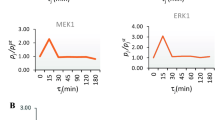
Abstract
Although epidermal growth factor receptor (EGFR; also called ErbB1) and its relatives initiate one of the most well-studied signalling networks, there is not yet a genome-wide view of even the earliest step in this pathway: recruitment of proteins to the activated receptors. Here we use protein microarrays comprising virtually every Src homology 2 (SH2) and phosphotyrosine binding (PTB) domain encoded in the human genome to measure the equilibrium dissociation constant of each domain for 61 peptides representing physiological sites of tyrosine phosphorylation on the four ErbB receptors. This involved 77,592 independent biochemical measurements and provided a quantitative protein interaction network that reveals many new interactions, including ones that fall outside of our current view of domain selectivity. By slicing through the network at different affinity thresholds, we found surprising differences between the receptors. Most notably, EGFR and ErbB2 become markedly more promiscuous as the threshold is lowered, whereas ErbB3 does not. Because EGFR and ErbB2 are overexpressed in many human cancers, our results suggest that the extent to which promiscuity changes with protein concentration may contribute to the oncogenic potential of receptor tyrosine kinases, and perhaps other signalling proteins as well.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yarden, Y. & Sliwkowski, M. X. Untangling the ErbB signalling network. Nature Rev. Mol. Cell Biol. 2, 127–137 (2001)
Sadowski, I., Stone, J. C. & Pawson, T. A noncatalytic domain conserved among cytoplasmic protein-tyrosine kinases modifies the kinase function and transforming activity of Fujinami sarcoma virus P130gag-fps. Mol. Cell. Biol. 6, 4396–4408 (1986)
Kavanaugh, W. M. & Williams, L. T. An alternative to SH2 domains for binding tyrosine-phosphorylated proteins. Science 266, 1862–1865 (1994)
Songyang, Z. et al. SH2 domains recognize specific phosphopeptide sequences. Cell 72, 767–778 (1993)
Songyang, Z. et al. Specific motifs recognized by the SH2 domains of Csk, 3BP2, fps/fes, GRB-2, HCP, SHC, Syk, and Vav. Mol. Cell. Biol. 14, 2777–2785 (1994)
Hatada, M. H. et al. Molecular basis for interaction of the protein tyrosine kinase ZAP-70 with the T-cell receptor. Nature 377, 32–38 (1995)
MacBeath, G. & Schreiber, S. L. Printing proteins as microarrays for high-throughput function determination. Science 289, 1760–1763 (2000)
Zhu, H. et al. Global analysis of protein activities using proteome chips. Science 293, 2101–2105 (2001)
Yan, K. S., Kuti, M. & Zhou, M. M. PTB or not PTB—that is the question. FEBS Lett. 513, 67–70 (2002)
Peri, S. et al. Development of human protein reference database as an initial platform for approaching systems biology in humans. Genome Res. 13, 2363–2371 (2003)
Yaffe, M. B. et al. A motif-based profile scanning approach for genome-wide prediction of signalling pathways. Nature Biotechnol. 19, 348–353 (2001)
Obenauer, J. C., Cantley, L. C. & Yaffe, M. B. Scansite 2.0: Proteome-wide prediction of cell signalling interactions using short sequence motifs. Nucleic Acids Res. 31, 3635–3641 (2003)
Bowers, G. et al. The relative role of ErbB1–4 receptor tyrosine kinases in radiation signal transduction responses of human carcinoma cells. Oncogene 20, 1388–1397 (2001)
Schoeberl, B., Eichler-Jonsson, C., Gilles, E. D. & Muller, G. Computational modeling of the dynamics of the MAP kinase cascade activated by surface and internalized EGF receptors. Nature Biotechnol. 20, 370–375 (2002)
Ponzetto, C. et al. A multifunctional docking site mediates signalling and transformation by the hepatocyte growth factor/scatter factor receptor family. Cell 77, 261–271 (1994)
Hendriks, B. S., Orr, G., Wells, A., Wiley, H. S. & Lauffenburger, D. A. Parsing ERK activation reveals quantitatively equivalent contributions from epidermal growth factor receptor and HER2 in human mammary epithelial cells. J. Biol. Chem. 280, 6157–6169 (2005)
Su, A. I. et al. Large-scale analysis of the human and mouse transcriptomes. Proc. Natl Acad. Sci. USA 99, 4465–4470 (2002)
Felder, S. et al. SH2 domains exhibit high-affinity binding to tyrosine-phosphorylated peptides yet also exhibit rapid dissociation and exchange. Mol. Cell. Biol. 13, 1449–1455 (1993)
Haugh, J. M. Localization of receptor-mediated signal transduction pathways: the inside story. Mol. Interv. 2, 292–307 (2002)
Burke, P., Schooler, K. & Wiley, H. S. Regulation of epidermal growth factor receptor signalling by endocytosis and intracellular trafficking. Mol. Biol. Cell 12, 1897–1910 (2001)
Gavin, A. C. et al. Functional organization of the yeast proteome by systematic analysis of protein complexes. Nature 415, 141–147 (2002)
Ho, Y. et al. Systematic identification of protein complexes in Saccharomyces cerevisiae by mass spectrometry. Nature 415, 180–183 (2002)
Li, S. et al. A map of the interactome network of the metazoan C. elegans. Science 303, 540–543 (2004)
Uetz, P. et al. A comprehensive analysis of protein–protein interactions in Saccharomyces cerevisiae. Nature 403, 623–627 (2000)
Gorgoulis, V. et al. Expression of EGF, TGF-α and EGFR in squamous cell lung carcinomas. Anticancer Res. 12, 1183–1187 (1992)
Irish, J. C. & Bernstein, A. Oncogenes in head and neck cancer. Laryngoscope 103, 42–52 (1993)
Moscatello, D. K. et al. Frequent expression of a mutant epidermal growth factor receptor in multiple human tumors. Cancer Res. 55, 5536–5539 (1995)
Ross, J. S. & Fletcher, J. A. The HER-2/neu oncogene in breast cancer: prognostic factor, predictive factor, and target for therapy. Stem Cells 16, 413–428 (1998)
Slamon, D. J. et al. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235, 177–182 (1987)
Wong, A. J. et al. Structural alterations of the epidermal growth factor receptor gene in human gliomas. Proc. Natl Acad. Sci. USA 89, 2965–2969 (1992)
Alimandi, M. et al. Cooperative signalling of ErbB3 and ErbB2 in neoplastic transformation and human mammary carcinomas. Oncogene 10, 1813–1821 (1995)
Wallasch, C. et al. Heregulin-dependent regulation of HER2/neu oncogenic signalling by heterodimerization with HER3. EMBO J. 14, 4267–4275 (1995)
Pinkas-Kramarski, R. et al. Diversification of Neu differentiation factor and epidermal growth factor signalling by combinatorial receptor interactions. EMBO J. 15, 2452–2467 (1996)
Woodring, P. J., Hunter, T. & Wang, J. Y. Regulation of F-actin-dependent processes by the Abl family of tyrosine kinases. J. Cell Sci. 116, 2613–2626 (2003)
Schultz, J., Milpetz, F., Bork, P. & Ponting, C. P. SMART, a simple modular architecture research tool: identification of signalling domains. Proc. Natl Acad. Sci. USA 95, 5857–5864 (1998)
Bateman, A. et al. The Pfam protein families database. Nucleic Acids Res. 28, 263–266 (2000)
Acknowledgements
We thank B. Schoeberl for comments on the manuscript, J. Grudzien for development work on the silicone gaskets, and the Bauer Center for Genomics Research at Harvard University for support with instrumentation and automation. This work was supported by awards from the W. M. Keck Foundation and the Arnold and Mabel Beckman Foundation. R.B.J. is the recipient of a Ruth L. Kirschstein National Research Service Award (NIH), A.G. is the recipient of an NSF Graduate Research Fellowship, and J.A.K. is the recipient of a Howard Hughes Medical Institute Predoctoral Fellowship in the Biological Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Figure 1
This figure shows multiple sequence alignments for human SH2 domains (a) and human PTB domains (b). (RTF 241 kb)
Supplementary Figure 2
This figure shows SDS-polyacrylamide gels of purified SH2 and PTB domains. (PDF 313 kb)
Supplementary Figure 3
This figure shows western blots of cells stimulated with EGF or HRGβ1. (PDF 84 kb)
Supplementary Table 1
This table provides a list of the SH2 domains (a) and PTB domains (b) used in this study. (RTF 156 kb)
Supplementary Table 2
This table provides an estimate of the percentage of each purified SH2 or PTB domain that was monomeric as judged by analytical size exclusion column chromatography. (RTF 104 kb)
Supplementary Table 3
This table provides a list of the peptides used in this study. (RTF 52 kb)
Supplementary Table 4
This table provides equilibrium dissociation constants for SH2/PTB interactions with peptides derived from the four human ErbB receptors. (XLS 46 kb)
Supplementary Table 5
This table provides a comparison of the interactions observed on the protein microarrays with previously reported interactions. (RTF 176 kb)
Supplementary Table 6
This table provides a list of the domains that bound to EGFR, ErbB2, and ErbB3 at four different affinity thresholds. (RTF 49 kb)
Supplementary Methods
This file contains technical details of experimental methods used in this study. (RTF 29 kb)
Supplementary Notes
Additional notes on the Supplementary Figures and Tables. (RTF 6 kb)
Rights and permissions
About this article
Cite this article
Jones, R., Gordus, A., Krall, J. et al. A quantitative protein interaction network for the ErbB receptors using protein microarrays. Nature 439, 168–174 (2006). https://doi.org/10.1038/nature04177
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04177
This article is cited by
-
Anticancer effect of zanubrutinib in HER2-positive breast cancer cell lines
Investigational New Drugs (2023)
-
MicroRNAs as the critical regulators of tyrosine kinase inhibitors resistance in lung tumor cells
Cell Communication and Signaling (2022)
-
TRAF4 hyperactivates HER2 signaling and contributes to Trastuzumab resistance in HER2-positive breast cancer
Oncogene (2022)
-
Rapid whole cell imaging reveals a calcium-APPL1-dynein nexus that regulates cohort trafficking of stimulated EGF receptors
Communications Biology (2021)
-
Effect of Epidermal Growth Factor on the Colony-formation Ability of Porcine Spermatogonial Germ Cells
Biotechnology and Bioprocess Engineering (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.