Abstract
Addictions are serious and common psychiatric disorders, and are among the leading contributors to preventable death. This selective review outlines and highlights the need for a multi-method translational approach to genetic studies of these important conditions, including both licit (alcohol, nicotine) and illicit (cannabis, cocaine, opiates) drug addictions and the behavioral addiction of disordered gambling. First, we review existing knowledge from twin studies that indicates both the substantial heritability of substance-specific addictions and the genetic overlap across addiction to different substances. Next, we discuss the limited number of candidate genes which have shown consistent replication, and the implications of emerging genomewide association findings for the genetic architecture of addictions. Finally, we review the utility of extensions to existing methods such as novel phenotyping, including the use of endophenotypes, biomarkers and neuroimaging outcomes; emerging methods for identifying alternative sources of genetic variation and accompanying statistical methodologies to interpret them; the role of gene–environment interplay; and importantly, the potential role of genetic variation in suggesting new alternatives for treatment of addictions.
Similar content being viewed by others
Introduction
The term ‘addiction’ covers a broad range of maladaptive aspects of drug use or other behaviors leading to clinically significant impairment or distress.1 Addiction includes alcohol use disorders, nicotine dependence, cannabis and cocaine use disorders as well as non-substance–related behaviors. These serious but common psychiatric disorders are among the leading contributors to morbidity and mortality worldwide.2, 3
This review discusses: (a) classification and diagnosis of addictions; (b) studies demonstrating the role of heritable variation in addiction and the overlap of heritable influences across drug classes; (c) putative candidate genes and emerging results from genomewide association studies; and (d) novel research methods to advance phenotyping of addictions, including the use of endophenotypes, biomarkers and imaging technology; (e) the role of alternative sources of genetic variants and bioinformatics; (f) gene–environment interplay; and (g) the emergence of pharmacogenomics. Finally, we discuss how the application of these various methods has begun to elucidate processes underlying nicotine addiction and the strategies this enables for new treatments.
Classification and diagnostic criteria
Addictions are primarily diagnosed using the Diagnostic and Statistical Manual of Mental Disorders (DSM, currently DSM-IV)1 according to which substance abuse and dependence are distinct categories—throughout this review, we use the term ‘addiction’ to refer to substance abuse or dependence. Proposed changes to the DSM system of nomenclature (DSM-5, http://www.dsm5.org/Pages/Default.aspx) may eliminate the distinction between abuse and dependence, replacing it with a single category of substance use disorder (see Box 1 for diagnostic criteria).4 Also proposed for DSM-5 is the addition of ‘behavioral addictions’.5 A number of candidate behavioral addictions were considered, such as compulsive internet use (for example, gaming), sexual activity or shopping, but the only behavioral addiction that is currently formally codified is disordered gambling. This decision was based, in part, on symptomatic and neurobiological similarities between disordered gambling and substance use disorders6 and the more developed research base for disordered gambling than for the other behavioral addictions.
Given the enormous public health burden of addictions, the delineation of their etiology has been of paramount importance. The next sections detail research findings outlining the magnitude of heritable influences on addictions.
Heritable influences on addiction
Numerous family, adoption and twin studies have identified the significant role of heritable influences on individual differences in addiction. Results from twin studies suggest that 33–71% of the variation in liability to nicotine dependence7, 8, 9, 10 can be attributed to heritable influences, while 48–66% of the variation in alcohol dependence11, 12, 13, 14 is heritable. Similarly, a recent meta-analysis of eight twin studies reported heritability estimates of 51–59% for cannabis addiction.15 Heritability estimates for cocaine use disorders range from 42 to 79%, with the lower estimates reported for females.16, 17, 18 Two large-scale studies have examined opioid addiction. Kendler et al.16 reported that 23% of the variation in opioid addiction in men was attributable to genetic factors, whereas Tsuang et al.19 reported a considerably higher estimate of 54% in male Vietnam Era twins. There have also been two major twin studies of disordered gambling, with consistent evidence for heritable (49%) variation.20, 21 Across these studies, there has been no consistent evidence for differences in the magnitude or nature of heritable influences on addiction in men and women. However, two important factors have been found to contribute to variation in heritability:
-
a)
Stages of addiction: Despite the typical clinical characterization of individuals as affected versus unaffected for addiction, research has shown that liability to addiction is a multi-stage process.7, 22, 23 The process of addiction begins with early stages of initiation of use, followed by escalation to regular and chronic use, which can become problematic and develop into addiction. Early stages are less heritable and more greatly influenced by familial environmental factors, whereas later stages, such as problem use and dependence are more strongly influenced by heritable factors. Multiple twin studies have examined the extent to which genetic influences on these later stages overlap with those influencing initiation and non-problem use.24 Although a significant proportion of genetic factors influencing problem use also influence earlier stages, there is also support for genetic factors that are specific to problem use and contribute to its increased heritability.
-
b)
Developmental course: Due to the natural course of addiction, studying the role of developmental stage on addiction is challenging. Initiation of drug use typically occurs during adolescence, whereas disorders emerge during early adulthood. Even within these stages, there is considerable variation in etiology, depending on whether adolescents or adults are being studied.25, 26 For instance, for a history of any alcohol use, heritability declines rapidly during emerging adulthood, likely due to the ubiquity of normative alcohol consumption. By contrast, for measures of quantity/frequency of use (for example, drinks/day),27 and the number of substances used,28, 29 heritable factors appear to have a stronger influence during adulthood. During adolescence, shared environmental factors contribute maximally to familial resemblance, but with the emergence of adulthood, genetic influences are unveiled and heritable variation explains up to 75% of individual differences.27 For problem use, however, there has been consistent evidence for heritable influences, even during adolescence.30
Drug-specific genetic influences
There is significant overlap in genetic influences on alcohol, nicotine and illicit drug addictions, as well as across addiction and other externalizing disorders. Particularly for illicit drugs, with the possible exception of opioids, twin studies have found only modest support for specific genetic factors,17, 31 supporting the possible role of common pathways (for example, via dopaminergic neurotransmission) that connect problem use of multiple drugs. However, for nicotine dependence, genetic factors shared with alcohol and illicit drugs are responsible for only 37% of heritable variation, indicating a considerable degree of genetic specificity.32 Given their very different pharmacologies, the reason as to why greater genetic specificity is not observed among illicit substances in twin designs may be attributable to several factors. Most notably, the high degree of comorbidity across addictions may amplify genetic overlap, and second, the power required to detect substance-specific genetic influences may be limited.
Decades of genetic epidemiological research have documented the importance of heritable influences on addiction. Multiple genetic variants of modest effect size contribute to this genetic architecture. The next section identifies genes that have been widely studied in the context of addictions.
Genes for addiction—candidate gene searches
Despite numerous studies examining putative candidate genes for addiction-related phenotypes, the field has been characterized by lack of replication and there are remarkably few genes that we can say with confidence are associated with addiction. Those genes with the strongest evidence for association are summarized in Table 1. In addition to these, there are a number of biologically plausible candidates for addiction for which there is some evidence for association (albeit without consistent replication).
Alcohol
In addition to widely studied variants in ADH1B and ALDH2, two functional polymorphisms (rs1693482 and rs698) in ADH1C are known to regulate alcohol metabolism and have been found to have a protective influence on alcohol consumption.33 Unlike rs1229984 in ADH1B and rs671 in ALDH2, which are uncommon and absent respectively, in non-Asian populations, the ADH1C polymorphisms are common. In addition to metabolism genes, serotonergic variants have also been implicated in the etiology of alcoholism.34 However, a meta-analysis of the commonly studied serotonin transporter gene (SLC6A4) polymorphism found only weak association (odds ratio (OR)=1.2, P<0.05) with alcoholism.35, 36, 37, 38, 39, 40, 41
Nicotine
Variants in the chromosome 15 cluster of genes encoding subunits of the nicotinic acetylcholine receptor, including CHRNA5/CHRNA3/CHRNB4, are among the most robustly replicated association signals for nicotine addiction. In addition, variants in CHRNA4 (encoding the α4 subunit of the neuronal nicotinic acetylcholine receptor) have also been suggested to influence various aspects of nicotine addiction,42, 43, 44 albeit inconsistently. A recent meta-analysis of linkage studies identified the CHRNA4 region on 20q13.2–q13.3 for maximum cigarettes smoked in a 24-h period. The DRD2/ANKK1 Taq1A allele, the subject of many studies on alcohol, has been studied in the context of nicotine addiction—a meta-analysis found support for an association for smoking initiation and current smoking but not for cigarettes/day.45
Cannabis
Inconsistent associations have been reported between variants in the cannabinoid receptor 1 gene (CNR1), to which cannabinoids putatively bind, and the fatty acid amide hydrolase gene (FAAH) and cannabis dependence symptoms (see Agrawal and Lynskey46 for a review). As with alcoholism, GABRA2 (encoding the α2 subunit of the GABA (gamma-amino-butyric acid) receptor) has been examined for cannabis dependence but with limited success.39, 47, 48
Cocaine
Several genes have been implicated in various aspects of cocaine addiction. These include dopaminergic single nucleotide polymorphisms (SNPs) in DRD2/ANKK149 as well as neighboring NCAM1 and TTC12, CALCYON,50 dopamine beta-hydroxylase (DBH)51 and catechol-O-methyltransferase (COMT);52 opioidergic genes such as POMC;53 CNR1;54 orthologs of genes regulating circadian rhythms (CLOCK, PER1, and PER2);55 tryptophan hydroxylase 2 (TPH2)56 and others gleaned from linkage studies (for example, alpha-endomannosidase (MANEA))57—a majority of these await replication. Of particular interest, the functional SNP in the CHRNA5/A3/B4 cluster on chromosome 15, rs16969968, (extensively discussed in later sections and in Table 1 with reference to nicotine dependence) has been found to be associated with cocaine dependence in two independent studies—paradoxically, the allelic variant of this marker that confers risk for nicotine dependence appears to afford protection from cocaine addiction.58, 59
Opioids
The gene encoding the mu-opioid receptor (OPRM1) to which opioids bind to produce their analgesic and rewarding effects is the most widely studied candidate gene for heroin and other opioid addictions.60 Functional OPRM1 polymorphisms identified in humans include the extensively-studied rs1799971 (A118G),61, 62, 63, 64 but a meta-analysis65 did not support its significant role in opioid addictions. Other aspects of the opioidergic system have also been queried. However, analyses involving prodynorphin (PDYN),66 proenkephalin (PENK),67 and the kappa (OPRK1)68, 69 and delta opioid receptors (OPRD1)69, 70, 71 have not produced consistent results.
Disordered gambling
A relatively novel subject of genetic enquiry, there have been only 10 candidate gene studies of disordered gambling.72 Two correlated variants in the gene encoding the dopamine receptor 1 (DRD1) have been reported to be associated with disordered gambling—rs4532 (-48G>A or -48DdeI)73 and rs265981 (5262T>C, -800HaeIII).74 However, a recent study by Lobo et al.75 did not replicate this association.
Putative mechanisms of action
The candidate genes described above can be broadly categorized into those with substance-specific influences (for example, ALDH2 and ADH1B for alcohol) and those that likely influence addiction liability via their relationship with a general predisposition to externalizing behaviors, including disinhibition, impulsivity and addiction (for example, DRD2/ANKK1).
Drug-specific effects
Genes associated with metabolism of psychoactive substances are anticipated to exert drug-specific effects. For instance, the association between variants in ADH1B (rs1229984) and ALDH2 (rs671) and alcohol consumption can be directly attributed to the effect of these SNPs on alcohol metabolism. rs1229984 affects the rate of conversion of alcohol to acetaldehyde, whereas rs671 substantially reduces conversion of this aversive acetaldehyde to acetate.33, 76 The accumulation of acetaldehyde is well recognized in the development of the flushing syndrome. The alcohol flush reaction is common in Asians, and involves facial reddening, accompanied by nausea, dizziness and headaches; and these experiences result in reduced alcohol intake. What is quite remarkable is that with increasing social pressures on Japanese and Koreans to consume alcohol, even individuals with protective ADH1B and ALDH2 alleles continue to drink.77, 78
General effects
Based on extensive twin studies, there is clear evidence that a large proportion of genetic factors influencing multiple drugs of addiction are shared. For example, two independent twin studies reported that significant proportions of genetic influence on addiction to a variety of illicit substances could be attributed to a common genetic vulnerability.19, 31 Liability to alcohol and nicotine (although, less so) also overlaps with this shared genetic influence.32 These shared genetic pathways likely reflect multiple processes. Koob and Volkow79 attribute addiction to cycles of impulsivity and compulsivity. Initial experimentation with drugs or escalation to chronic use at a young age has been found to accompany other behavioral indices of impulsivity (for example, conduct problems), and thus genes influencing impulsivity will likely have a general effect on multiple addictions. Multiple genes, including those in the dopamine reward circuits, have been widely studied in this regard.80 These reward circuits participate independently, and interactively with glutamatergic and GABA-ergic signaling, in the persistence of drug use and the development and maintenance of addiction. However, although there is clear evidence of central nervous system changes in these circuits related to impulsivity, disinhibition and drug use, the molecular mechanism of the action of gene variants in these pathways on specific drugs of addiction requires further consideration.
Candidate gene studies capitalize on a priori knowledge regarding the biological underpinnings of addiction. The next section summarizes the contributions of the more agnostic genomewide association study (GWAS) approach to the genetic study of addiction.
Genes for addiction—exploring the human genome
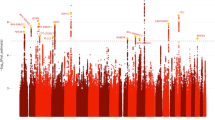
The challenge of non-replication in candidate gene studies, the desire for identification of novel variants for addiction and the decreasing costs and increasing efficiency of large-scale, high-density genotyping has led to the increasing use of GWAS to study addiction. The primary challenge in these studies is the profound burden of multiple testing, which requires gene variants to exceed a threshold P-value of 5 × 10−8 for statistical significance. Initial GWAS of smoking (for example, N=2000),81 alcohol dependence (N=1884–3865)82, 83, 84, 85 (and heavy consumption, for example, N=3400–400086) and cannabis dependence (for example, N=3054)87 failed to find any statistically significant associations. Immediately recognizing the need for considerably larger samples, multiple research groups combined GWAS data to produce large meta-analyses. Studying cigarettes smoked per day (an indicator of liability to nicotine dependence), the Tobacco and Genetics Consortium (N=74,053),88 Thorgeirsson et al. (N=10,995)89 and Liu et al. (N=41,150)90 simultaneously identified rs1051730, which is highly correlated with a functional missense polymorphism (rs16969968) in the nicotinic acetylcholine receptor gene cluster (CHRNA5/A3/B4) on 15q25. It is worth noting that although the first study to identify the role of rs16969968 on smoking behaviors was a study of nicotine dependence,91 these large meta-analyses have focused on cigarettes smoked per day as it is frequently assessed in a variety of genetic studies. Whether this SNP exerts as strong an effect on nicotine addiction as it does on smoking quantity/frequency is being investigated. Nonetheless, even the effect of this functional variant is modest and multiple other, as yet unidentified variants, are likely to be associated with smoking behavior and nicotine dependence.
Following this trend, a recent meta-analysis of alcohol consumption (N=47,500) identified a variant, rs6943555, in autism susceptibility candidate 2 (AUTS2) gene.92 Follow-up analyses found expression changes in AUTS2 in mice bred for alcohol preference.92 For alcohol dependence, although similar meta-analytic efforts are ongoing, Frank et al.83 recently identified rs1789891, which is a proxy for the functional Arg272Gln variant in ADH1C, to be associated with alcohol dependence at P=1.27 × 10−8. Given the clear biological significance of this variant, this finding is particularly encouraging.
Despite growing sample sizes and our ability to capture over 2.5 million SNPs across the genome, SNPs reaching formal significance levels in association studies typically explain <2% of the variance in addiction-related phenotypes. For instance, the effect associated with rs1051730 and liability for smoking was a β of 1.03. Thus, it was first thought, as has been suggested for other complex multifactorial traits, that most of the heritability of addiction is ‘missing’.93, 94 However, Visscher and colleagues95 point out that the emphasis on SNPs reaching only formal significance at 5 × 10−8 overlooks the evidence for polygenic association in the remainder of the GWAS data. For instance, studies of smoking and substance-related phenotypes have identified cell-adhesion genes as contributors although no signal has attained genomewide significance (for example, Bierut et al.81 and Johnson et al.96). In addition, recent studies have found that sets of SNPs of nominal significance might be used to identify genomic regions that are over-represented across independent GWAS97, 98—here as well, cell-adhesion genes, such as cadherins, are prominent, although only nominally significant in the individual studies. Although it is not possible to distinguish true signals from false positives with current sample sizes, we can be certain that even among SNPs that are less significantly associated, there are many SNPs that do influence the traits under study. Using a regression approach that simultaneously examines the contribution of all GWAS variants, Visscher and colleagues95 have shown that 43, 17 and 54% of total phenotypic variance in height, body mass index99 and intelligence quotient100 respectively, is due to causal variants in linkage disequilibrium with SNPs on commercial GWAS arrays. For height, they further showed that adjusting for imperfect linkage disequilibrium between typed SNPs and causal SNPs raised the estimate to 54%, and further adjustment for the gross under-representation of rare SNPs on commercial chips, raised the estimate to 70–80%, close to estimates of heritability of height from conventional twin and family studies. Such efforts are currently under way for studies of addiction.
A second, and perhaps more perplexing outcome of GWAS is the relative lack of findings that are biologically plausible. This has led investigators to question the content of current GWAS arrays which, despite their high density, can provide inadequate coverage of certain genomic regions, owing to fewer SNPs and absence of adequate linkage disequilibrium required to impute such variants. The ADH1B polymorphism, rs1229984, serves as an example: this functional variant is neither included on commercial GWAS arrays nor reliably imputed owing to its low allele frequency in non-Asian populations. However, by genotyping it in multiple large samples, Bierut et al.101 recently revealed a genomewide significant association finding for rs1229984 with alcohol dependence.
Both candidate gene studies and GWAS continue to provide important clues regarding the sources of genetic variation in addictions. However, unless a variant is functional, considerably more research is required to understand how a signal from one of these gene-finding methods actually affects the liability to addiction at a biological level. Even for missense mutations, further work is often required to quantify expression changes. Thus, genetic epidemiological studies, such as gene association, are only the beginning. Using select examples, the next sections illustrate the utility of novel approaches that extend the search for the genetic basis of addictions.
Phenotyping
The first major area of growth for research into addictions has been the systematic transition from diagnostic classification of affected versus unaffected to a dimensional conceptualization, such as measures of quantity/frequency (for example, cigarettes smoked per day), symptom counts, factor scores extracted from multiple indices of problem use and other latent variables representing continuously distributed quantitative measures of addiction vulnerability. Such continuous measures are gaining popularity in genetic studies102, 103, 104 as they are heritable and this heritability overlaps considerably with genetic influences on addiction.103, 105, 106 An additional advantage of such continuous phenotypes is that they are not limited by heterogeneity in those who are unaffected (for example, assigning the same unaffected value to those who have never had a drink of alcohol, those who are light drinkers and those who endorse 1–2 criteria for alcohol dependence but do not receive a diagnosis) or affected but at varying levels of severity, which can significantly reduce power for genetic association studies. This research is also encouraged by DSM-5, which proposes to define addiction as a multi-level disorder (absent, mild/moderate and severe).
Variations in assessments
Although addiction is clinically diagnosed using the DSM, genetic research has relied on additional assessment strategies that can yield higher heritability and reduce measurement heterogeneity. For instance, nicotine dependence can be diagnosed using the DSM but, for research purposes, quantitative indices (which can also be used to define affecteds and unaffecteds) such as the Heaviness of Smoking Index107 and the Fagerstrom Test for Nicotine Dependence (FTND)108 are frequently used. Interestingly, the overlap between FTND and DSM-diagnosed individuals is modest (kappa≈0.3)109 and one study found FTND-based nicotine dependence to be more heritable than DSM-based nicotine dependence.8
Challenge studies
Typically, for genetic research, addiction is assessed via self-report interviews and questionnaires and via clinical interview. However, laboratory-based measures can also be used to provide detailed assessments of individual differences in addiction using challenge paradigms in which acute doses of a drug are administered to participants in a controlled setting. Using this paradigm, Schuckit and Gold110 have developed the level of response phenotype for oral alcohol challenges—outcomes including positive (for example, high) and negative (for example, nausea) subjective feelings, body sway and various physiological and biomarker changes were used to identify low level of response individuals who are at increased risk for alcoholism111, 112 (although, in some studies, high level of response associates with alcoholism113, 114). These subjective responses to alcohol have been found to be associated with variation in GABRA2115, 116 (Table 1) and in SLC6A4.117
Endophenotypes
Level of response is considered by some to be an endophenotype. Defined by Gottesman and Gould118 as measurable indices of liability to a phenotype, these measures have gained popularity in genomic studies as they are heritable and assumed to be more proximal to the biological underpinnings of the behavior being studied.119 Although they may co-segregate with disease, they are more closely related to the causes than the consequence of disorder.120 There have been multiple putative endophenotypes proposed for addiction, including alcohol and drug-related attentional bias, frequently assessed using a modified version of the traditional Stroop task,121 and electro-encephalogram activity (for example, beta wave patterns, P300 amplitude).122, 123, 124 A number of these endophenotypes have been used in candidate gene efforts—for instance, SNPs in GABRA2 (Table 1) have been found to associate with resting electro-encephalogram beta waves.36, 125
Biomarkers
Similarly, biomarkers are intermediate phenotypes that are related to disorder and can be a consequence of it. For instance, liver function tests (for example, gammaglutamyl transferase) or carbohydrate-deficient transferrin are commonly used to examine the impact of prolonged alcohol use. Unlike endophenotypes, which can be used to putatively predict likelihood of disorder, these biomarkers are diagnostic aids that facilitate clinical management of addictions. They are also promising targets for gene association studies. Using various carbohydrate-deficient transferrin indices, and adjusting for alcohol intake, a recent GWAS isolated the highly significant independent effects of variants in the transferrin (TF, P=5.5 × 10−43), and phosphoglucomutase 1 (PGM1, P=2 × 10−9) genes on carbohydrate-deficient transferrin.126
Neuroimaging phenotypes
Neuroimaging outcomes are promising new endophenotypes for addiction. Although current sample sizes are modest, primarily due to costs and burden of imaging technology, promising results have begun to emerge. For example, although not directly addressing functionality, Villafuerte et al.127 recently found that in families of alcoholics, rs279826 and rs279858 in the GABRA2 gene, previously associated with alcohol dependence (Table 1), were associated, not only with alcoholism and self-reported impulsivity but also with insula cortex activation in women during anticipation of monetary reward. The insula cortex has been implicated in cue-induced drug craving and addiction and thus, this study provides a potential neurobiological perspective on the link between GABRA2 and alcoholism. Capitalizing on polygenic variation in addiction, another recent study used a sum score created from frequently studied variants in dopamine pathways genes (DAT1 9-repeat, DRD4 7-repeat, DRD2 -141C Del, DRD2 Taq1A C (A2), and COMT Val(158)Met). Although no variant was statistically significant on its own, the sum score was associated with monetary reward-related activation in the ventral striatum, explaining 10.9% of variance.128
Summary
How we measure addiction is likely to have a strong effect on the genes and genetic pathways that we identify. A multi-pronged approach to measurement, including multiple self-report assessments, laboratory-based measures and the collection of data on neurophysiological and neuroimaging endophenotypes and examining their correlations with each other, provide avenues for linking genes to behavior. To this end, resources such as the PhenX toolkit (Research Triangle Park, NC, USA),129 which provides unrestricted access to state-of-the-art assessments for research, are invaluable. Not only does the toolkit provide protocols for such multi-pronged measurement but the systematic use of identical protocols by multiple investigators will ultimately result in sample sizes large enough to detect even modest genetic effects.
Advances in genomics
Until recently, candidate gene studies and GWAS focused on common variation. However, three additional sources of genomic variation have the potential to further explain heritability in addiction. First, copy number variants—large segments of DNA that are deleted or duplicated producing considerable structural instability—need to be explored for addictions. Recent research has shown associations between rare copy number variants (mostly deletions) and several psychiatric disorders, including schizophrenia, autism and Parkinson disease (see Stankiewicz and Lupski130 for a review). Second, as discussed above, rare variants are inadequately captured on commercial GWAS arrays. Deeper sequencing of the human genome presents the opportunity to identify such rare SNPs (<1% minor allele frequency). Although this is being facilitated by the 1000 Genomes Project,131 the identification of disease-specific rare variants requires next-generation sequencing in samples ascertained for addiction. Finally, epigenetic modifications are implicated as contributors to and consequence of chronic substance use. Animal research shows that repeated drug use alters gene expression profiles in the brain reward system, through epigenetic mechanisms such as histone acetylation and methylation change (see Renthal and Nestler,132 Maze and Nestler133 and Wong et al.134 for reviews) and there is no doubt that epigenetic variation is an integral component of the biology of addiction. Although epigenomic methodologies (for example, whole genome methylation) grow increasingly accessible, the primary challenge remains tissue-specificity: epigenetic signatures in peripheral tissues (for example, leukocytes) may not correlate with those in the central nervous system. This has led to increasing investment in the NIH Roadmap Epigenomics Project,135 which aims to develop databases of human epigenomic maps (epigenome atlas) in a variety of healthy and tumor tissue and eventually, to provide a degree of cross-tissue correspondence in epigenomic profiles. Even in the absence of direct epigenomic typing, resources such as the Encyclopedia of DNA Elements136 allow flexible annotation of the functional landscape of the human genome, such as regions of potential epigenomic modification.
Summary
Despite some current disenchantment with studies of common variants, there is much work that is needed to understand the precise mechanisms by which candidate genes and their common SNPs influence addiction. Animal studies of gene manipulation afford opportunities to investigate this—for example, both Xenopus oocytes137 and rodents138 have been used to investigate the role of GABRA2 in alcohol intake (Table 1). An alternative method involves the use of annotation algorithms to identify the potential correspondence between variants and gene activity and to further enmesh these variants in biological pathways. These approaches are briefly reviewed next.
Advanced biostatistics and bioinformatics
All of the above phenotypic and genotypic approaches rely heavily on biostatistical advances and innovation in statistical methods. Biostatistics and computational biology have rapidly become the foundation of post-GWAS interpretation of results. Relying primarily on existing curated databases, these methods attempt to model the inherent and often non-linear complexity in biological processes. For instance, gene-based association studies (for example, PLINK set–based test,139 VEGAS,140 GRAIL141 and GATES142) combine information from several SNPs within each gene, identifying genes that show more signals of association than expected by chance. Pathway analysis also examines the combined effects of multiple genetic variants (that could be of small effect). By means of exploratory pathway analysis, it is possible to test whether associated genetic variants are more prevalent in any known biological pathway (see for example, IPA (Ingenuity Pathway Analysis; Ingenuity Systems, www.ingenuity.com)), or any known functional gene group.143 In a recent study, Reimers et al.144 performed a pathway analysis using SNPs within 48 addiction candidate genes in alcohol-dependent cases and controls. They tested seven gene sets (pathways), including various neurotransmitter systems. In line with previous findings, their results showed that four of the neurotransmitter pathways (corticotropin-releasing hormone, GABA, glutamate and norepinephrine) significantly contributed to alcohol-dependence risk. A number of these methods rely on the Gene Ontology project—a large bioinformatics project that combines the representation of genes and gene products across species as well as across different databases.145
Summary
There is a considerable need for computational approaches to generate and interpret results from genetic studies for addiction. Although several groups have attempted to model the complex pathways underlying addictions, there has been little consistency across studies, which is likely due to the inherent lack of replication for the individual genetic findings. One possible reason for this lack of consistency across studies, at individual SNP and pathway levels, might be the moderating role of environment.
Gene–environment interplay
Moderation of genetics by environment
Genetic studies typically assume homogeneity of effect size. However, for addiction, the relevance of genetic influences may depend on environmental contexts. This has resulted in recognition of the importance of gene–environment interplay (including both gene–environment (GE) correlation and gene–environment (G × E) interaction) in the etiology of addictions. GE correlation146 refers to genetic predispositions that influence the likelihood of being exposed to a certain environment (for example, heritable influences have been found to influence affiliations with delinquent or substance-using peers). Gene–environment interaction (G × E)147 refers to moderation of genetic predisposition as a consequence of environmental exposure—for example, studies of adolescent Finnish twins indicated that in less stable neighborhoods, there was greater evidence of genetic influence on alcohol use.148, 149 Conversely, in more supervised and restricted environments, there was less opportunity to express genetic predispositions to alcohol use and greater influence of environmental effects. Additionally, both low levels of parental monitoring150 and increasing affiliations with substance-using peers151, 152 have been found to augment the importance of genetic influences on drug use.
Studies examining potential interactions between measured genes and environment153, 154 are also becoming more common. For example, Dick et al.155, 156 reported that the association of GABRA2 and CHRM2 variants with externalizing trajectories diminished with high levels of parental monitoring. In addition to environment exacerbating genetic vulnerability, there is also evidence for the stress-buffering effects of genotype. Nelson et al.157 found that the effect of childhood sexual abuse on alcoholism was buffered in those carrying the H2 haplotype of the gene encoding the corticotrophin-releasing factor (CRHR1).
The role of environment after accounting for genetics
Although the previous section presents the notion of environmental moderation of genetic vulnerability, whether environmental and other risk factors (for example, comorbid psychiatric problems) continue to exert an influence on addiction after overlapping genetic risk factors are partialled out is of utmost interest to the prevention and treatment community. Twins, particularly MZ pairs discordant for environmental exposure, provide a fascinating demonstration of the constant interplay between genetic background and environmental exposures. For instance, examining pairs of twins discordant for exposure to childhood sexual abuse, Nelson et al.158 reported that the twin who had experienced abuse was considerably more likely to also report a lifetime history of addiction, even when compared with their genetically identical but unexposed co-twin. Interestingly, these genetically identical twin pairs can even have differing epigenetic profiles, with within-pair differences becoming more pronounced with increasing age.159 The informativeness of discordant MZ pairs applies to every research methodology described above and is only just being harnessed in genetic studies of addiction.
Summary
Recently, there have been several published critiques of the genotype–environment interplay methdology160, 161 and it is likely that some of the limitations noted by these investigators apply to studies of addiction. However, addiction is the most obvious example of a process that is subject to gene–environment interplay—even individuals with a family history of addiction may circumvent their genetic vulnerability by limiting drug exposure. There is now even emerging evidence that the efficacy of behavioral treatments may interact with genotype to predict outcomes, such as disruptive childhood behaviors,162 and the next section briefly reviews genotypic modification of drug therapies. Thus, although it is important to recognize the importance of the statistical caveats to modeling gene–environment interplay, it is necessary to continue conducting such studies on addiction.
Pharmacogenomics
The extent to which drug therapy, such as acamprosate and naltrexone for alcohol dependence and nicotine replacement therapy and other medications (for example, bupropion and varenicline) for nicotine dependence, may be more successful in individuals with certain genetic profiles is of considerable interest.163 These drugs target receptors encoded by genes of interest—for instance, baclofen (for alcohol) acts as an agonist at pre-synaptic GABA-B receptors, while varencline (for nicotine) is a partial agonist of the α4β2 nicotinic receptors. Although these drugs have shown promise in increasing rates of abstinence, typically only a minority of treated individuals discontinue drug use. For example, 12 months post-treatment, typically only 1 in 10 smokers treated with pharmacotherapies remain abstinent.164 Although such findings suggest that pharmacotherapy may be a promising avenue for treatment development, given the typically low rate of success for existing approaches to treating addictions, individual differences in the apparent effectiveness of these drug treatments has led to increasing interest in pharmacogenetics, the study of genetic variation underlying individual differences in both drug metabolism and response to the effects of drugs.165 Despite some non-replications, three independent studies have reported that carriers of the A118G polymorphism (rs1799971) in OPRM1 respond more positively to naltrexone treatment for alcohol dependence.166, 167, 168 Another promising treatment for alcoholism, ondansetron, a 5-HT3 (5-hydroxytryptamine (serotonergic) receptor 3) antagonist has been found to be a particularly useful treatment in early-onset (Type II) alcoholics, while the selective serotonin reuptake inhibitor (SSRI) sertraline has been found to be more efficacious in later-onset (Type I) alcoholics.169, 170
Summary
Whether treatment effects vary by genotype is an important area of further study. Growth in this area relies heavily on discoveries of common and rare variants and a continued effort to outline their biological function.
Genetics of addiction and related illness
Thus far, we have discussed genetic influences underlying addictions. However, some of these genes are responsible for the links between addictive behaviors and other forms of illness, particular cancer. For instance, rs1229984 in ADH1B and rs671 in ALDH2 have been implicated in the etiology of esophageal cancer.171 By modifying acetaldehyde accumulation and clearance, the enzymatic consequences of these variants result in increased exposure to ethanol and acetaldehyde, an effect that is exacerbated in individuals who drink alcohol despite carrying these protective variants.172, 173 Likewise, rs16969968 (and other variants) in the chromosome 15 gene cluster that is now widely recognized as a risk factor for nicotine addiction has also been found to confer risk for lung cancer,174, 175, 176 peripheral artery disease175 and chronic obstructive pulmonary disease.177 Whether this missense mutation has an independent effect on these diseases or whether its effect is mediated by its modulation of exposure to smoking continues to be explored.178 Finally, there appears to be an emergence of disordered gambling in patients receiving dopamine replacement therapy for Parkinson's disease—there is some evidence that dopaminergic stimulation interacts with variants in dopamine genes (for example, DAT, DRD3 and DRD4) to induce reward-seeking behaviors, particularly pathological gambling, but these genetic connections are speculative.179
Summary
Addictions, serious illnesses themselves, are linked with other diseases that may be consequences, and in rare instances, a potential cause, of the addictive behavior. Interplay between the addictive behavior and genetic predisposition (for example, increased risk of esophageal cancer in those who carry one copy of rs671 in ALDH2 and also continue to drink alcohol) is likely responsible for a majority of these relationships.
Can animal models inform human genetic studies of addiction?
Throughout this review, where possible, experimental manipulation in animals is used to highlight progress made in understanding the functional significance of genetic systems. There is also a long and distinguished tradition of animal models for addiction. A detailed discussion of these methods is beyond the scope of this review. Both rats and mice, for instance, can be selectively bred for alcohol preference (for example, alcohol-preferring,180 alko-alcohol181) and these animals can be trained to approximate aspects of addiction, such as binge drinking (for example, using the drinking-in-the-dark paradigm).182 Mutagenesis has been used to produce fruit flies (Drosophila melanogaster) that vary on their alcohol consumption and response to alcohol (for example, cheapdate, tipsy).183 For addiction research, the issue of consilience, or the relative similarities and differences between human behavior and animal phenotypes devised to study these behaviors, continues to be a challenge. Highlighting the need to reconcile these differences, Crabbe,184 for instance, notes that while rodents, even those with high alcohol preference, self-limit their alcohol ingestion and rarely induce intoxication, loss of control over alcohol intake is the cornerstone of alcoholism producing conceptual discrepancies across rodent and human behaviors.
Genetic studies of nicotine addiction
Genetic studies of addiction are at a watershed—we have clearly identified some genetic contributors to addiction and we continue to explore the role of others using multiple methods and considering the pivotal role of environmental variation. The research described so far is made possible by growing collaboration and intellectual sharing across investigators from varying disciplines—such collaboration is critical now as technological advances allow us to study addiction from multiple integrated perspectives. To highlight the enormous potential of using multiple translational methods, Table 2 summarizes the application of a number of approaches outlined above to the study of nicotine addiction.
Addiction, genetics and public health
Why study the genetics of addiction? Critics argue that it has modest benefit from a public health standpoint (e.g., ref. 185). They posit that (a) genetic variants, when and if they are discovered, have small effect sizes; (b) comparable environmental factors are easier to delineate and have stronger influence; and arguably (c) are more amenable to modification. These criticisms, by taking a short-sighted view, often obfuscate the true goal of genetic research—to provide improved therapeutic alternatives for individuals who, despite rigorous environmental modification (for example, increased taxation, reduced availability and even treatment) remain addicted to drugs. It is worth reiterating that the goal of genetic research into addiction is no different from that for Type 2 diabetes or cardiovascular disease and that addictions are also among the top contributors to preventable death worldwide. Perhaps this argues for an even more concentrated effort to understand the etiology of addiction.
In his commentary on genomic studies of complex traits, Hirschhorn186 notes that ‘The difficulty in translation is not unique to genetic discoveries: nearly a century and three Nobel Prizes separate the determination of the chemical composition of cholesterol from the development of statins. Each discovery of a biologically relevant locus is a potential first step in a translational journey, and some journeys will be shorter than others.’ Genetic factors are partly responsible for, not only, the comorbidity across addictions but also between addictions and other mental illness (for example, autism and bipolar disorder). The growth of these existing findings from genetic epidemiological studies into promising leads for treatment is necessary and with time, sustained funding and translational collaborations, this goal of a clear impact of genetic research on public health can and will be achieved.
References
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th edn, Revised edn. American Psychiatric Association: Washington, DC, 1994.
Centers for Disease Control (CDC). Annual smoking-attributable mortality, years of potential life lost, and economic costs-United States. MMWR 2002; 51: 300–303.
Centers for Disease Control (CDC). Alcohol-Attributable Deaths and Years of Potential Life Lost—United States, 2001. MMWR 2004; 53: 870.
O’Brien C . Addiction and dependence in DSM-V. Addiction 2011; 106: 866–867.
Petry NM . Gambling and substance use disorders: current status and future directions. Am J Addict 2007; 16: 1–9.
Frascella J, Potenza MN, Brown LL, Childress AR . Shared brain vulnerabilities open the way for nonsubstance addictions: carving addiction at a new joint? Ann N Y Acad Sci 2010; 1187: 294–315.
Kendler KS, Neale MC, Sullivan P, Corey LA, Gardner CO, Prescott CA . A population-based twin study in women of smoking initiation and nicotine dependence. Psychol Med 1999; 29: 299–308.
Lessov CN, Martin NG, Statham DJ, Todorov AA, Slutske WS, Bucholz KK et al. Defining nicotine dependence for genetic research: evidence from Australian twins. Psychol Med 2004; 34: 865–879.
Edwards AC, Maes HH, Pedersen NL, Kendler KS . A population-based twin study of the genetic and environmental relationship of major depression, regular tobacco use and nicotine dependence. Psychol Med 2011; 41: 395–405.
Lyons M, Hitsman B, Xian H, Panizzon MS, Jerskey BA, Santangelo S et al. A twin study of smoking, nicotine dependence, and major depression in men. Nicotine Tob Res 2008; 10: 97–108.
Prescott CA, Kendler KS . Genetic and Environmental Influences on Alcohol and Tobacco Dependence among Women. NIAAA Res Monogr 1995; 30: 59–87.
Kendler KS, Heath AC, Neale MC, Kessler RC, Eaves LJ . A population-based twin study of alcoholism in women. JAMA 1992; 268: 1877–1882.
Heath AC, Martin NG . Genetic influences on alcohol consumption patterns and problem drinking: results from the Australian NH&MRC twin panel follow-up survey. Ann N Y Acad Sci 1994; 708: 72–85.
Heath AC, Whitfield JB, Madden PA, Bucholz KK, Dinwiddie SH, Slutske WS et al. Towards a molecular epidemiology of alcohol dependence: analysing the interplay of genetic and environmental risk factors. Br J Psychiatry Suppl 2001; 40: s33–s40.
Verweij KJ, Zietsch BP, Lynskey MT, Medland SE, Neale MC, Martin NG et al. Genetic and environmental influences on cannabis use initiation and problematic use: a meta-analysis of twin studies. Addiction 2010; 105: 417–430.
Kendler KS, Karkowski LM, Neale MC, Prescott CA . Illicit psychoactive substance use, heavy use, abuse, and dependence in a US population-based sample of male twins. Arch Gen Psychiatry 2000; 57: 261–269.
Tsuang MT, Bar JL, Harley RM, Lyons MJ . The Harvard Twin Study of Substance Abuse: what we have learned. Harv Rev Psychiatry 2001; 9: 267–279.
van den Bree MB, Johnson EO, Neale MC, Pickens RW . Genetic and environmental influences on drug use and abuse/dependence in male and female twins. Drug Alcohol Depend 1998; 52: 231–241.
Tsuang MT, Lyons MJ, Meyer JM, Doyle T, Eisen SA, Goldberg J et al. Co-occurrence of abuse of different drugs in men: the role of drug- specific and shared vulnerabilities. Arch Gen Psychiatry 1998; 55: 967–972.
Slutske WS, Zhu G, Meier MH, Martin NG . Genetic and environmental influences on disordered gambling in men and women. Arch Gen Psychiatry 2010; 67: 624–630.
Slutske WS, Eisen S, True WR, Lyons MJ, Goldberg J, Tsuang M . Common genetic vulnerability for pathological gambling and alcohol dependence in men. Arch Gen Psychiatry 2000; 57: 666–673.
Heath AC, Martin NG, Lynskey MT, Todorov AA, Madden PA . Estimating two-stage models for genetic influences on alcohol, tobacco or drug use initiation and dependence vulnerability in twin and family data. Twin Res 2002; 5: 113–124.
Neale MC, Harvey E, Maes HH, Sullivan PF, Kendler KS . Extensions to the modeling of initiation and progression: applications to substance use and abuse. Behav Genet 2006; 36: 507–524.
Agrawal A, Neale M, Jacobson K, Prescott CA, Kendler KS . Illicit drug use and abuse/dependence: modeling of two-stage variables using the CCC approach. Addict Behav 2005; 30: 1043–1048.
Heath AC, Lynskey MT, Waldron M . Substance use. In: Rutter M, Taylor E (eds). Handbook of Child and Adolescent Psychiatry. Blackwell: London, 2007.
Dick DM, Pagan JL, Viken R, Purcell S, Kaprio J, Pulkkinen L et al. Changing environmental influences on substance use across development. Twin Res Hum Genet 2007; 10: 315–326.
Kendler KS, Schmitt E, Aggen SH, Prescott CA . Genetic and environmental influences on alcohol, caffeine, cannabis, and nicotine use from early adolescence to middle adulthood. Arch Gen Psychiatry 2008; 65: 674–682.
Derringer J, Krueger RF, McGue M, Iacono WG . Genetic and environmental contributions to the diversity of substances used in adolescent twins: a longitudinal study of age and sex effects. Addiction 2008; 103: 1744–1751.
Derringer J, Krueger RF, Iacono WG, McGue M . Modeling the impact of age and sex on a dimension of poly-substance use in adolescence: a longitudinal study from 11- to 17-years-old. Drug Alcohol Depend 2010; 110: 193–199.
Rhee SH, Hewitt JK, Young SE, Corley RP, Crowley TJ, Stallings MC . Genetic and environmental influences on substance initiation, use, and problem use in adolescents. Arch Gen Psychiatry 2003; 60: 1256–1264.
Kendler KS, Jacobson KC, Prescott CA, Neale MC . Specificity of genetic and environmental risk factors for use and abuse/dependence of cannabis, cocaine, hallucinogens, sedatives, stimulants, and opiates in male twins. Am J Psychiatry 2003; 160: 687–695.
Kendler KS, Myers J, Prescott CA . Specificity of genetic and environmental risk factors for symptoms of cannabis, cocaine, alcohol, caffeine, and nicotine dependence. Arch Gen Psychiatry 2007; 64: 1313–1320.
Edenberg HJ . The genetics of alcohol metabolism: role of alcohol dehydrogenase and aldehyde dehydrogenase variants. Alcohol Res Health 2007; 30: 5–13.
Dick DM, Bierut LJ . The genetics of alcohol dependence. Curr Psychiatry Rep 2006; 8: 151–157.
McHugh RK, Hofmann SG, Asnaani A, Sawyer AT, Otto MW . The serotonin transporter gene and risk for alcohol dependence: a meta-analytic review. Drug Alcohol Depend 2010; 108: 1–6.
Edenberg HJ, Dick DM, Xuei X, Tian H, Almasy L, Bauer LO et al. Variations in GABRA2, encoding the alpha 2 subunit of the GABA(A) receptor, are associated with alcohol dependence and with brain oscillations. Am J Hum Genet 2004; 74: 705–714.
Lappalainen J, Krupitsky E, Remizov M, Pchelina S, Taraskina A, Zvartau E et al. Association between alcoholism and gamma-amino butyric acid alpha2 receptor subtype in a Russian population. Alcohol Clin Exp Res 2005; 29: 493–498.
Fehr C, Sander T, Tadic A, Lenzen KP, Anghelescu I, Klawe C et al. Confirmation of association of the GABRA2 gene with alcohol dependence by subtype-specific analysis. Psychiatr Genet 2006; 16: 9–17.
Lind P, MacGregor S, Agrawal A, Heath AC, Martin NG, Whitfield JB . The role of GABRA2 in alcohol dependence, smoking and illicit drug use in an australian population sample. Alcohol Clin Exp Res 2008; 32: 1721–1731.
Covault J, Gelernter J, Hesselbrock V, Nellissery M, Kranzler HR . Allelic and haplotypic association of GABRA2 with alcohol dependence. Am J Med Genet B Neuropsychiatr Genet 2004; 129: 104–109.
Drgon T, D’Addario C, Uhl GR . Linkage disequilibrium, haplotype and association studies of a chromosome 4 GABA receptor gene cluster: candidate gene variants for addictions. Am J Med Genet B Neuropsychiatr Genet 2006; 141: 854–860.
Han S, Yang BZ, Kranzler HR, Oslin D, Anton R, Gelernter J . Association of CHRNA4 polymorphisms with smoking behavior in two populations. Am J Med Genet B Neuropsychiatr Genet 2011; 156B: 421–429.
Keskitalo-Vuokko K, Pitkaniemi J, Broms U, Heliovaara M, Aromaa A, Perola M et al. Associations of nicotine intake measures with CHRN genes in finnish smokers. Nicotine Tob Res 2011; 13: 686–690.
Xie P, Kranzler HR, Krauthammer M, Cosgrove KP, Oslin D, Anton RF et al. Rare nonsynonymous variants in Alpha-4 nicotinic acetylcholine receptor gene protect against nicotine dependence. Biol Psychiatry 2011; 70: 528–536.
Munafo MR, Timpson NJ, David SP, Ebrahim S, Lawlor DA . Association of the DRD2 gene Taq1A polymorphism and smoking behavior: a meta-analysis and new data. Nicotine Tob Res 2009; 11: 64–76.
Agrawal A, Lynskey MT . Candidate genes for cannabis use disorders: findings, challenges and directions. Addiction 2009; 104: 518–532.
Agrawal A, Edenberg HJ, Foroud T, Bierut LJ, Dunne G, Hinrichs AL et al. Association of GABRA2 with drug dependence in the collaborative study of the genetics of alcoholism sample. Behav Genet 2006; 36: 640–650.
Corley RP, Zeiger JS, Crowley T, Ehringer MA, Hewitt JK, Hopfer CJ et al. Association of candidate genes with antisocial drug dependence in adolescents. Drug Alcohol Depend 2008; 96: 90–98.
Gelernter J, Yu Y, Weiss R, Brady K, Panhuysen C, Yang BZ et al. Haplotype spanning TTC12 and ANKK1, flanked by the DRD2 and NCAM1 loci, is strongly associated to nicotine dependence in two distinct American populations. Hum Mol Genet 2006; 15: 3498–3507.
Luo X, Kranzler H, Lappalainen J, Rosenheck R, Charney D, Zuo L et al. CALCYON gene variation, schizophrenia, and cocaine dependence. Am J Med Genet B Neuropsychiatr Genet 2004; 125B: 25–30.
Kalayasiri R, Sughondhabirom A, Gueorguieva R, Coric V, Lynch WJ, Lappalainen J et al. Dopamine beta-hydroxylase gene (DbetaH) -1021C → T influences self-reported paranoia during cocaine self-administration. Biol Psychiatry 2007; 61: 1310–1313.
Ittiwut R, Listman JB, Ittiwut C, Cubells JF, Weiss RD, Brady K et al. Association between polymorphisms in catechol-O-methyltransferase (COMT) and cocaine-induced paranoia in European-American and African-American populations. Am J Med Genet B Neuropsychiatr Genet 2011; 156: 651–660.
Zhang H, Kranzler HR, Weiss RD, Luo X, Brady KT, Anton RF et al. Pro-opiomelanocortin gene variation related to alcohol or drug dependence: evidence and replications across family- and population-based studies. Biol Psychiatry 2009; 66: 128–136.
Zuo L, Kranzler HR, Luo X, Yang BZ, Weiss R, Brady K et al. Interaction between two independent CNR1 variants increases risk for cocaine dependence in European Americans: a replication study in family-based sample and population-based sample. Neuropsychopharmacology 2009; 34: 1504–1513.
Malison RT, Kranzler HR, Yang BZ, Gelernter J . Human clock, PER1 and PER2 polymorphisms: lack of association with cocaine dependence susceptibility and cocaine-induced paranoia. Psychiatr Genet 2006; 16: 245–249.
Dahl JP, Cubells JF, Ray R, Weller AE, Lohoff FW, Ferraro TN et al. Analysis of variations in the tryptophan hydroxylase-2 (TPH2) gene in cocaine dependence. Addict Biol 2006; 11: 76–83.
Farrer LA, Kranzler HR, Yu Y, Weiss RD, Brady KT, Anton R et al. Association of variants in MANEA with cocaine-related behaviors. Arch Gen Psychiatry 2009; 66: 267–274.
Grucza RA, Wang JC, Stitzel JA, Hinrichs AL, Saccone SF, Saccone NL et al. A risk allele for nicotine dependence in CHRNA5 is a protective allele for cocaine dependence. Biol Psychiatry 2008; 64: 922–929.
Sherva R, Kranzler HR, Yu Y, Logue MW, Poling J, Arias AJ et al. Variation in nicotinic acetylcholine receptor genes is associated with multiple substance dependence phenotypes. Neuropsychopharmacology 2010; 35: 1921–1931.
Uhl GR, Sora I, Wang Z . The mu opiate receptor as a candidate gene for pain: polymorphisms, variations in expression, nociception, and opiate responses. Proc Natl Acad Sci USA 1999; 96: 7752–7755.
Bond C, LaForge KS, Tian M, Melia D, Zhang S, Borg L et al. Single-nucleotide polymorphism in the human mu opioid receptor gene alters beta-endorphin binding and activity: possible implications for opiate addiction. Proc Natl Acad Sci U S A 1998; 95: 9608–9613.
Kroslak T, LaForge KS, Gianotti RJ, Ho A, Nielsen DA, Kreek MJ . The single nucleotide polymorphism A118G alters functional properties of the human mu opioid receptor. J Neurochem 2007; 103: 77–87.
LaForge KS, Yuferov V, Kreek MJ . Opioid receptor and peptide gene polymorphisms: potential implications for addictions. Eur J Pharmacol 2000; 410: 249–268.
Pan L, Xu J, Yu R, Xu MM, Pan YX, Pasternak GW . Identification and characterization of six new alternatively spliced variants of the human mu opioid receptor gene, Oprm. Neuroscience 2005; 133: 209–220.
Glatt SJ, Bousman C, Wang RS, Murthy KK, Rana BK, Lasky-Su JA et al. Evaluation of OPRM1 variants in heroin dependence by family-based association testing and meta-analysis. Drug Alcohol Depend 2007; 90: 159–165.
Ray R, Doyle GA, Crowley JJ, Buono RJ, Oslin DW, Patkar AA et al. A functional prodynorphin promoter polymorphism and opioid dependence. Psychiatr Genet 2005; 15: 295–298.
Comings DE, Blake H, Dietz G, Gade-Andavolu R, Legro RS, Saucier G et al. The proenkephalin gene (PENK) and opioid dependence. Neuroreport 1999; 10: 1133–1135.
Gerra G, Leonardi C, Cortese E, D’Amore A, Lucchini A, Strepparola G et al. Human kappa opioid receptor gene (OPRK1) polymorphism is associated with opiate addiction. Am J Med Genet B Neuropsychiatr Genet 2007; 144B: 771–775.
Zhang H, Kranzler HR, Yang BZ, Luo X, Gelernter J . The OPRD1 and OPRK1 loci in alcohol or drug dependence: OPRD1 variation modulates substance dependence risk. Mol Psychiatry 2008; 13: 531–543.
Franke P, Nothen MM, Wang T, Neidt H, Knapp M, Lichtermann D et al. Human delta-opioid receptor gene and susceptibility to heroin and alcohol dependence. Am J Med Genet 1999; 88: 462–464.
Mayer P, Rochlitz H, Rauch E, Rommelspacher H, Hasse HE, Schmidt S et al. Association between a delta opioid receptor gene polymorphism and heroin dependence in man. Neuroreport 1997; 8: 2547–2550.
Lobo DS, Kennedy JL . Genetic aspects of pathological gambling: a complex disorder with shared genetic vulnerabilities. Addiction 2009; 104: 1454–1465.
Comings DE, Gade R, Wu S, Chiu C, Dietz G, Muhleman D et al. Studies of the potential role of the dopamine D1 receptor gene in addictive behaviors. Mol Psychiatry 1997; 2: 44–56.
da Silva Lobo DS, Vallada HP, Knight J, Martins SS, Tavares H, Gentil V et al. Dopamine genes and pathological gambling in discordant sib-pairs. J Gambl Stud 2007; 23: 421–433.
Lobo DS, Souza RP, Tong RP, Casey DM, Hodgins DC, Smith GJ et al. Association of functional variants in the dopamine D2-like receptors with risk for gambling behaviour in healthy Caucasian subjects. Biol Psychol 2010; 85: 33–37.
Thomasson HR, Crabb DW, Edenberg HJ, Li TK . Alcohol and aldehyde dehydrogenase polymorphisms and alcoholism. Behav Genet 1993; 23: 131–136.
Higuchi S, Matsushita S, Imazeki H, Kinoshita T, Takagi S, Kono H . Aldehyde dehydrogenase genotypes in Japanese alcoholics. Lancet 1994; 343: 741–742.
Lee HC, Lee HS, Jung SH, Yi SY, Jung HK, Yoon JH et al. Association between polymorphisms of ethanol-metabolizing enzymes and susceptibility to alcoholic cirrhosis in a Korean male population. J Korean Med Sci 2001; 16: 745–750.
Koob GF, Volkow ND . Neurocircuitry of addiction. Neuropsychopharmacology 2010; 35: 217–238.
Volkow ND, Fowler JS, Wang GJ, Swanson JM, Telang F . Dopamine in drug abuse and addiction: results of imaging studies and treatment implications. Arch Neurol 2007; 64: 1575–1579.
Bierut LJ, Madden PA, Breslau N, Johnson EO, Hatsukami D, Pomerleau OF et al. Novel genes identified in a high-density genome wide association study for nicotine dependence. Hum Mol Genet 2007; 16: 24–35.
Treutlein J, Cichon S, Ridinger M, Wodarz N, Soyka M, Zill P et al. Genome-wide association study of alcohol dependence. Arch Gen Psychiatry 2009; 66: 773–784.
Frank J, Cichon S, Treutlein J, Ridinger M, Mattheisen M, Hoffmann P et al. Genome-wide significant association between alcohol dependence and a variant in the ADH gene cluster. Addict Biol 2012; 17: 171–180.
Edenberg HJ, Koller DL, Xuei X, Wetherill L, McClintick JN, Almasy L et al. Genome-wide association study of alcohol dependence implicates a region on chromosome 11. Alcohol Clin Exp Res 2010; 34: 840–852.
Bierut LJ, Agrawal A, Bucholz KK, Doheny KF, Laurie C, Pugh E et al. A genome-wide association study of alcohol dependence. Proc Natl Acad Sci U S A 2010; 107: 5082–5087.
Heath AC, Whitfield JB, Martin NG, Pergadia ML, Goate AM, Lind PA et al. A quantitative-trait genome-wide association study of alcoholism risk in the community: findings and implications. Biol Psychiatry 2011; 70: 513–518.
Agrawal A, Lynskey MT, Hinrichs A, Grucza R, Saccone SF, Krueger R et al. A genome-wide association study of DSM-IV cannabis dependence. Addict Biol 2011; 16: 514–518.
Tobacco and Genetics Consortium. Genome-wide meta-analyses identify multiple loci associated with smoking behavior. Nat Genet 2010; 42: 441–447.
Thorgeirsson TE, Gudbjartsson DF, Surakka I, Vink JM, Amin N, Geller F et al. Sequence variants at CHRNB3-CHRNA6 and CYP2A6 affect smoking behavior. Nat Genet 2010; 42: 448–453.
Liu JZ, Tozzi F, Waterworth DM, Pillai SG, Muglia P, Middleton L et al. Meta-analysis and imputation refines the association of 15q25 with smoking quantity. Nat Genet 2010; 42: 436–440.
Saccone SF, Hinrichs AL, Saccone NL, Chase GA, Konvicka K, Madden PA et al. Cholinergic nicotinic receptor genes implicated in a nicotine dependence association study targeting 348 candidate genes with 3713 SNPs. Hum Mol Genet 2007; 16: 36–49.
Schumann G, Coin LJ, Lourdusamy A, Charoen P, Berger KH, Stacey D et al. Genome-wide association and genetic functional studies identify autism susceptibility candidate 2 gene (AUTS2) in the regulation of alcohol consumption. Proc Natl Acad Sci U S A 2011; 108: 7119–7124.
Maher B . Personal genomes: the case of the missing heritability. Nature 2008; 456: 18–21.
Maher B . The search for the genome ‘Dark Matter’ moves closer. Nature News 2008; 1235, doi:10.1038/news.2008.1235.
Yang J, Benyamin B, McEvoy BP, Gordon S, Henders AK, Nyholt DR et al. Common SNPs explain a large proportion of the heritability for human height. Nat Genet 2010; 42: 565–569.
Johnson C, Drgon T, Liu QR, Zhang PW, Walther D, Li CY et al. Genome wide association for substance dependence: convergent results from epidemiologic and research volunteer samples. BMC Med Genet 2008; 9: 113.
Drgon T, Johnson CA, Nino M, Drgonova J, Walther DM, Uhl GR . ‘Replicated’ genome wide association for dependence on illegal substances: genomic regions identified by overlapping clusters of nominally positive SNPs. Am J Med Genet B Neuropsychiatr Genet 2011; 156: 125–138.
Johnson C, Drgon T, Walther D, Uhl GR . Genomic regions identified by overlapping clusters of nominally-positive SNPs from genome-wide studies of alcohol and illegal substance dependence. PLoS One 2011; 6: e19210.
Yang J, Manolio TA, Pasquale LR, Boerwinkle E, Caporaso N, Cunningham JM et al. Genome partitioning of genetic variation for complex traits using common SNPs. Nat Genet 2011; 43: 519–525.
Davies G, Tenesa A, Payton A, Yang J, Harris SE, Liewald D et al. Genome-wide association studies establish that human intelligence is highly heritable and polygenic. Mol Psychiatry 2011; 16: 996–1005.
Bierut LJ, Goate AM, Breslau N, Johnson EO, Bertelsen S, Fox L et al. ADH1B is associated with alcohol dependence and alcohol consumption in populations of European and African ancestry. Mol Psychiatry 2012; 17: 445–450.
Agrawal A, Pergadia ML, Saccone SF, Lynskey MT, Wang JC, Martin NG et al. An autosomal linkage scan for cannabis use disorders in the nicotine addiction genetics project. Arch Gen Psychiatry 2008; 65: 713–721.
Grant JD, Agrawal A, Bucholz KK, Madden PA, Pergadia ML, Nelson EC et al. Alcohol consumption indices of genetic risk for alcohol dependence. Biol Psychiatry 2009; 66: 795–800.
Agrawal A, Grant JD, Littlefield A, Waldron M, Pergadia ML, Lynskey MT et al. Developing a quantitative measure of alcohol consumption for genomic studies on prospective cohorts. J Stud Alcohol Drugs 2009; 70: 157–168.
Kendler KS, Myers J, Dick D, Prescott CA . The relationship between genetic influences on alcohol dependence and on patterns of alcohol consumption. Alcohol Clin Exp Res 2010; 34: 1058–1065.
Dick DM, Meyers JL, Rose RJ, Kaprio J, Kendler KS . Measures of current alcohol consumption and problems: two independent twin studies suggest a complex genetic architecture. Alcohol Clin Exp Res 2011; 35: 2152–2161.
Heatherton TF, Kozlowski LT, Frecker RC, Rickert W, Robinson J . Validity of the Fagerstrom test for nicotine dependence and of the Heaviness of Smoking Index among relatively light smokers. Br J Addict 1989; 84: 791–799.
Heatherton TF, Kozlowski LT, Frecker RC, Fagerstrom KO . The Fagerstrom test for nicotine dependence: a revision of the Fagerstrom Tolerance Questionnaire. Br J Addict 1991; 86: 1119–1127.
Moolchan ET, Aung AT, Henningfield JE . Treatment of adolescent tobacco smokers: issues and opportunities for exposure reduction approaches. Drug Alcohol Depend 2003; 70: 223–232.
Schuckit MA, Gold EO . A simultaneous evaluation of multiple markers of ethanol/placebo challenges in sons of alcoholics and controls. Arch Gen Psychiatry 1988; 45: 211–216.
Schuckit M . Vulnerability factors for alcoholism. In: Davis K (ed). Neuropsychopharmacology: The Fifth Generation of Progress. Lippincott Williams and Wilkins: Baltimore, 2002.
Schuckit MA, Smith TL, Kalmijn J . The search for genes contributing to the low level of response to alcohol: patterns of findings across studies. Alcohol Clin Exp Res 2004; 28: 1449–1458.
Vogel-Sprott M, Chipperfield B . Family history of problem drinking among young male social drinkers: behavioral effects of alcohol. J Stud Alcohol 1987; 48: 430–436.
Ramchandani VA, Flury L, Morzorati SL, Kareken D, Blekher T, Foroud T et al. Recent drinking history: association with family history of alcoholism and the acute response to alcohol during a 60 mg% clamp. J Stud Alcohol 2002; 63: 734–744.
Roh S, Matsushita S, Hara S, Maesato H, Matsui T, Suzuki G et al. Role of GABRA2 in moderating subjective responses to alcohol. Alcohol Clin Exp Res 2011; 35: 400–407.
Pierucci-Lagha A, Covault J, Feinn R, Nellissery M, Hernandez-Avila C, Oncken C et al. GABRA2 alleles moderate the subjective effects of alcohol, which are attenuated by finasteride. Neuropsychopharmacology 2005; 30: 1193–1203.
Hu X, Oroszi G, Chun J, Smith TL, Goldman D, Schuckit MA . An expanded evaluation of the relationship of four alleles to the level of response to alcohol and the alcoholism risk. Alcohol Clin Exp Res 2005; 29: 8–16.
Gottesman II, Gould TD . The endophenotype concept in psychiatry: etymology and strategic intentions. Am J Psychiatry 2003; 160: 636–645.
Kendler KS, Neale MC . Endophenotype: a conceptual analysis. Mol Psychiatry 2010; 15: 789–797.
Cannon TD, Keller MC . Endophenotypes in the genetic analyses of mental disorders. Annu Rev Clin Psychol 2006; 2: 267–290.
Field M, Cox WM . Attentional bias in addictive behaviors: a review of its development, causes, and consequences. Drug Alcohol Depend 2008; 97: 1–20.
Rangaswamy M, Porjesz B . Uncovering genes for cognitive (dys)function and predisposition for alcoholism spectrum disorders: a review of human brain oscillations as effective endophenotypes. Brain Res 2008; 1235: 153–171.
Porjesz B, Rangaswamy M . Neurophysiological endophenotypes, CNS disinhibition, and risk for alcohol dependence and related disorders. ScientificWorldJournal 2007; 7: 131–141.
Begleiter H, Porjesz B . Genetics of human brain oscillations. Int J Psychophysiol 2006; 60: 162–171.
Porjesz B, Almasy L, Edenberg HJ, Wang K, Chorlian DB, Foroud T et al. Linkage disequilibrium between the beta frequency of the human EEG and a GABAA receptor gene locus. Proc Natl Acad Sci U S A 2002; 99: 3729–3733.
Kutalik Z, Benyamin B, Bergmann S, Mooser V, Waeber G, Montgomery GW et al. Genome-wide association study identifies two loci strongly affecting transferrin glycosylation. Hum Mol Genet 2011; 20: 3710–3717.
Villafuerte S, Heitzeg MM, Foley S, Wendy Yau WY, Majczenko K, Zubieta JK et al. Impulsiveness and insula activation during reward anticipation are associated with genetic variants in GABRA2 in a family sample enriched for alcoholism. Mol Psychiatry 2012; 17: 511–519.
Nikolova YS, Ferrell RE, Manuck SB, Hariri AR . Multilocus genetic profile for dopamine signaling predicts ventral striatum reactivity. Neuropsychopharmacology 2011; 36: 1940–1947.
Hamilton CM, Strader LC, Pratt JG, Maiese D, Hendershot T, Kwok RK et al. The PhenX toolkit: get the most from your measures. Am J Epidemiol 2011; 174: 253–260.
Stankiewicz P, Lupski JR . Structural variation in the human genome and its role in disease. Annu Rev Med 2010; 61: 437–455.
1000 Genomes Project Consortium. A map of human genome variation from population-scale sequencing. Nature 2010; 467: 1061–1073.
Renthal W, Nestler EJ . Epigenetic mechanisms in drug addiction. Trends Mol Med 2008; 14: 341–350.
Maze I, Nestler EJ . The epigenetic landscape of addiction. Ann N Y Acad Sci 2011; 1216: 99–113.
Wong CC, Mill J, Fernandes C . Drugs and addiction: an introduction to epigenetics. Addiction 2011; 106: 480–489.
Bernstein BE, Stamatoyannopoulos JA, Costello JF, Ren B, Milosavljevic A, Meissner A et al. The NIH roadmap epigenomics mapping consortium. Nat Biotechnol 2010; 28: 1045–1048.
Myers RM, Stamatoyannopoulos J, Snyder M, Dunham I, Hardison RC, Bernstein BE et al. A user's guide to the encyclopedia of DNA elements (ENCODE). PLoS Biol 2011; 9: e1001046.
Hurley JH, Ballard CJ, Edenberg HJ . Altering the relative abundance of GABA A receptor subunits changes GABA- and ethanol-responses in Xenopus oocytes. Alcohol Clin Exp Res 2009; 33: 1089–1096.
Liu J, Yang AR, Kelly T, Puche A, Esoga C, June Jr HL et al. Binge alcohol drinking is associated with GABAA alpha2-regulated Toll-like receptor 4 (TLR4) expression in the central amygdala. Proc Natl Acad Sci U S A 2011; 108: 4465–4470.
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 2007; 81: 559–575.
Liu JZ, McRae AF, Nyholt DR, Medland SE, Wray NR, Brown KM et al. A versatile gene-based test for genome-wide association studies. Am J Hum Genet 2010; 87: 139–145.
Raychaudhuri S, Plenge RM, Rossin EJ, Ng AC, Purcell SM, Sklar P et al. Identifying relationships among genomic disease regions: predicting genes at pathogenic SNP associations and rare deletions. PLoS Genet 2009; 5: e1000534.
Li MX, Gui HS, Kwan JS, Sham PC . GATES: a rapid and powerful gene-based association test using extended Simes procedure. Am J Hum Genet 2011; 88: 283–293.
Ruano D, Abecasis GR, Glaser B, Lips ES, Cornelisse LN, de Jong AP et al. Functional gene group analysis reveals a role of synaptic heterotrimeric G proteins in cognitive ability. Am J Hum Genet 2010; 86: 113–125.
Reimers MA, Riley BP, Kalsi G, Kertes DA, Kendler KS . Pathway based analysis of genotypes in relation to alcohol dependence. Pharmacogenomics J 2011 (in press).
Harris MA, Clark J, Ireland A, Lomax J, Ashburner M, Foulger R et al. The Gene Ontology (GO) database and informatics resource. Nucleic Acids Res 2004; 32 (Database issue): D258–D261.
Scarr S, McCartney J . How people make their own environment: a theory of genotype greater than environmental effects. Child Dev 1983; 54: 424–435.
Heath AC, Nelson EC . Effects of the interaction between genotype and environment. Research into the genetic epidemiology of alcohol dependence. Alcohol Res Health 2002; 26: 193–201.
Dick DM, Rose RJ, Viken RJ, Kaprio J, Koskenvuo M . Exploring gene-environment interactions: socioregional moderation of alcohol use. J Abnorm Psychol 2001; 110: 625–632.
Dick DM, Bernard M, Aliev F, Viken R, Pulkkinen L, Kaprio J et al. The role of socioregional factors in moderating genetic influences on early adolescent behavior problems and alcohol use. Alcohol Clin Exp Res 2009; 33: 1739–1748.
Dick DM, Viken R, Purcell S, Kaprio J, Pulkkinen L, Rose RJ . Parental monitoring moderates the importance of genetic and environmental influences on adolescent smoking. J Abnorm Psychol 2007; 116: 213–218.
Button TM, Stallings MC, Rhee SH, Corley RP, Boardman JD, Hewitt JK . Perceived peer delinquency and the genetic predisposition for substance dependence vulnerability. Drug Alcohol Depend 2009; 100: 1–8.
Agrawal A, Balasubramanian S, Smith EK, Madden PA, Bucholz KK, Heath AC et al. Peer substance involvement modifies genetic influences on regular substance involvement in young women. Addiction 2010; 105: 1844–1853.
Caspi A, Moffitt TE . Gene-environment interactions in psychiatry: joining forces with neuroscience. Nat Rev Neurosci 2006; 7: 583–590.
Rutter M, Moffitt TE, Caspi A . Gene-environment interplay and psychopathology: multiple varieties but real effects. J Child Psychol Psychiatry 2006; 47: 226–261.
Dick DM, Latendresse SJ, Lansford JE, Budde JP, Goate A, Dodge KA et al. Role of GABRA2 in trajectories of externalizing behavior across development and evidence of moderation by parental monitoring. Arch Gen Psychiatry 2009; 66: 649–657.
Dick DM, Meyers JL, Latendresse SJ, Creemers HE, Lansford JE, Pettit GS et al. CHRM2, parental monitoring, and adolescent externalizing behavior: evidence for gene-environment interaction. Psychol Sci 2011; 22: 481–489.
Nelson EC, Agrawal A, Pergadia ML, Wang JC, Whitfield JB, Saccone FS et al. H2 haplotype at chromosome 17q21.31 protects against childhood sexual abuse-associated risk for alcohol consumption and dependence. Addict Biol 2010; 15: 1–11.
Nelson EC, Heath AC, Lynskey MT, Bucholz KK, Madden PA, Statham DJ et al. Childhood sexual abuse and risks for licit and illicit drug-related outcomes: a twin study. Psychol Med 2006; 36: 1473–1483.
Fraga MF, Ballestar E, Paz MF, Ropero S, Setien F, Ballestar ML et al. Epigenetic differences arise during the lifetime of monozygotic twins. Proc Natl Acad Sci U S A 2005; 102: 10604–10609.
Risch N, Herrell R, Lehner T, Liang KY, Eaves L, Hoh J et al. Interaction between the serotonin transporter gene (5-HTTLPR), stressful life events, and risk of depression: a meta-analysis. JAMA 2009; 301: 2462–2471.
Duncan LE, Keller MC . A critical review of the first 10 years of candidate gene-by-environment interaction research in psychiatry. Am J Psychiatry 2011; 168: 1041–1049.
van Ijzendoorn MH, Bakermans-Kranenburg MJ, Belsky J, Beach S, Brody G, Dodge KA et al. Gene-by-environment experiments: a new approach to finding the missing heritability. Nat Rev Genet 2011; 12: 881.
Schnoll RA, Lerman C . Current and emerging pharmacotherapies for treating tobacco dependence. Expert Opin Emerg Drugs 2006; 11: 429–444.
Hughes JR . How confident should we be that smoking cessation treatments work? Addiction 2009; 104: 1637–1640.
Sturgess JE, George TP, Kennedy JL, Heinz A, Muller DJ . Pharmacogenetics of alcohol, nicotine and drug addiction treatments. Addict Biol 2011; 16: 357–376.
Oslin DW, Berrettini W, Kranzler HR, Pettinati H, Gelernter J, Volpicelli JR et al. A functional polymorphism of the mu-opioid receptor gene is associated with naltrexone response in alcohol-dependent patients. Neuropsychopharmacology 2003; 28: 1546–1552.
Ray LA, Hutchison KE . Effects of naltrexone on alcohol sensitivity and genetic moderators of medication response: a double-blind placebo-controlled study. Arch Gen Psychiatry 2007; 64: 1069–1077.
Anton RF, Oroszi G, O’Malley S, Couper D, Swift R, Pettinati H et al. An evaluation of mu-opioid receptor (OPRM1) as a predictor of naltrexone response in the treatment of alcohol dependence: results from the Combined Pharmacotherapies and Behavioral Interventions for Alcohol Dependence (COMBINE) study. Arch Gen Psychiatry 2008; 65: 135–144.
Heilig M, Goldman D, Berrettini W, O’Brien CP . Pharmacogenetic approaches to the treatment of alcohol addiction. Nat Rev Neurosci 2011; 12: 670–684.
Kenna GA . Medications acting on the serotonergic system for the treatment of alcohol dependent patients. Curr Pharm Des 2010; 16: 2126–2135.
McKay JD, Truong T, Gaborieau V, Chabrier A, Chuang SC, Byrnes G et al. A genome-wide association study of upper aerodigestive tract cancers conducted within the INHANCE consortium. PLoS Genet 2011; 7: e1001333.
Brooks PJ, Enoch MA, Goldman D, Li TK, Yokoyama A . The alcohol flushing response: an unrecognized risk factor for esophageal cancer from alcohol consumption. PLoS Med 2009; 6: e50.
Brooks PJ, Goldman D, Li TK . Alleles of alcohol and acetaldehyde metabolism genes modulate susceptibility to oesophageal cancer from alcohol consumption. Hum Genomics 2009; 3: 103–105.
Wang Y, Broderick P, Webb E, Wu X, Vijayakrishnan J, Matakidou A et al. Common 5p15.33 and 6p21.33 variants influence lung cancer risk. Nat Genet 2008; 40: 1407–1409.
Thorgeirsson TE, Geller F, Sulem P, Rafnar T, Wiste A, Magnusson KP et al. A variant associated with nicotine dependence, lung cancer and peripheral arterial disease. Nature 2008; 452: 638–642.
Hung RJ, McKay JD, Gaborieau V, Boffetta P, Hashibe M, Zaridze D et al. A susceptibility locus for lung cancer maps to nicotinic acetylcholine receptor subunit genes on 15q25. Nature 2008; 452: 633–637.
Saccone NL, Culverhouse RC, Schwantes-An TH, Cannon DS, Chen X, Cichon S et al. Multiple independent loci at chromosome 15q25.1 affect smoking quantity: a meta-analysis and comparison with lung cancer and COPD. PLoS Genet 2010; 6, pii: e1001053.
Wacholder S, Chatterjee N, Caporaso N . Intermediacy and gene-environment interaction: the example of CHRNA5-A3 region, smoking, nicotine dependence, and lung cancer. J Natl Cancer Inst 2008; 100: 1488–1491.
Calandrella D, Antonini A . Pathological gambling in Parkinson's disease: disease related or drug related? Expert Rev Neurother 2011; 11: 809–814.
Bell RL, Rodd ZA, Lumeng L, Murphy JM, McBride WJ . The alcohol-preferring P rat and animal models of excessive alcohol drinking. Addict Biol 2006; 11: 270–288.
Sommer W, Hyytia P, Kiianmaa K . The alcohol-preferring AA and alcohol-avoiding ANA rats: neurobiology of the regulation of alcohol drinking. Addict Biol 2006; 11: 289–309.
Crabbe JC, Harris RA, Koob GF . Preclinical studies of alcohol binge drinking. Ann N Y Acad Sci 2011; 1216: 24–40.
Heberlein U . Genetics of alcohol-induced behaviors in Drosophila. Alcohol Res Health 2000; 24: 185–188.
Crabbe JC . Consilience of rodent and human phenotypes relevant for alcohol dependence. Addict Biol 2010; 15: 103–108.
Merikangas KR, Risch N . Genomic priorities and public health. Science 2003; 302: 599–601.
Hirschhorn JN . Genomewide association studies—illuminating biologic pathways. N Engl J Med 2009; 360: 1699–1701.
Oze I, Matsuo K, Wakai K, Nagata C, Mizoue T, Tanaka K et al. Alcohol drinking and esophageal cancer risk: an evaluation based on a systematic review of epidemiologic evidence among the Japanese population. Jpn J Clin Oncol 2011; 41: 677–692.
Luo X, Kranzler HR, Zuo L, Wang S, Schork NJ, Gelernter J . Diplotype trend regression analysis of the ADH gene cluster and the ALDH2 gene: multiple significant associations with alcohol dependence. Am J Hum Genet 2006; 78: 973–987.
Macgregor S, Lind PA, Bucholz KK, Hansell NK, Madden PA, Richter MM et al. Associations of ADH and ALDH2 gene variation with self report alcohol reactions, consumption and dependence: an integrated analysis. Hum Mol Genet 2009; 18: 580–593.
Li D, Zhao H, Gelernter J . Strong Association of the Alcohol Dehydrogenase 1B Gene (ADH1B) with alcohol dependence and alcohol-induced medical diseases. Biol Psychiatry 2011; 70: 504–512.
Soyka M, Preuss UW, Hesselbrock V, Zill P, Koller G, Bondy B . GABA-A2 receptor subunit gene (GABRA2) polymorphisms and risk for alcohol dependence. J Psychiatr Res 2008; 42: 184–191.
Noble EP, Blum K . Alcoholism and the D2 dopamine receptor gene. JAMA 1993; 270: 1547–1548.
Munafo MR, Matheson IJ, Flint J . Association of the DRD2 gene Taq1A polymorphism and alcoholism: a meta-analysis of case-control studies and evidence of publication bias. Mol Psychiatry 2007; 12: 454–461.
Smith L, Watson M, Gates S, Ball D, Foxcroft D . Meta-analysis of the association of the Taq1A polymorphism with the risk of alcohol dependency: a HuGE gene-disease association review. Am J Epidemiol 2008; 167: 125–138.
Bierut LJ, Stitzel JA, Wang JC, Hinrichs AL, Grucza RA, Xuei X et al. Variants in nicotinic receptors and risk for nicotine dependence. Am J Psychiatry 2008; 165: 1163–1171.
Kuryatov A, Berrettini W, Lindstrom J . Acetylcholine receptor (AChR) alpha5 subunit variant associated with risk for nicotine dependence and lung cancer reduces (alpha4beta2)alpha5 AChR function. Mol Pharmacol 2011; 79: 119–125.
Lessov-Schlaggar CN, Pergadia ML, Khroyan TV, Swan GE . Genetics of nicotine dependence and pharmacotherapy. Biochem Pharmacol 2008; 75: 178–195.
Heath AC . Persist or quit? Testing for a genetic contribution to smoking persistence. Acta Genet Med Gemellol (Roma) 1990; 39: 447–458.
Hughes JR . Should criteria for drug dependence differ across drugs? Addiction 2006; 101 (Suppl 1): 134–141.
McBride O, Strong DR, Kahler CW . Exploring the role of a nicotine quantity-frequency use criterion in the classification of nicotine dependence and the stability of a nicotine dependence continuum over time. Nicotine Tob Res 2010; 12: 207–216.
Strong DR, Kahler CW, Colby SM, Griesler PC, Kandel D . Linking measures of adolescent nicotine dependence to a common latent continuum. Drug Alcohol Depend 2009; 99: 296–308.
Bierut LJ, Dinwiddie SH, Begleiter H, Crowe RR, Hesselbrock V, Nurnberger Jr JI et al. Familial transmission of substance dependence: alcohol, marijuana, cocaine, and habitual smoking: a report from the Collaborative Study on the Genetics of Alcoholism. Arch Gen Psychiatry 1998; 55: 982–988.
Kaprio J, Hammar N, Koskenvuo M, Floderus-Myrhed B, Langinvainio H, Sarna S . Cigarette smoking and alcohol use in Finland and Sweden: a cross-national twin study. Int J Epidemiol 1982; 11: 378–386.
Pergadia ML, Heath AC, Martin NG, Madden PA . Genetic analysis of DSM-IV nicotine withdrawal in adult twins. Psychol Med 2006; 36: 963–972.
Han S, Gelernter J, Luo X, Yang BZ . Meta-analysis of 15 genome-wide linkage scans of smoking behavior. Biol Psychiatry 2010; 67: 12–19.
Boardman JD . State-level moderation of genetic tendencies to smoke. Am J Public Health 2009; 99: 480–486.
Johnson EO, Chen LS, Breslau N, Hatsukami D, Robbins T, Saccone NL et al. Peer smoking and the nicotinic receptor genes: an examination of genetic and environmental risks for nicotine dependence. Addiction 2010; 105: 2014–2022.
Chen LS, Johnson EO, Breslau N, Hatsukami D, Saccone NL, Grucza RA et al. Interplay of genetic risk factors and parent monitoring in risk for nicotine dependence. Addiction 2009; 104: 1731–1740.
Weiss RB, Baker TB, Cannon DS, von NA, Dunn DM, Matsunami N et al. A candidate gene approach identifies the CHRNA5-A3-B4 region as a risk factor for age-dependent nicotine addiction. PLoS Genet 2008; 4: e1000125.
Grucza RA, Johnson EO, Krueger RF, Breslau N, Saccone NL, Chen LS et al. Incorporating age at onset of smoking into genetic models for nicotine dependence: evidence for interaction with multiple genes. Addict Biol 2010; 15: 346–357.
Vink JM, Smit AB, de Geus EJ, Sullivan P, Willemsen G, Hottenga JJ et al. Genome-wide association study of smoking initiation and current smoking. Am J Hum Genet 2009; 84: 367–379.
Jackson KJ, Marks MJ, Vann RE, Chen X, Gamage TF, Warner JA et al. Role of alpha5 nicotinic acetylcholine receptors in pharmacological and behavioral effects of nicotine in mice. J Pharmacol Exp Ther 2010; 334: 137–146.
Fowler CD, Lu Q, Johnson PM, Marks MJ, Kenny PJ . Habenular alpha5 nicotinic receptor subunit signalling controls nicotine intake. Nature 2011; 471: 597–601.
Hong LE, Hodgkinson CA, Yang Y, Sampath H, Ross TJ, Buchholz B et al. A genetically modulated, intrinsic cingulate circuit supports human nicotine addiction. Proc Natl Acad Sci U S A 2010; 107: 13509–13514.
Swan GE, Javitz HS, Jack LM, Wessel J, Michel M, Hinds DA et al. Varenicline for smoking cessation: nausea severity and variation in nicotinic receptor genes. Pharmacogenomics J 2011 (in press).
Rose JE, Behm FM, Drgon T, Johnson C, Uhl GR . Personalized smoking cessation: interactions between nicotine dose, dependence and quit-success genotype score. Mol Med 2010; 16: 247–253.
Acknowledgements
The preparation of this review was supported by the National Institute on Drug Abuse (NIDA) Grant DA23668 to AA. AA receives support from ABMRF/Foundation for Alcohol Research. We also gratefully acknowledge current and past funding support from the National Institutes of Health and the National Health & Medical Research Council of Australia which has facilitated our research, some of which is showcased here. We acknowledge funding from the National Institute on Drug Abuse (NIDA) Grants DA25886 (AA), DA18660 (MTL), DA030976 (WSS co-I), DA17305 (ECN), DA027046 (CNLS) and R00DA023549 (NAG); National Institute on Alcohol Abuse and Alcoholism (NIAAA) Grants AA11998 (MTL, ACH, ECN), AA014041 (JBW), AA013326 (NGM) and AA17688 (ACH). KJHV is supported by an ANZ Trustees PhD scholarship in Medical Research. Funding bodies did not participate in the preparation of this review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under the Creative Commons Attribution-NonCommercial-No Derivative Works 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Agrawal, A., Verweij, K., Gillespie, N. et al. The genetics of addiction—a translational perspective. Transl Psychiatry 2, e140 (2012). https://doi.org/10.1038/tp.2012.54
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tp.2012.54
Keywords
This article is cited by
-
The neuropsychopharmacology of acetyl-L-carnitine (LAC): basic, translational and therapeutic implications
Discover Mental Health (2024)
-
The Genetically Informed Neurobiology of Addiction (GINA) model
Nature Reviews Neuroscience (2023)
-
Effect of human mesenchymal stem cell secretome administration on morphine self-administration and relapse in two animal models of opioid dependence
Translational Psychiatry (2022)