Abstract
Caspase-1, also known as interleukin-1β (IL-1β)-converting enzyme (ICE), regulates antimicrobial host defense, tissue repair, tumorigenesis, metabolism and membrane biogenesis. On activation within an inflammasome complex, caspase-1 induces pyroptosis and converts pro-IL-1β and pro-IL-18 into their biologically active forms. “ICE−/−” or “Casp1−/−” mice generated using 129 embryonic stem cells carry a 129-associated inactivating passenger mutation on the caspase-11 locus, essentially making them deficient in both caspase-1 and caspase-11. The overlapping and unique functions of caspase-1 and caspase-11 are difficult to unravel without additional genetic tools. Here, we generated caspase-1–deficient mouse (Casp1Null) on the C57BL/6 J background that expressed caspase-11. Casp1Null cells did not release IL-1β and IL-18 in response to NLRC4 activators Salmonella Typhimurium and flagellin, canonical or non-canonical NLRP3 activators LPS and ATP, Escherichia coli, Citrobacter rodentium and transfection of LPS, AIM2 activators Francisella novicida, mouse cytomegalovirus and DNA, and the infectious agents Listeria monocytogenes and Aspergillus fumigatus. We further demonstrated that caspase-1 and caspase-11 differentially contributed to the host defense against A. fumigatus infection and to endotoxemia.
Similar content being viewed by others
Introduction
Inflammatory caspases, include caspase-1 (human and mouse), caspase-4 (human), caspase-5 (human) and caspase-11 (mouse), contribute to a variety of biological functions1,2. Caspase-1, also known as interleukin-1β (IL-1β)-converting enzyme or ICE2,3,4,5, can directly cleave the precursor cytokines pro-IL-1β and pro-IL-18 at the sites Asp116–Ala1173,4,6,7 and Asp35–Asn365,8,9,10, respectively, generating a biologically active form of the cytokines for release by the cell. Characterization of the purified caspase-1 protein in 1992 revealed a heterodimeric cysteine protease composed of two subunits, p10 and p2011.
Caspase-1 is activated within an inflammasome complex12, a macromolecular protein complex formed in the cytoplasm of a cell on recognition of pathogen-associated molecular patterns and danger-associated molecular patterns by a NOD, LRR-containing protein (NLR), AIM2-like receptor (ALR) or pyrin13. In 1995, two independent groups each generated a mouse line lacking the gene encoding caspase-1, called “ICE−/−” or “Casp1−/−”, using embryonic stem cells obtained from the 129 mouse strain14,15. A later study revealed that these mouse lines lack caspase-11 expression due to a 129-associated passenger mutation on the caspase-11 locus that potentially mediated rapid decay of the caspase-11 mRNA16. The close proximity of the caspase-1 and caspase-11 loci prevented their segregation despite extensive backcrossing to the C57BL/6 background16. We will follow the existing convention of the published literature and call previously-generated “ICE−/−” and “Casp1−/−” strains as Casp1−/−Casp11−/− (also known as Casp1−/−Casp11129mt/129mt) mice16. The biological insights into caspase-1 gained from the use of Casp1−/−Casp11−/− mouse strains should be revisited.
Here, we generated caspase-1–deficient mouse strain on the C57BL/6 J background, referred to as Casp1Null, to overcome the confounding loss of caspase-11 in existing mouse strains. Bone marrow-derived macrophages (BMDMs) or dendritic cells (BMDCs) from Casp1Null mice expressed caspase-11 proteins and did not secrete IL-1β and IL-18 or undergo pyroptosis in response to canonical NLRP3, NLRC4 or AIM2 inflammasome activators. In addition, Casp1Null BMDMs failed to secrete IL-1β and IL-18, but underwent caspase-11–dependent pyroptosis, in response to non-canonical activation of the inflammasome. Further, both caspase-1 and caspase-11 contributed to the host defense against infection with A. fumigatus, whereas caspase-11 predominantly contributed to lethality during LPS-induced endotoxemia. This caspase-1–deficient mouse strain provides the scientific community with an exciting opportunity to refine the roles of caspase-1 and caspase-11 in health and disease.
Results
The Casp1Null mouse line expresses caspase-11
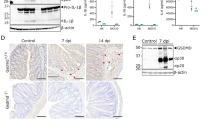
Previously generated caspase-1-deficient mouse strains using embryonic stem cells from the 129 strain lack caspase-11 expression16. To overcome this confounding factor, we used CRISPR-Cas9 technology and C57BL/6 J embryonic stem cells to generate a caspase-1–deficient mouse strain, which we designate “Casp1Null” mice (Supplementary Fig. 1). We generated primary bone marrow-derived macrophages (BMDMs) from Casp1Null mice and assessed the expression of caspase-11 proteins following stimulation with IFN-β, IFN-γ or LPS. We found inducible expression of caspase-11 in WT and Casp1Null BMDMs stimulated with IFN-β, IFN-γ or LPS, whereas Casp11−/− BMDMs, as expected, did not (Fig. 1). We also confirmed that caspase-1 expression was absent in Casp1Null BMDMs and intact in WT and Casp11−/− BMDMs (Fig. 1).
Inflammasome activities are impaired in Casp1Null BMDMs in response to canonical inflammasome activation
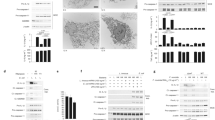
Caspase-1 is activated within an inflammasome following engagement of the inflammasome-initiating sensors NAIP–NLRC4, NLRP3 and AIM213. To systematically validate the Casp1Null line, we measured inflammasome responses from primary BMDMs generated from this line following stimulation with known inflammasome triggers. The NAIP–NLRC4 inflammasome can be activated by Salmonella enterica serovar Typhimurium (S. Typhimurium) which had been grown to a log-phase or through transfection of bacterial flagellin into the host cytoplasm17,18,19,20,21,22. Activation of the NLRC4 inflammasome using S. Typhimurium or transfection of flagellin from S. Typhimurium led to robust activation of caspase-1, release of IL-1β and/or IL-18, and cell death in WT and Casp11−/− BMDMs, but not in Casp1Null and Casp1−/−Casp11−/− BMDMs (Fig. 2A and B).
(A) Top, immunoblot analysis of pro-caspase-1 (Pro-Casp-1) and the caspase-1 subunit p20 (Casp-1 p20) in unprimed WT or mutant BMDMs left untreated (medium alone [Med]) or assessed 4 h after infection with Salmonella Typhimurium (MOI, 1; left) or 4 h after transfection of S Typhimurium flagellin (4 μg/ml; middle) or in LPS-primed BMDMs left untreated (Med) or assessed 30 min after stimulation with 5 mM ATP (LPS + ATP, right). Bottom, immunoblot analysis of pro-caspase-1 (Pro-Casp-1) and the caspase-1 subunit p20 (Casp-1 p20) in unprimed WT or mutant BMDMs left uninfected (medium alone [Med]) or assessed 20 h after infection with F. novicida (MOI, 100; left) or 5 h after transfection with poly(dA:dT) (5 μg/ml; middle) or 10 h after infection with mouse cytomegalovirus (MCMV, MOI, 10; right). (B–D) Release of IL-1β, IL-18, death of BMDMs, and release of TNF after treatment as in (A). Cell death indicates % of LDH release relative to total lysis, set at 100% (B–D). Data in (A–D) are representative of three independent experiments (mean and s.e.m. of values from three independent experiments in B–D).
The canonical NLRP3 inflammasome responds to a variety of activators, including ATP, nigericin, bacterial toxin, RNA and crystalline substances23,24,25,26. We found that while WT BMDMs secreted IL-1β and IL-18 and underwent cell death in response to LPS and ATP, Casp1Null and Casp1−/−Casp11−/− BMDMs failed to do so (Fig. 2A and B).
Activation of the DNA sensor AIM2 by infection with the Gram-negative bacterium Francisella novicida, transfection of dsDNA ligand poly(dA:dT) into the cytoplasm or infection by the DNA virus mouse cytomegalovirus (MCMV) leads to assembly of the inflammasome27,28,29,30,31,32,33,34. Engagement of AIM2 using these microbial and synthetic agents induced maturation of caspase-1, release of IL-1β or IL-18, and cell death in both WT and Casp11−/− BMDMs, whereas impaired secretion of IL-1β or IL-18 and cell death were observed in Casp1Null and Casp1−/−Casp11−/− BMDMs (Fig. 2A and B). Secretion of the inflammasome-independent cytokine TNF was normal in BMDMs of all genotypes (Fig. 2B).
The Gram-positive bacterium Listeria monocytogenes activates caspase-1 via multiple inflammasome-initiating sensors, including the AIM2, NLRP3 and NLRC425,29,35,36,37,38,39,40. We found that Casp1Null and Casp1−/−Casp11−/− BMDMs failed to secrete IL-1β or IL-18 and undergo cell death (Supplementary Fig. 2A and B). However, we found no role for caspase-11 in the activation of the inflammasome induced by the L. monocytogenes (Supplementary Fig. 2A and B)41, consistent with the notion that caspase-11 mediates recognition of LPS from Gram-negative bacteria16,42,43,44. We have also recently shown that Casp1Null mice, similar to Casp1−/−Casp11−/− mice45,46,47,48, are more susceptible to infection with F. novicida than WT mice, confirming the biological importance of caspase-1 in a mouse model of bacterial infection49.
Secretion of IL-1β and IL-18, but not pyroptosis, is impaired in Casp1Null BMDMs in response to non-canonical inflammasome activation
Gram-negative bacteria, including Escherichia coli and Citrobacter rodentium, introduce LPS into the host cytoplasm during infection and engage non-canonical activation of the NLRP3 inflammasome via caspase-1116. Casp1Null BMDMs did not secrete IL-1β and IL-18 in response to infection by E. coli and C. rodentium or transfection of LPS (Fig. 3A and B). However, cell death was observed in Casp1Null BMDMs in response to infection by E. coli and C. rodentium or transfection of LPS (Fig. 3B), which is consistent with the model that, in response to non-canonical activation of the NLRP3 inflammasome, pyroptosis is driven by caspase-11 rather than caspase-116,42,44,50.
(A) Immunoblot analysis of pro-caspase-1 (Pro-Casp-1) and the caspase-1 subunit p20 (Casp-1 p20) in unprimed WT or mutant BMDMs left untreated (medium alone [Med]) or assessed 20 h after infection with C. rodentium (MOI, 20, left), E. coli (MOI, 20, middle), or 10 h after LPS transfection (4 μg/ml, right). (B) Release of IL-1β, IL-18, death of BMDMs, and release of TNF after treatment as in (A). (C) Survival of 8-week-old WT and mutant mice injected intraperitoneally with 54 mg LPS per kg body weight. NS, not statistically significant, **P < 0.01 and ****P < 0.0001 (log-rank test). Data are representative of two (C) or three independent experiments (A and B; mean and s.e.m. are representative of values from three independent experiments in B).
Previous studies have demonstrated that Casp1−/−Casp11−/− mice are resistant to acute LPS endotoxemia15,16,51, suggesting that caspase-1 and/or caspase-11 mediate LPS-induced lethality. Further studies have shown that Casp11−/− mice are resistant to acute LPS endotoxemia42,44,52,53, arguing that caspase-11 is a main driver of LPS-induced lethality. To investigate the function of caspase-1 in this model, Kayagaki and colleagues microinjected a bacterial artificial chromosome transgene encoding caspase-11 into Casp1−/−Casp11−/− mouse embryos, re-establishing caspase-11 expression in this mouse line (referred to as the Casp1−/−Casp11Tg mouse strain)16. Using this mouse strain, they reported that Casp1−/−Casp11Tg mice were modestly resistant to LPS-induced lethality compared with WT mice16.
To investigate the contribution of caspase-1 in LPS endotoxemia in Casp1Null mouse strain, we injected LPS into WT, Casp1Null, Casp1−/−Casp11−/− and Casp11−/− mice and monitored their survival. We found that 86% of WT (12/14) and 93% Casp1Null (13/14) mice succumbed to LPS-induced endotoxemia (not statistically significant, P = 0.031) (Fig. 3C). Casp1Null mice succumbed with a slightly delayed kinetic compared with WT mice, consistent with previous observations16. In contrast, only 35% of the Casp11−/− mice (6/17, P < 0.0001 compared with WT) and 14% Casp1−/−Casp11−/− (1/7, P = 0.0014 compared with WT) mice succumbed to endotoxemia (Fig. 3C). These data suggested a dominant role of caspase-11 and a minor role of caspase-1 in mediating acute lethal endotoxemia. Taken together, we have validated our Casp1Null mice for use in the study of inflammasome biology.
Differential roles of caspase-1 and caspase-11 in response to infection with the fungal pathogen Aspergillus fumigatus
In addition to its function in the recognition of bacteria and viruses, inflammasomes have a central role in the control of fungal pathogens, including Aspergillus fumigatus54. We found that A. fumigatus failed to induce the release of IL-1β and IL-18 in Casp1Null and Casp1−/−Casp11−/− bone marrow-derived dendritic cells (BMDCs), whereas maturation of caspase-1 and the release of IL-1β and IL-18 in WT and Casp11−/− BMDCs were observed (Fig. 4A and B). This finding supported our previous observations showing that caspase-11 is dispensable for activation of the inflammasome induced by A. fumigatus infection55. Similar to BMDMs, Casp1Null BMDCs retained the ability to express the caspase-11 protein (Fig. 4A).
(A) Immunoblot analysis of pro-caspase-1 (Pro-Casp-1) and the caspase-1 subunit p20 (Casp-1 p20) and GAPDH (loading control) in unprimed WT or mutant bone marrow-derived dendritic cells left untreated (medium alone [Med]) or assessed 20 h after infection with A. fumigatus (MOI, 10). (B) Release of IL-1β and IL-18 after treatment as in (A). (C) Survival of 8-week-old WT and mutant mice infected with 5 × 105 A. fumigatus conidia after immunosuppression with cyclophosphamide and cortisone acetate. *P < 0.05, ****P < 0.0001 (log-rank test). Data are representative of two (C) or three independent experiments (A and B; mean and s.e.m. are representative of values from three independent experiments in B).
We have previously found that Casp1−/−Casp11−/− mice were extremely sensitive to infection by A. fumigatus compared with WT mice55. However, whether caspase-1 or caspase-11 contributed to the host defense against A. fumigatus infection in vivo has remained unclear. To investigate this, we immunocompromised WT, Casp1Null, Casp1−/−Casp11−/− and Casp11−/− mice with cyclophosphamide and cortisone acetate and intranasally infected these mice with A. fumigatus conidia. Immunosuppression procedures were used because immunocompetent WT mice and mice lacking components of the inflammasome do not succumb to infection with A. fumigatus55, which is in line with the observation that only immunocompromised individuals in large are susceptible to invasive pulmonary aspergillosis56.
Following intranasal infection with A. fumigatus conidia, Casp1Null mice were substantially more susceptible to A. fumigatus–induced mortality compared with WT mice (Fig. 4C). The hypersusceptibility of Casp1Null mice to A. fumigatus was phenocopied by Casp1−/−Casp11−/− mice (Fig. 4C). In addition, we found that Casp11−/− mice were also more susceptible to infection with A. fumigatus conidia compared with WT mice (Fig. 4C). However, Casp11−/− mice succumbed to infection with a delayed kinetic compared with Casp1Null mice or Casp1−/−Casp11−/− mice. Although caspase-11 had no role in the activation of the inflammasome in BMDCs in response to A. fumigatus, caspase-11 contributed to the host defense against A. fumigatus infection in vivo. It is possible that activation of caspase-11 might induce pyroptosis and/or actin-mediated phagosomal killing in a cell-type-specific manner in order to control A. fumigatus dissemination in vivo57,58,59. Indeed, the release of IL-18 via A. fumigatus-sensing NLRP3 and AIM2 inflammasomes induces production of IFN-γ, which might provide a priming signal for caspase-11 to clear A. fumigatus in vivo55. This IL-18–IFN-γ–Caspase-11 signaling pathway and defense strategy has been reported in the host clearance of the cytosolic bacterium Burkholderia thailandensis60. Overall, our study has generated and validated a valuable genetic tool to enable us to refine the differential contribution of caspase-1 and capsase-11 in health and disease in future studies.
Discussion
Inflammatory caspases are multi-functional proteins which mediate host defense to infectious diseases and regulate tumor development, metabolic syndromes, autoinflammatory disease, tissue repair, and cell survival1,2. Previously generated caspase-1–deficient mouse strains using embryonic stem cells of the 129 background lack caspase-11 expression, essentially rendering them deficient in both caspase-1 and caspase-1116. Therefore, the biological insights of caspase-1 gained from using Casp1−/−Casp11−/− mice should be re-examined. We generated caspase-1–deficient mouse strain on the C57BL/6 J background to provide the scientific community a genetic tool to revisit the biological functions of caspase-1 and caspase-11.
Studies into the molecular mechanisms regulating caspase-1 and caspase-11 have revealed important differences between these proteases. Mouse caspase-1 shares 46% amino acid sequence identity with mouse caspase-1161. Caspase-1 is unequivocally required for the proteolytic processing of pro-IL-1β and pro-IL-18 and for pyroptosis in response to canonical inflammasome activators2,3,4,5,8,9,10. Caspase-11 cannot directly proteolytically process pro-IL-1β and pro-IL-1861, although previous studies suggest that one of the caspase-11 homologs in humans, caspase-4, could cleave pro-IL-1β and pro-IL-1862,63. Caspase-11 itself is capable of driving pyroptosis in a caspase-1–independent manner in response to non-canonical activation of the inflammasome, that is, in response to transfection of LPS or during infection by certain Gram-negative bacteria, including C. rodentium, E. coli and Vibrio cholerae16. In this case, expression of caspase-11 is mediated by interferon signaling following TLR4–dependent recognition of extracellular LPS from most Gram-negative bacteria64,65,66,67,68. Caspase-11 recognizes LPS introduced into the host cytoplasm16,42,43,44, which induces caspase-11–dependent cleavage of the pro-pyroptotic factor gasdermin D69,70,71. The N-terminal fragment of gasdermin D mediates pore formation on the cell membrane that leads to pyroptosis and activation of caspase-1 via the NLRP3 inflammasome69,70,71,72,73,74,75,76. However, direct interaction between caspase-11 and caspase-1 resulting in the activation of caspase-1 has also been reported52,61,77. A more recent study also demonstrated that caspase-11 can be activated by host-derived oxidized phospholipids in dendritic cells to drive IL-1β release without pyroptosis78. In our study, the residual cell death observed in Casp1Null BMDMs in response to F. novicida or Casp11−/− BMDMs in response to C. rodentium might indicate a minor contribution from inflammasome-independent cell death pathways. F. novicida can engage apoptosis via a caspase-1-independent, caspase-8-dependent mechanism79,80,81. C. rodentium or its relative enteropathogenic E. coli and enterohemorrhagic E. coli encode the effector proteins NleB82,83 and NleH84 to actively suppress apoptosis and/or necroptosis. These data suggest that downregulation of NleB and NleH over the course of infection by these enteropathogens might engage cell death pathways other than pyroptosis.
Studies in mouse models have revealed differential contributions between caspase-1 and caspase-11 in infection and cancer. Casp1−/−Casp11−/− mice are hypersusceptible to infection by S. Typhimurium 18,65,85,86. A further study has found that the caspase-11-expressing mouse strain Casp1−/−Casp11Tg (generated via microinjection of a bacterial artificial chromosome transgene encoding caspase-11 into Casp1−/−Casp11−/− mouse embryos) revealed that they were more susceptible to infection with S. Typhimurium than Casp1−/−Casp11−/− mice65. However, this study reported that Casp11−/− mice were not more susceptible compared with wild-type mice65. These data would suggest that caspase-11 is detrimental to the host only in the absence of caspase-1. However, others have reported a protective role for caspase-11 during salmonellosis exclusively in the intestine63,87. In mouse macrophages or embryonic fibroblasts, both caspase-1 and caspase-11 contribute to cell-autonomous control of intracellular replication of S. Typhimurium and other bacteria21,58,88,89,90.
In the context of intestinal inflammation, Casp1−/−Casp11−/− mice are susceptible to colitis induced by the colitogen dextran sulfate sodium (DSS)91,92,93. More recent studies have now revealed that mice lacking caspase-11 alone are sensitive to DSS-induced colitis94,95,96. The susceptibility observed in Casp11−/− mice largely phenocopies that of Casp1−/−Casp11−/− mice94, suggesting that caspase-11 could be a dominant inflammatory caspase that drives a protective response in the intestine. This protective response in the intestine might be attributed to the ability of caspase-11 to mediate secretion of IL-1895,96, a cytokine generally considered to be protective in colitis. Caspase-1 could have non-redundant functions with caspase-11 in the intestine and that further studies are required to investigate their relative effect during intestinal inflammation.
The caspase-11 homologs in humans, caspase-4 and caspase-5, also recognize LPS and activate caspase-1, mediate cleavage of gasdermin D, and drive pyroptosis43,69,70. However, subtle differences have been reported between human caspase-4 and human caspase-5. Caspase-4, but not caspase-5, is required for cell death and IL-1β production in the human monocytic THP-1 cell line following transfection of LPS97,98. Caspase-4 also mediates secretion of IL-1β and IL-18 in LPS-stimulated mouse BMDMs engineered to express human caspase-499. However, other studies have suggested that both caspase-4 and caspase-5 are required for IL-1β release in human monocytes stimulated with LPS or in the THP-1 cell line infected with S. Typhimurium97,100, suggesting non-redundant activities between these caspases. A further study has demonstrated that caspase-4 is not necessary for IL-1β release in primary human macrophages infected with S. Typhimurium, L. pneumophila and Y. pseudotuberculosis101. The function of these inflammatory caspases is likely to be cell-type- and species-specific and influenced by the type of activators delivered into the cell.
Overall, our study has generated and validated a mouse strain for use in unraveling the specific function of caspase-1 without the confounding absence of caspase-11. We also showed the unique and overlapping functions of caspase-1 and caspase-11 during infection and inflammation. Two recent studies have now reported caspase-1-deficient mice on the C57BL/6 background102,103. Together with our study, the availability of caspase-1-deficient mice provides a valuable tool for the scientific community to propel investigations that aim to refine the differential functions of caspase-1 and caspase-11 in health and disease.
Methods
Mice
Casp1−/−Casp11−/− (also known as Casp1−/−Casp11129mt/129mt) and Casp11−/− mice have been described previously16. WT C57BL/6 J mice were purchased from the Jackson Laboratory and bred at St. Jude Children’s Research Hospital. Animal studies were conducted under protocols approved by the St. Jude Children’s Research Hospital on the Use and Care of Animals.
Generation of Casp1Null mice
Pronuclear-stage C57BL/6 J zygotes were injected with 2 single guide RNAs (sgRNAs) (Casp1-Guide-01: ATTCTTGACGTCTTAATCTC [125 ng/μL] and Casp1-Guide-02: TTGGGACATTGCAACGAACT [125 ng/μL]) designed to introduce DNA double strand breaks into intron 1 and intron 4 of the Casp1 gene, and a human codon optimized Cas9 mRNA transcript (50 ng/μL) (Fig. S1), and were subsequently surgically transplanted into the oviducts of pseudo pregnant CD1 females. Newborn mice bearing a null allele of Casp1 (Casp1Null) were identified by amplification of a 716 bp fragment using primers flanking the 2 break sites [Casp1-F51 and Casp1-R32 (Tables S1 and S2)]. Sanger sequencing of the 716 bp amplicon confirmed proper deletion of the ~3.8 kb fragment containing exons 2–4. sgRNAs were designed and generated as described previously104. The Cas9 mRNA transcript was generated as described previously104. Potential off-target sites were identified using Cas-OFFinder and each locus was PCR-amplified and sequenced (Table S3)105. No off-target site cleavage was observed.
PCR genotyping
Genotyping of the Casp1 locus was performed using primers flanking each sgRNA target site using the following primer pairs (Fig. S1, and Tables S1 and S2): Casp1-F51 and Casp1-R52 (5′ target site), Casp1-F31 and Casp1-R32 (3′ target site), and Casp1-F51 and Casp1-R32 (Casp1 allele).
Microbial culture
S. Typhimurium SL1344, Citrobacter rodentium (51459, American Type Culture Collection) and Escherichia coli (11775, American Type Culture Collection) were inoculated into Luria-Bertani media (3002–031, MP Biomedicals) and incubated under aerobic conditions overnight at 37 °C. S. Typhimurium SL1344 was subcultured (1:10) into fresh LB media for 3 h at 37 °C to generate log-phase grown bacteria. F. novicida strain U112 were grown in BBL™ Trypticase™ Soy Broth (TSB) (211768, BD) supplemented with 0.2% L-cysteine (BP376-100, ThermoFisher Scientific) under aerobic conditions overnight at 37 °C. F. novicida was subcultured (1:10) in fresh TSB supplemented with 0.2% L-cysteine for 4 h and resuspended in PBS. L. monocytogenes was cultured in brain heart infusion broth (211059, BD) overnight. Aspergillus fumigatus CBS144-89 was grown on 2% (wt/vol) malt− 2% (wt/vol) agar slants for 1 week at room temperature, and conidia were harvested in water containing 0.05% (vol/vol) Tween 80106.
Cultivation and stimulation of bone marrow-derived macrophages and dendritic cells
BMDMs and BMDCs were cultured as described previously46,55. In brief, BMDMs were generated from mouse bone marrow cells grown after 5–6 days in DMEM (11995073, ThermoFisher Scientific) supplemented with 10% FBS (TMS-013-B, Millipore), 30% L929 conditioned media and 1% penicillin and streptomycin (15070-063, ThermoFisher Scientific). BMDMs were seeded in antibiotic–free media at a concentration of 1 × 106 cells onto 12-well plates and incubated overnight. BMDCs were generated from mouse bone marrow cells grown over 7 days in RPMI 1640 (10-040-CV, Corning) supplemented with 10% FBS, 1% penicillin-streptomycin, 1% non-essential amino acid (11140, ThermoFisher Scientific), 1% sodium pyruvate (11360, ThermoFisher Scientific), and 20 ng/ml GM-CSF.
The following conditions were used to stimulate BMDMs: F. novicida (MOI 100 and 20 h for caspase-1 activation), S. Typhimurium (MOI 1, 4 h), C. rodentium (MOI 20 for 20 h), E. coli (MOI 20 for 20 h) and L. monocytogenes (MOI 20, 8 h). 50 μg/ml gentamicin (15750-060, ThermoFisher Scientific) was added after 2 h (S. Typhimurium), 4 h (C. rodentium, E. coli and L. monocytogenes), and 8 h (F. novicida) post-infection to kill extracellular bacteria. To activate the canonical NLRP3 inflammasome, BMDMs were primed using 500 ng/ml ultrapure LPS from Salmonella minnesota R595 (tlrl-smlps, InvivoGen) for 4 h and stimulated with 5 mM ATP (10127531001, Roche) for 45 min. The MCMV Smith MSGV strain (VR-1399™, American Type Culture Collection) was obtained from P.G. Thomas (St. Jude Children’s Research Hospital). MCMV was added to unprimed BMDMs at an MOI of 10 for 10 h. For DNA transfection, 2.5 μg of poly(dA:dT) (tlrl-patn, InvivoGen) were resuspended in PBS and mixed with 0.3 μl of Xfect polymer in Xfect reaction buffer (631318, Clontech Laboratories, Inc.) per reaction. After 10 min, 50 μl of the DNA–Xfect complex was added to BMDMs in 500 μl Opti-MEM (31985-070, ThermoFisher Scientific) and incubated for 5 h. For LPS transfection, 2 μg of ultrapure LPS from Salmonella minnesota R595 (tlrl-smlps, InvivoGen) was resuspended in PBS and mixed with 0.3 μl of Xfect polymer in Xfect reaction buffer per reaction. After 10 min, 50 μl of the LPS–Xfect complex was added to BMDMs in 500 μl Opti-MEM and incubated for 6 h. For flagellin transfection, 2 μg of ultrapure flagellin from S. Typhimurium (tlrl-epstfla-5, InvivoGen) was resuspended in PBS and mixed with 20 μl of DOTAP (D1163, Sigma) per reaction. The reaction mixture was incubated for 20 min and added to BMDMs in 500 μl Hank’s Balanced Salt Solution (SH30031.02, HyClone, GE Healthcare Life Sciences). BMDCs were infected with A. fumigatus conidia (MOI, 10) for 20 h.
Lactate dehydrogenase assay
Levels of lactate dehydrogenase released by cells were determined using the CytoTox 96 Non-Radioactive Cytotoxicity Assay according to the manufacturer’s instructions (G1780, Promega). Cell culture supernatants were collected for ELISA.
Immunoblotting analysis
Cells and supernatant were lysed in RIPA buffer and sample loading buffer containing SDS and 100 mM DTT. Proteins were separated on 8–12% polyacrylamide gels. Following electrophoretic transfer of protein onto PVDF membranes (IPVH00010, Millipore), membranes were blocked in 5% skim milk and incubated with primary antibodies against caspase-1 (1:3,000 dilution, AG-20B-0042, Adipogen), caspase-11 (1:1,000 dilution, NB120-10454, Novus) and GAPDH (1:10,000 dilution, #5174, Cell Signaling Technologies). Membranes were then incubated with HRP-conjugated secondary antibody for 1 h and proteins were visualized using Super Signal Femto substrate (34096, ThermoFisher Scientific).
Cytokine analysis
Cytokine levels were determined using a multiplex ELISA (MCYTOMAG-70K, Millipore) or IL-18 ELISA (BMS618/3TEN, Affymetrix eBioscience) according to the manufacturers’ instructions.
LPS-induced endotoxemia
Male or female mice were injected intraperitoneally with 54 mg per kg body weight of LPS (L2630, Sigma) and monitored throughout the day for 5 days.
A. fumigatus infection in vivo
Cyclophosphamide monohydrate (C0768, Sigma) was dissolved in sterile PBS and given by intraperitoneal injection (150 mg per kg of body weight). Cortisone 21-acetate (C3130, Sigma) was suspended in 0.05% Tween 80 in PBS and administered by subcutaneous injection (112 mg per kg of body weight). Mice were given a combination of cyclophosphamide and cortisone acetate 2 day prior to infection and the day of infection. Mice were anesthetized by isoflurane inhalation and inoculated intranasally with 5 × 105 conidia from A. fumigatus strain CBS144.89 in 30 μl of 0.05% Tween 80 in PBS.
Statistical analysis
GraphPad Prism 6.0 software was used for data analysis. Data are shown as mean ± s.e.m. Statistical significance was determined by a log-rank test. P < 0.05 was considered statistically significant.
Additional Information
How to cite this article: Ming Man, S. et al. Differential roles of caspase-1 and caspase-11 in infection and inflammation. Sci. Rep. 7, 45126; doi: 10.1038/srep45126 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Martinon, F. & Tschopp, J. Inflammatory caspases: linking an intracellular innate immune system to autoinflammatory diseases. Cell 117, 561–574 (2004).
Man, S. M. & Kanneganti, T. D. Converging roles of caspases in inflammasome activation, cell death and innate immunity. Nature reviews. Immunology 16, 7–21 (2016).
Kostura, M. J. et al. Identification of a monocyte specific pre-interleukin 1 beta convertase activity. Proceedings of the National Academy of Sciences of the United States of America 86, 5227–5231 (1989).
Black, R. A., Kronheim, S. R. & Sleath, P. R. Activation of interleukin-1 beta by a co-induced protease. FEBS letters 247, 386–390 (1989).
Ghayur, T. et al. Caspase-1 processes IFN-gamma-inducing factor and regulates LPS-induced IFN-gamma production. Nature 386, 619–623 (1997).
Auron, P. E. et al. Nucleotide sequence of human monocyte interleukin 1 precursor cDNA. Proceedings of the National Academy of Sciences of the United States of America 81, 7907–7911 (1984).
March, C. J. et al. Cloning, sequence and expression of two distinct human interleukin-1 complementary DNAs. Nature 315, 641–647 (1985).
Ushio, S. et al. Cloning of the cDNA for human IFN-gamma-inducing factor, expression in Escherichia coli, and studies on the biologic activities of the protein. Journal of immunology 156, 4274–4279 (1996).
Okamura, H. et al. Cloning of a new cytokine that induces IFN-gamma production by T cells. Nature 378, 88–91 (1995).
Gu, Y. et al. Activation of interferon-gamma inducing factor mediated by interleukin-1 beta converting enzyme. Science 275, 206–209 (1997).
Thornberry, N. A. et al. A novel heterodimeric cysteine protease is required for interleukin-1 beta processing in monocytes. Nature 356, 768–774 (1992).
Martinon, F., Burns, K. & Tschopp, J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Molecular cell 10, 417–426 (2002).
Man, S. M. & Kanneganti, T. D. Regulation of inflammasome activation. Immunological reviews 265, 6–21 (2015).
Kuida, K. et al. Altered cytokine export and apoptosis in mice deficient in interleukin-1 beta converting enzyme. Science 267, 2000–2003 (1995).
Li, P. et al. Mice deficient in IL-1 beta-converting enzyme are defective in production of mature IL-1 beta and resistant to endotoxic shock. Cell 80, 401–411 (1995).
Kayagaki, N. et al. Non-canonical inflammasome activation targets caspase-11. Nature 479, 117–121 (2011).
Broz, P. et al. Redundant roles for inflammasome receptors NLRP3 and NLRC4 in host defense against Salmonella . The Journal of experimental medicine 207, 1745–1755 (2010).
Man, S. M. et al. Inflammasome activation causes dual recruitment of NLRC4 and NLRP3 to the same macromolecular complex. Proceedings of the National Academy of Sciences of the United States of America 111, 7403–7408 (2014).
Miao, E. A. et al. Cytoplasmic flagellin activates caspase-1 and secretion of interleukin 1beta via Ipaf. Nature immunology 7, 569–575 (2006).
Franchi, L. et al. Cytosolic flagellin requires Ipaf for activation of caspase-1 and interleukin 1beta in salmonella-infected macrophages. Nature immunology 7, 576–582 (2006).
Amer, A. et al. Regulation of Legionella phagosome maturation and infection through flagellin and host Ipaf. The Journal of biological chemistry 281, 35217–35223 (2006).
Mariathasan, S. et al. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 430, 213–218 (2004).
Mariathasan, S. et al. Cryopyrin activates the inflammasome in response to toxins and ATP. Nature 440, 228–232 (2006).
Kanneganti, T. D. et al. Critical role for Cryopyrin/Nalp3 in activation of caspase-1 in response to viral infection and double-stranded RNA. The Journal of biological chemistry 281, 36560–36568 (2006).
Kanneganti, T. D. et al. Bacterial RNA and small antiviral compounds activate caspase-1 through cryopyrin/Nalp3. Nature 440, 233–236 (2006).
Martinon, F., Petrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006).
Man, S. M., Karki, R. & Kanneganti, T. D. AIM2 inflammasome in infection, cancer, and autoimmunity: Role in DNA sensing, inflammation, and innate immunity. European journal of immunology 46, 269–280 (2016).
Man, S. M., Karki, R. & Kanneganti, T. D. DNA-sensing inflammasomes: regulation of bacterial host defense and the gut microbiota. Pathogens and disease 74, ftw028(2016).
Rathinam, V. A. et al. The AIM2 inflammasome is essential for host defense against cytosolic bacteria and DNA viruses. Nature immunology 11, 395–402 (2010).
Fernandes-Alnemri, T., Yu, J. W., Datta, P., Wu, J. & Alnemri, E. S. AIM2 activates the inflammasome and cell death in response to cytoplasmic DNA. Nature 458, 509–513 (2009).
Fernandes-Alnemri, T. et al. The AIM2 inflammasome is critical for innate immunity to Francisella tularensis . Nature immunology 11, 385–393 (2010).
Hornung, V. et al. AIM2 recognizes cytosolic dsDNA and forms a caspase-1-activating inflammasome with ASC. Nature 458, 514–518 (2009).
Roberts, T. L. et al. HIN-200 proteins regulate caspase activation in response to foreign cytoplasmic DNA. Science 323, 1057–1060 (2009).
Burckstummer, T. et al. An orthogonal proteomic-genomic screen identifies AIM2 as a cytoplasmic DNA sensor for the inflammasome. Nature immunology 10, 266–272 (2009).
Wu, J., Fernandes-Alnemri, T. & Alnemri, E. S. Involvement of the AIM2, NLRC4, and NLRP3 inflammasomes in caspase-1 activation by Listeria monocytogenes . J Clin Immunol 30, 693–702 (2010).
Tsuchiya, K. et al. Involvement of absent in melanoma 2 in inflammasome activation in macrophages infected with Listeria monocytogenes . Journal of immunology 185, 1186–1195 (2010).
Sauer, J. D. et al. Listeria monocytogenes triggers AIM2-mediated pyroptosis upon infrequent bacteriolysis in the macrophage cytosol. Cell host & microbe 7, 412–419 (2010).
Meixenberger, K. et al. Listeria monocytogenes-infected human peripheral blood mononuclear cells produce IL-1beta, depending on listeriolysin O and NLRP3. Journal of immunology 184, 922–930 (2010).
Kim, S. et al. Listeria monocytogenes is sensed by the NLRP3 and AIM2 inflammasome. European journal of immunology 40, 1545–1551 (2010).
Warren, S. E. et al. Cutting edge: Cytosolic bacterial DNA activates the inflammasome via Aim2. Journal of immunology 185, 818–821 (2010).
Mueller, N. J., Wilkinson, R. A. & Fishman, J. A. Listeria monocytogenes infection in caspase-11-deficient mice. Infection and immunity 70, 2657–2664 (2002).
Kayagaki, N. et al. Noncanonical inflammasome activation by intracellular LPS independent of TLR4. Science 341, 1246–1249 (2013).
Shi, J. et al. Inflammatory caspases are innate immune receptors for intracellular LPS. Nature 514, 187–192 (2014).
Hagar, J. A., Powell, D. A., Aachoui, Y., Ernst, R. K. & Miao, E. A. Cytoplasmic LPS activates caspase-11: implications in TLR4-independent endotoxic shock. Science 341, 1250–1253 (2013).
Mariathasan, S., Weiss, D. S., Dixit, V. M. & Monack, D. M. Innate immunity against Francisella tularensis is dependent on the ASC/caspase-1 axis. The Journal of experimental medicine 202, 1043–1049 (2005).
Man, S. M. et al. The transcription factor IRF1 and guanylate-binding proteins target activation of the AIM2 inflammasome by Francisella infection. Nature immunology 16, 467–475 (2015).
Meunier, E. et al. Guanylate-binding proteins promote activation of the AIM2 inflammasome during infection with Francisella novicida . Nature immunology 16, 476–484 (2015).
Jones, J. W. et al. Absent in melanoma 2 is required for innate immune recognition of Francisella tularensis. Proceedings of the National Academy of Sciences of the United States of America 107, 9771–9776 (2010).
Man, S. M. et al. IRGB10 Liberates Bacterial Ligands for Sensing by the AIM2 and Caspase-11-NLRP3 Inflammasomes. Cell 167(382–396), e317 (2016).
Ruhl, S. & Broz, P. Caspase-11 activates a canonical NLRP3 inflammasome by promoting K(+) efflux. European journal of immunology 45, 2927–2936 (2015).
Lamkanfi, M. et al. Inflammasome-dependent release of the alarmin HMGB1 in endotoxemia. Journal of immunology 185, 4385–4392 (2010).
Wang, S. et al. Murine caspase-11, an ICE-interacting protease, is essential for the activation of ICE. Cell 92, 501–509 (1998).
Yang, D., He, Y., Munoz-Planillo, R., Liu, Q. & Nunez, G. Caspase-11 Requires the Pannexin-1 Channel and the Purinergic P2X7 Pore to Mediate Pyroptosis and Endotoxic Shock. Immunity 43, 923–932 (2015).
van de Veerdonk, F. L., Joosten, L. A. & Netea, M. G. The interplay between inflammasome activation and antifungal host defense. Immunological reviews 265, 172–180 (2015).
Karki, R. et al. Concerted Activation of the AIM2 and NLRP3 Inflammasomes Orchestrates Host Protection against Aspergillus Infection. Cell host & microbe 17, 357–368 (2015).
Segal, B.H. Aspergillosis. The New England journal of medicine 360, 1870–1884 (2009).
Vande Walle, L. & Lamkanfi, M. Pyroptosis. Current Biology 26, R568–R572 (2016).
Akhter, A. et al. Caspase-11 promotes the fusion of phagosomes harboring pathogenic bacteria with lysosomes by modulating actin polymerization. Immunity 37, 35–47 (2012).
Caution, K. et al. Caspase-11 and caspase-1 differentially modulate actin polymerization via RhoA and Slingshot proteins to promote bacterial clearance. Sci Rep-Uk 5 (2015).
Aachoui, Y. et al. Canonical Inflammasomes Drive IFN-gamma to Prime Caspase-11 in Defense against a Cytosol-Invasive Bacterium. Cell host & microbe 18, 320–332 (2015).
Wang, S. et al. Identification and characterization of Ich-3, a member of the interleukin-1beta converting enzyme (ICE)/Ced-3 family and an upstream regulator of ICE. The Journal of biological chemistry 271, 20580–20587 (1996).
Fassy, F. et al. Enzymatic activity of two caspases related to interleukin-1beta-converting enzyme. European journal of biochemistry / FEBS 253, 76–83 (1998).
Knodler, L. A. et al. Noncanonical Inflammasome Activation of Caspase-4/Caspase-11 Mediates Epithelial Defenses against Enteric Bacterial Pathogens. Cell host & microbe 16, 249–256 (2014).
Gurung, P. et al. Toll or interleukin-1 receptor (TIR) domain-containing adaptor inducing interferon-beta (TRIF)-mediated caspase-11 protease production integrates Toll-like receptor 4 (TLR4) protein- and Nlrp3 inflammasome-mediated host defense against enteropathogens. The Journal of biological chemistry 287, 34474–34483 (2012).
Broz, P. et al. Caspase-11 increases susceptibility to Salmonella infection in the absence of caspase-1. Nature 490, 288–291 (2012).
Rathinam, V. A. et al. TRIF Licenses Caspase-11-Dependent NLRP3 Inflammasome Activation by Gram-Negative Bacteria. Cell 150, 606–619 (2012).
Schauvliege, R., Vanrobaeys, J., Schotte, P. & Beyaert, R. Caspase-11 gene expression in response to lipopolysaccharide and interferon-gamma requires nuclear factor-kappa B and signal transducer and activator of transcription (STAT) 1. The Journal of biological chemistry 277, 41624–41630 (2002).
Sander, L. E. et al. Detection of prokaryotic mRNA signifies microbial viability and promotes immunity. Nature 474, 385–389 (2011).
Shi, J. et al. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature 526, 660–665. (2015).
Kayagaki, N. et al. Caspase-11 cleaves gasdermin D for non-canonical inflammasome signaling. Nature 526, 666–671 (2015).
He, W. T. et al. Gasdermin D is an executor of pyroptosis and required for interleukin-1beta secretion. Cell research 25, 1285–1298 (2015).
Ding, J. et al. Pore-forming activity and structural autoinhibition of the gasdermin family. Nature 535, 111–116 (2016).
Aglietti, R. A. et al. GsdmD p30 elicited by caspase-11 during pyroptosis forms pores in membranes. Proceedings of the National Academy of Sciences of the United States of America(2016).
Liu, X. et al. Inflammasome-activated gasdermin D causes pyroptosis by forming membrane pores. Nature 535, 153–158 (2016).
Sborgi, L. et al. GSDMD membrane pore formation constitutes the mechanism of pyroptotic cell death. The EMBO journal(2016).
Chen, X. et al. Pyroptosis is driven by non-selective gasdermin-D pore and its morphology is different from MLKL channel-mediated necroptosis. Cell research 26, 1007–1020 (2016).
Kang, S. J. et al. Dual role of caspase-11 in mediating activation of caspase-1 and caspase-3 under pathological conditions. The Journal of cell biology 149, 613–622 (2000).
Zanoni, I. et al. An endogenous caspase-11 ligand elicits interleukin-1 release from living dendritic cells. Science 352, 1232–1236 (2016).
Pierini, R. et al. AIM2/ASC triggers caspase-8-dependent apoptosis in Francisella-infected caspase-1-deficient macrophages. Cell death and differentiation 19, 1709–1721 (2012).
Lai, X. H., Golovliov, I. & Sjostedt, A. Francisella tularensis induces cytopathogenicity and apoptosis in murine macrophages via a mechanism that requires intracellular bacterial multiplication. Infection and immunity 69, 4691–4694 (2001).
Wickstrum, J. R. et al. Francisella tularensis induces extensive caspase-3 activation and apoptotic cell death in the tissues of infected mice. Infection and immunity 77, 4827–4836 (2009).
Pearson, J. S. et al. A type III effector antagonizes death receptor signalling during bacterial gut infection. Nature 501, 247–251 (2013).
Li, S. et al. Pathogen blocks host death receptor signalling by arginine GlcNAcylation of death domains. Nature 501, 242–246 (2013).
Hemrajani, C. et al. NleH effectors interact with Bax inhibitor-1 to block apoptosis during enteropathogenic Escherichia coli infection. Proceedings of the National Academy of Sciences of the United States of America 107, 3129–3134 (2010).
Lara-Tejero, M. et al. Role of the caspase-1 inflammasome in Salmonella typhimurium pathogenesis. The Journal of experimental medicine 203, 1407–1412 (2006).
Raupach, B., Peuschel, S. K., Monack, D. M. & Zychlinsky, A. Caspase-1-mediated activation of interleukin-1beta (IL-1beta) and IL-18 contributes to innate immune defenses against Salmonella enterica serovar Typhimurium infection. Infection and immunity 74, 4922–4926 (2006).
Sellin, M. E. et al. Epithelium-Intrinsic NAIP/NLRC4 Inflammasome Drives Infected Enterocyte Expulsion to Restrict Salmonella Replication in the Intestinal Mucosa. Cell host & microbe 16, 237–248 (2014).
Man, S. M. et al. Actin polymerization as a key innate immune effector mechanism to control Salmonella infection. Proceedings of the National Academy of Sciences of the United States of America 111, 17588–17593 (2014).
Thurston, T. L. et al. Growth inhibition of cytosolic Salmonella by caspase-1 and caspase-11 precedes host cell death. Nature communications 7, 13292 (2016).
Sokolovska, A. et al. Activation of caspase-1 by the NLRP3 inflammasome regulates the NADPH oxidase NOX2 to control phagosome function. Nature immunology 14, 543–553 (2013).
Zaki, M. H. et al. The NLRP3 inflammasome protects against loss of epithelial integrity and mortality during experimental colitis. Immunity 32, 379–391 (2010).
Allen, I. C. et al. The NLRP3 inflammasome functions as a negative regulator of tumorigenesis during colitis-associated cancer. The Journal of experimental medicine 207, 1045–1056 (2010).
Dupaul-Chicoine, J. et al. Control of intestinal homeostasis, colitis, and colitis-associated colorectal cancer by the inflammatory caspases. Immunity 32, 367–378 (2010).
Demon, D. et al. Caspase-11 is expressed in the colonic mucosa and protects against dextran sodium sulfate-induced colitis. Mucosal immunology 7, 1480–1491 (2014).
Williams, T. M. et al. Caspase-11 Attenuates Gastrointestinal Inflammation and Experimental Colitis Pathogenesis. American journal of physiology. Gastrointestinal and liver physiology 308, G139–150 (2014).
Oficjalska, K. et al. Protective Role for Caspase-11 during Acute Experimental Murine Colitis. Journal of immunology 194, 1252–1260 (2014).
Baker, P. J. et al. NLRP3 inflammasome activation downstream of cytoplasmic LPS recognition by both caspase-4 and caspase-5. European journal of immunology 45, 2918–2926 (2015).
Schmid-Burgk, J. L. et al. Caspase-4 mediates non-canonical activation of the NLRP3 inflammasome in human myeloid cells. European journal of immunology 45, 2911–2917 (2015).
Kajiwara, Y. et al. A critical role for human caspase-4 in endotoxin sensitivity. Journal of immunology 193, 335–343 (2014).
Vigano, E. et al. Human caspase-4 and caspase-5 regulate the one-step non-canonical inflammasome activation in monocytes. Nature communications 6, 8761 (2015).
Casson, C. N. et al. Human caspase-4 mediates noncanonical inflammasome activation against gram-negative bacterial pathogens. Proceedings of the National Academy of Sciences of the United States of America(2015).
Case, C. L. et al. Caspase-11 stimulates rapid flagellin-independent pyroptosis in response to Legionella pneumophila . Proceedings of the National Academy of Sciences of the United States of America 110, 1851–1856 (2013).
Hu, B. et al. The DNA-sensing AIM2 inflammasome controls radiation-induced cell death and tissue injury. Science 354, 765–768 (2016).
Pelletier, S., Gingras, S. & Green, D. R. Mouse Genome Engineering via CRISPR-Cas9 for Study of Immune Function. Immunity 42, 18–27 (2015).
Bae, S., Park, J. & Kim, J. S. Cas-OFFinder: a fast and versatile algorithm that searches for potential off-target sites of Cas9 RNA-guided endonucleases. Bioinformatics 30, 1473–1475 (2014).
Girardin, H., Latge, J. P., Srikantha, T., Morrow, B. & Soll, D. R. Development of DNA probes for fingerprinting Aspergillus fumigatus . J Clin Microbiol 31, 1547–1554 (1993).
Acknowledgements
We would like to thank the Transgenic and Gene Knockout Shared Resource (St. Jude Children’s Research Hospital) for technical assistance. We thank V.M. Dixit and N. Kayagaki (Genentech) for applying mutant mouse strains. Work from our laboratory is supported by the US NIH (AI101935, AI124346, AR056296, and CA163507 to T.-D.K.), the American Lebanese Syrian Associated Charities (to T.-D.K.), and the R.G. Menzies Early Career Fellowship from the National Health and Medical Research Council of Australia (to S.M.M.).
Author information
Authors and Affiliations
Contributions
S.M.M., R.K., B.B., A.B., S.G., and S.P. performed the research and analyzed the data; S.M.M., and T.-D.K. wrote the paper. T.-D.K. provided overall supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Man, S., Karki, R., Briard, B. et al. Differential roles of caspase-1 and caspase-11 in infection and inflammation. Sci Rep 7, 45126 (2017). https://doi.org/10.1038/srep45126
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep45126
This article is cited by
-
Cross-species transcriptomics identifies obesity associated genes between human and mouse studies
Journal of Translational Medicine (2024)
-
Gasdermin D independent canonical inflammasome responses cooperate with caspase-8 to establish host defense against gastrointestinal Citrobacter rodentium infection
Cell Death & Disease (2023)
-
Doxorubicin-Induced Cardiotoxicity: An Overview on Pre-clinical Therapeutic Approaches
Cardiovascular Toxicology (2022)
-
TYK2 licenses non-canonical inflammasome activation during endotoxemia
Cell Death & Differentiation (2021)
-
Alendronate Augments Lipid A–Induced IL-1α Release via Activation of ASC but Not Caspase-11
Inflammation (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.