Abstract
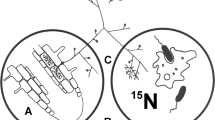
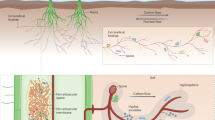
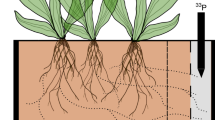
Arbuscular mycorrhizal fungi (order Glomales), which form mycorrhizal symbioses with two out of three of all plant species1, are believed to be obligate biotrophs that are wholly dependent on the plant partner for their carbon supply2. It is thought that they possess no degradative capability and that they are unable to decompose complex organic molecules, the form in which most soil nutrients occur. Earlier suggestions that they could exist saprotrophically were based on observation of hyphal proliferation on organic materials3,4. In contrast, other mycorrhizal types have been shown to acquire nitrogen directly from organic sources5,6,7. Here we show that the arbuscular mycorrhizal symbiosis can both enhance decomposition of and increase nitrogen capture from complex organic material (grass leaves) in soil. Hyphal growth of the fungal partner was increased in the presence of the organic material, independently of the host plant.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Trappe, J. M. in Ecophysiology of VA Mycorrhizal Plants (ed. Safir, G.) 5–25 (CRC Press, Boca Raton, 1987).
Smith, S. E. & Read, D. J. Mycorrhizal Symbiosis (Academic, San Diego, 1996).
Mosse, B. Observations on the extramatrical mycelium of a vesicular-arbuscular endophyte. Trans. Brit. Mycol. Soc. 42, 439–448 (1959).
St. John, T. V., Coleman, D. C. & Reid, C. P. P. Association of vesicular-arbuscular mycorrhizal hyphae with soil organic particles. Ecology 64, 957–959 (1983).
Abuzinadah, R. & Read, D. J. The role of proteins in the nitrogen nutrition of ectomycorrhizal plants V. Nitrogen transfer in birch (Betula pendula L. ) infected with different mycorrhizal fungi. New Phytol. 112, 55–60 (1989).
Hodge, A., Alexander, I. J. & Gooday, G. W. Chitinolytic enzymes of pathogenic and ectomycorrhizal fungi. Mycol. Res. 99, 935–941 (1995).
Leake, J. R. & Read, D. J. Proteinase activity in mycorrhizal fungi II. The effects of mineral and organic nitrogen sources on induction of extracellular proteinase in Hymenoscyphus ericae (Read) Korf and Kernan. New Phytol. 116, 123–128 (1990).
Vitousek, P. M. & Howarth, R. W. Nitrogen limitation on land and in the sea: how can it occur? Biogeochemistry 13, 87–115 (1991).
Chapin, F. S., Moilanen, L. & Kielland, K. Preferential use of organic nitrogen for growth by a non-mycorrhizal arctic sedge. Nature 361, 150–153 (1993).
Jones, D. L. & Darrah, P. R. Re-sorption of organic components by roots of Zea mays L. and its consequences in the rhizosphere. I. Re-sorption of 14C labelled glucose, mannose and citric acid. Plant Soil 143, 259–266 (1992).
Read, D. J. Mycorrhizas in ecosystems. Experientia 47, 376–391 (1991).
Sanders, F. E. & Tinker, P. B. Mechanism of absorption of phosphate from soil by Endogone mycorrhizas. Nature 233, 278–279 (1971).
Newsham, K. K., Fitter, A. H. & Watkinson, A. R. Multi-functionality and biodiversity in arbuscular mycorrhizas. Trends Ecol. Evol. 10, 407–411 (1995).
Tinker, P. B. H. & Nye, P. H. Solute Transport in the Rhizosphere (Oxford Univ. Press, Oxford, 2000).
Tobar, R. M., Azcón, R. & Barea, J. M. The improvement of plant N acquisition from an ammonium-treated, drought-stressed soil by the fungal symbiont in arbuscular mycorrhizae. Mycorrhiza 4, 105–108 (1994).
Mäder, P. et al. Transport of 15N from a soil compartment separated by a polytetrafluoroethylene membrane to plant roots via the hyphae of arbuscular mycorrhizal fungi. New Phytol. 146, 155–161 (2000).
Van Vuuren, M. M. I., Robinson, D. & Griffiths, B. S. Nutrient inflow and root proliferation during the exploitation of a temporally and spatially discrete source of nitrogen in soil. Plant Soil 178, 185–192 (1996).
Fransen, B., de Kroon, H. & Berendse, F. Root morphological plasticity and nutrient acquisition of perennial grass species from habitats of different nutrient availability. Oecologia 115, 351–358 (1998).
Hodge, A., Stewart, J., Robinson, D., Griffiths, B. S. & Fitter, A. H. Root proliferation, soil fauna and plant nitrogen capture from nutrient-rich patches in soil. New Phytol. 139, 479–494 (1998).
Bever, J. D. Dynamics within a mutualism and the maintenance of diversity: inference from a model of interguild frequency dependence. Ecol. Lett. 2, 52–61 (1999).
Stark, J. M. in Exploitation of Environmental Heterogeneity by Plants (eds Caldwell, M. M. & Pearcy, R. W.) 255–284 (Academic, San Diego, 1994).
Koide, R. T. & Li, M. Appropriate controls for vesicular arbuscular mycorrhizal research. New Phytol. 111, 35–44 (1989).
van der Heijden, M. G. A. et al. Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature 396, 69–72 (1998).
Kormanik, P. P. & McGraw, A.-C. in Methods and Principles of Mycorrhiza Research (ed. Schenck, N. C.) 37–46 (American Phytopathol. Soc., St Paul, Minnesota, USA, 1982).
Staddon, P. L., Fitter, A. H. & Graves, J. D. Effect of elevated atmospheric CO2 on mycorrhizal colonization, external mycorrhizal hyphal production and phosphorus inflow in Plantago lanceolata and Trifolium repens in association with the arbuscular mycorrhizal fungus Glomus mosseae. Global Change Biol. 5, 347–358 (1999).
Bligh, E. G. & Dyer, W. J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917 (1959).
Frostegård, A., Tunlid, A. & Bååth, E. Changes in microbial community structure during long-term incubation in two soils experimentally contaminated with metals. Soil Biol. Biochem. 28, 55–63 (1996).
Acknowledgements
We thank C. Scrimgeour and W. Stein for conducting the mass spectrometry analysis; P. Roberts for drawing Fig. 1; and D. Robinson and P. Young for comments on the manuscript. This work was supported by the Biotechnology and Biological Sciences Research Council (BBSRC).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hodge, A., Campbell, C. & Fitter, A. An arbuscular mycorrhizal fungus accelerates decomposition and acquires nitrogen directly from organic material. Nature 413, 297–299 (2001). https://doi.org/10.1038/35095041
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35095041
This article is cited by
-
Concentration characteristics and the drivers of soluble components in freshly fallen plant litter
Plant Ecology (2024)
-
Mycorrhiza-mediated recruitment of complete denitrifying Pseudomonas reduces N2O emissions from soil
Microbiome (2023)
-
Analysis of the AMT gene family in chili pepper and the effects of arbuscular mycorrhizal colonization on the expression patterns of CaAMT2 genes
BMC Genomics (2023)
-
Field inoculation by arbuscular mycorrhizal fungi with contrasting life-history strategies differently affects tomato nutrient uptake and residue decomposition dynamics
Plant and Soil (2023)
-
Arbuscular mycorrhiza enhances maize grain yield and nitrogen uptake during the grain filling stage with contrasting nitrogen status in two types of soils
Plant Growth Regulation (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.