Abstract
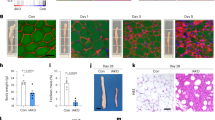
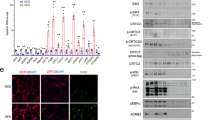
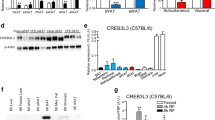
The adipose-derived hormone leptin maintains energy balance in part through central nervous system-mediated increases in sympathetic outflow that enhance fat burning. Triggering of β-adrenergic receptors in adipocytes stimulates energy expenditure by cyclic AMP (cAMP)-dependent increases in lipolysis and fatty-acid oxidation. Although the mechanism is unclear, catecholamine signalling is thought to be disrupted in obesity, leading to the development of insulin resistance. Here we show that the cAMP response element binding (CREB) coactivator Crtc3 promotes obesity by attenuating β-adrenergic receptor signalling in adipose tissue. Crtc3 was activated in response to catecholamine signals, when it reduced adenyl cyclase activity by upregulating the expression of Rgs2, a GTPase-activating protein that also inhibits adenyl cyclase activity. As a common human CRTC3 variant with increased transcriptional activity is associated with adiposity in two distinct Mexican-American cohorts, these results suggest that adipocyte CRTC3 may play a role in the development of obesity in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Flegal, K. M., Carroll, M. D., Ogden, C. L. & Curtin, L. R. Prevalence and trends in obesity among US adults, 1999-2008. J. Am. Med. Assoc. 303, 235–241 (2010)
Cowie, C. C. et al. Full accounting of diabetes and pre-diabetes in the U.S. population in 1988-1994 and 2005-2006. Diabetes Care 32, 287–294 (2009)
Bartness, T. J. & Song, C. K. Thematic review series: adipocyte biology. Sympathetic and sensory innervation of white adipose tissue. J. Lipid Res. 48, 1655–1672 (2007)
Lafontan, M. & Langin, D. Lipolysis and lipid mobilization in human adipose tissue. Prog. Lipid Res. 48, 275–297 (2009)
Bachman, E. S. et al. βAR signaling required for diet-induced thermogenesis and obesity resistance. Science 297, 843–845 (2002)
Soloveva, V., Graves, R. A., Rasenick, M. M., Spiegelman, B. M. & Ross, S. R. Transgenic mice overexpressing the β1-adrenergic receptor in adipose tissue are resistant to obesity. Mol. Endocrinol. 11, 27–38 (1997)
Hagiwara, M. et al. Transcriptional attenuation following cAMP induction requires PP-1-mediated dephosphorylation of CREB. Cell 70, 105–113 (1992)
Michael, L. F., Asahara, H., Shulman, A., Kraus, W. & Montminy, M. The phosphorylation status of a cyclic AMP-responsive activator is modulated via a chromatin-dependent mechanism. Mol. Cell. Biol. 20, 1596–1603 (2000)
De Cesare, D. & Sassone-Corsi, P. Transcriptional regulation by cyclic AMP-responsive factors. Prog. Nucleic Acid Res. Mol. Biol. 64, 343–369 (2000)
Gonzalez, G. A. & Montminy, M. R. Cyclic AMP stimulates somatostatin gene transcription by phosphorylation of CREB at Serine 133. Cell 59, 675–680 (1989)
Chrivia, J. C. et al. Phosphorylated CREB binds specifically to the nuclear protein CBP. Nature 365, 855–859 (1993)
Radhakrishnan, I. et al. Solution structure of the KIX domain of CBP bound to the transactivation domain of CREB: a model for activator–coactivator interactions. Cell 91, 741–752 (1997)
Screaton, R. A. et al. The CREB coactivator TORC2 functions as a calcium- and cAMP-sensitive coincidence detector. Cell 119, 61–74 (2004)
Bittinger, M. A. et al. Activation of cAMP response element-mediated gene expression by regulated nuclear transport of TORC proteins. Curr. Biol. 14, 2156–2161 (2004)
Liu, Y. et al. A fasting inducible switch modulates gluconeogenesis via activator/coactivator exchange. Nature 456, 269–273 (2008)
Altarejos, J. Y. et al. The Creb1 coactivator Crtc1 is required for energy balance and fertility. Nature Med. 14, 1112–1117 (2008)
Koo, S. H. et al. The CREB coactivator TORC2 is a key regulator of fasting glucose metabolism. Nature 437, 1109–1111 (2005)
Saberi, M. et al. Novel liver-specific TORC2 siRNA corrects hyperglycemia in rodent models of type 2 diabetes. Am. J. Physiol. Endocrinol. Metab. 297, E1137–E1146 (2009)
Wang, Y. et al. Targeted disruption of the CREB coactivator Crtc2 increases insulin sensitivity. Proc. Natl Acad. Sci. USA 107, 3087–3092 (2010)
Katoh, Y. et al. Silencing the constitutive active transcription factor CREB by the LKB1-SIK signaling cascade. FEBS J. 273, 2730–2748 (2006)
Fu, A. & Screaton, R. A. Using kinomics to delineate signaling pathways: control of CRTC2/TORC2 by the AMPK family. Cell Cycle 7, 3823–3828 (2008)
Ahn, S. et al. A dominant negative inhibitor of CREB reveals that it is a general mediator stimulus-dependent transcription of c-fos. Mol. Cell. Biol. 18, 967–977 (1998)
Iourgenko, V. et al. Identification of a family of cAMP response element-binding protein coactivators by genome-scale functional analysis in mammalian cells. Proc. Natl Acad. Sci. USA 100, 12147–12152 (2003)
Conkright, M. D. et al. TORCs: transducers of regulated CREB activity. Mol. Cell 12, 413–423 (2003)
Lumeng, C. N., Deyoung, S. M., Bodzin, J. L. & Saltiel, A. R. Increased inflammatory properties of adipose tissue macrophages recruited during diet-induced obesity. Diabetes 56, 16–23 (2007)
Landsberg, L. Feast or famine: the sympathetic nervous system response to nutrient intake. Cell. Mol. Neurobiol. 26, 497–508 (2006)
Carmen, G. Y. & Victor, S. M. Signalling mechanisms regulating lipolysis. Cell. Signal. 18, 401–408 (2006)
Cannon, B. & Nedergaard, J. Brown adipose tissue: function and physiological significance. Physiol. Rev. 84, 277–359 (2004)
Seale, P., Kajimura, S. & Spiegelman, B. M. Transcriptional control of brown adipocyte development and physiological function—of mice and men. Genes Dev. 23, 788–797 (2009)
Cederberg, A. et al. FOXC2 is a winged helix gene that counteracts obesity, hypertriglyceridemia, and diet-induced insulin resistance. Cell 106, 563–573 (2001)
Qi, L. et al. Adipocyte CREB promotes insulin resistance in obesity. Cell Metab. 9, 277–286 (2009)
Wu, Z. et al. Transducer of regulated CREB-binding proteins (TORCs) induce PGC-1α transcription and mitochondrial biogenesis in muscle cells. Proc. Natl Acad. Sci. USA 103, 14379–14384 (2006)
Roy, A. A. et al. RGS2 interacts with Gs and adenylyl cyclase in living cells. Cell. Signal. 18, 336–348 (2006)
Salim, S., Sinnarajah, S., Kehrl, J. H. & Dessauer, C. W. Identification of RGS2 and type V adenylyl cyclase interaction sites. J. Biol. Chem. 278, 15842–15849 (2003)
Sinnarajah, S. et al. RGS2 regulates signal transduction in olfactory neurons by attenuating activation of adenylyl cyclase III. Nature 409, 1051–1055 (2001)
Rohlfs, E. M., Daniel, K. W., Premont, R. T., Kozak, L. P. & Collins, S. Regulation of the uncoupling protein gene (Ucp) by β1, β2, and β3-adrenergic receptor subtypes in immortalized brown adipose cell lines. J. Biol. Chem. 270, 10723–10732 (1995)
Granneman, J. G. Expression of adenylyl cyclase subtypes in brown adipose tissue: neural regulation of type III. Endocrinology 136, 2007–2012 (1995)
Freson, K. et al. -391 C to G substitution in the regulator of G-protein signalling-2 promoter increases susceptibility to the metabolic syndrome in white European men: consistency between molecular and epidemiological studies. J. Hypertens. 25, 117–125 (2007)
Dentin, R., Hedrick, S., Xie, J., Yates, J., III & Montminy, M. Hepatic glucose sensing via the CREB coactivator CRTC2. Science 319, 1402–1405 (2008)
Qi, L. et al. TRB3 links the E3 ubiquitin ligase COP1 to lipid metabolism. Science 312, 1763–1766 (2006)
Goodarzi, M. O. et al. Lipoprotein lipase is a gene for insulin resistance in Mexican Americans. Diabetes 53, 214–220 (2004)
Goodarzi, M. O. et al. Determination and use of haplotypes: ethnic comparison and association of the lipoprotein lipase gene and coronary artery disease in Mexican-Americans. Genet. Med. 5, 322–327 (2003)
Bild, D. E. et al. Multi-ethnic study of atherosclerosis: objectives and design. Am. J. Epidemiol. 156, 871–881 (2002)
Livak, K. J. Allelic discrimination using fluorogenic probes and the 5′ nuclease assay. Genet. Anal. 14, 143–149 (1999)
Choudhry, S. et al. Population stratification confounds genetic association studies among Latinos. Hum. Genet. 118, 652–664 (2006)
Heard-Costa, N. L. et al. NRXN3 is a novel locus for waist circumference: a genome-wide association study from the CHARGE Consortium. PLoS Genet. 5, e1000539 (2009)
Willer, C. J. et al. Six new loci associated with body mass index highlight a neuronal influence on body weight regulation. Nature Genet. 41, 25–34 (2009)
Zeger, S. L. & Liang, K. Y. Longitudinal data analysis for discrete and continuous outcomes. Biometrics 42, 121–130 (1986)
Acknowledgements
This study was supported in part by National Institutes of Health grants R01-DK049777, R01-DK083834, P30-DK063491, R01-HL088457, R01-DK79888, R01-HL071205, N01-HC95159, N02-HL64278and M01-RR00425 (General Clinical Research Center Grant from the National Center for Research Resources, The J.W. Kieckhefer Foundation, The Clayton Medical Research Foundation, Inc., The Helmsley Foundation, the Cedars-Sinai Winnick Clinical Scholars Award (to M.O.G.) and the Cedars-Sinai Board of Governor’s Chair in Medical Genetics (J.I.R.). We thank B. Beutler and O. Siggs (The Scripps Research Institute) for help with macrophage studies.
Author information
Authors and Affiliations
Contributions
Y.S., J.A. and M.M. conceived the project and experimental design. M.O.G. analysed human data. Y.S., J.A., H.I., R.B., J.K., J.G., M.I., J.P., M.F.H., P.K.S., N.G., L.V. and N.M. performed experiments and data analysis. X.G. designed and supervised human data analysis. J.C. performed statistical analysis. M.R.G. performed genotyping experiments. Y.-D.I.C. was involved in study design. K.D.T. designed and supervised human genotyping experiments. W.A.H. and J.I.R. designed and conceived the MACAD study. Y.S., M.O.G. and M.M. wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-18 with legends and Supplementary Tables 1-3. (PDF 1105 kb)
Rights and permissions
About this article
Cite this article
Song, Y., Altarejos, J., Goodarzi, M. et al. CRTC3 links catecholamine signalling to energy balance. Nature 468, 933–939 (2010). https://doi.org/10.1038/nature09564
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09564
This article is cited by
-
In vitro CRISPR screening uncovers CRTC3 as a regulator of IFN-γ-induced ferroptosis of hepatocellular carcinoma
Cell Death Discovery (2023)
-
Amphiregulin normalizes altered circuit connectivity for social dominance of the CRTC3 knockout mouse
Molecular Psychiatry (2023)
-
Biological functions of CRTC2 and its role in metabolism-related diseases
Journal of Cell Communication and Signaling (2023)
-
Comprehensive evaluation of the metabolic effects of porcine CRTC3 overexpression on subcutaneous adipocytes with metabolomic and transcriptomic analyses
Journal of Animal Science and Biotechnology (2021)
-
Activation of the adipocyte CREB/CRTC pathway in obesity
Communications Biology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.