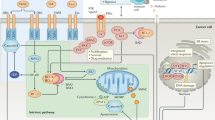
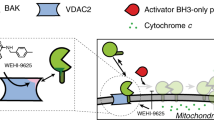
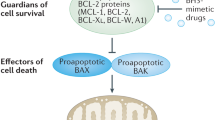
Abstract
A retrospective look at the basis of human disease pathogenesis almost always reveals an apoptotic component that either contributes to disease progression or accounts for it. What makes this field particularly exciting is the breadth of therapeutic opportunities that are on offer. The pace of apoptosis research has raised expectations that therapeutics will follow soon. But many of the organizations that are best placed to take advantage of these discoveries consider the ability to modulate the life or death of a cell for the purpose of disease treatment as perhaps being 'too good to be true'. Nevertheless, practical therapeutics that modulate apoptosis will no doubt appear in the clinic or on the shelf in the next few years.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ellis, R. E., Yuan, J. Y. & Horvitz, H. R. Mechanisms and functions of cell death. Annu. Rev. Cell Biol. 7, 663–698 (1991).
Wang, J. L. et al. Structure-based discovery of an organic compound that binds bcl-2 protein and induces apoptosis of tumor cells. Proc. Natl Acad. Sci. USA 97, 7124–7129 (2000).
Adams, J. M. & Cory, S. The Bcl-2 protein family: arbiters of cell survival. Science 281, 1322– 1326 (1998).
Reed, J. C. Bcl-2 family proteins. Oncogene 17, 3225 –3236 (1998).
Veis, D. J., Sorenson, C. M., Shutter, J. R. & Korsmeyer, S. J. Bcl-2-deficient mice demonstrate fulminant lymphoid apoptosis, polycystic kidneys, and hypopigmented hair. Cell 75, 229–240 (1993).
Cory, S. Regulation of lymphocyte survival by the bcl-2 gene family. Annu. Rev. Immunol. 13, 513–543 ( 1995).
Schlagbauer-Wadl, H. et al. Bcl-2 antisense oligonucleotides (G3139) inhibit Merkel cell carcinoma growth in SCID mice. J. Invest. Dermatol. 114, 725–730 (2000).
Jansen, B. et al. bcl-2 antisense therapy chemosensitizes human melanoma in SCID mice. Nature Med. 4, 232– 234 (1998).
Jansen, B. et al. Bcl-2 antisense plus dacarbazine therapy for malignant melanoma . Proc. Am. Assoc. Cancer Res. Conf. Programmed Cell Death Regul. A59 (2000).
Waters, J. S. et al. Phase I clinical and pharmacokinetic study of bcl-2 antisense oligonucleotide therapy in patients with non-Hodgkin's lymphoma . J. Clin. Oncol. 18, 1812– 1823 (2000).
Zangemeister-Wittke, U. et al. A novel bispecific antisense oligonucleotide inhibiting both bcl-2 and bcl-xL expression efficiently induces apoptosis in tumor cells. Clin. Cancer Res. 6, 2547– 2555 (2000).
Reed, J. C. Splicing and dicing apoptosis genes. Nature Biotechnol. 17, 1064–1065 (1999).
Taylor, J. K., Zhang, Q. Q., Wyatt, J. R. & Dean, N. M. Induction of endogenous Bcl-xS through the control of Bcl-x pre-mRNA splicing by antisense oligonucleotides. Nature Biotechnol. 17 , 1097–1100 (1999).
Que, F. G. et al. Cholangiocarcinomas express Fas ligand and disable the Fas receptor. Hepatology 30, 1398– 1404 (1999).
Ashkenazi, A. & Dixit, V. M. Death receptors: signaling and modulation. Science 281, 1305– 1308 (1998).
Schneider, P. & Tschopp, J. Apoptosis induced by death receptors . Pharm. Acta Helv. 74, 281– 286 (2000).
Boldin, M. P., Goncharov, T. M., Goltsev, Y. V. & Wallach, D. Involvement of MACH, a novel MORT1/FADD-interacting protease, in Fas/APO-1- and TNF receptor-induced cell death. Cell 85, 803–815 (1996).
Muzio, M. et al. FLICE, a novel FADD-homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/APO-1) death-inducing signaling complex. Cell 85, 817–827 ( 1996).
Kischkel, F. C. et al. Apo2L/TRAIL-dependent recruitment of endogenous FADD and caspase-8 to death receptors 4 and 5. Immunity 12, 611–620 (2000).
Sprick, M. R. et al. FADD/MORT1 and caspase-8 are recruited to TRAIL receptors 1 and 2 and are essential for apoptosis mediated by TRAIL receptor 2. Immunity 12, 599–609 ( 2000).
Bodmer, J. L. et al. TRAIL receptor-2 signals apoptosis through FADD and caspase-8 . Nature Cell Biol. 2, 241– 243 (2000).
Thome, M. et al. Viral FLICE-inhibitory proteins (FLIPs) prevent apoptosis induced by death receptors. Nature 386, 517– 521 (1997).
Rasper, D. M. et al. Cell death attenuation by 'Usurpin', a mammalian DED-caspase homologue that precludes caspase-8 recruitment and activation by the CD-95 (Fas, APO-1) receptor complex. Cell Death Differ. 5 , 271–288 (1998).
Ashkenazi, A. et al. Safety and antitumor activity of recombinant soluble Apo2 ligand. J. Clin. Invest. 104, 155– 162 (1999).
Walczak, H. et al. Tumoricidal activity of tumor necrosis factor-related apoptosis-inducing ligand in vivo. Nature Med. 5, 157– 163 (1999).
Marsters, S. A., Pitti, R. A., Sheridan, J. P. & Ashkenazi, A. Control of apoptosis signaling by Apo2 ligand. Recent Prog. Horm. Res. 54, 225–234 ( 1999).
Griffith, T. S. & Lynch, D. H. TRAIL: a molecule with multiple receptors and control mechanisms. Curr. Opin. Immunol. 10, 559–563 ( 1998).
Zhang, X. D., Franco, A. V., Nguyen, T., Gray, C. P. & Hersey, P. Differential localization and regulation of death and decoy receptors for TNF-related apoptosis-inducing ligand (TRAIL) in human melanoma cells. J. Immunol. 164, 3961–3970 (2000).
Kim, K., Fisher, M. J., Xu, S. Q. & el-Deiry, W. S. Molecular determinants of response to TRAIL in killing of normal and cancer cells. Clin. Cancer Res. 6, 335–346 (2000).
Hollstein, M., Sidransky, D., Vogelstein, B. & Harris, C. C. p53 mutations in human cancers. Science 253, 49–53 (1991).
Takimoto, R. & El-Deiry, W. S. Wild-type p53 transactivates the KILLER/DR5 gene through an intronic sequence-specific DNA-binding site . Oncogene 19, 1735–1743 (2000).
Ashkenazi, A. The Apo2L/TRAIL system: therapeutic opportunities. Proc. Am. Assoc. Cancer Res. Conf. Programmed Cell Death Regul. (2000 ).
Nagata, S. Steering anti-cancer drugs away from the TRAIL. Nature Med. 6, 502–503 (2000).
Jo, M. et al. Apoptosis induced in normal human hepatocytes by tumor necrosis factor-related apoptosis-inducing ligand. Nature Med. 6, 564–567 (2000).
Hymowitz, S. G. et al. A unique zinc-binding site revealed by a high-resolution X-ray structure of homotrimeric Apo2L/TRAIL. Biochemistry 39, 633–640 (2000).
Hymowitz, S. G. et al. Triggering cell death: the crystal structure of Apo2L/TRAIL in a complex with death receptor 5. Mol. Cell 4, 563–571 (1999).
Alnemri, E. S. et al. Human ICE/CED-3 protease nomenclature. Cell 87, 171 (1996).
Thornberry, N. A. et al. A novel heterodimeric cysteine protease is required for interleukin-1β processing in monocytes. Nature 356, 768 –774 (1992).
Cerretti, D. P. et al. Molecular cloning of the interleukin-1β converting enzyme . Science 256, 97–100 (1992).
Yuan, J., Shaham, S., Ledoux, S., Ellis, H. M. & Horvitz, H. R. The C. elegans cell death gene ced-3 encodes a protein similar to mammalian interleukin-1β-converting enzyme . Cell 75, 641–652 (1993).
Nicholson, D. W. et al. Identification and inhibition of the ICE/CED-3 protease necessary for mammalian apoptosis. Nature 376, 37– 43 (1995).
Kuida, K. et al. Decreased apoptosis in the brain and premature lethality in CPP32-deficient mice. Nature 384, 368–372 (1996).
Nicholson, D. W. Caspase structure, proteolytic substrates, and function during apoptotic cell death. Cell Death Differ. 6, 1028– 1042 (1999).
Earnshaw, W. C., Martins, L. M. & Kaufmann, S. H. Mammalian caspases: structure, activation, substrates, and functions during apoptosis. Annu. Rev. Biochem. 68, 383–424 (1999).
Thornberry, N. A. et al. A combinatorial approach defines specificities of members of the caspase family and granzyme B. Functional relationships established for key mediators of apoptosis. J. Biol. Chem. 272, 17907–17911 (1997).
Garcia-Calvo, M. et al. A. Inhibition of human caspases by peptide-based and macromolecular inhibitors. J. Biol. Chem. 273, 32608– 32613 (1998).
Cursio, R. et al. A caspase inhibitor fully protects rats against lethal normothermic liver ischemia by inhibition of liver apoptosis. FASEB J. 13, 253–261 (1999).
Mocanu, M. M., Baxter, G. F. & Yellon, D. M. Caspase inhibition and limitation of myocardial infarct size: protection against lethal reperfusion injury. Br. J. Pharmacol. 130, 197–200 ( 2000).
Farber, A. et al. A specific inhibitor of apoptosis decreases tissue injury after intestinal ischemia–reperfusion in mice. J. Vasc. Surg. 30, 752–760 ( 1999).
Daemen, M. A. et al. Inhibition of apoptosis induced by ischemia–reperfusion prevents inflammation. J. Clin. Invest. 104, 541–549 (1999).
Endres, M. et al. Attenuation of delayed neuronal death after mild focal ischemia in mice by inhibition of the caspase family. J. Cereb. Blood Flow Metab. 18, 238–247 ( 1998).
Yakovlev, A. G. et al. Activation of CPP32-like caspases contributes to neuronal apoptosis and neurological dysfunction after traumatic brain injury. J. Neurosci. 17, 7415–7424 (1997).
Kondratyev, A. & Gale, K. Intracerebral injection of caspase-3 inhibitor prevents neuronal apoptosis after kainic acid-evoked status epilepticus. Brain Res. Mol. Brain Res. 75, 216–224 (2000).
Li, M. et al. Functional role of caspase-1 and caspase-3 in an ALS transgenic mouse model. Science 288, 335– 339 (2000).
Schierle, G. S. et al. Caspase inhibition reduces apoptosis and increases survival of nigral transplants. Nature Med. 5, 97 –100 (1999).
Braun, J. S. et al. Neuroprotection by a caspase inhibitor in acute bacterial meningitis. Nature Med. 5, 298– 302 (1999).
Grobmyer, S. R. et al. Peptidomimetic fluoromethylketone rescues mice from lethal endotoxic shock. Mol. Med. 5, 585– 594 (1999).
Hotchkiss, R. S. et al. Prevention of lymphocyte cell death in sepsis improves survival in mice. Proc. Natl Acad. Sci. USA 96, 14541 –14546 (1999).
Sanchez, I. et al. Caspase-8 is required for cell death induced by expanded polyglutamine repeats. Neuron 22, 623– 633 (1999).
Goldberg, Y. P. et al. Cleavage of huntingtin by apopain, a proapoptotic cysteine protease, is modulated by the polyglutamine tract. Nature Genet. 13, 442–449 ( 1996).
Gervais, F. G. et al. Involvement of caspases in proteolytic cleavage of Alzheimer's amyloid-beta precursor protein and amyloidogenic A beta peptide formation . Cell 97, 395–406 (1999).
Vocero-Akbani, A. M., Heyden, N. V., Lissy, N. A., Ratner, L. & Dowdy, S. F. Killing HIV-infected cells by transduction with an HIV protease-activated caspase-3 protein. Nature Med. 5, 29–33 (1999).
Lee, D. et al. Potent and selective nonpeptide inhibitors of caspases 3 and 7 inhibit apoptosis and maintain cell functionality. J. Biol. Chem. 275, 16007–16014 ( 2000).
Acknowledgements
I thank S. Roy, S. Xanthoudakis, D. McKay, C. Bayly and K. Clark (Merck) and A. Ashkenasi (Genentech) for assistance with this manuscript and its figures.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nicholson, D. From bench to clinic with apoptosis-based therapeutic agents. Nature 407, 810–816 (2000). https://doi.org/10.1038/35037747
Issue Date:
DOI: https://doi.org/10.1038/35037747
This article is cited by
-
Survivin and Caspase-3 Cannot Predict Recurrence for Urinary Bladder Carcinoma
SN Comprehensive Clinical Medicine (2021)
-
MLL-Rearranged Acute Lymphoblastic Leukemia
Current Hematologic Malignancy Reports (2020)
-
7-Chloroquinoline-1,2,3-triazoyl carboxamides induce cell cycle arrest and apoptosis in human bladder carcinoma cells
Investigational New Drugs (2020)
-
Mitochondrial mechanisms and therapeutics in ischaemia reperfusion injury
Pediatric Nephrology (2019)
-
Glutaredoxin 1 mediates the protective effect of steady laminar flow on endothelial cells against oxidative stress-induced apoptosis via inhibiting Bim
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.