Abstract
Pterosaurs are extinct flying reptiles, the first vertebrates to achieve powered flight. Our understanding of the evolutionary transition between basal, predominantly long-tailed forms to derived short-tailed pterodactyloids remained poor until the discovery of Wukongopterus and Darwinopterus in western Liaoning, China. In this paper we report on a new genus and species, Douzhanopterus zhengi, that has a reduced tail, 173% the length of the humerus, and a reduced fifth pedal digit, whose first phalange is ca. 20% the length of metatarsal III, both unique characters to Monofenestra. The morphological comparisons and phylogenetic analysis presented in this paper demonstrate that Douzhanopterus is the sister group to the ‘Painten pro-pterodactyloid’ and the Pterodactyloidea, reducing the evolutionary gap between long- and short-tailed pterosaurs.
Similar content being viewed by others
Introduction
Pterosaurs are extinct flying reptiles that achieved powered flight by developing an entirely distinctive anatomy comparable to any animals alive today. These reptiles are traditionally divided into two groups, short-tailed pterodactyloids and long-tailed ‘rhamphorhynchoids’1. In previous phylogenetic studies, the long-tailed ones have been shown to be a paraphyletic group2,3,4,5. However, our understanding of the evolutionary transition between the two groups was severly limited until the discovery of Wukongopterus and Darwinopterus from western Liaoning, China. In addition to Kunpengopterus, these transitional forms have derived cranial characters and primitive postcranial features6,7,8,9,10,11, alongside Cuspicephalus scarfi, from Dorset, England, which is also considered to be a basal monofenestratan12. Two further specimens, informally named ‘Rhamphodactylus’ and the ‘Painten pro-pterodactyloid’ have also been collected from the southern Franconian region of Germany and are relevant to this question. Both these pterosaurs exhibit mosaic characters, considered as the last step on the road from non-pterodactyloids to pterodactyloids13,14. Recently, Allkaruen koi, found in northern central Chubut of Argentina, was also reported to be the sister group of monofenestratan (Wukongopteridae + Pterodactyloidea) pterosaurs15. Here, we report on a new fossil specimen found in western Liaoning, China, which sheds new light on the origin of the Pterodactyloidea.
The new specimen reported here is housed in the Shandong Tianyu Museum of Nature; it was bought from a local farmer, who said that it had been collected in Toudaoyingzi, Jiangchang. However, based on the matrix surrounding the fossil as well as our own field work in this area, we are sure that this specimen was collected from Tiaojishan Formation in Linglongta. The age of these strata are disputed as some early researchers regarded them as ranging between the Middle and Late Jurassic7,16,17. Recently, however, this unit, especially the beds bearing Darwinopterus, have been considered early Late Jurassic in age18,19,20,21. Thus, the geological age of the specimen discussed here is considered to be early Late Jurassic16,17.
Result
Systematic palaeontology
Pterosauria Kaup, 1834, Monofenestrata Lü, Unwin, Jin, Liu et Ji, 2010, Douzhanopterus zhengi gen. et sp. nov.
Etymology
Douzhan, Chinese pinyin, the name of a buddha granted by Wukong, the Monkey King in the Chinese legend, indicating the relationship between this new pterosaur and other non-pterodactyloid monofenestratans, such as Wukongopterus, pterus, Greek, referring wings; species name in honor of Professor Xiaoting Zheng, who supported our research on this specimen.
Holotype
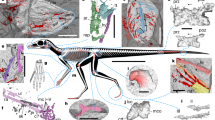
One complete postcranial skeleton housed at Shangdong Tianyu Museum of Nature, Pingyi, China (STM 19–35A & B) (Figs 1 and 2, Tables 1, 2 and 3).
This illustration is based mostly on the part of the holotype, while dark grey areas are based on the counterpart. Abbreviations: ca, carpus; cdv, caudal vertebra; cv3–7, third to seventh cervical vertebra; dca, distal syncarpal; dlca, distal lateral carpal; dpc, deltopectoral crest; dta, distal tarsal; dv, dorsal vertebra; fe, femur; fi, fibula; hu, humerus; il, ilium; mcI-IV, metacarpal I-IV; mtI-V, metatarsal I-V; pca, proximal syncarpal; pphd, phalanges of pedal digit; pph1d4, first phalanx of pedal digit IV; pph1/2d5, first or second phalanx of pedal digit V; phd1–3, phalanges of manual digit I-III; ph1-4d4, first to fourth phalanx of manual digit IV; ppu, prepubis; pt, pteroid; ra, radius; sca-cor, scapulocoracoid; ses, sesamoid; st, sternum; sc, sacrum; ti, tibia; ul, ulna; l, left; r, right; ?, unknown element. Scale bar, 50 mm.
Locality and Horizon
Linglongta, Jianchang, Liaoning, China; Daohugou Bed (or Tiaojishan Formation), Late Jurassic.
Diagnosis
Monofenestratan pterosaur diagnosed by autapomorphies including a reduced tail that is 173% the length of the humerus as well as a reduced fifth pedal digit, whose first phalange is ca. 20% length of metatarsal III. It can be further distinguished from other monofenestratan pterosaurs on the basis of the following combination of characters: the length of the mid-cervical 2.5–3.5 times of width; 22 caudal vertebrae with elongated zygapophyses and chevrons; pteroid over half length of ulna; tibia ca. 180% length of femur, and; fifth pedal digit V having two phalanges.
Description
This new specimen is preserved in part and counterpart, and nearly all the postcranial elements are complete and articulated. Most of the bones are broken horizontally through the middle, and nearly all shafts of long bones are hollow inside, with thin cortices. Some cervical vertebrae are argillic, and only the outlines can be distinguished. The description below is mainly based on the part of the specimen (STM 19–35A, Figs 1 and 2). Elements only preserved on the counterpart (STM 19–35B) will be mentioned in the following text.
This new material comprises a small-sized pterosaur, with a wingspan of 0.74 m, twice the total length of coracoid, humerus, ulna, metacarpal IV, and four wing phalanges. Based on the fusion of syncarpals, scapulocoracoid, tibia and fibula, as well as the fact that the extensor tendon process is fused with the first wing phalange, this specimen is an osteologically mature individual22,23.
The first seven cervical vertebrae are preserved in STM 19–35, while additional elements preserved on the counterpart might include a fused atlas-axis. This section of the series has a distinct centrum condyle and posterodorsal directed neural spine, while the other part is obscured. Each mid-cervical vertebra has a pair of developed pre- and post-zygapophyses. The vertebrae are elongated, and the ratio of length to width is 2.5–3.5 (Table 1). There are nine dorsal vertebrae preserved, suggesting a total of 13 due to the distance between the last preserved dorsal vertebra and the sacrum. Six sacral vertebrae form a sacrum, and their transverse processes are directed lateroposteriorly, thicker than those of the of dorsal vertebrae. The caudal vertebrae of STM 19–35 are complete, even the distal most caudal, which was buried in the part before preparation. There are 22 caudal vertebrae, with a total length of 83.86 mm (Fig. 1b). These approximately elongate from the first to the sixth, and shorten to the last (Table 2). The zygapophyses and chevrons of the anterior caudal vertebrae are extremely elongated, at least three times length of centra (Fig. 1b), while the last elongated zygapophysis or chevron is present at the position of the sixteenth caudal. The last caudal vertebrae only have tiny centra. The length of tail is moderately reduced relative to the size of this animal (Fig. 3a; Supplementary Table 1), intermediate between long-tailed forms and short-tailed pterodactyloids.
(a) Relative length of the tail plotted against a proxy for body size (length of the humerus); (b) ratio of metacarpal to ulna against a proxy for body size (length of the humerus); (c) relative length of first phalange of pedal digit V plotted against a proxy for foot size (length of the metatarsal III). The data used here are listed in Supplementary Tables 1–3. Filled stars, Douzhanopterus zhengi; filled circles, ‘Painten pro-pterodactyloid’; triangles, pterodactyloids; positive crosses, anurognathids; oblique crosses, non-pterodactyloids (except anurognathids in (a)).
The sternum of STM 19–35 is not complete, broken into two pieces. The cristospine is not preserved, but the left part of the sternal plate is nearly complete and no keel can be seen. The length of the sternum is similar to the width of the plate; the anterior margin is inclined posteriorly, and the posterior margin is nearly straight.
The scapula is slender. It is elongated and approximately 40% longer than coracoid (Table 3). No ventral expansion is present on the coracoid. The humerus is straight. It has a short deltopectoral crest, which is trapezoid and placed proximally, and it has a distinct ulnar crest, which directs posteriorly. No pneumatic foramen can be observed because of preservation, and the ulna is slightly thicker in mid-shaft than the radius. The pteroid is elongated, reaching half the length of ulna, and the outlines of the right carpals can be distinguished. The proximal and distal syncarpals formed and one small sesamoid attaches to the distal lateral carpal. The metacarpals are moderately elongated, around 65% the length of humerus and 53% the length of ulna, comprising a ratio between those basal and derived pterosaurs (Fig. 3b; Supplementary Table 2). Metacarpals I-III are articulated with the carpal as well as the wing metacarpal, and manual digits I-III bear large claws. The second wing phalange is the longest one; the first and third phalanges have similar length; the fourth one is slightly shorter than the others, but still over 80% length of the longest one (Table 3).
The pelvic girdles are fused with the sacrum; the preacetabular process is obscured but the postacetabular process is complete and short. Anterior to the acetabular fossa, two pieces of bones are present which we interpret as prepubes. These are longer than wide, and not fused in the middle. The femur of STM 19–35 is slightly curved and has a distinct head and constricted neck, forming a 150° angle with its shaft. The tibia is straight, and fused with the proximal and distal ends of the fibula, which reach up to 45% the length of the tibia (Table 3). Rectangular lateral and medial tarsals are preserved in each hind limb; metatarsal II is the longest, while metatarsal IV is shorter than metatarsals I-III. Metatarsal III is 31% the length of the tibia, while metatarsal V is the shortest (Table 3). The fifth pedal digit still has two phalanges; the first of these is straight while the second is slightly curved (Fig. 1c). The first pedal phalange is only 20% to 23% the length of metatarsal III, larger than those of pterodactyloids, but shorter than non-pterodactyloids (Fig. 3c; Supplementary Table 3).
Systematics of Douzhanopterus zhengi sp. et gen. nov
In order to access the position of Douzhanopterus zhengi gen. et sp. nov. within the Pterosauria, we carried out a new phylogenetic analysis (see Methods and Supplementary Methods). This analysis was mainly based on the previous work of Wang et al.6, consisting of 64 species (two outgroups and 62 ingroups) and 89 characters (including two continuous characters, modified from Andres et al.5). We also added Darwinopterus, Kunpengopterus sinensis, Cuspicephalus scarfi, the ‘Painten pro-pterodactyloid’, Allkaruen koi, Changchengopterus pani, Douzhanopterus zhengi, and Kryptodrakon progenitor, used TNT for the analysis, and generated 114 most parsimonious trees (MPTs), 253.383 steps in length (CI = 0.600; RI = 0.852). The strict consensus of these MPTs shows that the Monofenetrata and Pterodactyloidea are monophyletic, with the support of 12 and 18 characters, respectively (see the synapomorphies listed in the Supplementary Information). However, the relationship of non-pterodactyloid monofenestratans in this group are still not well-resolved. Through the comparison of 114 MPTs, two unstable taxa were identified, Cuspicephalus scarfi and Allkaruen koi. Both of them lie in basal positions of Monofenestrata. The reduced strict consensus tree without these two taxa is shown in Fig. 4. In this reduced tree, the ‘Painten pro-pterodactyloid’ is the sister group to the Pterodactyloidea, while Douzhanopterus occupies a more basal position, the sister group to the clade including the ‘Painten pro-pterodactyloid’ and the Pterodactyloidea. The ‘Wukongopteridae’ is not resolved as a monophyletic group in this analysis and Wukongopterus lii is the most basal monofenestran.
Discussion
Relationships among basal monofenestratans
Some basal monofenestratans have been considered transitional forms between non-pterodactyloids and pterodactyloids, including Wukongopterus6, Darwinopterus7,8,9,10, Kunpengopterus8, Cuspicephalus12, Allkaruen15, ‘Rhamphodactylus’13, and the ‘Painten pro-pterodactyloid’14. However, just the upper jaw of Cuspicephalus are preserved12, and it is absent in Douzhanopterus. The overlap with Allkaruen and Douzhanopteus is linited to cervical vertebrae15, which means that too limited information is available to distinguish these taxa from one another.
Compared with Wukongopterus, Darwinopterus, and Kunpengopterus6,7,8,9, Douzhanopterus occupies a derived position as it possesses a number of advanced postcranial ratios (Table 4). For example, the cervicals in Douzhanopterus are more elongated than those of the other three taxa, while the tail is less than half their length relative to body size. In addition, the fifth pedal digit of Douzhanopterus retains two much reducted pedal phalanges.
The description of ‘Rhamphodactylus’ is quite limited13, but the ‘Painten pro-pterodactyloid’ has been discussed in more detail14. Both these animals are young juvenile individuals because no fusion of postcranial elements can be found. They are in the same size and have quite similar ratios of postcranial elements, although the measurements of ‘Rhamphodactylus’ were calculated in this study based on figures. Indeed, these two were not treated as the same taxon because of differences in their geological ages14. Compared with the new Chinese material, both these enigmatic taxa share the presence of an elongated metacarpal IV (62–66% the length of the humerus, and 46–53% the length of the ulna), reduced first (20% to 31% the length of metatarsal III) and second phalanges of pedal digit V, as well as a reduced tail (0.8–1.7 the length of the humerus) with elongated zygapophyses and chevrons, demonstrating the transitional position of these three specimens between other non-pterodactyloid mononfenestratans and pterodactyloids. However, Douzhanopterus varies compared to the two German specimens in a number of postcranial ratios (Table 4). For example, the new Chinese taxon has a larger pteroid/ulna length ratio than that of the two German specimens (Table 4), while its tibia/femur length ratio is much larger than that of the other two non-pterodactyloid monofenestratans (Table 4). Douzhanopterus and the ‘Painten pro-pterodactyloid’ have two reduced phalanges of the fifth pedal digit relative to other non-pterodactyloids, while our new Chinese taxon boasts a smaller ratio between the first phalange of pedal digit V to metatarsal III (20% and 23%) compared to the ‘Painten pro-pterodactyloid’ (31%). The tails of the two German pterosaurs are also incomplete, lacking their distal-most ends. Thus, assuming that these distal-most caudal vertebrae exhibit the same average lengths as the anterior parts of the ‘Painten pro-pterodactyloid’, the whole tail of the ‘Painten pro-pterodactyloid’ would have a maximum length of 29.6 mm, shorter than its humerus. Douzhanopterus also has 22 caudal vertebrae, more than the ‘Painten pro-pterodactyloid’ which has an estimated 1714; the former also has much larger tail/humerus length ratio, nearly twice than that of the latter, but still less than half that of other non-pterodactyloid monofenestratans (Table 4). The centra, rod-like zygaporphyses, and chevrons of the caudal vertebrae in Douzhanopterus are slightly more elongated than those of the German specimens, but not so much as in other non-pteroadctyloids. Based on our as yet unpublished study of young basal monofenestratan specimens, tail length relative to body size changes, but the number of caudal vertebrae and the elongation of fifth pedal digits remain stable during ontogeny. Hence, Douzhanopterus has a primitive tail but a slightly derived foot relative to the German specimens; thus, as the result of our phylogenetic analysis, Douzhanopterus lies in a more basal position than the ‘Painten pro-pterodactyloid’.
The discovery of Douzhanopterus enhances our understanding of the origin of the Pterodactyloidea. Indeed, Wukongopterus, Darwinopterus, and Kunpengopterus, known on the basis of abundant specimens from the early Oxfordian (160–161 Ma)17, are the first step in this transition, and evidence mainly cranial changes6,7,8,9. Specimens of Douzhanopterus, however, from the same deposits, are much rarer than these taxa, which may indicate that these basal monofenestratans were dominant at the time while Douzhanopterus occupied an inferior position. Hence, monofenestratans including Wukongopterus and Darwingopterus, may first appear further back in time into the Middle or even Early Jurassic15, becoming more diverse and successful during the early Late Jurassic. We hypothesize that Douzhanopterus likely originated adjacent to, or around this time.
Basing on a study of Darwinopterus modularis, modular evolution was first proposed by Lü et al.7. Building on this, the intermediate characters seen in Douzhanopterus can also be divided into two modules, an anterior part encompassing the postcranium, mainly cervical vertebrae and metacarpals, and a posterior part, mainly encompassing caudal vertebra and feet. In the anterior part, elongation of cervicals is seen, as was the case in Wukongopterus and Darwinopterus, whose cervical length/diameter ratios are no more than 2.56,7,8,9. In Douzhanopterus, this ratio becomes slightly larger, suggesting that this trend in cervical elongation was maintained. This effect was also seen in the metacarpals6,8, and elongation was again maintained in Douzhanopterus. The posterior parts of the skeleton change significantly; tail length relative to body size in Douzhanopterus was reduced by at least 50% compared to Wukongopterus and Darwinopterus. The new Chinese pterosaur also possesses by far the oldest record of a reduced fifth digit, and it is shorter than third of that in Wukongopterus and Darwinopterus relative to its body size. This is corroborated by the fact that both ‘Rhamphodactylus’ and the ‘Painten pro-pterodactyloid’ were collected from early Late Tithonian-aged deposits (ca. 150 Ma)14, about 10 million years younger than the age of Douzhanopterus. These two German specimens have shorter tails than Douzhanopterus, which are quite similar to pterodactyloids. The other postcranial changes between Douzhanopterus and the two German pterosaurs are not significant, and even some characters, such as reduction of fifth pedal digits and elongation of metacarpals, are more primitive in the latter specimens than in Douzhanopterus. Some researchers have suggested that modularity may play an important role at one anatomical level, while at a finer level, evolution within a module may follow a mosaic pattern15. The distinctions we report between Douzhanopterus and two the German pterosaurs are consistent with this hypothesis.
Methods
The phylogenetic analysis was conducted with the software Tree Analysis Using New Technology (TNT) version 1.124. The ‘Max.trees’ setting was set to 10,000 and zero-length branches were collapsed using the rule ‘min. length = 0’. Two ‘Traditional search’ rounds were used, first starting with Wagner trees and duplicating 1,000 times, while the second round was built on trees from memory. The command ‘best’ was used to filter the trees for the best score, and all others were set as default. As discussed in the text, we added Douzhanopterus zhengi gen. et sp. nov., the ‘Painten pro-pterodactyloid’, Allkaruen koi, Kryptodrakon progenitor, Cuspicephalus scarfi, Changchengopterus pani, Darwinopterus modularis, D. linglongtaensis, D. robustdens, and Kunpengopterus sinensis to the matrix of Wang et al.6, as well as two continuous characters, the ratio between the metacarpal/humerus length and caudal length/diameter (modified from Andres et al.5). Another new character relating to the length of the first phalanx of pedal digit V compared to metatarsal III was added as character 88, while characters that relate to the humerus/metacarpal-IV, femur/metacarpal-IV, and ulna/metacarpal-IV length ratios as used by Wang et al.6were deleted, to avoid artificially upweighting the ratio of metacarpal IV to other elements. We also re-coded Wukongopterus lii for character 50; this character relates to peg-like teeth. In the holotype and the only specimen of Wukongopterus lii, the skull is incomplete, and there are less than 15 peg-like teeth preserved on either side. Thus, the inferred number of teeth for each side in upper and lower jaws are 16 and 12, respectively6. If this is correct, 16 teeth should be scored as state 2 and 12 teeth is state 1; we consider that it is better to choose the two states, but not just state 2. The complete characters and synapomorphies of nodes present in the strict consensus tree are listed in Supplementary Methods. The file with complete matrix, which can be run directly in TNT, is provided as Supplementary Dataset.
Additional Information
How to cite this article: Wang, X. et al. New evidence from China for the nature of the pterosaur evolutionary transition. Sci. Rep. 7, 42763; doi: 10.1038/srep42763 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Wellnhofer, P. The Illustrated Encyclopedia of Pterosaurs. (Salamander Books, 1991).
Bennett, S. C. Taxonomy and systematics of the late Cretaceous pterosaur Pteranodon (Pterasauria, Pterodactyloidea). Occas. Pap. Mus. Nat. Hist. Univ. Kansas 169, 1–70 (1994).
Kellner, A. W. A. In: Evolution and Palaeobiology of Pterosaurs (eds Buffetaut, E. & Mazin, J. M. ) 105–137 (Geological Society, 2003).
Unwin, D. M. In: Evolution and Palaeobiology of Pterosaurs (eds Buffetaut, E. & Mazin, J. M. ) 139–190 (Geological Society, 2003).
Andres, B., Clark, J. & Xu, X. The earliest pterodactyloid and the origin of the group. Curr. Biol. 24, 1011–1016 (2014).
Wang, X., Kellner, A. W. A., Jiang, S. & Meng, X. An unusual long-tailed pterosaur with elongated neck from western Liaoning of China. An. Acad. Bras. Cienc. 81, 793–812 (2009).
Lü, J., Unwin, D. M., Jin, X., Liu, Y. & Ji, Q. Evidence for modular evolution in a long-tailed pterosaur with a pterodactyloid skull. Proc. Roy. Soc. B Biol. Sci. 277, 383–389 (2010).
Wang, X. et al. New long-tailed pterosaurs (Wukongopteridae) from western Liaoning, China. An. Acad. Bras. Cienc. 82, 1045–1062 (2010).
Lü, J., Xu, L., Chang, H. & Zhang, X. A new darwinopterid pterosaur from the Middle Jurassic of western Liaoning, northeastern China and its ecological implications. Acta Geol. Sin. 85, 507–514 (2011).
Lü, J. et al. An egg-adult association, gender, and reproduction in pterosaurs. Science, 331, 321–324 (2011).
Wang. X. et al. Eggshell and histology provide insight on the life history of a pterosaur with two functional ovaries. An. Acad. Bras. Cienc. 87, 1599–1609 (2015).
Martill, D. M. & Etches, S. A new monofenestratan pterosaur from the Kimmeridge Clay Formation (Kimmeridgian, Upper Jurassic) of Dorset, England. Act. Pal. Pol. 58, 285–294 (2012).
Rauhut, O. W. M. Ein “Rhamphodactylus” aus der Mörnsheim-Formation von Mühlheim. Jahresbericht 2011 und Mitteilungen der Freunde der Bayerischen Staatssammlung für Paläontologie und Historische Geologie München e.V. 40, 69–74 (2012).
Tischlinger, H. & Frey, E. A new pterosaur with mosaic characters of basal and pterodactyloid pterosauria from the Upper Kimmeridgian of Painten (Upper Palatinate, Germany). Archaeopteryx 31, 1–13 (in German with English abstract) (2013).
Codorniú, L., Carabajal, A. P., Pol, D., Unwin, D. & Rauhut, O. W. M. A Jurassic pterosaur from Patagonia and the origin of the pterodactyloid neurocranium. PeerJ 4, e2311 (2016).
Liu, Y. Q. et al. U-Pb zircon age for the Daohugou Biota at Ningcheng of Inner Mongolia and comments on related issues. Chin. Sci. Bull. 51, 2634–2644 (2006).
Zhang, H., Wang, M. & Liu, X. Constraints on the upper boundary age of the Tiaojishan Formation volcanic rocks in West Liaoning-North Hebei by LA-ICP-MS dating. Chin. Sci. Bull. 53, 3574–3584 (2008).
Liu, Y. et al. Timing of the earliest known feathered dinosaurs and transitional pterosaurs older than the Jehol Biota. Paleo-3, 323, 1–12 (2012).
Sullivan, C. et al. The vertebrates of the Jurassic Daohugou Biota of northeastern China. J. Vert. Paleont. 34, 243–280 (2014).
Wang, X. et al. Timing of Linglongta Pterosaur Fauna from Western Liaoning and Zhejiangopterus: a summary of geochronology and stratigraphic sequence of pterosaur fossil-bearing beds in China. Earth. Sci. Front. 21, 157–184 (in Chinese with English abstract) (2014).
Chu et al. High-precision U-Pb geochronology of the Jurassic Yanliao Biota from Jianchang (western Liaoning Province, China): Age constraints on the rise of feathered dinosaurs and eutherian mammals. Geochem. Geophys. Geosyst. 17, doi: 10.1002/2016GC006529 (2016).
Bennett, S. C. The ontogeny of Pteranodon and other pterosaurs. Paleobiology 19, 92–106 (1993).
Kellner, A. W. A. Comments on Triassic pterosaurs with discussion about ontogeny and description of new taxa. An. Acad. Bras. Cienc. 87, 667–689 (2015).
Goloboff, P. A., Farris, J. S. & Nixon, K. C. TNT, a free program for phylogenetic analysis. Cladistics 24, 774–786 (2008).
Wellnhofer, P. Die Pterodactyloidea (Pterosauria) der Oberjura-Plattenkalke Suddentschlands, Bayer. Akad. Wiss. Math. Nat. Klasse Nf. H. 141, 1–133 (in German) (1970).
Wang, X. & Zhou, Z. A new pterosaur (Pterodactyloidea, Tapejaridae) from the Early Cretaceous Jiufotang Formation of western Liaoning, China and its implications for biostratigraphy. Chinese Sci. Bull. 48, 16–23 (2003).
Acknowledgements
We thank X. Zheng for supporting this research, and H. Zhou for preparing the specimen. TNT was provided by the Willi Hennig Society, and we also thank A.W.A. Kellner for his comments that improved an earlier version of this manuscript. This study was supported by the National Natural Science Foundation of China (41572020, 41472023, 41372014, 41602011, 41688103), the Major Research plan of the National Natural Science Foundation of China (91514302), the Strategic Priority Research Program (B) of CAS (XDB18000000), the Key Laboratory of Economic Stratigraphy and Palaeogeography, Nanjing Institute of Geology and Palaeontology (CAS), the Hundred Talents Project of CAS, the National Science Fund for Distinguished Young Scholars (40825005), and the National Key Basic Research Program of China (2012CB821900).
Author information
Authors and Affiliations
Contributions
X.-L.W. and S.-X.J. designed the research plan. X.-L.W., S.-X.J., J.-Q.Z., and X.C. performed analysis. X.-L.W., S.-X.J., and X.C. wrote the manuscript. All authors discussed and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Wang, X., Jiang, S., Zhang, J. et al. New evidence from China for the nature of the pterosaur evolutionary transition. Sci Rep 7, 42763 (2017). https://doi.org/10.1038/srep42763
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep42763
This article is cited by
-
Dietary diversity and evolution of the earliest flying vertebrates revealed by dental microwear texture analysis
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.