Abstract
Some species of Trichoderma are fungicolous on fungi and have been extensively studied and commercialized as biocontrol agents. Multigene analyses coupled with morphology, resulted in the discovery of T. hypoxylon sp. nov., which was isolated from surface of the stroma of Hypoxylon anthochroum. The new taxon produces Trichoderma- to Verticillium-like conidiophores and hyaline conidia. Phylogenetic analyses based on combined ITS, TEF1-α and RPB2 sequence data indicated that T. hypoxylon is a well-distinguished species with strong bootstrap support in the polysporum group. Chemical assessment of this species reveals a richness of secondary metabolites with trichothecenes and epipolythiodiketopiperazines as the major compounds. The fungicolous life style of T. hypoxylon and the production of abundant metabolites are indicative of the important ecological roles of this species in nature.
Similar content being viewed by others
Introduction
Traditionally the taxonomy of species of Trichoderma was based on morphology. Most species in this genus are usually fast growing, produce highly branched conidiophores with cylindrical to nearly subglobose phialides and ellipsoidal to globose conidia1,2,3,4. However, high morphological homoplasy in sexual state makes identification difficult, and the importance of sequence data have been increased5,6. Based on the combined phenotypic and phylogenetic analysis, about 260 species have been recognized and accepted5,6,7,8,9,10. The internal transcript spacers (ITS), translation elongation factor 1-alpha (TEF1-α) and largest subunit of RNA polymerase II (RBP2) genes are more available to recognize species within Trichoderma5,9,10. Phylogenetic analysis based on a combination of ITS, TEF1-α and RBP2 are recommended solve the problem of Trichoderma species complex and reveal taxonomic deversity2,6,7,8,9,10,11.
Fungi associated with other fungi as saprobes, commensals or parasites are termed fungicolous fungi12. These fungi usually produce rich secondary metabolites, which have been reported as an important resource for bioactive small molecule discovery such as anti-fungi, tumors, nematodes and bacteria13,14. Some metabolites produced by the species in this genus play very important ecological roles in nature15. Trichoderma species are most frequently found in vegetable matter, decaying wood and soil, plant rhizosphere, as well as on other fungi5,6,16. Fungicolous Trichoderma species comprise diversity and abundant genes associated with secondary metabolites productivity15,16. These genes are responsible to a number of secondary metabolites with pharmaceutical and biotechnological importance including peptides, peptaibols, poliketides, pyrones, siderophores and nonvolatile terpenes14,15,16,17,18,19,20,21,22. Some of secondary metabolites are responsible for survival and adaption of their habitat15,16. For example, peptaibols are small peptides of non-ribosomal origin produced by Trichoderma15 and have the ability to induce systemic resistance in plants against microbial invasion. Harzianum A is a growth-promoting trichothecene produced by Trichoderma arundinaceum17.
In the search of novel bioactive compounds, we carried out resource and diversity investigation of fungicolous fungi in China and Thailand. Two isolates of Trichoderma were obtained from stroma of Hypoxylon anthochroum. They are described, illustrated and named as a new species Trichoderma hypoxylon. Its phylogenetic positions were also explored, which inferred from sequence analyses of the combined internal transcribed spacer (ITS), partial RNA polymerase II subunit (RPB2) and translation elongation factor 1 alpha (TEF1-α) exon genes. Detailed comparisons were made between the new taxa and their related fungi. Considering T. hypoxylon is a new fungicolous species and its special life style, we reasoned that it is valued to study the chemical profiles to get the linkage to the biological roles.
Results
Phylogenetic analyses
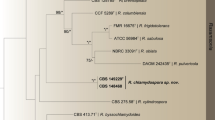
Alignment results show that the sequences ITS, RPB2 and TEF1-α of T. hypoxylon are less than 97% similar to other Trichoderma species. Based on phylogenetic analysis of single gene of ITS, RPB2 and TEF1-α, T. hypoxylon formed a clade with Trichoderma taxi and Trichoderma rubi. The position of this clade showed closed relationship with section Hypoceanum, Polysporum, Psychrophila and other species in Trichoderma (Fig. S1, and Fig. S2). The phylogenetic analysis of RPB2 showed that T. hypoxylon grouped with the Polysporum section (Fig. S3). Therefore, the combination matrix included 60 ingroup taxa of Trichoderma which are phylogenetically close to T. hypoxylon. This data matrix comprised 2839 characters. Bayesian inference (BI), maximum likelihood (ML) and maximum parsimony (MP) trees generated shared the same topology. In MP analyses, 1642 (57.8%) characters are constant, 216 (7.6%) characters are parsimony-uninformative, and 981 (34.6%) characters are parsimony informative. In MP analyses, 1642 characters were constant, 981 were parsimony-informative, and 216 variable characters were parsimony-uninformative. Maximum likelihood tree was Presented (Fig. 1).
Based on the analyses, 60 strains of Trichoderma, including our new species T. hypoxylon, formed a strongly supported group (MPBP/MLBP/BIPP = 100%/100%/1.00). They also clustered in 7 recognized subclades and a new subclade including T. hypoxylon, Brevicompactum (MLBP/BIPP = 94%/1.00), Deliquescens (MPBP/BIPP = 100%/1.00), Hypoceanum (MPBP/MLBP/BIPP = 100%/99%/1.00), Polysporum (BIPP = 1.00), Psychrophila (MPBP/MLBP/BIPP = 98%/96%/1.00), and green spore sections (MPBP/MLBP/BIPP = 100%/100%/1.00), whereas two strain of Trichoderma atroviride Bissett positioned inside Polysporum section. The tree topology is basically congruent with previous reports6.
The new species T. hypoxylon (CGMCC 3.17906, CGMCC 3.17907), and T. taxi and T. rubi formed a clade independent from other Trichoderma species with strong support (MPBP/MLBP/BIPP = 100%/100%/1.00). However, T. hypoxylon isolates were distinguished from T. axi and T. rubi with high support (MPBP/MLBP/BIPP = 100%/100%/1.00).
Taxonomy
Trichoderma hypoxylon Jing Z. Sun, Xing Z. Liu & K.D. Hyde, sp. nov.
Index Fungorum number: IF552046, Facesoffungi number: FoF: 02075, Figs 2, 3, 4.
Type:—HMAS 246918
Colonizing or hyperparasitic on stroma of Hypoxylon anthochroum. Sexual morph Undetermined. Asexual morph forming Acremonium- to Verticillium-like conidiophores and hyaline conidia. Phialides lageniform to cylindrical, hyaline, 4.5–12 × 3–3.5 μm ( = 6.5 ± 1.4 × 3.2 ± 0.25 μm, n = 30); l/w 2.5–4(−4.5) (n = 30). Mature conidia forming a head at the apex of conidiophores, hyaline, smooth-walled, unicellular, oboviod and mostly 2.8–4.3 × 2.1–2.4 μm (
= 6.5 ± 1.4 × 3.2 ± 0.25 μm, n = 30); l/w 2.5–4(−4.5) (n = 30). Mature conidia forming a head at the apex of conidiophores, hyaline, smooth-walled, unicellular, oboviod and mostly 2.8–4.3 × 2.1–2.4 μm ( = 3.8 ± 0.4 × 2.2 ± 0.10 μm, n = 30), l/w 1–2(−2.2) (n = 30). Slow growing on PDA, SNA and CMD; PDA colonies especially dense, whitish; SNA and CMA colonies consisting of concentric rings with irregular outline when cultured in darkness at 30 °C.
= 3.8 ± 0.4 × 2.2 ± 0.10 μm, n = 30), l/w 1–2(−2.2) (n = 30). Slow growing on PDA, SNA and CMD; PDA colonies especially dense, whitish; SNA and CMA colonies consisting of concentric rings with irregular outline when cultured in darkness at 30 °C.
Colonies on PDA after 10 d at 20, 25 and 30 °C dense (Fig. 4a–c), with a thick white layer of cotton-like aerial mycelia, forming concentric rings at 20 and 25 °C, however not forming concentric rings at 30 °C, odour woody, and agar not pigmented. Conidia formed within 10 d in the aerial mycelium, mature conidia gathered at the apex of conidiophores, hyaline, smooth walled, 1-celled, oboviod and mostly 2.8–4.3 × 2.1–2.4 μm ( = 3.8 ± 0.4 × 2.2 ± 0.10 μm, n = 30), l/w 1.2–2.2 (−2.5) (n = 30).
= 3.8 ± 0.4 × 2.2 ± 0.10 μm, n = 30), l/w 1.2–2.2 (−2.5) (n = 30).
Colony on CMD after 10 d at 20, 25 and 30 °C flat (Fig. 4d–f), with a thin white layer of mycelia, not forming concentric rings and conidia at 20 and 25 °C, however forming concentric rings at 30 °C, no distinctive odour, agar not pigmented. Conidiophores Acremonium- to irregularly Verticillium-like; phialides lageniform to cylindrical, hyaline, 8.5–19.5 × 2–2.51 μm ( = 13.5 ± 1.4 × 2.1 ± 0.18 μm n = 30), l/w 3.5–9(−9.5) (n = 30); Conidia formed after 20 d, mature conidia gathered at the apex of conidiophores hyaline, smooth walled, one-celled, oval, 4.8–6.3 × 3.0–3.4 μm (
= 13.5 ± 1.4 × 2.1 ± 0.18 μm n = 30), l/w 3.5–9(−9.5) (n = 30); Conidia formed after 20 d, mature conidia gathered at the apex of conidiophores hyaline, smooth walled, one-celled, oval, 4.8–6.3 × 3.0–3.4 μm ( = 4.9 ± 0.4 × 3.1 ± 0.1 μm, n = 30), l/w 1.2–1.6 (−2.0) (n = 30).
= 4.9 ± 0.4 × 3.1 ± 0.1 μm, n = 30), l/w 1.2–1.6 (−2.0) (n = 30).
Colony on SNA after 10 d at 20, 25 and 30 °C flat (Fig. 4g–i), with a thin white layer of mycelia, not forming concentric rings and conidia at 20 and 25 °C, however forming concentric rings at 30 °C, no distinctive odor, agar not pigmented.
Colony radius on PDA after 72 h at 20 °C, 28.2–30.9 mm; 25 °C, 33.4–36.3 mm; 30 °C, 0.32–1 mm; and 35 °C, 0 mm (n = 5). Colony radius on CMD after 72 h at 20 °C. 41.2–46.1 mm; 25 °C. 37.4–38.3 mm; 30 °C, 8.32–9.12 mm; and 35 °C 0 mm (n = 5). Colony radius on SNA after 72 h at 20 °C, 24.9–26.8 mm; 25 °C, 27.1–29.2 mm; 30 °C, 10.4–12.1 mm, and 35 °C, 0 mm (n = 5).
Etymology:—hypoxylon refers to the genus of host fungus Hypoxylon anthochroum.
Distribution:—Chiang Rai, Thailand
Host:—Hypoxylon anthochroum, a saprobic fungus on dead wood.
Material examination: Thailand, Chiang Rai Province, isolated from the stroma of Hypoxylon anthochroum, 5 May, 2014. MFLU16–1263 (Holotype, dried culture MFLU 16–1263!), HMAS 246918 (Isotype, dried culture HMAS 246918!) ex-type living culture, MFUCC 15–0683, CGMCC 3.17906, CGMCC 3.17907.
Notes: The asexual morph of Trichoderma hypoxylon produces Acremonium- to Verticillium-like conidiophores and hyaline conidia. Phylogenetically T. hypoxylon is related to T. taxi and T. rubi and together formed a new independent clade distinguished other Trichoderma species (Fig. 1). However, T. hypoxylon can be phylogenetically distinguished from these species. The conidia of T. hypoxylon (2.8–4.3 × 2.1–2.4 μm) are also longer than T. taxi (2.4–3.1 × 2.0–2.5 μm)23 and T. rubi (2.3–3.3 × 2.0–2.7 μm)6. Meanwhile, T. hypoxylon did not produce pigment on medium, whereas T. rubi produced brownish pigment and yellow crystals6.
Secondary metabolite analysis and characterization of compounds
HPLC analyses of extracts of T. hypoxylon cultivated on PDA medium were carried out to assess the production of secondary metabolites. Four major peaks with high yields were obtained by HPLC chromatography (Fig. 5A). To characterize compounds from T. hypoxylon, 1 liter fermentation on PDA medium were performed. After the semi-preparative reversed-phase HPLC separation step, we isolated four known compounds trichodermamide A (1), aspergillazine A (2), aspergillazine C (3) and harzianum B (4) (Fig. 5B). The assignments of four compounds were based on the published data of proton NMR24,25,26,27. Notably, the yields of harzianum B were 50 mg out of 330 mg crude extracts.
Secondary metabolite assessment of Trichoderma hypoxylon.
(a) HPLC analysis of secondary metabolite production under 330 nm wavelength. The strain was grown for 14 days at 25 °C on PDA media. (b) Four characterized compounds in this study. 1: trichodermamide A, 2: aspergillazine A, 3: aspergillazine C, 4: harzianum B.
Discussion
Traditionally, delimitation of Trichoderma species was mainly based on the morphology1, although, it could not well explain the taxonomic position of these species6,9. Phylogenetic analysis has resulted in the discovery of many new species and has been extensively used in fungal taxonomy28. Presently, more than 258 species of Trichoderma are accepted based on phylogenetic analysis5,10. However, many species of Trichoderma remain to be discovered and described5,6. To explore the taxonomic position of T. hypoxylon, phylogenetic tree containing all species of Trichoderma species was constructed and the putative position of T. hypoxylon was shown (Figs S1–S3). The single gene of ITS, RPB2 and TEF1-α could distinguish T. hypoxylon from other Trichoderma species, the results suggested that these three genes are effective in taxonomy of Trichoderma5,6,9. However, the single gene could not well delimit Longibrachiatum, Viride and some other sections in genus of Trichoderma. Multigene analysis was a popular and feasible approach to solve the problem5,6,10.
Based on analyses of the combined sequences of ITS, RPB2 and TEF1-α, 56 currently known species in Trichoderma clustered together (Fig. 1). Nine subclades, Brevicompactum, Deliquescens, Green spore group, Hypoceanum, Longibrachiatum, Polysporum, Psychrophilum, Viride and a new subclade including Trichoderma hypoxylon were recognized, which is basically congruent with the results by Jaklitsch and Voglmayr6. The new subclade contained T. hypoxylon, T. taxi and T. rubi together readily distinguished from other Trichoderma species6. Trichoderma hypoxylon clearly differed from T. taxi and T. rubi, resulting the sequences similarity of RPB2 and TEF1-α are less than 97%.
Trichoderma rubi was found as a new saprobe on stems of Rubus ulmifolius, T. taxi was reported as new endophyte of Taxus mairei, whereas T. hypoxylon was an inhabitant on stroma of Hypoxylon anthochroum. In spite of their ecological niches, it showed closed phylogenetic relationship (Fig. 1, Figs S1–S3) and morphological similarity. However, they are a little different in conidiophores and conidia size, and T. hypoxylon did not produce pigment on PDA medium, whereas T. rubi produced brownish pigment and yellow crystals on PDA medium6.
Trichoderma species are a rich source of secondary metabolites14,15,16,17,18,19,20,21,22,29, probably resulting their environments adaptation and lifestyles15,16,30. Comparative genome analysis revealed that fungicolous T. atroviride and T. virens are enriched in secondary metabolism-related genes compared with the biomass-degrading Trichoderma reesei30. Chemical analysis also showed that these fungicolous species could produce more peptaibols, peptides, polyketides, pyrones, siderophores, terpenoids/steroids than those non-fungicolous Trichoderma15,30. These compounds are ecologically and commercially important for their antimicrobial and anti-cancer properties, as well as their ability to induce systemic resistance in plants against microbial invasion15,17,31. As a new fungicolous species, the chemical diversity of T. hypoxylon aroused our attentions. Therefore, the secondary metabolites of this fungus were evaluated. Four major compounds were characterized (Fig. 5). Trichodermamide and aspergillazines are two kind of modified dipeptides15,25. Trichodermamide A has been found from a marine-derived T. virens24, it is also be obtained from marine-derived fungi Spicaria elegans25 and Neosartorya pseudofischeri32 and endophytic fungus Trichoderma spirale33. The yield of trichodermamide A produced by these fungi were 1.58 mg/L24, 0.73 mg/L25, 0.12 mg/L32 and 8.98 mg/L33 individually. In this study, we found that trichodermamide A was one of major compounds in the terrestrial fungus T. hypoxylon. It suggested that Trichoderma species are important resource for exploration of trichodermamide. Aspergillazines were firstly reported from a soil fungi Aspergillus unilateralis (MST-F867)26, it showed antibacterial and anti-cancer activity15,19. Aspergillazines A been found from two marine-derived fungi Spicaria elegans26 and T. virides25, in which the productive rate of aspergillazines A was 3.17 mg/L26 and 0.47 mg/L25. This compound has been found co-occurred with trichodermamide A in T. virides25. Trichothecenes are a well-studied class of sesquiterpene-based mycotoxins34. They are potent cytotoxins to eukaryotic cells which are mainly produced by fungal species of Fusarium, Myrothecium and Trichoderma in order Hypocreales15,34. Harzianum B is one of trichothecenes, which inhibit eukaryotic cell growth27 and have the cytotoxic, antibiotic, and anthelmintic activities35. It is found that in a Hypocrea sp. (sexual morph of Trichoderma) strain F000527 yield of 18.13 mg/L27, while our results showed harzianum B was extremely high in T. hypoxylon with yield of 50 mg/L. The high amount of trichothecenes in T. hypoxylon, indicating that it has antagonistic potential against fungal hosts.
Materials and Methods
Isolates and specimens
Samples were collected on 5 May 2014 in Chiang Mai Province, Thailand. Trichoderma strains were isolated from the host by single spore isolation as detailed in Chomnunti et al.36. The holotype is deposited in the Herbarium of Mae Fah Luang University and the isotype in the Herbarum of Mycology, Chinese Academy of Science (HMAS, Beijing, China). Ex-type living cultures are deposited in the Culture Collection of Mae Fah Luang University (MFLUCC, Chiang Rai, Thailand) and the China General Microbiological Culture Collection Center (CGMCC, Beijing, China). Facesoffungi and Index Fungorum numbers are registered as explained in Jayasiri et al.37 and Index Fungorum38.
Morphological characterization
Methods and morphology were described basically following counterparts by Jaklitsch & Voglmayr6 Colony radius and characteristics were determined on PDA as detailed in Manamgoda et al.6 cornmeal dextrose agar (CMD; Difco cornmeal agar + 2% w/v dextrose)10 and a defined low nutrient agar (SNA)4 at 20, 25 and 30 °C in darkness for 7 d, then exposed to artificial light to stimulate conidia formation until 10 d. Microscopic observations and measurements were made from preparations mounted in 50% lactic acid. Photographs were taken with a Nikon DS–Fi2 CCD (Nikon, Japan) connected to a Nikon 80i microscope (Nikon, Japan) for anatomical structures. The statistics presented here are based on measurement of 30 mature conidia (±S.D.) and 30 phialides (±S.D.) at 100× magnification. To assess and describe their structure and morphology of conidiophores were taken from the edge of conidiogenous pustules or fascicles. Conidia were studied from cultures after 10 d of incubation.
DNA extraction, PCR amplification and sequencing
Approximately 50 mg of fungal material from each culture was placed in 600 μl of 2% CTAB buffer and ground with a plastic pestle. Genomic DNA was extracted using a modified CTAB extraction protocol39. Three primer pairs, ITS5 and ITS440, fRPB2–5 f and fRPB2–7cr23, EF983F and EF2218R41 were separately used to amplify fragments of ITS, RPB2 and TEF1-α. Each PCR was performed on a Votix thermal cycler (Bio-Rad, CA, USA) using easy tag (Tiangen, Beijing, China) in a final volume of 50 μl containing 10 μmol of each primer and 2 μl of DNA (10 ng/L). Reactions were run with positive and negative controls to ensure accuracy and to detect contamination. Automated sequencing was performed by Sino Geno Max Co. (Beijing, China). The sequences used in this study are deposited in GenBank under the accession number provided in Table S1.
Phylogenetic analysis
The ITS and TEF1-α data sets used sequences of isolates CGMCC 3.17906, CGMCC 3.17907 and reference sequences were downloaded from GenBank (Table S1). The ITS, TEF1-α and RPB2 data sets were aligned by MAFFT ver.7.03 using the Q-INS-I strategy, individually42. The ambiguous areas of alignment were located and removed using Gblocks 0.91b43. Previous phylogenetic analysis of ITS, RPB2 and TEF1-α sequence data from 260 Trichoderma species was conducted seperately with N. berolinensis and N. eustromatica as outgroup. Then, single and combined genes analyses of ITS, TEF1-α and RPB2 sequence data of 60 phylogenetic closed Trichoderma species in Brevicompactum, Deliquescens, Hypoceanum, Longibrachiatum, Polysporum, Psychrophila and green spore sections were carried out. N. berolinensis and N. eustromatica were arranged as outgroup taxa.
Maximum parsimony (MP) analysis was conducted by PAUP 4.0b1044 using a heuristic search with tree-bisection-reconnection branch swapping. All characters were treated as unordered and unweighted, gaps were treated as missing data, sequences were auto-increased and Maxtrees was 5,000. Topological confidence of resulted trees was tested by bootstrap proportion with 1,000 replicates, each with 100 replicates of random addition. Bootstrap proportion (BP) higher than 50% from maximum parsimony analysis from PAUP are given.
Maximum-likelihood (ML) analysis was performed in RAxML45 implemented in raxml GUI v.1.346. GTRGAMMAI was specified as the model. The analysis was run with a rapid bootstrap analysis using a random start with rapid bootstrap analysis with 1,000 replicates. Bootstrap proportion (BP) higher than 50% from maximum likelihood analysis from RAxML are given.
Bayesian Inference (BI) analysis was performed with MrBayes 3.1.247 using Markov chain Monte Carlo (MCMC) algorithm. Appropriate nucleotide substitution models was determined by MrModeltest 2.348 and the best fit model “GTR + I + G” was selected by Akaike Information Criterion for the investigated data set. Six chains (one cold and three heated) of 135,000 Markov chain Monte Carlo generations were run, sampling every 100 generation resulting in 1,350 total trees (in two simultaneous analyses). The initial 337 trees (25%) were discarded as burn-in phase of the analyses, and the remaining trees in each analysis were used to calculate posterior probabilities (PP) in the majority rule consensus tree49, posterior probabilities greater than 0.95 are given.
All trees were viewed in TreeView 1.6.650 and revised in Adobe Illustrator CS5.
Analytical methods and equipment overview
1H-NMR spectra were recorded on a Bruker Avance-500 spectrometer using TMS as internal standard, and chemical shifts were recorded as δ values. ESI-MS utilized on an Agilent Accurate-Mass-QTOF LC/MS 6520 instrument. HPLC analysis was performed on a Waters HPLC system (Waters e2695, Waters 2998, Photodiode Array Detector) using an ODS column (C18, 250 × 4.6 mm, YMC Pak, 5 μm) with a flow rate of 1 mL/min.
Fermentation and isolation
The fungal strain was cultured on 20 slants of potato dextrose agar at 25 °C for seven days. The fermented PDA substrate was extracted repeatedly with ethyl acetate by exhaustive maceration (4 × 200 mL), and the organic solvent was evaporated to dryness under vacuum to afford the crude extract (330 mg). The residue was fractionated by Sephadex LH-20 CC using CH2Cl2: Acetone (V:V = 1:1) elution to obtain fifteen fractions 1–15. The fractions 3–7 (200 mg) was separated by semi-preparative RP-HPLC (Waters Symmetry PrepTM C18 column; 7 μm; 7.8 × 300 mm; 45% MeOH in H2O over 45 min; 2 mL/min) to afford 4 (tR 34.5 min; 50.0 mg). The fraction 11 (35 mg) was separated by semi-preparative RP-HPLC (Waters Symmetry PrepTM C18 column; 7 μm; 7.8 × 300 mm; from 22% CH3CN to 30% CH3CN in H2O over 45 min; 2 ml/min) to afford 1 (tR 18.8 min; 5.0 mg), 2 (tR 22.4 min; 8.0 mg), and 3 (tR 41.1 min; 4.5 mg).
Trichodermamide A (1)
White powder (MeOH)25,26; 1H NMR (500 MHz, in Acetone-d6) δH: 2.72 (dd, J = 19.4, 2.2, H-3a); 2.28 (d, J = 19.4, H-3b); 4.48 (m, H-5); 5.60 (ddd, J = 10.4, 2.0, 2.0, H-6); 5.55 (ddd, J = 10.4, 2.0, 2.0, H-7); 4.24 (m, H-8); 4.15 (dd, J = 7.8, 2.2, H-9); 8.58 (s, H-3′); 7.41 (d, J = 8.8, H-5′); 7.12 (d, J = 8.8, H-6′); 3.96 (s, 7′-OCH3); 3.91 (s, 8′-OCH3); 9.40 (s, CONH); Positive ESIMS: m/z 433.1 [M+H]+.
Aspergillazine A (2)
Yellow powder (MeOH)26,27; 1H NMR (500 MHz, in Acetone-d6) δH: 3.14 (d, J = 11.7, H-3a); 2.42 (d, J = 11.7, H-3b); 4.16 (d, J = 4.9, H-5); 5.95 (dd, J = 10.0, 4.9, H-6); 6.07 (dd, J = 10.0, 4.9, H-7); 4.33 (m, H-8); 4.23 (br s, H-9); 6.90 (s, H-3′); 7.09 (d, J = 8.8, H-5′); 6.69 (d, J = 8.8, H-6′); 3.90 (s, 7′-OCH3); 3.83 (s, 8′-OCH3); 9.38 (s, CONH); 9.90 (s, -O-NH); Positive ESIMS: m/z 449.1 [M + H]+.
Aspergillazine C (3)
Yellow powder (MeOH)27; 1H NMR (500 MHz, in Acetone-d6) δH: 2.96 (d, J = 13.9, H-3a); 2.40 (d, J = 13.9, H-3b); 4.31 (m, H-5); 5.42 (dd, J = 10.0, 1.8, H-6); 5.49 (dd, J = 10.0, 1.9, H-7); 4.16 (dd, J = 7.8, 1.2, H-8); 3.77 (d, J = 8.0, H-9); 6.85 (s, H-3′); 7.11 (d, J = 8.8, H-5′); 6.69 (d, J = 8.8, H-6′); 3.90 (s, 7′-OCH3); 3.84 (s, 8′-OCH3); Positive ESIMS: m/z 451.1 [M + H]+.
Harzianum B (4)
White powder (MeOH)28; 1H NMR (500 MHz, in CDCl3) δH: 3.84 (d, J = 5.2, H-2); 2.57 (dd, J = 15.5, 7.8, H-3a); 2.03 (ddd, J = 15.5, 5.3, 3.6, H-3b); 5.65 (dd, J = 7.8, 3.6, H-4); 1.92‒1.96 (m, H-7a); 1.40‒1.43 (m, H-7b); 1.97‒1.99 (m, H-8); 5.41 (d, J = 5.8, H-10); 3.63 (d, J = 5.8, H-11); 3.13 (d, J = 4.0, H-13a); 2.83 (d, J = 4.0, H-13b); 0.72 (s, H3-14); 0.95 (s, H3-15); 1.71 (s, H3-16); 6.03 (d, J = 15.4, H-2′); 7.32 (dd, J = 15.4, 10.2, H-3′); 6.61 (dd, J = 10.2, 10.2, H-4′); 6.64 (dd, J = 10.2, 10.2, H-5′); 7.32 (dd, J = 15.4, 10.2, H-6′); 6.07 (d, J = 15.4, H-7′); Positive ESIMS: m/z 401.2 [M + H]+.
Additional Information
How to cite this article: Sun, J. et al. A new species of Trichoderma hypoxylon harbours abundant secondary metabolites. Sci. Rep. 6, 37369; doi: 10.1038/srep37369 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Samuels, G. J. Trichoderma: a review of biology and systematics of the genus. Microbiol. Res. 100, 923–935 (1996).
Zhu, Z. X. & Zhuang, W. Y. Three new species of Trichoderma with hyaline ascospores from China. Mycologia 107, 328–345 (2015).
Jaklitsch, W. M. European species of Hypocrea part II: species with hyaline ascospores. Fungal Divers. 48, 1–250 (2011).
Jaklitsch, W. M. European species of Hypocrea Part I. The green-spored species. Stud. Mycol. 63, 1–91 (2009).
Qin, W. T. & Zhuang, W. Y. Seven wood-inhabiting new species of the genus Trichoderma (Fungi, Ascomycota) in Viride clade. Sci. Rep- UK 6, 27074 (2016).
Jaklitsch, W. M. & Voglmayr, H. Biodiversity of Trichoderma (Hypocreaceae) in Southern Europe and Macaronesia. Stud. Mycol. 80, 1–87 (2015).
Samuels, G. J. et al. The Longibrachiatum clade of Trichoderma: a revision with new species. Fungal Div. 55, 77–108 (2012).
Chaverri, P., Castlebury, L. A., Samuels, G. J. & Geiser, D. M. Multilocus phylogenetic structure within the Trichoderma harzianum/Hypocrea lixiicomplex. Mol. Phylogenet. Evol. 27, 302–313 (2003).
Druzhinina, I. S., Kopchinskiy, A. G. & Kubicek, C. P. The first 100 Trichoderma species characterized by molecular data. Mycoscience 47, 55–64 (2006).
Bissett, J., Gams, W., Jaklitsch, W. & Samuels, G. J. Accepted Trichoderma names in the year 2015. IMA fungus 6, 263 (2015).
Qin, W. T. & Zhuang, W. Y. Two new hyaline-ascospored species of Trichoderma and their phylogenetic positions. Mycologia 107, 205–214 (2016).
Kirk, P. M., Cannon, P. F., Minter, D. W., Stalpers, J. A. & Ainsworth, G. C. Ainsworth & Bisby’s dictionary of the fungi. 10 edition. p227 (CABI, 2008).
Schmidt, L. E., Gloer, J. B. & Wicklow, D. T. Solanapyrone analogues from a Hawaiian fungicolous fungus. J. Nat. Prod. 70, 1317–1320 (2007).
Reino, J. L., Guerrero, R. F., Hernandez-Galan, R. & Collado, I. G. Secondary metabolites from species of the biocontrol agent Trichoderma. Phytochem. Rev. 7, 89–123 (2008).
Contreras-Cornejo, H. A., Macías-Rodríguez, L., del-Val, E. & Larsen, J. Ecological functions of Trichoderma spp. and their secondary metabolites in the rhizosphere: interactions with plants. FEMS Microbiol. Ecol. 92, fiw036 (2016).
Druzhinina, I. S. et al. Trichoderma: the genomics of opportunistic success. Nat. Microbiol. Rev. 16, 749–759 (2011).
Malmierca, M. G. et al. Involvement of Trichoderma trichothecenes in the biocontrol activity and induction of plant defense-related genes. Appl. Environ. Microb. 78, 4856–4868 (2012).
Vinale, F. et al. Trichoderma secondary metabolites active on plants and fungal pathogens. The Open Mycol. J. 8, 127–139 (2014).
Patil, A. S., Patil, S. R. & Paikrao, H. M. Trichoderma secondary metabolites: their biochemistry and possible role in disease management. In Microbial-mediated induced systemic resistance in plants (eds Choudhary, D. K. & Varma, A. ) Ch. 6, 69–102 (Springer Singapore, 2016).
Degenkolb, T., Von Doehren, H., Fog Nielsen, K., Samuels, G. J. & Brueckner, H. Recent advances and future prospects in peptaibiotics, hydrophobin, and mycotoxin research, and their importance for chemotaxonomy of Trichoderma and Hypocrea. Chem. Biodivers. 5, 671–680 (2008).
Vinale, F. et al. Trichoderma–plant–pathogen interactions. Soil Biol. Biochem. 40, 1–10 (2008).
Velázquez-Robledo, R. et al. Role of the 4-phosphopantetheinyl transferase of Trichoderma virens in secondary metabolism and induction of plant defense responses. Mol. Plant Microbe In. 24, 1459–1471 (2011).
Zhang, C. L., Liu, S. P., Lin, F. C., Kubicek, C. P. & Druzhinina, I. S. Trichoderma taxi sp. nov., an endophytic fungus from Chinese yew Taxus mairei. FEMS Microbiol. Lett. 270, 90–96 (2007).
Garo, E. et al. Trichodermamides A and B, cytotoxic modified dipeptides from the marine-derived fungus Trichoderma virens. J. Nat. Prod. 66, 423–426 (2003).
Liu, R., Gu, Q. Q., Zhu, W. M., Cui, C. B. & Fan, G. T. Trichodermamide A and aspergillazine A, two cytotoxic modified dipeptides from a marine-derived fungus Spicaria elegans. Arch. Pharm. Res. 28, 1042–1046 (2005).
Capon, R. J. et al. Aspergillazines A–E: novel heterocyclic dipeptides from an Australian strain of Aspergillus unilateralis. Org. Biomol. Chem. 3, 123–129 (2005).
Jin, H. Z. et al. Harzianums A and B produced by a fungal strain, Hypocrea sp. F000527, and their cytotoxicity against tumor cell lines. J. Asian Nat. Prod. Res. 9, 203–207 (2007).
Taylor, J. W. et al. Phylogenetic species recognition and species concepts in fungi. Fungal Genet. Biol. 31, 21–32 (2000).
Mukherjee, P. K., Horwitz, B. A. & Kenerley, C. M. Secondary metabolism in Trichoderma–a genomic perspective. Microbiology 158, 35–45 (2012).
Kubicek, C. P. et al. Comparative genome sequence analysis underscores mycoparasitism as the ancestral life style of Trichoderma. Genome boil. 12, R40 (2011).
Yang, D. et al. Genome sequence and annotation of Trichoderma parareesei, the ancestor of the cellulase producer Trichoderma reesei. Genome Announ. 3, e00885–15 (2015).
Lan, W. J. et al. Five new cytotoxic metabolites from the marine fungus Neosartorya pseudofischeri. Mar Drugs 14(1), 18 (2016).
Li, D. L., Chen, Y. C., Tao, M. H., Li, H. H. & Zhang, W. M. Two new octahydronaphthalene derivatives from Trichoderma spirale, an endophytic fungus derived from Aquilaria sinensis. Helvetica Chimica Acta 95, 805–809 (2012).
Ueno, Y. Trichothecenes. Chemical, biological and toxicological aspects. (Elsevier, 1983).
Ayers, S. et al. Peptaibols from two unidentified fungi of the order Hypocreales with cytotoxic, antibiotic, and anthelmintic activities. J Pept Sci. 18, 500–510 (2012).
Chomnunti, P. et al. The sooty moulds. Fungal Divers. 66, 1–36 (2014).
Jayasiri, S. C. et al. The Faces of Fungi database: fungal names linked with morphology, phylogeny and human impacts. Fungal Divers. 74, 3–18 (2015).
Index Fungorum (2016) http://www.indexfungorum.org/Names/Names. asp. Accessed 21 March (2016).
Rogers, S. O. & Bendich, A. J. Extraction of total cellular DNA from plants, algae and fungi. In Plant molecular biology manual (eds Stanton, B. G. et al.) 183–190 (Springer, 1994).
Samuels, G. J. et al. Trichoderma stromaticum sp. nov., a parasite of the cacao witches broom pathogen.Mycol. Res. 104, 760–764 (2000).
Overton, B. E., Stewart, E. L., Geiser, D. M. & Jaklitsch, W. M. Systematics of Hypocrea citrina and related taxa. Stud. Mycol. 56, 1–38 (2006).
Katoh, K. & Standley, D. M. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Bio. Evol. 30, 772–780 URL http://mafft.cbrc.jp/alignment/software/ (2013).
Castresana, J. Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol. Bio. Evol. 17, 540–552 URL http://molevol.cmima.csic.es/castresana/Gblocks.html (2000).
Swofford, D. L. PAUP*. Phylogenetic analysis using parsimony (*and other methods). Version 4. 1–142. URL http://www.paup.csit.fsu.edu/ (2002).
Stamatakis, A. RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinfo. 22, 2688–2690 (2006).
Silvestro, D. & Michalak, I. RaxmlGUI: a graphical front-end for RAxML. Org. Divers. Evol. 12, 335–337 URL https://github.com/dsilvestro/raxmlGUI (2012).
Ronquist, F. et al. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Bio. 61, 539–542 URL http://mrbayes.sourceforge.net/ (2012).
Nylander, J. MrModeltest 2.3 README, Uppsala University, Uppsala, Sweden. URL https://github.com/nylander/MrModeltest2 (2008).
Cai, L., Jeewon, R. & Hyde, K. D. Phylogenetic investigations of Sordariaceae based on multiple gene sequences and morphology. Mycol. Res. 110, 137–150 (2006).
Rambaut, A. FigTree v1. 4.2. Edinburgh: University of Edinburgh. URL http://tree.bio.ed.ac.uk/software/figtree/ (2014).
Acknowledgements
We thank Dr. Shaun Pennycook for advising on the fungal name, and Prof. Eric H. C. McKenzie for revision of manuscript. This research was jointly supported by grants from “the 100 Talents Project” of Chinese Academy of Sciences and Natural Science Foundation of China (No. 31600024 and No. 21302216).
Author information
Authors and Affiliations
Contributions
X.L., W.Y. and K.H. initiated and coordinated the project, and also edited the manuscript. J.S. isolated the fungus and did the morphology and phylogenetic analyses and initially wrote the manuscript. Y.P. extracted genomic DNA and amplified genes. E.L. and W.L. studied the metabolites.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Sun, J., Pei, Y., Li, E. et al. A new species of Trichoderma hypoxylon harbours abundant secondary metabolites. Sci Rep 6, 37369 (2016). https://doi.org/10.1038/srep37369
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37369
This article is cited by
-
Discovery and genetic identification of amphiphilic coprogen siderophores from Trichoderm hypoxylon
Applied Microbiology and Biotechnology (2021)
-
Fungicolous fungi: terminology, diversity, distribution, evolution, and species checklist
Fungal Diversity (2019)
-
Discovery from a large-scaled survey of Trichoderma in soil of China
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.