Abstract
Increasing levels of pCO2 within the oceans will select for resistant organisms such as anemones, which may thrive under ocean acidification conditions. However, increasing pCO2 may alter the bacterial community of marine organisms, significantly affecting the health status of the host. A pH gradient associated with a natural volcanic vent system within Levante Bay, Vulcano Island, Italy, was used to test the effects of ocean acidification on the bacterial community of two anemone species in situ, Anemonia viridis and Actinia equina using 16 S rDNA pyrosequencing. Results showed the bacterial community of the two anemone species differed significantly from each other primarily because of differences in the Gammaproteobacteria and Epsilonproteobacteria abundances. The bacterial communities did not differ within species among sites with decreasing pH except for A. viridis at the vent site (pH = 6.05). In addition to low pH, the vent site contains trace metals and sulfide that may have influenced the bacteria community of A. viridis. The stability of the bacterial community from pH 8.1 to pH 7.4, coupled with previous experiments showing the lack of, or beneficial changes within anemones living under low pH conditions indicates that A. viridis and A. equina will be winners under future ocean acidification scenarios.
Similar content being viewed by others
Introduction
Ocean acidification, or an increase in oceanic pCO2, may have significant effects on organisms within the marine environment. The increase in oceanic pCO2 could profoundly affect organismal physiology, the carbonate structure of the substrate, and overall ecosystem functioning1,2. Under this changing environment certain species may thrive while others may suffer negative consequences. Identifying the winners and losers of changing oceanic pCO2 will provide insight into future shifts in community assemblages within the marine environment.
While ocean acidification may cause reduced rates of survival, growth, calcification and reproduction on several different calcifying species, from corals to coccolithophores3, non-calcifying organisms, especially ones that photosynthesize, may be positively affected by ocean acidification. Therefore, reduced oceanic pH may be beneficial for some organisms like anemones3,4,5. For example, prolonged exposure to high pCO2 showed an increase in the growth of the algal symbiont within the anemone Anthopleura elegantissima6.
Anemonia viridis is a symbiotic sea anemone found within the northeastern Atlantic and Mediterranean Sea and is often used as a model for cnidarian studies7. Interestingly, A. viridis has a high capacity to buffer intracellular pH conditions even when exposed to low pH seawater8 suggesting that overall host physiology may not be severely impacted by ocean acidification. Several studies have been conducted on A. viridis in natural volcanic CO2 seeps, specifically off the coast of Vulcano, (Aeolian Islands) Italy. At this location Suggett et al.4 showed that A. viridis increased productivity (measured as gross photosynthesis:respiration ratios) with increasing pCO2 along a natural gradient, suggesting that this particular anemone may thrive under future pCO2 scenarios. Within the same site, Horwitz et al.5 showed that autotrophy of A. viridis increased with increasing pCO2, which illustrates the overall flexibility of this anemone species. Interestingly, A. viridis in sites with varying levels of pCO2 showed no significant differences in chlorophyll a concentrations, Symbiodinium densities or protein concentrations although the Symbiodinium mitotic index within anemones increased under high pCO24,5. In light of these previous physiological studies, A. viridis appears to be either resistant to or positively affected by ocean acidification and may be considered a “winner” under acidified conditions4.
In concordance with changes in the physiological state of A. viridis, ocean acidification conditions may alter the microbial communities of this anemone. Indeed, physiological changes of the host because of declining pH may lead to altered states within the bacterial communities. Marine microbes respond rapidly to environmental shifts9, are key components of the oceanic nutrient cycles10, and are important members of the host microbiome, providing essential functions for host health11. Some studies indicate that microbial communities will not be altered by ocean acidification conditions12,13. For example, Oliver et al.13 showed that there was no change in community abundance, structure or composition of the bacterial fraction of microbial plankton under ocean acidification conditions. Bacterial communities associated with oxic sediments, however, appear to significantly differ at sites with naturally low pH compared with control sites14,15. Furthermore, other studies indicate that microbial communities associated with host organisms may in fact change under elevated pCO2. Webster et al.16 documented differences in microbial communities associated with the coral species Acropora millipora, the foraminifera Marginopora vertebralis, the crustose coralline algae Hydrolithon onkodes, and reef biofilms between treatments of pH 8.1 and 7.9 conditions. Alternatively, a more recent study by Webster et al.17 showed that the coral species Acropora millipora and Seriatopora hystrix, the foraminifera Marginopora vertebralis and Heterostegina depressa, the CCA Hydrolithon onkodes and the sea urchin Echinometra sp. showed no difference in bacterial community composition between pH 8.1 and 7.9 treatments, and only detected differences in the bacterial communities when temperature treatments were high and pH was low17. A study of A. millipora and Porites cylindrica at a natural CO2 seep in Papua New Guinea, however, indicated differences in the bacterial communities of these corals at the low pH site, predominantly caused by a loss of the bacterial symbiont Endozoicomonas18. Analysis of the bacteria associated with A. viridis from Ischia Island (Gulf of Naples), Italy showed that the microbial communities of this species were more diverse in samples from a low pH site (7.0) compared with samples from a reference site (8.1), but there was no difference in antibiotic activity of associated bacteria when comparing the two sites19. The lack of consistency and preliminary nature of the research testing the effects of ocean acidification on bacterial communities indicates a significant knowledge gap in need of study.
Actinia equina is a non-photosymbiotic sea anemone commonly found in the rocky intertidal of the North Atlantic Ocean and the Mediterranean Sea7. This species of anemone can survive for prolonged periods of time out of the water and are even found covered in sand20. Along the pH gradient in Levante Bay, A. equina is very common but is exposed to high pCO2 conditions only when fully submerged, while during low tide it is often exposed to atmospheric CO2 levels. The continuous diurnal exposure to a variable environment may suggest that this species is more resistant to environmental changes than others21. A study on the lysozyme activity of A. equina mucus, however, showed a peak in activity at pH 622. These results suggest that one of the best defenses against bacterial invasion, activity of the enzyme lysozyme, will increase for A. equina under reduced pH, thereby protecting the resident microbiome of the anemone leading to overall maintenance in host health. How the resident microbiome, including the bacterial communities of A. equina, will be impacted by ocean acidification is unknown.
The objectives of the present study were to i) characterize and compare the microbial communities of two anemone species with different life history strategies found along a natural pH gradient within the sublittoral zone of Levante Bay, Vulcano Island, Italy (A. viridis, a subtidal species with an endosymbiont, and A. equina, an intertidal species with no endosymbiont), and ii) to determine whether the bacterial assemblages of these two species differed with decreasing levels of pH.
Methods
Sample collection
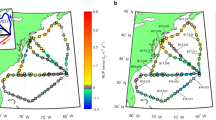
Anemone samples were collected on June 21, 2013 between 10:00 and 11:00 hrs at sites along the sublittoral between 0.25 and 2 m depth in Levante Bay, North Vulcano Island (38° 25′N, 14° 57′E). Water temperature was 23 °C and did not vary within the sampled area. Samples were taken along the gradient from five sites: the reference site (500 m northeast of the vent; pH 8.15), site 1 (390 m from the vent, pH 8.03), site 2 (300 m from the vent, pH 7.86), site 3 (240 m from the vent, pH 7.44), and the vent site (15 m from the vent; pH 6.05; Fig. 1). Although extensive physical parameters were not collected simultaneously with the anemone samples, thorough geochemical surveys have been conducted previously that characterize Levante Bay spatially and temporally23,24,25. These surveys indicate that sites 1, 2, 3 and the reference site differ little except for within their carbonate chemistry composition (see Boatta et al.23 and Johnson et al.24 for further details in the carbonate chemistry within this area). Temperature, salinity, total alkalinity, sulfur concentrations, nitrate, phosphate, sediment texture, and total nitrogen and total organic carbon content within the sediment do not significantly differ along the north shore of Levante Bay14,23,24,25. However, nitrite and silicate have shown significant differences between site 3 and the reference site location25. The vent site, in addition to having extremely low pH conditions (6.05), also contains elevated levels of trace elements, iron, and hydrogen sulfide compared with other sites further from the CO2 vent23,26. Anemone samples for the present study included ~300 μg of tentacles from three randomly selected individuals of A. viridis at each site, and ~300 μg of both tentacles and the body column from three randomly selected A. equina. Samples were collected with sterile scissors by collectors wearing sterile nitrile gloves. Once the anemone sample was collected it was removed from the water and placed within a sterile 10 ml polystyrene tube. Samples of A. equina were not collected from the vent site because this species was absent within this location. All samples were stored in 10 ml of RNA Later (Qiagen, Inc), cryopreserved using liquid nitrogen within 1 hour of collection27, and transported back to Mote Marine Laboratory (Sarasota FL) in a liquid nitrogen dry shipper for subsequent analyses.
Map of sites sampled along the shoreline of Levante Bay, Vulcano Island Italy.
Figure modified from Horwitz et al.5.
Next Generation Sequencing
Source DNA was extracted from each anemone sample using the MoBio Powersoil DNA isolation kit with a modified protocol (MoBio Inc., Carlsbad, CA). The microbiome of each sample was analyzed using 16 S rDNA 454 pyrosequencing. A modified amplicon pyrosequencing (bTEFAP) procedure was performed with 16 S universal Eubacterial primers, a modified 27 F and 519 R primer. Polymerase chain reaction was carried out using a single-step 30 cycle HotStarTaq Plus Master Mix Kit (Qiagen, Valencia, CA) under the following conditions: 94 °C for 3 minutes, 28 cycles of 94 °C for 30 seconds; 53 °C for 40 seconds and 72 °C for 1 minute with a final elongation step at 72 °C for 5 minutes. Amplicon products from different samples were combined equally and purified using Agencourt Ampure beads (Agencourt Bioscience Corporation, MA, USA). Samples were sequenced via Roche 454 FLX titanium instruments and reagents following manufacturer’s guidelines. The sequence data was processed at MRDNA laboratory (www.mrdnalab.com, Shallowater, TX) using a standardized analysis pipeline developed and implemented by MRDNA laboratory. Briefly, sequences were depleted of barcodes and primers and then short sequences <200 bp were removed. Sequences with ambiguous base calls as well as sequences with homopolymer runs exceeding 6 bp were removed. Sequences were then de-noised and chimeras were removed. Operational taxonomic units (OTUs) were defined after removal of singleton sequences, clustering at 3% divergence (97% similarity)28. Final OTUs were taxonomically classified using BLASTn against a curated database derived from GreenGenes, RDPII and NCBI (www.ncbi.nlm.nih.gov; http://rdp.cme.msu.edu)29 and compiled into each taxonomic level (Supplementary Table 1).
The sequencing data from this study were submitted to GenBank within the National Center for Biotechnology Information (http://www.ncbi.nlm.nih.gov) under Accession numbers KX149130 – KX150371 for 16 S rRNA gene pyrosequencing.
Statistics
Rarefaction curves, which estimated the coverage of species richness, were created from the average number of reads within each bacteria species detected using pyrosequencing for each anemone species. Coverage, the probability of occurrence of the bacterial species observed in the sample within each site, was calculated using the Zhang-Huang estimator from the ‘entropart’ package in R30,31,32. The Shannon, Simpson, inverse Simpson diversity indices, and Pielou’s richness of the bacterial community were calculated at the species taxonomic level for each sample. Differences in diversity or richness among sites were tested for each anemone species using analysis of variance (ANOVA), after passing assumptions of normality and homoscedasticity. However, a Kruskal Wallis test was used for the Simpson and inverse Simpson index of the A. viridis data because of non-normal distributions within the dataset. The percent composition of bacterial groups from each sample was also analyzed at the OTU level using a factorial permutation multivariate analysis of variance (PERMANOVA) with anemone species and sites designated as the two independent variables. PERMANOVA was also used to determine if there were differences among sites within species. Pairwise PERMANOVAs were used to determine which sites where different between and within anemone species when significant differences were detected within the omnibus test. A similarity percentages (SIMPER) analysis within the ‘vegan’ package of the statistical program R33 was used to provide a relative dissimilarity index among sites and also provided the percent dissimilarity caused by each bacterial OTU. The top five contributors to dissimilarity were tested for differences among sites using Kruskal Wallis tests. Dunn’s test with a Bonferroni correction was used for posthoc comparisons when significant differences occurred using the ‘dunn.test’ package in R34. Bacterial OTU data were then processed through non-metric multidimensional scaling (NMDS), which applied the rank orders of data to represent the position of communities in multidimensional space using a reduced number of dimensions that can be easily plotted and visualized. The NMDS results were then plotted in two-dimensional ordination space using the ‘vegan’ package in R33. The relative abundance of bacterial classes and the relative abundances of genera within major classes were also plotted for visualization. A heatmap of the most common genera, those found in >1% abundance, was created using the R packages ‘Heatplus’35, ‘gplots’36, ‘RColorBrewer’37 and ‘vegan’33.
Results
No visual difference in size, color or health was observed in Anemonia viridis or Actinia equina individuals from different sites. Both species were plentiful along the shoreline of Levante Bay and appeared robust regardless of the location. There was however, no A. equina within the vent site due to a lack of intertidal substrate and habitat.
Metadata from pyrosequencing
Pyrosequencing resulted in an average 21,530 (±2,339) sequence reads per A. viridis sample, and 21,367 (±5,160) reads per A. equina sample (see GenBank accession numbers for sequencing information). For A. viridis, the average base pair length detected for each read was 444 (±4) bp and average GC content was 57% (±0.1). For A. equina, the average base pair length per sequence was 459 (±2) bp and average GC content was 57% (±0.1). Prior to analyses, the data were standardized using a subset of 6,167 random sequence reads per replicate sample, the lowest coverage found within a sample after sequencing.
Rarefaction curve
The rarefaction curve from the A. viridis samples indicated that a substantial portion of the bacterial species were detected within our dataset (Supplementary Figure 1). Coverage analyses indicated that >99% of the bacterial species within each site were detected even after subsetting samples to standardize for sequencing reads (Supplementary Table 2).
Bacterial diversity
Statistical analyses showed no difference in diversity or richness within the bacterial species among sites for either A. viridis or A. equina (Supplementary Table 3; Fig. 2). Overall, diversity was lower in site 1 and site 3 for both A. viridis and A. equina.
Shannon diversity index of bacterial species found within samples of Anemonia viridis and Actinia equina along the pH gradient of Levante Bay, Vulcano, Italy (R = reference site, S1 = site 1, S2 = site 2, S3 = site 3, V = vent site).
ND represents no data because A. equina was not found within the vent site.
Comparison between anemone species
The bacterial community of A. viridis significantly differed from the bacterial community of A. equina (F(1,25) = 5.798, R2: 0.188, p < 0.001). The NMDS ordination plot indicates little to no overlap in the relative abundance of bacteria from these two anemone species (Fig. 3). Comparisons of the bacterial class composition showed that there were significantly more Gammaproteobacteria (X2(1) = 9.152, p = 0.003) and significantly less Epsilonproteobacteria (X2(1) = 17.626, p < 0.001) within A. viridis compared with A. equina. Overall, A. viridis had a relative bacterial abundance of 27.69% (±6.4% SE) Gammaproteobacteria, whereas A. equina contained only 5.26% (±1.18%) Gammaproteobacteria (Fig. 4). A. viridis had on average only 0.17% (±0.07%) Epsilonproteobacteria, whereas A. equina had on average 19.13% (±6.77%) Epsilonproteobacteria (Fig. 4). Importantly, a significant difference remained between species even when the vent site data of A. viridis was removed from the comparison (F(1,22) = 5.848, R2: 0.210, p < 0.001). The factorial PERMANOVA also showed that when using both anemone species data there were no differences detected among sites, nor was there an interaction effect between sites and anemone species (sites: F(4,18) = 1.359, R2 = 0.168, p = 0.065; species x site: F(3,18) = 0.964, R2 = 0.089, p = 0.523).
Relative abundances of bacterial classes present in (a) Anemonia viridis and (b) Actinia equina collected at different sites along a pH gradient in Levante Bay, Vulcano, Italy (R = reference site, S1 = site 1, S2 = site 2, S3 = site 3, V = vent site). Bacteria present in <3% relative abundance are grouped within ‘others’.
Bacterial community analysis within anemone species
There was a significant difference in bacterial communities of A. viridis among sites (F(4,10) = 1.47; R2: 0.370, p = 0.046; Fig. 5a) when this anemone species was analyzed independently. Pairwise PERMANOVAs, however, showed no significant differences between sites tested likely because of high levels of variation within sites (Supplementary Table 4). The heatmap created of the most dominant genera visually displays the high levels of variation among the samples, little convergence within sites, and the dominance by a few bacterial genera, mostly within the Alphaprotebacteria class (Supplementary Figure 2). SIMPER analysis of the Bray Curtis dissimilarity index (Table 1) showed that the most dissimilar site was the vent site compared with all other sites sampled and Site 3 compared with the reference site. The NMDS ordination plot of the A. viridis data illustrates the separation of the vent site compared with all other sites (Fig. 5a). The SIMPER analysis also showed that differences between the vent site and all other sites were primarily driven by five OTUs: 1) an OTU with 85% similarity to Candidatus Cryptoprodotis (Fig. 6a), 2) an OTU with 83% similarity to the genus Anaplasma (Fig. 6a), 3) Spiroplasma spp (homology identified as 83%; data not shown), 4) Vibrio (homology identified as 92%; Fig. 6b), and 5) Thiomicrospira (homology identified as 94%; Fig. 6b). Comparisons of these genera among sites using nonparametric Kruskal-Wallis tests showed significant differences for the OTU similar to Candidatus Cryptoprodotis (X2(4) = 9.43, p = 0.049; Supplementary Figure 3), for Spiroplasma (X2 (4) = 12.897, p = 0.012; Supplementary Figure 3), and also for Thiomicrospira (X2 (4) = 13.796, p = 0.008; Supplementary Figure 3). The posthoc Dunn’s test with a Bonferroni correction showed the OTU similar to Candidatus Cryptoprodotis was significantly higher at the vent site compared with site 3 (Z = −2.647, p = 0.041; Fig. 6a; Supplementary Figure 3). However, there were no differences detected in Spiroplasma spp. among sites using this conservative posthoc test (Supplementary Figure 3). Thiomicrospira was significantly more prevalent at the vent site compared with all other sites (Z = −2.936, p = 0.017; Fig. 6b; Supplementary Figure 3).
NMDS ordination plots of bacterial community composition by OTUs for (a) Anemonia viridis (stress = 0.13; n = 3 per site, R = reference site, S1 = site 1, S2 = site 2, S3 = site 3, V = vent site) and (b) Actinia equina (stress = 0.11; n = 3 per site, R = reference site, S1 = site 1, S2 = site 2, S3 = site 3). Open black ovals represent 95% confidence intervals of the NMDS analysis.
Relative abundance of (a) Alphaproteobacteria, (b) Gammaproteobacteria, (c) Bacilli, and (d) Epsilonproteobacteria present in Anemonia viridis. Parentheses after the Genus name indicate percent homology of identification (R = reference site, S1 = site 1, S2 = site 2, S3 = site 3, V = vent site). Bacteria present in <3% relative abundance are grouped within ‘others’.
There were no significant differences in the bacterial communities of A. equina among sites (PERMANOVA F(3,8) = 0.844, R2 = 0.245, p = 0.627; Fig. 5b). Furthermore, the SIMPER dissimilarity index (Table 1) showed less dissimilarity among sites in the A. equina samples when compared with the A. viridis samples.
Relative bacterial abundance
The relative abundance of bacterial classes in A. viridis showed variations among sites, although there were no significant differences detected among sites for any of the dominant bacterial classes (Fig. 4a; Supplementary Table 5). Qualitative trends among bacterial classes and the genera that dominate those classes are reported. Alphaproteobacteria showed reduced abundance in the reference site as well as site 2, as compared with other sites (Fig. 4a). When examining the Alphaproteobacteria separately, the relative abundance of this class was dominated by an OTU 83% similar to Anaplasma in the reference site, as well as sites 1, 2, and 3 (Fig. 6a). However, the dominant Alphaproteobacteria at the vent site was the OTU 85% similar to Candidatus Cryptoprodotis. As noted previously, the Dunn’s posthoc test showed that the vent site had a significantly higher abundance of this OTU, when compared with site 3 (Z = −2.647, p = 0.041; Supplementary Figure 3). At the reference site there was a noticeable increase in Mollicutes (Fig. 4a), which was dominated by the genus Spiroplasma (Supplementary Figure 3). Gammaproteobacteria was the most common bacterial class in sites 2 and 3 (Fig. 4a). When qualitatively examining this class separately, the shift in Gammaproteobacteria was driven by an increase in Vibrio at these two sites (Fig. 6b), although no significance was detected statistically. The bacterial class Bacilli, which were dominated by Staphylococcus (Fig. 6c) had the highest relative abundance in the reference site samples, but were present in decreased levels at all other sites. Although Epsilonproteobacteria was generally rare in A. viridis (Fig. 4a) this class was dominated by chemolithoautotrophs that oxidize sulfur and showed a marked increase in the vent site compared with all other sites (Fig. 6d; data not statistically significant).
There were no significant differences among sites detected for the most dominant bacterial classes found within A. equina (Supplementary Table 6). Qualitative trends among bacterial classes and the genera that dominate those classes are reported. The relative abundance of bacterial classes of A. equina showed that Alphaproteobacteria was one of the most common classes of bacteria in the samples (Fig. 4b). Alphaproteobacteria abundances were highest at site 1 compared with all other sites, although this change was not statistically significant. The increase in relative abundance for Alphaproteobacteria at site 1 was a result of higher abundances of the OTU 83% similar to Anaplasma (Fig. 7a). Qualitatively, the relative amount of Rickettsiaceae within the Alphaproteobacteria class increased in site 2 compared with the other sites. Gammaproteobacteria remained relatively stable among sites (Fig. 7b), but the relative amount of Pseudomonas within this class increased substantially at sites closest to the vent (Fig. 7b). Mollicutes, dominated by the genus Mycoplasma (data not shown) tended to increase as pH decreased (i.e., at sites closest to the vent; Fig. 4b). Bacilli, which was dominated by Staphylococcus (Fig. 7c) was highest in the reference site compared with all of the other sites (Fig. 7c). Interestingly, Epsilonproteobacteria was the second most common class within Actinia (Fig. 7d); Arcobacter dominated this class (Fig. 7d).
Relative abundance of (a) Alphaproteobacteria, (b) Gammaproteobacteria, (c) Bacilli, (d) Epsiolonproteobacteria present in Actinia equina. Parentheses after the Genus name indicate percent homology of identification (R = reference site, S1 = site 1, S2 = site 2, S3 = site 3). Bacteria present in <3% relative abundance are grouped within ‘others’.
Discussion
The bacterial composition of the two anemone species, A. viridis and A. equina, was significantly different from each other, which may have been driven by variations in interspecific traits between host genera or because of differences in the tissues sampled between A. viridis and A. equina. Samples of A. viridis contained only tentacles, whereas A. equina samples included part of the body column because tentacles within this species are extremely small. Therefore, the difference in bacterial assemblages between the species may also be because A. equina samples contained tentacles, body wall, and potentially gonads and guts. Previous studies indicate that bacterial aggregates may be specific to certain tissue types within anemones. For example, Vibrio-like aggregates appear to congregate on the tentacles and oral disc of Aiptasia pallida38, whereas bacterial aggregates in Metridium senile were found on the tentacles39. Furthermore, another study showed bacterial aggregates only within the mesenteries of Nematostella vectensis40. Future studies should focus on whether differences detected within the present study were from host species specificity, tissue type, or both factors.
While both anemone species showed an abundance of Alphaproteobacteria, there were significantly more Gammaproteobacteria within A. viridis compared with A. equina. A. viridis is known to harbor Symbiodinium of clade A1941 as a symbiotic dinoflagellate partner, whereas A. equina does not harbor dinoflagellate endosymbionts. Bourne et al.42 showed that invertebrates containing photosynthetic symbionts often have more Gammaproteobacteria, many of which produce dimethylsulfoniopropionate (DMSP). Additionally, Ritchie43 suggests there are obligate symbioses between Symbiodinium and bacteria within the Alphaproteobacterial class, such as Ruegeria spp. and Roseobacteriales spp. Alphaproteobacteria were a dominant component of the A. viridis bacterial assemblage, further exemplifying the relationship between photosymbiosis and anemone-bacterial communities. Furthermore, the lack of Symbiodinium may require that A. equina host a more diverse assemblage of bacterial functional groups.
An additional difference in bacterial communities detected between these two species of anemones was driven by the relative abundances of the Epsilonproteobacteria class. Epsilonproteobacteria were heavily abundant in all A. equina samples, even those from the reference site. However, these bacteria were minimally present in A. viridis. The Epsilonproteobacteria within both anemones were primarily composed of sulfide oxidizing bacteria such as Sulfurimonas, Helicobacter, Sulfurovum, and Sulfospirillum. Epsilonproteobacteria such as these have been found in hydrothermal vents off of Okinawa and deep vents off the northeast Pacific Ocean, with optimal growth rates at approximately pH 6.544,45. Boatta et al.23 showed that at sites along the north shore of Levante Bay (>300 m from the vent) sulfide was absent and sulfate levels were typical for oceanic waters. The results of the present study, however, suggest that there may be some sulfide present within the seawater along the northern shore of Levante Bay that is influencing the bacterial community of the anemones. However, the low abundance of Epsilonproteobacteria within A. viridis and the high abundance of this class within A. equina may suggest that other factors may be affecting the microbial community. A. equina is often exposed to the atmospheric conditions during low tide, which may influence the bacterial assemblages found associated with this species. Atmospheric gas composition is known to affect microbial growth and abundance, knowledge that has been utilized for extending the shelf-life of perishable foods by slowing bacterial growth46. Since A. equina is exposed to air often during low tides, bacterial communities may be shaped by the atmospheric environment as well as the seawater. Arcobacter was also a large component of the Epsilonproteobacteria community of A. equina. Acrobacter are often pathogenic bacteria and aerotolerant, likely an important trait for inhabiting an intertidal anemone that is exposed to air during low tide.
There was a significant difference detected within the bacterial communities of A. viridis among sites. However, all sites showed similar bacterial communities except for samples from the vent site. Community-based analyses on the OTU data showed no difference in the bacterial assemblages when comparing the reference site, site 1, site 2, and site 3 even though the pH within these sites ranged from 8.15 to 7.44. Additionally, statistical differences detected within the genus level showed changes only within the vent sites. This lack of change within all other sampled sites indicates a high level of stability in the microbiome of A. viridis. The bacterial community of the vent site, pH of 6.05, distinctly separated from the other sites in the NMDS plot and SIMPER analysis showed that the vent site was the most different from all other sites sampled. In 2100 the pH of the oceans is predicted to decline to 7.747, a value much higher than that within site 3 (7.44), and substantially higher than the vent site (6.05). Differences that were detected within the vent site appeared to be particularly driven by the changes in composition within the Alphaproteobacteria and Gammaproteobacteria classes. Within the Alphaproteobacteria class there was an increase in the abundance of the OTU 85% similar to Candidatus Cryptoprodotis within the vent site. Candidatus Cryptoprodotis is a Rickettsia-like bacterium that has been associated with the ciliate species Pseudomicrothorax dubius48. Rickettsia-like organisms are obligate intracellular gram-negative bacterial parasites associated with a variety of vertebrate and invertebrate hosts48. Low homology within this particular OTU, however, suggests a previously undescribed genus.
The Gammaproteobacteria in A. viridis showed high diversity within sites, but mainly consisted of Vibrio spp, a known marine pathogen. This bacterium, however, did not show a trend with changing pH and increased at site 2 compared with all other sites. The vent site showed a different relative abundance of bacteria within the Gammaproteobacteria group with less Vibrio present and a high relative abundance of Thiomicrospira, an obligate chemolithoautotrophic sulfur oxidizer39. Again, the presence of Thiomicrospira is likely directly related to the high amount of hydrogen sulfide present within the vent site23,49,50,51.
Although there were no differences in the bacterial community among sites with pH values above 6.05, the vent site did harbor a significantly different bacterial community within A. viridis. The difference in the bacterial community of A. viridis at the vent site could be attributed to additional different characteristics found within that location. Extremely low pH conditions of 6.05 consistently recorded within this site may have significantly altered the microbial communities. Variations in pCO2 resulted in significantly different microbial communities of biofilms grown on glass slides52 as well as the microphytobenthic assemblage within Levante Bay25. Indeed, environmental influences shape microbial communities and drive spatial variation of microbial diversity53. Perhaps within A. viridis there is a very low pH threshold that was exceeded within the vent site, which led to altered microbial communities at this location. Alternatively, the presence of elevated trace elements may have influenced the bacterial community of the anemones at the vent site. Horwitz et al.26 showed there were differences in trace elements associated with the environment near the vent with higher levels of trace metals such as Fe, Mn, and Cr. Additionally, Fe, Cu, Pb, Co, and Sr increased in both the pedal disk and tentacles of A. viridis when trace elements were high26. Mn, Fe, Co, Ni, Zn, Cu, and Cd are considered trace elements essential for the proper function of marine microorganisms54. Indeed, these trace elements and their bio-availability may be particularly influential in changing the bacterial communities of A. viridis at the vent site. Further studies that isolate the influence of pCO2 from these trace elements are needed to completely understand the contribution each may play in controlling bacterial abundances on anemones.
Microbes living within host macroorganisms such as anemones may also be influenced by intrinsic factors. At the vent site, under high pCO2, A. viridis has significantly greater levels of autotrophy5. Suggett et al.4 also showed higher levels of photosynthesis and respiration in A. viridis under high pCO2 conditions compared with control sites. Increased levels of photosynthesis may also lead to elevated levels of photoassimilates available to bacterial communities, causing shifts at the community level. Studies of the bacterial communities within the rhizosphere of plants showed increasing sugars led to significant changes within the assemblage55. Additionally, saltwater mesocosm studies showed the addition of glucose led to significant changes in the bacterial community, specifically within the Alphaproteobacteria and Gammaproteobacteria classes56. Borell et al.41 also showed a decrease in dimethylsulfoniopropionate (DMSP) as well as superoxide dismutase (SOD) in A. viridis at sites within Levante Bay that had elevated pCO2 compared with the control site. Marine bacteria, such as Roseobacters found within the Alphaproteobacteria class, consume DMSP and catabolize DMSP to DMS, the main natural source of reduced sulfur released to the atmosphere57,58,59. These additional changes to the intrinsic composition of the host anemone and bioavailability of DMSP for catabolism, may in turn affect the bacterial community found within A. viridis. It is critical to note, however, that most studies found these physiological differences within the pH range of 8.2 to 7.44. The present study showed no differences in the bacterial assemblages of A. viridis within this range, and found that only within an extreme environment, such as the vent site, was there significant changes within the microbiome of this tolerant anemone species.
Spatial differences of the bacterial community within A. viridis must also be considered when interpreting the results of the present study. Meron et al.19 characterized the bacterial groups of A. viridis of Ischia Island, Italy, utilizing another natural CO2 vent. Interestingly, the bacterial groups that dominated the Ischia samples showed different relative abundances compared with the present samples from a nearby location. In Ischia, samples at the ambient (8.1) pH site were dominated by Firmicutes and Gammaproteobacteria. The present Vulcano samples did not pick up any OTUs within the Firmicute class. Alphaproteobacteria composed <10% of the bacteria present in Ischia, whereas our samples showed between 10 and 50% relative abundance of this bacterial group. Diversity also increased for samples under a pH of 7 in Ischia, whereas the Vulcano samples showed no significant change in diversity among sites tested. Differences in the bacterial communities between these two relatively close locations indicate that local factors causing different oceanographic characteristics are significantly influencing the bacterial communities of the A. viridis between these two locations.
There were no significant differences detected in the bacterial communities of A. equina among the different sites. The lack of change in bacterial community composition of the sampled sites for A. equina, suggests that this species, like A. viridis, may be tolerant of large variations in pCO2. Since A. equina is an intertidal species, bacteria associated with this anemone may be additionally tolerant to recurrent variations in temperature, irradiance, and salinity between tides, which could be reflected in the microbial group stability. The high relative abundance of sulfur-oxidizing bacteria, which characteristically have high growth rates at low pH conditions44,45 also, suggests an innate tolerance of the bacterial communities to low pH conditions. Site 3 regularly has a pH value of 7.44, levels much lower than predicted even 100 years from now for the oceans47, yet the bacterial communities of these A. equina samples were not different than those found at pH of 8.1. The results of the present study suggest that the bacterial community of A. equina may be unchanged as pH continues to decrease, and suggest a high tolerance of the A. equina holobiont to ocean acidification conditions.
The present study showed that there were high levels of bacterial community stability within the anemones sampled along a CO2 gradient in Levante Bay, Italy. Differences between species, however, demonstrate the significance the host plays in structuring the bacterial community, a function that is maintained throughout the pH gradient. The ambient conditions in Levante Bay are formed by a combination of the incoming seawater driven by the tides and weather conditions, and the vent emissions, which includes CO2 and trace metals. The lack of a difference of the bacterial communities in all sites other than the vent site, again indicate substantial resilience of community structure for these two anemones as pCO2 increases. In fact, the lack of a change in the bacterial community among all other sites tested suggests that these assemblages are resilient to changes in pH alone. Little difference in bacterial communities among sites outside of the vent also indicates that similar dynamics are influencing the bacterial communities even though pH is significantly different among these sites. Previous studies show that ocean acidification significantly changes the bacterial assemblages found in biofilms52 and within the sediments14 of Levante Bay. The high stability of the microbial community of both anemones tested in the present study indicates that the host itself may buffer the impact of ocean acidification on the anemones’ microbiome. Changes within the host physiology along the pCO2 gradient also did not influence bacterial communities within the host. Therefore, living within the tissues of the anemone offers stability and independence from ambient conditions at least until extreme thresholds are reached such as at the vent site. Scleractinian corals contain mechanisms within the fluid at the tissue-skeleton interface that mitigates the effects of ocean acidification60. The host anemones within the present study also provide a protective environment for the bacterial assemblages perhaps through the mitigation on intracellular pH8. Within the host, the bacteria are provided nutrition, a hospitable environment, and potentially protection from changes within the ambient environment. These results emphasize the resilience of anemones at both the macro- and micro-scale, under changing pCO2 conditions and indicate that A. viridis and A. equina will be winners under future oceanic conditions.
Additional Information
Accession Codes: Accession file SUB1478661: KX149130 - KX150371.
How to cite this article: Muller, E. M. et al. The stable microbiome of inter and sub-tidal anemone species under increasing pCO2. Sci. Rep. 6, 37387; doi: 10.1038/srep37387 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Hoegh-Guldberg, O. et al. Coral reefs under rapid climate change and ocean acidification. Science. 318, 1737–1742 (2007).
Doney, S. C., Fabry, V. J., Feely, R. A. & Kleypas, J. A. Ocean Acidification: The other CO2 problem. Ann. Rev. Mar. Sci. 1, 169–192 (2009).
Kroeker, K. J., Kordas, R. L., Crim, R. N. & Singh, G. G. Meta-analysis reveals negative yet variable effects of ocean acidification on marine organisms. Ecol. Lett. 13, 1419–1434 (2010).
Suggett, D. J. et al. Sea anemones may thrive in a high CO2 world. Glob. Chang. Biol. 18, 3015–3025 (2012).
Horwitz, R., Borell, E. M., Yam, R., Shemesh, A. & Fine, M. Natural high pCO2 increases autotrophy in Anemonia viridis (Anthozoa) as revealed from stable isotope (C, N) analysis. Sci. Rep. 5, 8779 (2015).
Towanda, T. & Thuesen, E. V. Prolonged exposure to elevated CO2 promotes growth of the algal symbiont Symbiodinium muscatinei in the intertidal sea anemone Anthopleura elegantissima. Biol. Open 1, 615–621 (2012).
van Ofwegen, L., Grasshoff, M. & van der Land, J. Octocorallia (excl. Pennatulacea) In European Register of Marine Species: a Check-list of the Marine Species in Europe and a Bibliography of Guides to Their Identification (eds. Costello MJ, Emblow C, White RJ ), 463 pp. Collection Patrimoines Naturels, 50. (Museum national d’Histoire naturelle: Paris, 2001).
Laurent, J. et al. Regulation of intracellular pH in cnidarians: Response to acidosis in Anemonia viridis. FEBS J. 281, 683–695 (2014).
Hagström, Å., Pinhassi, J. & Zweifel, U. L. Marine bacterioplankton show bursts of rapid growth induced by substrate shifts. Aquat. Microb. Ecol. 24, 109–115 (2001).
Fuhrman, J. A., Cram, J. A. & Needham, D. M. Marine microbial community dynamics and their ecological interpretation. Nat. Rev. Microbiol. 13, 133–146 (2015).
McFall-Ngai, M. et al. Animals in a bacterial world, a new imperative for the life sciences. Proc. Natl. Acad. Sci. 110, 3229–3236 (2013).
Roy, A. S. et al. Ocean acidification shows negligible impacts on high-latitude bacterial community structure in coastal pelagic mesocosms. Biogeosciences Discuss. 9, 13319–13349 (2012).
Oliver, A. E., Newbold, L. K., Whiteley, A. S. & van der Gast, C. J. Marine bacterial communities are resistant to elevated carbon dioxide levels. Environ. Microbiol. Rep. 6, 574–582 (2014).
Kerfahi, D. et al. Shallow Water marine sediment bacterial community shifts along a natural CO2 gradient in the Mediterranean Sea off Vulcano, Italy. Microb. Ecol. 67, 819–828 (2014).
Raulf, F. F. et al. Changes in microbial communities in coastal sediments along natural CO2 gradients at a volcanic vent in Papua New Guinea. Environ. Microbiol., doi: 10.1111/1462-2920.12729 (2015).
Webster, N. S. et al. Near-future ocean acidification causes differences in microbial associations within diverse coral reef taxa. Environ. Microbiol. Rep. 5, 243–251 (2013).
Webster, N. S. et al. Host-associated coral reef microbes respond to the cumulative pressures of ocean warming and ocean acidification. Nat. Publ. Gr. 1–9, doi: 10.1038/srep19324 (2016).
Morrow, K. M. et al. Natural volcanic CO2 seeps reveal future trajectories for host-microbial associations in corals and sponges. ISME J. 9, 894–908 (2015).
Meron, D., Buia, M. C., Fine, M. & Banin, E. Changes in Microbial Communities Associated with the Sea Anemone Anemonia viridis in a Natural pH Gradient. Microb. Ecol. 65, 269–276 (2013).
Banister, K. & Campbell, A. The Encyclopedia of Aquatic Life. (Equinox, 1985).
Oliver, T. A. & Palumbi, S. R. Do fluctuating temperature environments elevate coral thermal tolerance? Coral Reefs 30, 429–440 (2011).
Stabili, L., Schirosi, R., Parisi, M., Piraino, S. & Cammarata, M. The mucus of Actinia equina (Anthozoa, Cnidaria): an unexplored resource for potential applicative purposes. Mar. Drugs 13, 5276–5296 (2015).
Boatta, F. et al. Geochemical survey of Levante Bay, Vulcano Island (Italy), a natural laboratory for the study of ocean acidification. Mar. Pollut. Bull. 73, 485–494 (2013).
Johnson, V. R., Brownlee, C., Rickaby, R. E. M., Graziano, M. & Milazzo, M. Responses of marine benthic microalgae to elevated CO2 . Mar. Biol. 160, 1813–1824 (2013).
Johnson, V., Brownlee, C., Milazzo, M. & Hall-Spencer, J. Marine Microphytobenthic assemblage shift along a natural shallow-water CO2 gradient aubjected to multiple environmental stressors. J. Mar. Sci. Eng. 3, 1425–1447 (2015).
Horwitz, R., Borell, E. M., Fine, M. & Shaked, Y. Trace element profiles of the sea anemone Anemonia viridis living nearby a natural CO2 vent. PeerJ 2, e538 (2014).
Rocha, J., Coelho, F. J. R. C., Peixe, L., Gomes, N. C. M. & Calado, R. Optimization of preservation and processing of sea anemones for microbial community analysis using molecular tools. Sci. Rep. 4, 6986 (2014).
Dowd, S. E., Callaway, T. R., Wolcott, R. D. & Sun, Y. Evaluation of the bacterial diversity in the feces of cattle using 16 S rDNA bacterial tag-encoded FLX amplicon pyrosequencing (bTEFAP). BMC Microbiol. 8, 125 (2008).
DeSantis, T. Z. et al. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl. Environ. Microbiol. 72, 5069–5072 (2006).
R Development Core Team. R: A Language and Environment for Statistical Computing. R Found. Stat. Comput. Vienna Austria., doi: 10.1038/sj.hdy.6800737 (2011).
Zhang, Z. & Huang, H. Turing’s formula revisited. J. Quant. Linguist. 14, 222–241 (2007).
Marcon, E. & Hérault, B. entropart: An R Package to Measure and Partition Diversity. J. Stat. Softw. 67, 1–26 (2015).
Oksanen, J. et al. Vegan: Community Ecology Package. R package version 2.0–10. http://cran.r-project.org/package=vegan. R Packag. ver. 2.0–8 254 (2013).
Dinno, A. dunn.test: Dunn’s Test of Multiple Comparisons Using Rank Sums. R Packag. version 1.3.2. at http://cran.r-project.org/package=dunn.test (2016).
Ploner, A. Heatplus: Heatmaps with row and/or column covariates and colored clusters. R Packag. version 2.16.0. (2015).
Warnes, G. R. et al. gplots: Various R programming tools for plotting data. R Packag. version 2.17.0., doi: 10.1111/j.0022-3646.1997.00569.x (2015).
Neuwirth, E. RColorBrewer: ColorBrewer palettes. https://cran.r-project.org/web/packages/RColorBrew. R Packag. version 1.1-2. at https://cran.r-project.org/web/packages/RColorBrewer/index.html (2014).
Palincsar, E. E., Jones, W. R., Palincsar, J. S., Glogowski, M. A. & Mastro, J. L. Bacterial aggregates within the epidermis of the sea anemone Aiptasia pallida. Biol. Bull. 177, 130 (1989).
Schuett, C., Doepke, H., Grathoff, A. & Gedde, M. Bacterial aggregates in the tentacles of the sea anemone Metridium senile. Helgol. Mar. Res. 61, 211–216 (2007).
Har, J. Introducing the starlet sea anemone Nematostella vectensis as a model for investigating microbial mediation of health and disease in hexicorals. (2009).
Borell, E. M., Steinke, M., Horwitz, R. & Fine, M. Increasing pCO2 correlates with low concentrations of intracellular dimethylsulfoniopropionate in the sea anemone Anemonia viridis. Ecol. Evol. 4, 441–449 (2014).
Bourne, D. G. et al. Coral reef invertebrate microbiomes correlate with the presence of photosymbionts. ISME J. 7, 1452–1458 (2013).
Ritchie, K. B. Bacterial symbionts of corals and Symbiodinium. Benef. Microorg. Multicell. Life Forms 139–150, doi: 10.1007/978-3-642-21680-0 (2011).
Inagaki, F., Takai, K., Kobayashi, H., Nealson, K. H. & Horikoshi, K. Sulfurimonas autrotrophica gen. nov., sp. nov., a novel sulfur-oxidizing E-proteobacterium isolated from hydrothermal sediments in the Mid-Okinawa Trough. Int. J. Syst. Evol. Microbiol. 53, 1801–1805 (2003).
Huber, J. A. et al. Microbial population structures in the deep marine biosphere. Science. 318, 97–100 (2007).
Daniels, J., Krishnamurthi, R. & Rizvi, S. A review of effects of carbon dioxide on microbial growth and food quality. J. Food Prot. 48, 532–537 (1985).
IPCC. IPCC Fifth Assessment Report (AR5). IPCC (2013).
Ferrantini, F. et al. ‘candidatus Cryptoprodotis polytropus,’ A novel Rickettsia-like organism in the ciliated protist pseudomicrothorax dubius (ciliophora, nassophorea). J. Eukaryot. Microbiol. 56, 119–129 (2009).
Jannasch, H. W., Wirsen, C. O., Nelson, D. C. & Robertson, L. a. Thiomicrospira crunogena sp. nov., a colorless, sulfur-oxidizing bacterium from a deep-sea hydrothermal vent. Int. J. Syst. Bacteriol. 35, 422–424 (1985).
Sedwick, P. & Stuben, D. Chemistry of shallow submarine warm springs in an arc-volcanic setting: Vulcano Island, Aeolian Archipelago, Italy. Mar. Chem. 53, 147–161 (1996).
Amend, J. P., Rogers, K. L., Shock, E. L., Gurrieri, S. & Inguaggiato, S. Energetics of chemolithoautotrophy in the hydrothermal system of Vulcano Island, southern Italy. Geobiology 1, 37–58 (2003).
Lidbury, I., Johnson, V., Hall-Spencer, J. M., Munn, C. B. & Cunliffe, M. Community-level response of coastal microbial biofilms to ocean acidification in a natural carbon dioxide vent ecosystem. Mar. Pollut. Bull. 64, 1063–1066 (2012).
Martiny, J. B. H. et al. Microbial biogeography: putting microorganisms on the map. Nat. Rev. Microbiol. 4, 102–112 (2006).
Morel, F. M. M., Milligan, A. J. & Saito, M. A. Marine bioinorganic chemistry: The role of trace metals in the oceanic cycles of major nutrients, in The Oceans and Marine Geochemistry (ed. Elderfield, H. ) 113–143, (Elsevier, New York, 2004).
Baudoin, E., Benizri, E. & Guckert, A. Impact of artificial root exudates on the bacterial community structure in bulk soil and maize rhizosphere. Soil Biol. Biochem. 35, 1183–1192 (2003).
Øvreås, L. et al. Response of bacterial and viral communities to nutrient manipulations in seawater mesocosms. Aquat. Microb. Ecol. 31, 109–121 (2003).
Bates, T. S., Charlson, R. J. & Gammon, R. H. Evidence for the climatic role of marine biogenic sulphur. Nature 329, 319–321 (1987).
Kettle, A. J. & Andreae, M. O. Flux of dimethylsulfide from the oceans: A comparison of updated data sets and flux models. J. Geophys. Res. 105, 26, 793–26,808 (2000).
Raina, J. B., Dinsdale, E. A., Willis, B. L. & Bourne, D. G. Do the organic sulfur compounds DMSP and DMS drive coral microbial associations? Trends Microbiol. 18, 101–108 (2010).
Venn, A. A. et al. Impact of seawater acidification on pH at the tissue–skeleton interface and calcification in reef corals. Proc. Natl. Acad. Sci. 110, 1634–1639 (2013).
Acknowledgements
We thank Marco Milazzo, Riccardo Alessandro and Simone Fontana (University of Palermo), for logistical support and use of laboratories. Thank you to Joseph Mueller for sample processing and initial statistical analyses. Thank you also to Scot Dowd at MRDNA for intellectual assistance with the sequencing results. This study was funded by the Dart Foundation (KBR), Mote Marine Laboratory’s Postdoctoral Fellowship Program (EMM), and MedSeA (MF).
Author information
Authors and Affiliations
Contributions
E.M.M. collected and analyzed samples, conducted statistical analyses and wrote the main manuscript text; K.B.R. contributed to the design of the experiment, sample collection, and manuscript text; M.F. contributed to the design of the experiment, sample collection, and manuscript text. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Muller, E., Fine, M. & Ritchie, K. The stable microbiome of inter and sub-tidal anemone species under increasing pCO2. Sci Rep 6, 37387 (2016). https://doi.org/10.1038/srep37387
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37387
This article is cited by
-
Microbiomes of stony and soft deep-sea corals share rare core bacteria
Microbiome (2019)
-
Sea anemone and clownfish microbiota diversity and variation during the initial steps of symbiosis
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.