interactions with auxin and other hormones. Plant Cell 22, 623–639 (2010).
Marrison, J.L. & Leech, R.M. The subcellular and intra-organelle recognition of nuclear and chloroplast transcripts in developing leaf cells. Plant J. 6, 605–614 (1994).
Sun, Y. et al. Brassinosteroid regulates fiber development on cultured cotton ovules. Plant Cell Physiol. 46, 1384–1391 (2005).
Acknowledgements
We are indebted to X. Jiang (Beijing Forestry University, China) for technical assistance on plant hormone analysis, and to D. Zhang (USDA/ARS, BARC/PSI/SPCL) for his kind help in the statistical analysis of field data. We are grateful to X. Qiu (Department of Food & Bioproduct Sciences, University of Saskatchewan, Canada), J. Huang (Monsanto Company, USA) and J. Jacobs (Bayer CropScience, Belgium), Z. Pei (Department of Biology, Duke University, USA) and C. Lin (Department of Molecular, Cell, and Developmental Biology, University of California, USA), G. Xia (Institute of Microbiology, Chinese Academy Of Science) for their critical comments on the manuscript. This work has been supported by grant 2010CB126000 (to Y.P.) from the Chinese Ministry of Science and Technology, and by grant 30530490 (to Y.P.) from the National Natural Sciences Foundation of China.
Author information
Authors and Affiliations
Contributions
M.Z. performed field trials, in situ hybridization and quantified IAA content; X.Z. performed greenhouse experiments and quantified IAA content; S.S. performed cotton transformation and field trials; Q.Z. and Y.W. performed cotton transformation; J.Z. and X.L. performed field trials; L.H., D.L., M.L., Y.X. and X.L. performed vector construction and promoter analysis; J.Z. performed field data analysis; C.X. designed the hybridization experiment; Y.P. designed experiments and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–6 and Supplementary Figs. 1–10 (PDF 896 kb)
Rights and permissions
About this article
Cite this article
Zhang, M., Zheng, X., Song, S. et al. Spatiotemporal manipulation of auxin biosynthesis in cotton ovule epidermal cells enhances fiber yield and quality. Nat Biotechnol 29, 453–458 (2011). https://doi.org/10.1038/nbt.1843
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1843
This article is cited by
-
Comparative proteomic analysis identified proteins and the phenylpropanoid biosynthesis pathway involved in the response to ABA treatment in cotton fiber development
Scientific Reports (2023)
-
A comprehensive overview of cotton genomics, biotechnology and molecular biological studies
Science China Life Sciences (2023)
-
Genetic dissection of lint percentage in short-season cotton using combined QTL mapping and RNA-seq
Theoretical and Applied Genetics (2023)
-
Fine mapping and candidate gene analysis of qFL-A12-5: a fiber length-related QTL introgressed from Gossypium barbadense into Gossypium hirsutum
Theoretical and Applied Genetics (2023)
-
Coordination of floral and fiber development in cotton (Gossypium) by hormone- and flavonoid-signalling associated regulatory miRNAs
Plant Molecular Biology (2023)