Abstract
Geese are extremely adept in utilizing plant-derived roughage within their diet. However, the intestinal microbiome of geese remains limited, especially the dietary effect on microbial diversity. Cassava foliage was widely used in animal feed, but little information is available for geese. In this study, the geese were fed with control diet (CK), experimental diet supplemented with 5% cassava foliage (CF5) or 10% (CF10) for 42 days, respectively. The cecal samples were collected after animals were killed. High-throughput sequencing technology was used to investigate the microbial diversity in the caecum of geese with different dietary supplements. Taxonomic analysis indicated that the predominant phyla were distinct with different dietary treatments. The phyla Firmicutes (51.4%), Bacteroidetes (29.55%) and Proteobacteria (7.90%) were dominant in the CK group, but Bacteroidetes (65.19% and 67.29%,) Firmicutes (18.01% and 17.39%), Proteobacteria (8.72% and 10.18%), Synergistete (2.51% and 1.76%) and Spirochaetes (2.60% and 1.46%) were dominant in CF5 and CF10 groups. The abundance of Firmicutes was negatively correlated with the supplementation of cassava foliage. However, the abundance of Bacteroidetes and Proteobacteria were positively correlated with the supplementation of cassava foliage. Our study also revealed that the microbial communities were significantly different at genus levels. Genes related to nutrient and energy metabolism, immunity and signal transduction pathways were primarily enriched by the microbiome.
Similar content being viewed by others
Introduction
Microbial community in gastrointestinal tract (GIT) related to the functions of host, including maintaining host health, improving performance, reducing environmental pollution, and ensuring food and animal products’ safety1,2,3,4,5. Meanwhile, fecal microbiome also reflects feed conversion, gut pathogen and parasite colonization and immune system activity of host6,7,8. Therefore, both gut and fecal microbiome play an important role for animals. However, feces are the final products of digestion, and fecal microbiome mainly affects the environment rather than host which composition is mainly determined by GIT original microbiome9. Furthermore, gut microbiome and host have interaction effect, and nutrition means could regulate gut microbiome in animal production. The small intestine, colon and caecum have similar roles in digestion and absorption of nutrient components. However, there are more microbes in caecum than other GIT2. The mechanism of caecum fiber digestion is similar with rumen, fiber fermentation by microbes eventually produces volatile fatty acids (mainly acetic acid, propionic acid and butyric acid) and ammonia, and then the intestinal epithelium absorbs these for host. Therefore, analyses of caecum microbiome represent a key area of nutrition research in poultry, and it would lead to an additional understanding of the microbial biodiversity and interactions with hosts.
Compared with duck or other family Anatidae poultry, geese have excellent ability of roughage utilization and adaptability. Even if comparable proventriculus, small intestine and caecum, geese’s GIT has a bigger and more powerful gizzard. The powerful gizzards can generate greatest forces for breaking down the roughage with high fiber content, such as forage, corn straw silage, wheat straw and rice byproducts10,11,12. Another pivotal reason is that geese have increasing effective caecum fermentation. The microbiome in caecum actively ferment and convert fiber contents into digestible components for animal hosts. The caecum is a complex ecosystem that harbors a wide variety of microbiome, which is an important factor for animal production. Therefore, it is necessary to study the gut microbial diversity in geese. However, only few studies have investigated the microbial diversity in the caecum of geese. Wang et al.13 reported that the dominant bacteria in the caecum of geese are related to Pseudomonas sp. and Bifidobacterium sp. by denaturing gradient gel electrophoresis (DGGE) fingerprints. Liu et al.14 analyzed the geese caecum microbial diversity using 16S rRNA clone library approach, which are dominantly occupied by Clostridia-related species (58.7%), followed by Bacteroidetes (26.9%) and Erysipelotrichi (11.2%). However, the DGGE technique fails to accurately represent the gut microbiome due to its low coverage, throughput and semi-quantitative features. In addition, the DGGE technique is time-consuming and insufficient to reflect the true diversity of a diverse gut microbiota. The 16S rRNA has been widely used to study and characterize bacterial community compositions because it can maximize the bacterial classification. However, the deficiencies of this technology are very obvious, and bacterial diversity is limited by depth of sequencing and cost. In recent years, high-throughput next-generation sequencing (HT-NGS) has become more sophisticated, and it provides large-scale analysis with unprecedented depths and coverages. Compared with conventional 16S rRNA technique, HT-NGS 16S rRNA technique can achieve high coverage, which can reflect the microbiome structure more accurately. HT-NGS technology has been used to study the intestinal microbiome of chickens, rabbits, goats and dairy cows15,16,17,18. However, to date, there are only few studies reported the microbial community in the caecum of geese by HT-NGS technology.
Cassava foliage is known as an agricultural by-product with high protein contents, gross energy and mineral elements, and it can be used as animal feed. It has been used as dietary in chickens and ducks with positive effects on the growth performance19. From the perspective of feed resource, little information is available regarding the utilization of cassava foliage for geese. Therefore, we aimed to investigate the effects of cassava foliage as a dietary supplement on microbial diversity in the caecum of geese using HT-NGS technology.
Results
Sequences
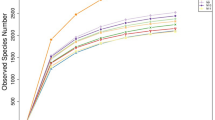
A total of 712,593,146 qualified sequences were obtained from the 18 samples from 18 geese caecum. These sequences included an average of 77,100 reads per caecum sample, and the length of unique tag N50 is 460 bp. (Table 2 and Table S1 and 2). Shannon value and Shannon rarefaction curves for each sample reached the saturation plateau (Table 2 and Fig. 1), indicating that the sampling effort had sufficient sequence coverage to accurately describe the bacterial composition of each group. Indices of bacterial richness based on OTUs were estimated by the method of Ace and Chao, and indices of bacterial diversity were determined using the method of Simpson and Shannon (Table 2). Among the 18 samples, a total of 200,365 OTUs were detected by our analysis, with an average of 11,131 OTUs per sample (Table 2).
Taxonomic composition
All sequences were classified from phylum to species based on the SILVA taxonomic database and using the analytical program QIIME. A total of 24 different phyla were detected from these samples. The Fig. 2 shows that the three groups had very dissimilar taxonomic compositions, even at the phylum-level distribution. Firmicutes (51.4%), Bacteroidetes (29.55%) and Proteobacteria (7.90%) were the dominant phyla in the CK (control diet) group. Bacteroidetes, Firmicutes, Proteobacteria, Synergistete and Spirochaetes were the dominant phyla in the CF5 (experimental diet was supplemented with 5% cassava foliage on the base of control diet) group, representing 65.19%, 18.01%, 8.72%, 2.51% and 2.60% of the total reads, respectively. Bacteroidetes, Firmicutes, Proteobacteria, Synergistete and Spirochaetes were the most commonly detected phyla in the CF10 (experimental diet was supplemented with 10% cassava foliage on the base of control diet) group, accounting for 67.29%, 17.39%, 10.18%, 1.76% and 1.46% of the total reads, respectively.
Effects of cassava foliage on the relative abundance (%reads) of (A) the most dominant phylum and (B) the most dominant genus in the cecal microbiome of geese. Error bars represent the SD of three samples. Boxes with a different letter above the error bars are significantly different at P < 0.01 by t-test analyses.CK represents control diet group; CF5 represents experimental diet which supplemented with 5% cassava foliage on the base of control diet; CF10 represents experimental diet which supplemented with 10% cassava foliage on the base of control diet. The same as below.
At the genus level, the detected sequences could be assigned into 163 different genera. The most abundant genera (the relative abundance of more than 1% of the three libraries) among the libraries suggested the importance of bacteria (Figs 2, 3 and 4). In the CK group, Bacteroides, Oscillospira, Faecalibacterium, Desulfovibrio, Megamonas, Dorea, Peptococcus, Prevotella, Treponema, Collinsella, Blautia, Parabacteroides and Ruminococcus were the dominant genera, representing 16%, 7.73%, 6.02%, 5.43%, 3.66%, 2.04%, 1.96%, 1.71%, 1.14%, 1.09%, 1.07%, 1 and 1% of the total sequences, respectively. In the CF5 group, Bacteroides, Prevotella, Desulfovibrio, Oscillospira, Phascolarctobacterium, Treponema, Parabacteroides and Faecalibacterium were the dominant genera, representing 18.05%, 16.86%, 7.11%, 6.42%, 2.01%, 1.83%, 1.43% and 1.09% of the total sequences, respectively. In the CF10 group, the most abundant sequences were related to Bacteroides, Desulfovibrio, Prevotella, Oscillospira, Phascolarctobacterium and Treponema, representing 22.94% 8.72% 6.89% 5.56% 1.65% and 1.38% of the sequences, respectively. We also noticed that there were many unclassified and uncultured bacteria in the samples from the CK, CF5 and CF10 groups, representing 57.63%, 55.50% and 52.03% of the total sequences, respectively.
To identify the specific bacterial taxa associated with cassava foliage, we compared the caecum microbiota in CK and CF5, CK and CF10, and CF5 and CF10 using LEfSe. Figure 5 shows a representative cladogram of the structure of the caecum microbiota and their predominant bacteria, showing the greatest differences in taxa between the three groups. The data indicated that two Order (Bacteroidales and Aeromonadales) and three Family (Elusimicrobiaceae, Erysipelotrichaceae and Succinivibrionaceae) belonged to the four dominant phyla (Bacteroidetes, Elusimicrobia, Firmicutes and Proteobacteria) in CK and CF5 (Fig. 5A1). One genus (Coprococcus) and one Family (Elusimicrobiaceae) belonged to the two dominant phyla (Elusimicrobia and Firmicutes) in CK and CF10 groups (Fig. 5A2). One Family (Methanocorpusculaceae) belonged to dominant phyla Euryarchaeota of Archaea in CF5 and CF10 groups (Fig. 5 A3). These different taxa could be used as distinguishing biomarkers. The changes in the caecum microbiota treated with cassava foliage were also explored using the Mann-Whitney U test at different taxon levels. At the phylum level, Bacteroidetes, Firmicutes, Proteobacteria, Synergistete and Spirochaetes were the most predominant phyla in the cassava foliage treated samples.
LEfSe identified the most differentially abundant taxons between CK and CF5 (A1,B1), CK and CF10 (A2,B2), CF5 and CF10 (A3,B3). Taxonomic cladogram obtained from LEfSe analysis of 16 S sequences (relative abundance ≥0.5%). (Red) CF5-enriched taxa, (Green) taxa enriched in CK (B1); (Red) CK-enriched taxa (B2); (Red) CF5-enriched taxa (B3). The brightness of each dot is proportional to its effect size. CK-enriched taxa are indicated with a positive LDA score (green), and taxa enriched in CF5 have a negative score (red) (A1); taxa enriched in CK have a negative score (red) (A2); taxa enriched in CF5 have a negative score (red) (A3). Only taxa meeting an LDA significant threshold >2 are shown.
The effect of cassava foliage on the relative abundance and diversity of microbial communities
Figure 2 reveals a profound change in microbial composition at the phylum and genus levels was induced by different treatments. The treatments of cassava foliage showed a statistically significant effect on the relative abundance of Bacteroidetes (P < 0.05), which was the most abundant phylum in cecal microbiome and increased with the addition of cassava foliage. In contrast, the relative abundance of Firmicutes, the second abundant phylum, was significantly decreased with the addition of cassava foliage (P < 0.05). The same trend was observed in Actinobacteria, the abundance of which was significantly decreased with the addition of cassava foliage (P < 0.05). The relative abundances of Proteobacteria, Synergistetes and Spirochaetes were increased with the addition of cassava foliage. The relative abundance of Proteobacteria in the CF10 group was significantly higher than that of the CK group (P < 0.05), and the relative abundances of Synergistetes and Spirochaetes in the CF5 group were significantly higher compared with the CK group (P < 0.05).
In the present study, we found that the relative abundances of genera were also affected by the addition of cassava foliage. Figure 2 shows the 14 most abundant genera. The relative abundances of Bacteroides, Prevotella, Desulfovibrio, Treponema and Phascolarctobacterium were significantly increased with the addition of cassava foliage. However, the relative abundances of Oscillospira, Faecalibacterium, Megamonas, Dorea, Peptococcus, Collinsella and Ruminococcus were significantly decreased (P < 0.05). The relative abundance of Parabacteroides in the CF5 group was significantly higher compared with the CK and CF10 groups (P < 0.05). However, the relative abundance of Sutterella was not significantly different among all groups. Based on the functionality prediction, we detected a clear difference in the KEGG Orthologs (KO) composition in caecum. There were 104,109 and 87 differential enrichment KEGG pathways between CK and CF5, CK and CF10, CF5 and CF10, respectively. Figure 6 shows the top 20 pathway enrichment of different groups. Cecal microbial pathways were also detected, which were related to amino acid metabolism pathways (e.g. valine, leucine and isoleucine degradation, phenylalanine metabolism, glycine, serine and threonine metabolism, glutathione metabolism, arginine biosynthesis, taurine and hypotaurine metabolism, histidine metabolism, glutathione metabolism, nitrogen metabolism, lysine degradation), sugar and nucleotide sugar metabolism pathways (e.g. pentose phosphate pathway, glycosaminoglycan degradation, ribosome biogenesis in eukaryotes, phosphotransferase system (PTS), glycolysis/gluconeogenesis, galactose metabolism), fatty acid degradation pathways (e.g. fatty acid degradation, butanoate metabolism, degradation of aromatic compounds, glyoxylate and dicarboxylate metabolism), immunity and signal transduction (e.g. cationic antimicrobial peptide (CAMP), ascorbate and aldarate metabolism, African trypanosomiasis, two-component system, inositol phosphate metabolism, carotenoid biosynthesis, seleno compound metabolism), bacterial proliferation and colonization (e.g. bacterial secretion system and bacterial chemotaxis), sulfur metabolism pathways (e.g. sulfur relay system and sulfur metabolism), energy metabolism (methane metabolism), respectively. Notably, the two-component system pathway showed significant difference in the three groups, and the ABC transporter pathway showed significant difference in CF5 and CF10 groups.
(A): CK-VS-CF5; (B): CK-VS-CF10; (C): CF5-VS-CF10. Rich Factor: The ratio of the number of differentially expressed genes and the total number genes which located in the pathway genes. The greater the Factor Rich, the higher the degree of enrichment. P-value closer to 0, the more significant enrichment.
Discussion
Many studies have used high-throughput sequencing to investigate the gut microbial diversity of poultry. These studies on microbiome have shown that Firmicutes, Bacteroidetes and Proteobacteria are numerically the most dominant phyla in the cecal microbiome of chickens20,21,22,23. In this study, for the first time, we identified the microbial diversity of cecal samples from geese using high-throughput sequencing, and then the effect of cassava foliage on such diversity was studied. At the phylum level, Bacteroidetes, Firmicutes, Proteobacteria, Synergistete, Spirochaetes and Actinobacteria were identified as the dominant bacteria in the cecal microbiome of geese. However, Liu et al.14 analyzed the microbial diversity of geese caecum at the class level using 16S rRNA clone library approach, and the cecal microbiome of geese are dominantly occupied by 58.7% Clostridia (Firmicutes), 26.9% Bacteroidetes (Bacteroidetes) and 11.2% Erysipelotrichi (Firmicutes), showing similar results with the cecal microbiome of chickens. Despite the limited number of analyzed sequences, the results provided a valuable insight into a poorly understood microbial ecosystem of goose caecum. Interestingly, Bacteroidetes is found as the most abundant bacteria in the duck caecum, and the dominant phyla high to low are Bacteroidetes, Firmicutes and Proteobacteria24. Similar result has been reported in caecum 16 S rRNA clone library approach of turkeys, and the dominant phyla high to low are Bacteroidetes (54%), Firmicutes (30%), Proteobacteria (3%) and Deferribacteres (3%)25. The microbial diversity of goose caecum was similar with that of ducks and turkeys, but it was different from that of chickens. From the perspective of evolution, geese and ducks are both poultry of Anseriformes, and they have closer relationship than chickens and turkeys. However, from the perspective of feeding, the geese and turkeys have stronger ability of roughage utilization compared with chickens and ducks. Furthermore, geese and ducks live in water. Therefore, the microbial diversity of gut could be affected by all genetic, dietary and environmental conditions.
In the present study, there were evident differences in the microbial composition among different treatments of cassava foliage. The phylum Bacteroidetes was the most abundant bacteria and its abundance was increased with the addition of cassava foliage. The abundance of phylum Bacteroidetes in the CF5 and CF10 groups was significantly higher than that in the CK group, whereas the abundance of Firmicutes in the CF5 and CF10 groups was significantly lower than that in the CK group. The cassava foliage diet contained higher fiber content than the control diet, leading to increased abundance of Bacteroides and decreased abundance of Firmicutes. This result was consistent with the study on intestinal microbiome in ducks, showing increased abundance of Bacteroides and decreased abundance of Firmicutes in the caecum24. The similar dietary effects on intestinal microbiome with a higher abundance of Bacteroidetes and a lower abundance of Firmicutes have been reported in rabbits, goats and humans, and the different nutrient compositions might lead to the different biodiversities16,17,26. In the present study, the abundance of Proteobacteria in the CF5 and CF10 groups was significantly higher than that in the CK group. This finding was consistent with the studies on ducks and goats17,24, but inconsistent with the studies on chickens. Such a discrepancy could be caused by species-specific differences. The phylum Synergistetes, which is known for its ability to degrade amino acids and pyruvate, was one of the most abundant bacteria in our study. To date, no study has reported Synergistetes as the dominant bacteria in the gut microbiome of chickens or ducks. Our finding indicated that Synergistetes was unique to geese and could play a key role in cecal digestion. However, further studies are still necessary to thoroughly understand the effects of cassava foliage on the abundance of these bacteria.
In our study, we investigated the effect of cassava foliage on the population structure at the genus level. Among the different genera, Bacteroides was the most abundant bacteria in goose caecum. Previous studies have shown that the microbial diversity in chicken caecum was mainly dominated by Bacteroides, which was consistent with our study12,23. Bacteroides is thought to play a fundamental role in the breakdown of complex polysaccharides, starch and cellulose into simpler compounds27. The treatments of cassava foliage increased the abundance of Bacteroides. Prevotella was another abundant genus in our study. Previous studies have shown that Prevotella is the most abundant bacterial genus in the rumens of goats and bovine, both of which have strong ability of roughage utilization17,28. Here, we found that the abundance of Prevotella tended to increase with the addition of cassava foliage, which was consistent with previous studies. Additionally, this observation was in agreement with a recent study by Jami et al.29, which shows that the genus Prevotella becomes the dominant bacteria in the bovine rumen with high-fiber diets. Prevotella has important role in the utilization of carbohydrates within the gut microbial ecosystem, and its fermented products are mainly acetic acid, succinic acid, isobutyric acid, isovaleric acid and lactic acid, which can be used by the animal hosts. This might explain why the abundance of Prevotella was changed by treatments of cassava foliage.
Notably, we detected a large number of microbiome in goose caecum, which belonged to unclassified and uncultured genera based on the current 16 S RNA gene sequence database. This finding suggested that geese might possess specific intestinal microbiome, reflecting the fact that few studies of this type of poultry have been previously conducted. Additionally, the relative abundances of unclassified and uncultured bacteria were decreased with the addition of cassava foliage, indicating that the microbiome of geese with more roughage could be better classified compared with the geese with less roughage. Further studies are required to better characterize these unknown bacteria and their special functions in the hosts.
The microbial diversity of geese gut may present many important functions, which are essential to geese life. In this study, we made prediction based on KO. The results indicated that the most abundant functional categories were associated with amino acid metabolism, sugar and nucleotide sugar metabolism, fatty acid degradation, energy metabolism, immunity and signal transduction, bacterial proliferation and colonization. Utilization of amino, sugar and nucleotide sugar is important for geese metabolism and growth. The amino acid metabolism pathway is specifically responsible for breaking down protein to amino acids or peptides. The sugar metabolism pathway and fatty acid degradation pathway are specifically responsible for digesting cellulose or other dietary fiber to volatile fatty acids and absorbing. The nucleotide sugar metabolism is crucial for purine and pyrimidine generation, which is vital substrate for DNA or RNA synthesis. These biochemical processes might be related to the high-energy requirements and high metabolism rate, which are responsible for energy metabolism pathway. The pathways of bacterial proliferation and colonization play crucial roles in gut colonization and host adhesion, infection and biosynthesis of fimbriae, flagella, outer membrane, metabolic and lipopolysaccharides. In the present study, the two-component system pathway showed significant difference in the three groups, which was one of immunity and signal transduction pathways. The two-component system with sensor kinase and response regulator modulates gene expression based on environmental stimulus (e.g. temperature, pH, osmotic level, toxicity and nutrients)2,30. Cassava foliage treatments up-regulated the expressions of genes responsible for two-component system pathway compared with control, and the differentiation may be caused by composition and amount of carbohydrate. In addition, the ABC transporter pathway showed significant difference in two cassava foliage treatments. Major functions of ABC transporters include the transport of lipids, bile salts, toxic compounds, and peptides, which maintain host health and boost the immune31. However, the effects of cassava foliage on the cecal microbial and functions remain unclear, and the actual actions may wait for more efforts.
Taken together, our study, based on 16 S rRNA gene sequencing, reported the overall composition of the microbial ecosystem in the caecum of geese with different diets. Our data revealed that cassava foliage treatment had significant effects on the microbial community in the caecum of geese. Genes related to nutrient and energy metabolism, immunity and signal transduction pathways were significantly expressed by the microbiome. These observations provided a better understanding of how the microbial ecology in the caecum of geese was affected by diet.
Materials and Methods
Experimental design and sampling
All protocols of animal handling and sampling were approved by the Animal Care and Use Committee of Chinese Academy of Tropical Agricultural Sciences (CATAS), and all efforts were made to minimize the suffering of animals according to recommendations proposed by the European Commission (1997). The study was carried out in accordance with the approved protocol. All methods were conducted in accordance with relevant guidelines. A total of 108 male Hainan indigenous geese (28 days old) with similar body weight were randomly divided into three groups with six cages of six geese per group. The geese were fed for 42 days with different diets in this trail, and Table 1 lists the dietary compositions. CK group was fed with control diet, and CF5 or CF10 group was fed with experimental diet supplemented with 5% cassava foliage (CF5) or 10% cassava foliage (CF10), respectively (Table 1). For the consistency of energy and protein levels, we adjusted several components, such as fish meal. After 12-h starvation at age of 70 days old, all the birds were individually weighed, and one bird per cage (with body weight closest to the mean cage weight) was selected and sacrificed. Birds were euthanized by cervical dislocation. Samples were aseptically scrapped from caecum and placed into a sterile glass slides. All samples were immediately stored at −80 °C until further analysis.
DNA extraction and 16S rRNA gene sequencing
Total genomic DNA was extracted from cecal samples using a Stool DNA Kit (OMEGA Bio-Tek, Norcross, GA, USA) according to the manufacturer’s instructions. V3-V4 regions of bacterial 16S rRNA gene (from 341 to 806) were amplified from extracted DNA using barcoded primers 349 F (5′- CCTACGGGNBGCASCAG -3′) and 806 R (5′-GACTACNVGGGTATCTAATCC-3′). PCR was performed in a 20-μL reaction system containing 0.8 μL of each primer, 10 ng template DNA, 4 μL 5 × FastPfu buffer, 2 μL 2.5 mM dNTPs and 0.4 μL FastPfu polymerase, and experiments were conducted in triplicate. Briefly, following an initial denaturation step at 95 °C for 5 min, the amplifications were carried out with 27 cycles at a melting temperature of 95 °C for 30 sec, an annealing temperature of 50 °C for 30 sec, and an extension temperature of 72 °C for 45 sec. Finally, an extra extension step at 72 °C for 10 min was performed. The amplicons were pooled, purified and then quantified using Nanodrop (Thermo Scientific, USA). Subsequently, next-generation sequencing was performed by Illumina Hiseq 2500 PE250, which was conducted by Genedenovo Inc. (Guangzhou, China).
Quality control
Reads filtering: (1) removing reads containing more than 10% of unknown nucleotides (N); (2) removing reads containing less than 80% of bases with quality (Q-value) > 20.
Tag assembling and abundance statistics: The filtered reads were then assembled into tags according to overlap between paired-end reads with more than 10-bp overlap and less than 2% mismatch. The software MOTHUR32 was used to remove the redundant tags to get unique tags. The obtained unique tags were then used to calculate the abundance.
Bioinformatics and statistical analysis
The high-quality sequences were clustered into operational taxonomic units (OTUs) defined at 97% similarity. These OTUs were applied for diversity (Shannon and Simpson), richness (Ace and Chao) and rarefaction curve analyses using MOTHUR32. Taxonomic assignments of OTUs that reached the 97% similarity level were made using (quantitative insights into microbial ecology) QIIME software package through comparison with the SILVA33, Greengene34 and RDP35 databases. A heat map was generated using the heat map function of the R (http://www.r-project.org/) and genus information for the three groups. Linear discriminant analysis (LDA) effect size (LEfSe) method was used to identify the most differentially abundant taxons between groups, which would help discover biomarkers36. The predicted KOs were summarized to functional categories at the genus level. Groups were compared using the Statistical Analysis of Metagenomic Profile package STAMP (http://kiwi.cs.dal.ca/Software/STAMP)37. Statistical analysis was performed using t-tests with the SPSS software (version 19.0 for Windows, SPSS Inc., Chicago, IL, USA). P < 0.05 was considered statistically significant.
Nucleotide sequence accession numbers
Sequences of this project have been deposited into the NCBI nucleotide database under accession number SRA544802.
Additional Information
How to cite this article: Li, M. et al. Cassava foliage affects the microbial diversity of Chinese indigenous geese caecum using 16S rRNA sequencing. Sci. Rep. 7, 45697; doi: 10.1038/srep45697 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
15 June 2017
A correction has been published and is appended to both the HTML and PDF versions of this paper. The error has not been fixed in the paper.
15 June 2017
Scientific Reports 7: Article number: 45697; published online: 06 April 2017; updated: 15 June 2017. The Acknowledgements section in this Article is incomplete. “This study was funded by the Central Public-interest Scientific Institution Basal Research Fund for Chinese Academy of Tropical Agricultural Sciences (No.
References
Sekirov, I., Russell, S. L., Antunes, L. C. et al. Gut microbiota in health and disease. Physiological Reviews 90, 859–904 (2010).
Shaufi, M. A. M., Sieo, C. C., Chong, C. W. et al. Deciphering chicken gut microbial dynamics based on high-throughput 16S rRNA metagenomics analyses. Gut Pathogens 7, 1–12 (2015).
Zhao, L., Wang, G., Siegel, P. et al. Quantitative Genetic Background of the Host Influences Gut Microbiomes in Chickens. Scientific Reports 3, 1970–1970 (2013).
Chung, H. et al. Gut immune maturation depends on colonization with a host specific microbiota. Cell 149, 1578–1593 (2012).
Zhang, C. et al. Structural modulation of gut microbiota in life-long calorierestricted mice. Nat Commun 4, 2163 (2013).
Singh, K. M., Shah, T., Deshpande, S. et al. High through put 16S rRNA gene-based pyrosequencing analysis of the fecal microbiota of high FCR and low FCR broiler growers. Molecular Biology Reports 39, 10595–602 (2012).
Vulevic, J., Juric, A., Tzortzis, G. et al. A mixture of trans-galactooligosaccharides reduces markers of metabolic syndrome and modulates the fecal microbiota and immune function of overweight adults. Journal of Nutrition 143, 324–31 (2013).
Reyes, A., Haynes, M., Hanson, N. et al. Viruses in the faecal microbiota of monozygotic twins and their mothers. Nature 466, 334–338 (2010).
Sekelja, M., Rud, I., Knutsen, S. H. et al. Abrupt temporal fluctuations in the chicken fecal microbiota are explained by its gastrointestinal origin. Applied & Environmental Microbiology 78, 2941–8 (2012).
Wang, Z. Y., Yang, H. M., Lu, J. et al. Influence of whole hulled rice and rice husk feeding on the performance, carcass yield and digestive tract development of geese. Animal Feed Science & Technology 194, 99–105 (2014).
Jin, L., Gao, Y. Y., Hui, Y. E. et al. Effects of Dietary Fiber and Grit on Performance, Gastrointestinal Tract Development, Lipometabolism, and Grit Retention of Goslings. Journal of Integrative Agriculture 13, 2731–2740 (2014).
He, L. W., Meng, Q. X., Li, D. Y. et al. Effect of different fibre sources on performance, carcass characteristics, and gastrointestinal tract development of growing Greylag geese. British Poultry Science 56, 88–93 (2015).
Wang, Z. Y., Shi, S. R., Xu, M. J. & Yang, H. M. 16S rRNA-based analysis of bacterial diversity in the microbial flora of the goose intestinal tract. J. Anim. Feed Sci. 18, 531–540 (2009).
Liu, B. Y. et al. Molecular profiling of bacterial species in the caecum of geese. Czech J. Anim. Sci. 56, 192–203 (2011).
Sergeant, M. J. et al. Extensive microbial and functional diversity within the chicken cecal microbiome. Plos One 9, e91941 (2014).
Bo, Z. et al. The bacterial communities associated with fecal types and body weight of rex rabbits. Sci. Rep. 5, 9342 (2015).
Han, X. et al. Rumen bacterial diversity of 80 to 110-day-old goats using 16S rRNA sequencing. PloS One 10, e0117811 (2015).
Kumar, S., Indugu, N., Vecchiarelli, B. & Pitta, D. W. Associative patterns among anaerobic fungi, methanogenic archaea, and bacterial communities in response to changes in diet and age in the rumen of dairy cows. Front. in Microbiol. 6, 781 (2015).
Borin, K., Lindberg, J. E. & Ogle, R. B. Digestibility and digestive organ development in indigenous and improved chickens and ducks fed diets with increasing inclusion levels of cassava leaf meal. J Anim Physiol A Anim Nutr 90, 230–237 (2006).
Corrigan, A., Horgan, K. P., Clipson, N. & Murphy, R. Effect of dietary supplementation with a Saccharomyces cerevisiae mannan oligosaccharide on the bacterial community structure of broiler cecal contents. Appl. Environ. Microbiol. 77, 6653–6662 (2011).
Luo, Y. H. et al. Broilers fed dietary vitamins harbor higher diversity of cecal bacteria and higher ratio of Clostridium, Faecalibacterium, and Lactobacillus than broilers with no dietary vitamins revealed by 16S rRNA gene clone libraries. Poultry Sci. 92, 2358–2366 (2013).
Oakley, B. B. et al. The chicken gastrointestinal microbiome. Fems Microbiol. Let. 360, 100–112 (2014).
Shaufi, M. A. M., Sieo, C. C., Chong, C. W., Gan, H. M. & Ho, Y. W. Deciphering chicken gut microbial dynamics based on high-throughput 16S rRNA metagenomics analyses. Gut Pathogens 7, 1–12 (2015).
Vasaï, F. et al. Overfeeding and genetics affect the composition of intestinal microbiota in Anas platyrhynchos, (Pekin) and Cairina moschata, (Muscovy) ducks. Fems Microbiol. Ecol. 87, 204–216 (2014).
Scupham, A. J., Patton, T. G., Bent, E. & Bayles, D. O. Comparison of the cecal microbiota of domestic and wild turkeys. Microbial. Ecology 56, 322–331 (2008).
De Filippo, C. et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 107, 14691–14696 (2010).
Lan, P. T., Sakamoto, M., Sakata, S. & Benno, Y. Bacteroides barnesiae sp. nov. Bacteroides salanitronis sp. nov. and Bacteroides gallinarum sp. nov. isolated from chicken caecum. Inter. J. Syst. Evol. Microbiol. 56, 2853–2859 (2006).
Jami, E. & Mizrahi, I. Composition and similarity of bovine rumen microbiota across individual animals. Plos One 7, e33306 (2012).
Jami, E., Israel, A., Kotser, A. & Mizrahi I. Exploring the bovine rumen bacterial community from birth to adulthood. Isme J. 7, 1069–1079 (2013).
Mikkelsen, H., Sivaneson, M. & Filloux, A. Key two-component regulatory systems that control biofilm formation in Pseudomonas aeruginosa. Environmental Microbiology 7, 1666–1681 (2011).
Borst, P. & Elferink, R. O. Mammalian ABC transporters in health and disease. Annual Review of Biochemistry 71, 537–592 (2002).
Schloss, P. D. et al. Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 75, 7537–7541 (2009).
Quast, C. et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res. 41, 590–596 (2013).
Desantis, T. Z. et al. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl. Environ. Microbiol. 72, 5069–5072 (2006).
Cole, J. R. et al. The Ribosomal Database project: improved alignments and new tools for rRNA analysis. Nucleic Acids Res. 37, 141–145 (2009).
Segata, N. et al. Metagenomic biomarker discovery and explanation. Genome Biol. 12, R60 (2011).
Parks, D. H. & Beiko, R. G. Identifying biologically relevant differences between metagenomic communities. Bioinformatics 26, 715–721 (2010).
Acknowledgements
This study was funded by the Central Public-interest Scientific Institution Basal Research Fund for Chinese Academy of Tropical Agricultural Sciences (No.17CXTD-18, 1630032016020), the Special Fund for Agro-Scientific Research in the Public Interest, China (grant no. 201303143) and the China Agriculture Research System (grant no. CARS-12).
Author information
Authors and Affiliations
Contributions
X.Zi, Y.Jiang and H.Zhou conceived and designed the experiments. M.Li and X.Zi executed the experiment and analyzed the samples. M.Li, Z.Zhang, T.Xu, Y.Jiang and X.Pan analyzed the data. M.Li and X.Pan write manuscript. All authors interpreted the data, critically revised the manuscript for important intellectual contents and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Li, M., Zhou, H., Pan, X. et al. Cassava foliage affects the microbial diversity of Chinese indigenous geese caecum using 16S rRNA sequencing. Sci Rep 7, 45697 (2017). https://doi.org/10.1038/srep45697
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep45697
This article is cited by
-
Impact of environmental factors on diversity of fungi in sediments from the Shenzhen River Estuary
Archives of Microbiology (2023)
-
Effects of maize silage feeding on growth performance, carcass characteristics, digestive system length, chemical composition, and meat quality of domestic geese
Tropical Animal Health and Production (2022)
-
Effects of king grass and sugarcane top in the absence or presence of exogenous enzymes on the growth performance and rumen microbiota diversity of goats
Tropical Animal Health and Production (2021)
-
Fungal diversity in deep-sea sediments from Magellan seamounts environment of the western Pacific revealed by high-throughput Illumina sequencing
Journal of Microbiology (2020)
-
Silage fermentation and ruminal degradation of cassava foliage prepared with microbial additive
AMB Express (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.