Abstract
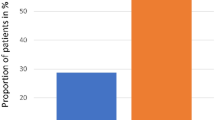
Oral mucositis (OM) is a complication of high-dose chemotherapy (HDC) followed by hematopoietic SCT (HSCT) with few effective treatments. Selenium has a cytoprotective role via the glutathione peroxidase (Glu.Px) enzyme and prevents chemotherapy-induced toxicities. We performed a double‐blind, randomized, placebo-controlled study to evaluate the efficacy of selenium on the prevention of OM in 77 patients with leukemia, undergoing allogeneic HSCT. Thirty-seven patients received oral selenium tablets (200 mcg twice daily) from the starting day of HDC to 14 days after transplantation. OM was evaluated daily for 21 days after transplantation according to World Health Organization oral toxicity scale. The incidence of severe OM (grades 3–4) was significantly lower in the selenium group (10.8% vs 35.1%, P<0.05). We noted that the duration of objective OM (grades 2–4), excluding patient’s self-declaration (grade 1), was significantly shorter in the selenium group (3.6±1.84 vs 5.3±2.2 days, P=0.014). Significant elevations in serum selenium level and plasma Glu.Px activity were observed 7 and 14 days after transplantation compared with baseline in the selenium group. We conclude that selenium can reduce the duration and severity of OM after HDC. Clinicaltrial.org ID: NCT01432873.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
D’Hondt LLC, Marc A, Jean-Luc C . Oral mucosit is induced by anticancer treatments: physiopathology and treatments. Ther Clin Risk Manag 2006; 2: 159–168.
Worthington HV, Clarkson JE, Eden O . Interventions for preventing oral mucositis for patients with cancer receiving treatment. Cochrane Database Syst Rev 2011; 4: CD000978.
Sonis ST . A biological approach to mucositis. J Support Oncol 2004; 2: 21–32.
Lalla RV, Schubert MM, Bensadoun RJ, Keefe D . Anti-inflammatory agents in the management of alimentary mucositis. Support Care Cancer 2006; 14: 558–565.
Porta C, Moroni M, Nastasi G . Allopurinol mouthwashes in the treatment of 5-fluorouracil-induced stomatitis. Am J Clin Oncol 1994; 17: 246–247.
Epstein JB, Silverman S, Paggiarino DA, Crockett S, Schubert MM, Senzer NN et al. Benzydamine HCl for prophylaxis of radiation-induced oral mucositis. Cancer 2001; 92: 875–885.
Thieblemont C, Dumontet C, Saad H, Roch N, Bouafia F, Arnaud P et al. Amifostine reduces mucosal damage after high-dose melphalan conditioning and autologous peripheral blood progenitor cell transplantation for patients with multiple myeloma. Bone Marrow Transplant 2002; 30: 769–775.
Ertekin MV, Koē M, Karslioglu I, Sezen O . Zinc sulfate in the prevention of radiation-induced oropharyngeal mucositis: a prospective, placebo-controlled, randomized study. Int J Radiat Oncol Biol Phys 2004; 58: 167–174.
Ryan-Harshman M, Aldoori W . The relevance of selenium to immunity, cancer, and infectious/inflammatory diseases. Can J Diet Pract Res 2005; 66: 98–102.
Buntzel J, Micke O, Mucke R, Glatzel M . Amifostine and selenium during simultaneous radiochemotherapy in head and neck cancer - redox status data. Trace Elem Electrolytes 2005; 22: 211–215.
Al-Moutairy AR, Tariq M . Effect of vitamin E and selenium on hypothermic restraint stress and chemically-induced ulcers. Dig Dis Sci 1996; 41: 1165–1171.
Koyuturk M, Bolkent S, Ozdil S, Arbak S, Yanardag R . The protective effect of vitamin C, vitamin E and selenium combination therapy on ethanol-induced duodenal mucosal injury. Hum Exp Toxicol 2004; 23: 391–398.
Buntzel J, Glatzel M, Kisters K, Mucke R, Bruns F, Schonekaes K et al. Selenium as radioprotector in head and neck cancer patients-First clinical results. Trace Elem Electrolytes 2006; 23: 178–180.
Fakih M, Cao S, Durrani FA, Rustum YM . Selenium protects against toxicity induced by anticancer drugs and augments antitumor activity: a highly selective, new, and novel approach for the treatment of solid tumors. Clin Colorectal Cancer 2005; 5: 132–135.
Yao X, Panichpisal K, Kurtzman N, Nugent K . Cisplatin nephrotoxicity: a review. Am J Med Sci 2007; 334: 115–124.
Camargo SMR, Francescato HDC, Lavrador MAS, Bianchi MLP . Oral administration of sodium selenite minimizes cisplatin toxicity on proximal tubules of rats. Biol Trace Elem Res 2001; 83: 251–262.
Hu YJ, Chen Y, Zhang YQ, Zhou MZ, Song XM, Zhang BZ et al. The protective role of selenium on the toxicity of cisplatin-contained chemotherapy regimen in cancer patients. Biol Trace Elem Res 1997; 56: 331–341.
Hadjibabaie M, Iravani M, Shamshiri AR, Zaker Z, Mousavi A, Alimoghaddam K et al. The prevalence of low selenium levels in adult patients undergoing bone marrow transplantation: a brief communication. Nutr Cancer 2008; 60: 837–839.
Mansouri A, Hadjibabaie M, Iravani M, Shamshiri AR, Hayatshahi A, Javadi MR et al. The effect of zinc sulfate in the prevention of high-dose chemotherapy-induced mucositis: a double-blind, randomized, placebo-controlled study. Hematol Oncol 2012; 30: 22–26.
Sonis ST . Oral mucositis. Anticancer Drugs 2011; 22: 607–612.
Paglia DE, Valentine WN . Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 1967; 70: 158–169.
Lilleby K, Garcia P, Gooley T, McDonnnell P, Taber R, Holmberg L et al. A prospective, randomized study of cryotherapy during administration of high-dose melphalan to decrease the severity and duration of oral mucositis in patients with multiple myeloma undergoing autologous peripheral blood stem cell transplantation. Bone Marrow Transplant 2006; 37: 1031–1035.
Spielberger R, Stiff P, Bensinger W, Gentile T, Weisdorf D, Kewalramani T et al. Palifermin for oral mucositis after intensive therapy for hematologic cancers. N Engl J Med 2004; 351: 2590–2598.
Keefe D, Lees J, Horvath N . Palifermin for oral mucositis in the high-dose chemotherapy and stem cell transplant setting: the Royal Adelaide Hospital Cancer Centre experience. Support Care Cancer 2006; 14: 580–582.
Keefe DM, Schubert MM, Elting LS, Sonis ST, Epstein JB, Raber-Durlacher JE et al. Updated clinical practice guidelines for the prevention and treatment of mucositis. Cancer 2007; 109: 820–831.
Sieja K, Talerczyk M . Selenium as an element in the treatment of ovarian cancer in women receiving chemotherapy. Gynecol Oncol 2004; 93: 320–327.
Nasilowska-Adamska B, Rzepecki P, Manko J, Czyz A, Markiewicz M, Federowicz I et al. The influence of palifermin (Kepivance) on oral mucositis and acute graft versus host disease in patients with hematological diseases undergoing hematopoietic stem cell transplant. Bone Marrow Transplant 2007; 40: 983–988.
Horsley P, Bauer JD, Mazkowiack R, Gardner R, Bashford J . Palifermin improves severe mucositis, swallowing problems, nutrition impact symptoms, and length of stay in patients undergoing hematopoietic stem cell transplantation. Support Care Cancer 2007; 15: 105–109.
Muecke R, Schomburg L, Glatzel M, Berndt-Skorka R, Baaske D, Reichl B et al. Multicenter, phase 3 trial comparing selenium supplementation with observation in gynecologic radiation oncology. Int J Radiat Oncol Biol Phys 2010; 78: 828–835.
Lymbury R, Tinggi U, Griffiths L, Rosenfeldt F, Perkins AV . Selenium status of the Australian population: effect of age, gender and cardiovascular disease. Biol Trace Elem Res 2008; 126: 1–10.
Hurst R, Armah CN, Dainty JR, Hart DJ, Teucher B, Goldson AJ et al. Establishing optimal selenium status: results of a randomized, double-blind, placebo-controlled trial. Am J Clin Nutr 2010; 91: 923–931.
Reid ME, Stratton MS, Lillico AJ, Fakih M, Natarajan R, Clark LC et al. A report of high-dose selenium supplementation: response and toxicities. J Trace Elem Med Biol 2004; 18: 69–74.
Acknowledgements
We express our gratitude to the staff of BMT wards 1 and 2, especially Mrs Mousavi and Mrs Shahriari, at the Hematology-Oncology and Stem Cell Transplantation Research Center, Shariati Hospital.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Jahangard-Rafsanjani, Z., Gholami, K., Hadjibabaie, M. et al. The efficacy of selenium in prevention of oral mucositis in patients undergoing hematopoietic SCT: a randomized clinical trial. Bone Marrow Transplant 48, 832–836 (2013). https://doi.org/10.1038/bmt.2012.250
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2012.250
Keywords
This article is cited by
-
Azithromycin oral suspension in prevention and management of oral mucositis in patients undergoing hematopoietic stem cell transplantation: a randomized controlled trial
Supportive Care in Cancer (2022)
-
Comparative Safety and Pharmacokinetic Evaluation of Three Oral Selenium Compounds in Cancer Patients
Biological Trace Element Research (2019)
-
Systematic review of natural and miscellaneous agents for the management of oral mucositis in cancer patients and clinical practice guidelines—part 1: vitamins, minerals, and nutritional supplements
Supportive Care in Cancer (2019)
-
Selenium supplementation in patients undergoing hematopoietic stem cell transplantation: effects on pro-inflammatory cytokines levels
DARU Journal of Pharmaceutical Sciences (2014)
-
N-acetyl cysteine for prevention of oral mucositis in hematopoietic SCT: a double-blind, randomized, placebo-controlled trial
Bone Marrow Transplantation (2014)