Abstract
The emerging concept of psychobiotics—live microorganisms with a potential mental health benefit—represents a novel approach for the management of stress-related conditions. The majority of studies have focused on animal models. Recent preclinical studies have identified the B. longum 1714 strain as a putative psychobiotic with an impact on stress-related behaviors, physiology and cognitive performance. Whether such preclinical effects could be translated to healthy human volunteers remains unknown. We tested whether psychobiotic consumption could affect the stress response, cognition and brain activity patterns. In a within-participants design, healthy volunteers (N=22) completed cognitive assessments, resting electroencephalography and were exposed to a socially evaluated cold pressor test at baseline, post-placebo and post-psychobiotic. Increases in cortisol output and subjective anxiety in response to the socially evaluated cold pressor test were attenuated. Furthermore, daily reported stress was reduced by psychobiotic consumption. We also observed subtle improvements in hippocampus-dependent visuospatial memory performance, as well as enhanced frontal midline electroencephalographic mobility following psychobiotic consumption. These subtle but clear benefits are in line with the predicted impact from preclinical screening platforms. Our results indicate that consumption of B. longum 1714 is associated with reduced stress and improved memory. Further studies are warranted to evaluate the benefits of this putative psychobiotic in relevant stress-related conditions and to unravel the mechanisms underlying such effects.
Similar content being viewed by others
Introduction
Recent years have seen growing research interest in the possibility of targeting the gut microbiome to beneficially impact on brain and behavior. A promising strategy in this field is that of psychobiotics—live microorganisms that convey a benefit upon the host’s mental health when consumed in adequate quantities.1 There is increasing interest in the impact of putative psychobiotics upon central nervous system processes, especially stress, mood, anxiety and cognition.2, 3 Preclinical research has indicated that chronic probiotic administration can reduce anxiety-like and depressive-like behavior, and can normalize associated physiological outputs such as corticosterone, noradrenaline, brain-derived neurotrophic factor and immune function.4, 5, 6, 7
Although most of the evidence in this area comes from animal studies, specific probiotic strains have shown potential for symptom alleviation in irritable bowel syndrome (IBS),8, 9 a stress-related brain–gut axis disorder associated with high rates of psychopathology10 as well as altered hypothalamic–pituitary–adrenal axis activity11 and cognition.12, 13 A number of encouraging proof-of-principle studies in healthy human volunteers have now demonstrated that multistrain probiotics, fermented drinks containing probiotics or prebiotics can alter resting brain activity, cognitive performance, baseline physiological stress outputs and self-reported psychological variables.5, 14, 15, 16, 17, 18 It is, however, unclear whether the use of single putative psychobiotic strains is also a viable approach in humans and whether a discovery pipeline can be developed to inform superior candidate strain selection as well as evaluation across a broader range of relevant parameters.
We have recently proposed a precision strategy in the use of psychobiotics for targeting stress-related central nervous system disorders including anxiety and depression.1 The rational selection of candidate strains identified in well-validated preclinical screening platforms is a more logical approach to guide the challenging move toward clinically useful psychobiotics. Accordingly, in preclinical studies, we have identified the Bifidobacterium longum 1714 strain, which selectively improves stress-related behaviours, physiology and cognitive performance.19, 20 The current study investigated whether these preclinical effects could be translated to healthy human volunteers.
We examined the effects of the 1714 strain compared with placebo on daily reported stress and the psychobiological response to an acute, controlled stressor.21, 22 We also assessed cognitive performance; in addition to our previous preclinical findings indicating improved memory, there is existing evidence that consumption of probiotics can affect performance on sustained attention performance,18 as well as in brain activity during social cognition.15 We thus assessed cognitive performance on tests assessing memory, sustained attention, social cognition and emotional processing. We also assessed brain activity in frontal, parietal and central regions using electroencephalography (EEG) following 4-week supplementation with the 1714 strain in comparison with placebo, as these regions have been associated with memory and sustained attention23, 24 and are sensitive to anxiolytics.25
Materials and methods
The research described received approval from the Clinical Research Ethics Committee of the Cork Teaching Hospitals (Protocol Number: APC044). Informed consent was obtained from all participants, who were free to withdraw from the study at any time.
Design
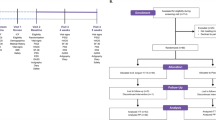
A repeated measures, placebo-controlled design was employed. A repeated measures design was employed in order to exclude the effects of individual differences across variables. Participants were screened at an initial visit for psychiatric disorder using the MINI International Neuropsychiatric Interview (MINI26), and demographic and baseline psychological information was collected. Following screening, participants completed neurocognitive visits and acute stress visits. Participants were then administered placebo for four weeks followed by the 1714 strain for four weeks, with the second and third set of visits following the placebo and probiotic phases. Participants also completed a 2-week post-probiotic follow-up. Participants filled in daily online questionnaires from the end of the first set of visits to the end of follow-up.
Participants
With a power of 0.8 for a one-way analysis of variance, a minimum sample size of 20 was required to demonstrate an effect sized f=0.3 at α=0.05.27 The study was completed by 22 healthy male volunteers (see Table 1 for detailed participant characteristics). Male participants were selected to avoid the need to control for menstrual cycle, which can impact upon cortisol output and other readouts. Participants were aged between 18 and 40 years of age. Exclusion criteria were as follows: having a significant acute or chronic illness; having a condition, following a diet or taking a medication that would interfere with study objectives, pose a safety risk or confound the interpretation of the study results; evidence of immunodeficiency, bleeding disorder or coagulopathy; English not being participant’s first language; colour blindness, dyslexia or dyscalculia; smoking; self-report habitually taking any probiotic products; receiving any treatment involving experimental drugs (see Supplementary Figure 1 for recruitment flowchart).
Materials
Both placebo and probiotic sticks contained maltodextrin and magnesium stearate; probiotic sticks additionally contained 1 × 109 colony-forming units per stick of the 1714 strain. Participants were instructed to take one stick each morning by mixing the contents of the stick into milk and drinking.
Self-report
Online questionnaires were completed each day: the Cohen Perceived Stress Scale,28 and a question on whether participants had taken their probiotic that day. Online questionnaires were administered using limesurvey software.29
Cognitive tasks
Tests from the Cambridge Neuropsychological Test Automated Battery (CANTAB30) were presented on a touch-screen monitor, Sahara i440D Slate Tablet PC (Sand Dune Ventures, Tablet Kiosk, Torrance, CA, USA) running CANTABeclipse software (Cambridge, UK). The researcher provided verbal instructions to participants from a standardised script. Tests were presented in different orders for different participants, using a Latin square design, to avoid effects of fatigue for tests completed later in the session. The test battery lasted ~45 min in total. The Paired Associate Learning (PAL) test was used to assess conditional learning of pattern–location associations. Paired associate learning performance has shown sensitivity to functional changes in the hippocampus31 and frontal lobes.23 The parallel mode (which presents different shapes at each visit) was used in order to avoid practice effects.
Rapid visual information processing was used to assess sustained attention. Performance on this task activates a frontoparietal network of brain regions,24 and a modified version of this task is sensitive to changes in quantitative frontal EEG.32
Emotion Recognition Task was used to assess social cognition. Functional magnetic resonance imaging has previously demonstrated probiotic effects on a network of brain regions involved in emotional and viscerosensory processing in healthy controls.15
In addition to tests from the CANTAB battery, we assessed emotional processing using an Emotional Stroop.33 The Stroop test was presented on the same high-resolution touch-screen monitor used with the CANTAB battery. The emotional Stroop is associated with activation in the anterior cingulate cortex.34, 35 Positively, negatively and neutrally valenced words were presented, matched for length in letters, orthographic neighbourhood size (that is, the number of words which differ from a given word by only one letter) and frequency of use.36
We used an auditory oddball task to study the P300 event-related potential, which has been shown to be sensitive to anxiolytic effects at the cerebral electrode locations assessed in the current study.25 See Supplementary Table 1 for detailed description of the cognitive tasks.
Neurocognitive assessment
Prior to EEG testing participants were asked to refrain from caffeine on the morning of their experimental session, as well as ensuring they got a good night’s sleep, to remove any piercings and avoid wearing hair gel. All EEG measurements were made using a Neuroscan, SynAmps 2 Amplifier and Neuroscan 4.3.1 acquisition software (Compumedics, Abbotsford, VIC, Australia). EEG data were recorded at a sampling rate of 1000 Hz. Scalp electrodes were attached at midline positions Fz, Pz and Cz, according to the international 10/20 system, as well as mastoid electrodes and a reference electrode on the nose. Vertical eye movements were detected using electrodes attached above and below the orbit of the left eye and horizontal eye movements were monitored by electrodes at the right and left outer canthi. Electrodes were applied to the scalp using soft paste and secured using tape and an elasticated hat.
Following a resting EEG recording, the cognitive tasks were completed (see Cognitive tasks). EEG was recorded continuously during performance of the auditory oddball task, but was not measured during the other cognitive tasks to avoid artefacts in the EEG readings.
Resting EEG
EEG measures of absolute power in the delta (1.5–3.5 Hz), theta (4–7.5 Hz), alpha1 (8–9.5 Hz), alpha2 (10–12.5 Hz), beta1 (13–17.5 Hz) and beta2 (18–25.5 Hz) frequency bands were taken for an initial five minute period with eyes closed (based on Romano-Torres et al.37). Participants were requested to relax and sit still with their eyes closed while resting EEG was recorded. Resting EEG has been shown to be consistent across repeated visits,38 and so should not be vulnerable to carryover effects.
Acute stress procedure
We employed the socially evaluated cold pressor test (SECPT)21 as a combined psychological and physiological stressor procedure, which has been shown not to induce hypothalamic–pituitary–adrenal axis habituation across repeated exposures.39 Participants were required to avoid alcohol for 24 h prior to the visit, as well as caffeinated beverages on the day of the stress procedure and strenuous exercise from 1400 hours the day before, and to fast for 2 h prior to testing. Pre-stress, the participant completed the state items from the state-trait anxiety inventory,40 and a baseline saliva sample was taken. The baseline measures were followed by a 5-min resting phase. Following the 5-min resting phase, another saliva sample was taken. The participant read the instructions for the SECPT and the experimenter answered any questions. The participant then completed the SECPT (see Supplementary Table 2 for full details). Following completion of the SECPT the participant completed the post-stress state anxiety questionnaire. Further saliva samples were taken 1 min after the cessation of the stressor, as well as 10, 20, 30, and 60 min post-stressor cessation.
Analysis
EEG analysis
Resting EEG, frequency domain Data were examined for eyeblink artefacts. After correction for eyeblinks, data were epoched, with an epoch duration of 2 s. A linear detrend based on the entire sweep was applied to the epoched file, which was then baseline corrected for the pre-stimulus interval. A band pass filter (0.1–35 Hz, IIR, 24 dB/octave) was applied. Following a further visual inspection for artefacts and baseline correction the epochs were averaged. Multiplication of data by a 25% cosine window was applied, as this has been used previously in examining anxiolytic effects.41 EEG measures of absolute power were extracted in the delta (1.5–3.5 Hz), theta (4–7.5 Hz), alpha1 (8–9.5 Hz), alpha2 (10–12.5 Hz), beta1 (13–17.5 Hz) and beta2 (18–25.5 Hz) frequency bands.
Resting EEG, time domain The EEG signal was downsampled from 1000 Hz to 256 Hz with an antialiasing filter set at 128 Hz. The filtered EEG signal was segmented into 1 s window without overlap. Curve length, number of maxima and minima, root mean squared amplitude, Hjorth parameters 42 (activity, mobility and complexity), zero crossings (raw epoch, Δ, ΔΔ), autoregressive modelling error (model order 1–9), nonlinear energy, variance (Δ, ΔΔ) were calculated using MATLAB. Mobility is an estimate of root-mean-square frequency. Mobility=std(y(t)dy/dt)/(y(t)), where the y(t) is the EEG amplitude at time, t.
Event-related potential Data were divided into epochs of 300 ms duration (200 to 500 ms post-oddball stimulus). Following a linear detrend and a baseline correction, data were visually examined for any artefacts and these were removed. Oddball epochs were averaged, and a band pass filter and further baseline correction were applied. The peak frequency was identified between 200 ms and 500 ms post-oddball for the P300.
Sample analysis
Salivary cortisol was analysed using Enzo Life Sciences (Exeter, UK) enzyme-linked immunosorbent assay (ELISA) kits (Catalogue no: ADI-901-071) according to manufacturer’s instructions. Lower limit of detection=0.16 nmol l−1. Inter and intra-assay coefficients of variability were 11.24% and 8.2%, respectively.
Statistical analysis
Data were analysed using SPSS 21 (IBM, Armonk, NY, USA). Repeated measures analysis of variance and t-tests (two-sided) were used to examine differences between conditions, and non-parametric equivalents (Friedman and Wilcoxon, respectively) were used where parametric assumptions were violated. Areas under the curve with respect to ground (AUCg) were also calculated,43 and analysed in the same manner. EEG data for three participants were removed (two had excessive levels of artefacts and one participant suffered a tension headache when the EEG equipment was attached). Daily questionnaire data for three participants were removed due to lack of response to online questionnaires.
Results
Acute stress response to socially evaluated cold pressor test
The stressor significantly increased salivary cortisol at all visits. Salivary cortisol was significant elevated at its peak (30 min following onset of stress) compared with immediately pre-stress (time 0) for all three conditions (visit one: T=5, P<0.001, r=0.8, post-placebo: T=4.67, P<0.001, r=0.78, post-1714: T=2, P<0.001, r=0.94), but there was not an interaction between probiotic condition and stressor time point, F(1.86, 37.11)=1.81, P=0.18 (Greenhouse-Geisser adjusted; Figure 1a). Nonetheless, total cortisol output, as measured by AUCg, was significantly affected by probiotic condition, χ2(2)=8.67, P<0.05, although area under the curve with respect to increase (AUCi) was not affected, χ2(2)=3.71, P>0.05 (Figure 1b). For AUCg, cortisol output was lower post-1714, both compared with the post-placebo, T=10.69, P=0.05, r=0.42, and compared with the first visit, T=10.94, P<0.05, r=0.45.
The socially evaluated cold pressor test (SECPT) elevated cortisol, and cortisol output and the increase in anxiety were reduced post-1714. (a) Salivary cortisol during socially evaluated cold pressor for each visit. (b) Total cortisol output for each visit, as measured by area under the curve with respect to ground (AUCg) and area under the curve with respect to increase (AUCi). (c) State anxiety (STAI) pre- and post-stressor at each visit. Lower scores indicate lower subjective anxiety. Error bars represent the s.e.m. *P<0.05, **P<0.001. HPA, hypothalamic-pituitary-adrenal.
There was a main effect of stressor on reported state anxiety, F(1, 21)=6.39, P=0.02. State anxiety was significantly elevated post-stressor at both visit one, T=8.58, P<0.05, r=0.43, and post-placebo, T=7.7, P<0.01, r=0.57. However, this was not the case post-1714, T=9.13, P>0.05, r=0.12 (Figure 1c). The mean time participants kept their hand in the water was M=157.5 s, s.d.=47.3 for visit 1, M=167.7, s.d.=39.5 post-placebo and M=170.2, s.d.=38 post-1714.
Reported daily stress and bowel satisfaction
Daily stress levels were broadly similar in both conditions during their first week, but became marginally lower in the 1714 condition by week 4, t(18)=1.95, P=0.07, Cohen’s d=0.44. Further, stress levels returned to a higher level during the 2-week follow-up period (Figure 2a). Overall stress, as measured with AUCg, was significantly lower during the 1714 condition compared with the placebo, t(18)=2.32, P=0.03, Cohen’s d=0.53, and AUCi was lower in the 1714 condition, although this was not statistically significant, t(18)=1.44, P>0.05, Cohen’s d=0.33 (Figure 2b). There was reduced AUCg and AUCi for bowel satisfaction in the 1714 condition (Supplementary Figures 2A and B), due to a brief, non-significant reduction in reported bowel habit satisfaction at week 2 of the 1714 condition, but this did not persist (Supplementary Figure 2C).
The probiotic condition was associated with reduced daily stress. (a) Daily stress levels during placebo phase, 1714 phase, and follow-up period. (b) Overall stress, as measured with AUCg and AUCi, during placebo phase, 1714 phase. *P<0.05. AUCg, area under the curve with respect to ground; AUCi, area under the curve with respect to increase.
Neurocognitive performance
Total errors on the paired associate learning task were significantly affected by condition, χ2(2)=10.46, P<0.01. Compared with the first visit, participants made fewer errors on the paired associate learning test both at post-placebo, (T=7.5, P<0.05, r=0.5) and post-1714, (T=6.63, P<0.01, r=0.59), a subtle effect at post-1714 that was greater than the placebo response (Figure 3). See Supplementary Table 3 for full details of results.
EEG
Fz mobility differed significantly across conditions, χ2(2)=13.37, P=0.01 (see Figures 4a for electrode positions). Mobility was significantly higher post-1714 compared with both post-probiotic, T=37, P=0.02, r=0.31 and visit one, T=39, P=0.02, r=0.3 (Figure 4b). Cz theta power differed significantly across 1714 conditions, χ2(2)=10.31, P<0.01. Cz theta power was significantly lower post-1714 compared with post-placebo, T=8, P<0.05, r=0.57, although theta power at post-1714 did not differ from visit one (Figure 4c). There was no impact of 1714 on P300 latency or amplitude (Supplementary Figure 3).
Discussion
The current research indicates that a putative psychobiotic, the B. longum 1714 strain, which has shown anti-stress and procognitive effects in healthy mice19, 20 can also ameliorate both the physiological and psychological response to an acute stressor, as well as longer-term daily self-reported psychological stress, in healthy human adults. Furthermore, there was a subtle improvement over placebo in visuospatial memory performance post-psychobiotic, as well as an EEG profile consistent with improved memory. Whereas previous studies in healthy humans have often examined probiotic cocktails, we believe this is the first study to examine the effect of a single psychobiotic strain upon a wide range of stress outcomes and neurocognitive evaluations informed by a preclinical screening platform.19, 20 We employed a repeated measures design to control for potential effects of individual differences, with carefully selected cognitive and stress challenges that are not associated with habituation over repeated testing within this timeframe.
Following the psychobiotic intervention, cortisol output during the SECPT was lower, and the increase in self-reported anxiety was no longer significant. Interestingly, in our prior animal studies we also showed there was a blunted physiological response to stress, albeit manifested as a reduction in stress-induced hyperthermia, without any alteration in corticosterone output.19 This difference highlights the challenges in translating the findings from animal studies to human populations and reinforces the need for multiple stress and behavioural readouts in preclinical screening platforms. The psychobiotic effect on the acute stress response is complemented by a reduction in daily perceived stress that is consistent with previous findings that a probiotic intervention can affect subjective everyday stress.5
The subtle improvement in visuospatial memory following probiotic administration is consistent with preclinical findings of enhanced learning and memory in response to this strain.20 This cognitive effect was further supported by the observation of enhanced frontal midline (Fz) mobility using EEG. Activity at Fz is representative of prefrontal cortex activity,44 and the prefrontal cortex is associated with paired associate learning.23 As prefrontal activity appears to be specifically associated with top–down processing during associative memory,23 the current EEG findings thus offer a potential insight into the timing of cognitive processes underlying the observed changes in paired associate performance, by suggesting that the impact of the psychobiotic on cognitive performance may be due to an effect on top–down processing, rather than simply affecting retention. Mobility has previously been shown to be higher in healthy controls compared with patients with Alzheimer’s disease,45 a neurodegenerative disorder with pronounced and progressive memory deficits.46 Although Alzheimer’s disease is a severe clinical condition, it is worth noting that the observed effects in Alzheimer’s patients were stronger frontocentrally, which is where the change in mobility was observed post-psychobiotic. The reduction in theta power post-psychobiotic compared with post-placebo is also consistent with improved memory performance.47
Further research is warranted to further elucidate mechanisms by which the 1714 strain could impact upon stress and neurocognition. It is possible that the reduction in daily stress and acute stress response may account for the observed effect on visuospatial memory. PAL test performance is dependent upon the hippocampus,31, 48 which has a high proportion of glucocorticoid receptors,49 and PAL test performance is poorer in subjects with stress-related disorders with associated hypothalamic–pituitary–adrenal axis dysfunction, such as IBS.13 B. longum NCC3001 enhanced hippocampal brain-derived neurotrophic factor messenger RNA and reduced anxiety-like behavior in mice infected with the non-invasive parasite T Muris50 and B. breve 6330 increased brain-derived neurotrophic factor total variants in healthy rats.51 It is thus of interest whether brain-derived neurotrophic factor levels and neurogenesis are heightened in humans following psychobiotic ingestion, thus having a key role in the impact of psychobiotic consumption upon the central nervous system. Furthermore, given previous evidence that psychobiotic effects in rodents were dependent upon the vagus nerve,7 it is possible that vagal activity may mediate such psychobiotic effects. However, strain-specific mechanisms are likely and need to be investigated on a case by case basis.
These results provide the impetus for further evaluation of this psychobiotic in pathological conditions. This could initially target stress-related brain–gut axis disorders with a cognitive component such as IBS, which has been found to be associated with a deficit in visuospatial memory performance, evident in PAL test performance,12, 13 and we have shown PAL performance to be subtly improved by the 1714 strain. Furthermore, the observed psychological effects of psychobiotics warrant research in other stress-related disorders including depression and anxiety.
In line with the predicted impact from preclinical research in this specific strain, the 1714 strain is a promising candidate psychobiotic strain associated with attenuated responses to psychological and physiological stress and a modest improvement over placebo in cognitive performance, as well as with altered resting EEG output in healthy volunteers. Further studies are warranted to evaluate the benefits of this putative psychobiotic in relevant stress-related conditions and to unravel the mechanisms underlying such effects.
References
Dinan TG, Stanton C, Cryan JF . Psychobiotics: a novel class of psychotropic. Biol Psychiatry. 2013; 74: 720–726.
Cryan JF, Dinan TG . Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci 2012; 13: 701–712.
Dinan TG, Stilling RM, Stanton C, Cryan JF . Collective unconscious: How gut microbes shape human behavior. J Psychiatr Res 2015; 63: 1–9.
Desbonnet L, Garrett L, Clarke G, Kiely B, Cryan JF, Dinan TG . Effects of the probiotic Bifidobacterium infantis in the maternal separation model of depression. Neuroscience 2010; 170: 1179–1188.
Messaoudi M, Lalonde R, Violle N, Javelot H, Desor D, Nejdi A et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br J Nutr 2011; 105: 755–764.
Bercik P, Denou E, Collins J, Jackson W, Lu J, Jury J et al. The intestinal microbiota affect central levels of brain-derived neurotropic factor and behavior in mice. Gastroenterology 2011; 141: 599–609.e3.
Bravo JA, Forsythe P, Chew MV, Escaravage E, Savignac HM, Dinan TG et al. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc Natl Acad Sci USA 2011; 108: 16050–16055.
Whorwell PJ, Altringer L, Morel J, Bond Y, Charbonneau D, O'Mahony L et al. Efficacy of an encapsulated probiotic Bifidobacterium infantis 35624 in women with irritable bowel syndrome. Am J Gastroenterol 2006; 101: 1581–1590.
O’Mahony L, McCarthy J, Kelly P, Hurley G, Luo F, Chen K et al. Lactobacillus and bifidobacterium in irritable bowel syndrome: Symptom responses and relationship to cytokine profiles. Gastroenterology 2005; 128: 541–551.
Whitehead WE, Palsson O, Jones KR . Systematic review of the comorbidity of irritable bowel syndrome with other disorders: What are the causes and implications? Gastroenterology 2002; 122: 1140–1156.
Kennedy PJ, Cryan JF, Quigley EMM, Dinan TG, Clarke G . A sustained hypothalamic–pituitary–adrenal axis response to acute psychosocial stress in irritable bowel syndrome. Psychol Med 2014; 44: 3123–3134.
Kennedy PJ, Allen AP, O’Neill A, Quigley EM, Cryan JF, Dinan TG et al. Acute tryptophan depletion reduces kynurenine levels: implications for treatment of impaired visuospatial memory performance in irritable bowel syndrome. Psychopharmacology (Berl) 2015; 232: 1357–1371.
Kennedy PJ, Clarke G, O‘Neill A, Groeger JA, Quigley EMM, Shanahan F et al. Cognitive performance in irritable bowel syndrome: evidence of a stress-related impairment in visuospatial memory. Psychol Med 2014; 44: 1553–1566.
Benton D, Williams C, Brown A . Impact of consuming a milk drink containing a probiotic on mood and cognition. Eur J Clin Nutr 2007; 61: 355–361.
Tillisch K, Labus J, Kilpatrick L, Jiang Z, Stains J, Ebrat B et al. Consumption of fermented milk product with probiotic modulates brain activity. Gastroenterology 2013; 144: 1394–401.e4.
Schmidt K, Cowen PJ, Harmer CJ, Tzortzis G, Errington S, . Burnet PWJ. Prebiotic intake reduces the waking cortisol response and alters emotional bias in healthy volunteers. Psychopharmacology (Berl) 2014; 232: 1793–1801.
Steenbergen L, Sellaro R, van Hemert S, Bosch JA, Colzato LS . A randomized controlled trial to test the effect of multispecies probiotics on cognitive reactivity to sad mood. Brain Behav Immun 2015; 48: 258–264.
Chung Y-C, Jin H-M, Cui Y, Kim DS, Jung JM, Park J-I et al. Fermented milk of Lactobacillus helveticus IDCC3801 improves cognitive functioning during cognitive fatigue tests in healthy older adults. J Func Foods 2014; 10: 465–474.
Savignac HM, Kiely B, Dinan TG, Cryan JF . Bifidobacteria exert strain-specific effects on stress-related behavior and physiology in BALB/c mice. Neurogastroenterol Motil 2014; 26: 1615–1627.
Savignac HM, Tramullas M, Kiely B, Dinan TG, Cryan JF . Bifidobacteria modulate cognitive processes in an anxious mouse strain. Behav Brain Res 2015; 287: 59–72.
Schwabe L, Haddad L, Schachinger H . HPA axis activation by a socially evaluated cold-pressor test. Psychoneuroendocrinology 2008; 33: 890–895.
Schwabe L, Wolf OT . Socially evaluated cold pressor stress after instrumental learning favors habits over goal-directed action. Psychoneuroendocrinology 2010; 35: 977–986.
Hales JB, Israel SL, Swann NC, Brewer JB . Dissociation of frontal and medial temporal lobe activity in maintenance and binding of sequentially presented paired associates. J Cogn Neurosci 2009; 21: 1244–1254.
Coull JT, Frith CD, Frackowiak RSJ, Grasby PM . A fronto-parietal network for rapid visual information processing: a PET study of sustained attention and working memory. Neuropsychologia 1996; 34: 1085–1095.
Fukami G, Hashimoto T, Shirayama Y, Hasegawa T, Watanabe H, Fujisaki M et al. Effects of etizolam and ethyl loflazepate on the P300 event-related potential in healthy subjects. Ann Gen Psychiatry 2010; 9: 37.
Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E et al. The Mini-International Neuropsychiatric Interview (MINI): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 1998; 59: 22–33.
Faul F, Erdfelder E, Lang AG, Buchner A . G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 2007; 39: 175–191.
Cohen S, Kamarck T, Mermelstein R . A global measure of perceived stress. J Health Soc Behav 1983; 24: 385–396.
Schmitz LPTC. . LimeSurvey: an Open Source Survey Tool. Limesurvey Project: Hamburg, Germany, 2015.
Robbins TW, Sahakian B. Computer methods of assessment of cognitive function. In Copeland JRM, Abou-Saleh MT, Blazer DG (eds). Principles and Practice of Geriatric Psychiatry. John Wiley & Sons Ltd: Chichester, UK, 1994 pp 205–209.
de Rover M, Pironti VA, McCabe JA, Acosta-Cabronero J, Arana FS, Morein-Zamir S et al. Hippocampal dysfunction in patients with mild cognitive impairment: a functional neuroimaging study of a visuospatial paired associates learning task. Neuropsychologia 2011; 49: 2060–2070.
Allen AP, Jacob TJC, Smith AP . Effects and after-effects of chewing gum on vigilance, heart rate, EEG and mood. Physiol Behav 2014; 133: 244–251.
Strauss E, Sherman EMS, Spreen O . A Compendium of Neuropsychological Tests: Administration, Norms, & Commentary. 3rd edn, Oxford University Press: New York, USA, 2006.
Bush G, Luu P, Posner MI . Cognitive and emotional influences in anterior cingulate cortex. Trends Cogn Sci 2000; 4: 215–222.
Etkin A, Egner T, Peraza DM, Kandel ER, Hirsch J . Resolving emotional conflict: a role for the rostral anterior cingulate cortex in modulating activity in the amygdala. Neuron 2006; 51: 871–882.
Larsen RJ, Mercer KA, Balota DA, Strube MJ . Not all negative words slow down lexical decision and naming speed: Importance of word arousal. Emotion 2008; 8: 445–452.
Romano-Torres M, Borja-Lascurain E, Chao-Rebolledo C, del-Rio-Portilla Y, Corsi-Cabrera M . Effect of diazepam on EEG power and coherent activity: sex differences. Psychoneuroendocrinology 2002; 27: 821–833.
Gudmundsson S, Runarsson TP, Sigurdsson S, Eiriksdottir G, Johnsen K . Reliability of quantitative EEG features. Clin Neurophysiol 2007; 118: 2162–2171.
Minkley N, Schröder TP, Wolf OT, Kirchner WH . The socially evaluated cold-pressor test (SECPT) for groups: Effects of repeated administration of a combined physiological and psychological stressor. Psychoneuroendocrinology 2014; 45: 119–127.
Spielberger CD, Gorsuch RL, Lushene RE . The State-trait Anxiety Inventory. Consulting Psychology Press: Palo Alto, CA, USA, 1970.
Barbanoj MJ, Anderer P, Antonijoan RM, Torrent J, Saletu B, Jané F . Topographic pharmaco-EEG mapping of increasing doses of buspirone and its comparison with diazepam. Hum Psychopharmacol 1994; 9: 101–109.
Hjorth B . EEG analysis based on time domain properties. Electroencephalogr Clin Neurophysiol 1970; 29: 306–310.
Pruessner JC, Kirschbaum C, Meinlschmid G, Hellhammer DH . Two formulas for computation of the area under the curve represent measures of total hormone concentration versus time-dependent change. Psychoneuroendocrinology 2003; 28: 916–931.
Asada H, Fukuda Y, Tsunoda S, Yamaguchi M, Tonoike M . Frontal midline theta rhythms reflect alternative activation of prefrontal cortex and anterior cingulate cortex in humans. Neurosci Lett 1999; 274: 29–32.
Martin-Loeches M, Garcia-Trapero J, Gil P, Rubia FJ . Topography of mobility and complexity parameters of the EEG in Alzheimer's disease. Biol Psychiatry 1991; 30: 1111–1121.
Sperling RA, Dickerson BC, Pihlajamaki M, Vannini P, LaViolette PS, Vitolo OV et al. Functional alterations in memory networks in early Alzheimer’s disease. Neuromolecular Med 2010; 12: 27–43.
Klimesch W . EEG alpha and theta oscillations reflect cognitive and memory performance: A review and analysis. Brain Res Rev 1999; 29: 169–195.
Eichenbaum H, Bunsey M . On the binding of associations in memory: clues from studies on the role of the hippocampal region in paired-associate learning. Curr Direct Psychol Sci 1995; 4: 19–23.
McEwen BS . Stress and the aging hippocampus. Front Neuroendocrinol 1999; 20: 49–70.
Bercik P, Verdu EF, Foster JA, Macri J, Potter M, Huang X et al. Chronic gastrointestinal inflammation induces anxiety-like behavior and alters central nervous system biochemistry in mice. Gastroenterology 2010; 139: 2102–12.e1.
O'Sullivan E, Barrett E, Grenham S, Fitzgerald P, Stanton C, Ross R et al. BDNF expression in the hippocampus of maternally separated rats: does Bifidobacterium breve 6330 alter BDNF levels? Benef Microbes 2011; 2: 199–207.
Beck AT, Steer RA, Brown GK . Manual for the Beck Depression Inventory-II. Psychological Corporation: San Antonio, USA, 1996.
Nelson H, Willison J . National adult reading test (NART): test manual, 2nd edn. NFER Nelson: Windsor, ON, Canada, 1991.
Acknowledgements
Research was conducted in the APC Microbiome Institute, a research centre funded by Science Foundation Ireland (SFI), through the Irish Government’s National Development Plan (grant number SFI/12/RC/2273). GB is based in the INFANT research centre, also funded by the SFI (grant number SFI/12/RC/2272). The authors and their work are also funded by the Health Research Board (HRB) through Health Research Awards (grants no HRA_POR/2011/23; TGD, JFC and GC, HRA_POR/2012/32; JFC, TGD and HRA-POR-2-14-647: GC, TGD) and through EU GRANT 613979 (MYNEWGUT FP7-KBBE-2013-7: TGD, JFC). The APC Microbiome Institute has conducted studies in collaboration with several companies including Mead Johnson, Cremo, Suntory Wellness, Nutricia and 4D Pharma. GC is supported by a NARSAD Young Investigator Grant from the Brain and Behavior Research Foundation (Grant Number 20771). The placebo and probiotic containing B. longum 1714 was kindly provided by Alimentary Health. 1714 is a trademark of Alimentary Health. Purchase of the CANTAB software used in the study was supported by UCC’s strategic research fund. We thank Anne-Marie Cusack and Katie Power for assistance with sample collection, Dr Gerard Moloney and Katie Togher for assistance with sample preparation, as well as Anna Connolly, Ana Paula Costa, Eileen Curran, Maria Donovan, Jennifer Keyes, Pauline Luczinski, Dr Áine Duggan, Dr Karen-Anne Neufeld McVey and Dr Karen Scott for acting as confederates for the socially evaluated cold pressor test.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
EM is Research Director at Alimentary Health. Alimentary Health provided the placebo and probiotic containing the B. longum 1714 strain. The APC Microbiome Institute has conducted studies in collaboration with several companies including Mead Johnson, Cremo, Suntory Wellness, Nutricia and 4D Pharma. The remaining authors declare no conflicts of interest.
Additional information
Supplementary Information accompanies the paper on the Translational Psychiatry website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Allen, A., Hutch, W., Borre, Y. et al. Bifidobacterium longum 1714 as a translational psychobiotic: modulation of stress, electrophysiology and neurocognition in healthy volunteers. Transl Psychiatry 6, e939 (2016). https://doi.org/10.1038/tp.2016.191
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tp.2016.191
This article is cited by
-
Efficacy of an eHealth self-management program in reducing irritable bowel syndrome symptom severity: a randomized controlled trial
Scientific Reports (2024)
-
The effect of psychoactive bacteria, Bifidobacterium longum Rosell®-175 and Lactobacillus rhamnosus JB-1, on brain proteome profiles in mice
Psychopharmacology (2024)
-
Alterations in human health parameters during stressful ship voyage to Antarctica: effects of probiotics intervention
Polar Biology (2024)
-
Butterflies in the gut: the interplay between intestinal microbiota and stress
Journal of Biomedical Science (2023)
-
Association of probable post-traumatic stress disorder with dietary pattern and gut microbiome in a cohort of women
Nature Mental Health (2023)