Abstract
A photo/thermal-switchable supramolecular nanoparticles assembly has been constructed based on an inclusion complex between anionic pillar[5]arene 2C-WP5A and azobenzene derivative Azo-py-OMe (G). The novel anionic pillar[5]arene-based host-guest inclusion complexation was investigated by the 1H NMR titration, 2D ROESY and isothermal titration microcalorimetry (ITC) showing high association constant (Ka) of (2.60 ± 0.06) × 104 M−1 with 1:1 binding stoichiometry. Furthermore, the supramolecular nanoparticles assembly can be conveniently obtained from G and a small amount of 2C-WP5A in aqueous solution, which was so-called “host induced aggregating (HIA)”. The size and morphology of the supramolecular nanoparticles assembly were characterized by TEM and DLS. As a result of the photo/thermal-isomerization of G included in the cavity of 2C-WP5A, the size of these nanoparticles could reversibly change from ~800 nm to ~250 nm, which could switch the solution of this assembly from turbid to clear.
Similar content being viewed by others
Introduction
Recently, size switchable materials have attracted much attention, as size is the fundamental and crucial factor that determined the properties of materials, for example as the noble metal nanoparticles1, polymeric microparticles2 or colloidal superparticles3. The stimuli-responsive molecules might be very useful to build up the size switchable systems4,5. Azobenzene is one of the most attractive stimuli-responsive molecules6,7, because of its reversible isomerization between trans and cis conformation upon external stimuli, such as mechanical stress8, electrostatic stimulation9,10, as well as light irradiation11,12,13. So the supramolecular assemblies involved azobenzene have various applications in intelligent membranes14,15, optical memories16,17,18 and biological systems19,20,21 and so on. A large number of photo-responsive systems based on the azobenzene and macrocycles have been reported22,23,24,25,26, such as, a photo regulable ion-extraction and ion-transport based on azobenzene-crown ethers27, a photo-responsive artificial muscle based on host-guest complex of α-cyclodextrin and azobenzene28,29, and a dual stimuli-responsive self-assembly supramolecular nanoparticles based on the ternary host-guest complexation between cucurbit[8]uril, a methyl viologen polymer, and mono- and multivalent azobenzene functionalized molecules30.
As a new class of macromolecules, pillar[n]arenes (5–15) have been extensively studied from their synthesis, functionalization and supramolecular chemistry since 200831,32,33,34,35,36. The first photo-responsive host-guest complexation based on pillar[6]arene and azobenzene-guest was reported by Huang in 201237. Thereafter, the author constructed a dual-responsive supra-amphiphilic polypseudo-rotaxane based on pillar[7]arene and azobenzene derivative in aqueous solution38. Meanwhile, Ogoshi also reported a photoreversible switching of the lower critical solution temperature (LCST) in host-guest system of pillar[6]arene and azobenzene derivative39. Up to now, most reported photo-responsive systems based on azobenzene and pillar[n]arene are mainly focus on pillar[6,7]arene40,41,42. Nevertheless, the diameter of the internal cavity of pillar[5]arene is ~4.7 Å, which is equal to α-cyclodextrin (~4.7 Å)43. In accordance with common sense, it is reasonable that the pillar[5]arene and azobenezene derivative could form host-guest inclusion complexation. Ogoshi has studied the threading process of pillar[5]arene onto a viologen derivative containing an azobenzene end-group44. But there has been no direct proof to reveal the host-guest complexation between anionic pillar[5]arene and photo-responsive azobenzene derivatives. Considering the easier access to pillar[5]arene compared with pillar[6,7]arene and the aforementioned reasons, it is necessary to investigate the host-guest complexation and assembly behavior based on pillar[5]arene and azobenzene derivatives. In addition, to the best of our knowledge, size switchable supramolecular nanoparticles formed by host-guest interactions between anion pillar[5]arene and azobenzene derivatives have been underexplored.
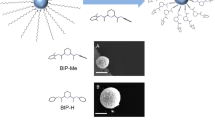
Herein, we present a feasible strategy for fabricating photo/thermal-responsive size switchable supramolecular nanoparticles assembly based on the unprecedented host-guest inclusion complex between anionic pillar[5]arene 2C-WP5A and azobenzene derivation G in aqueous solution (Fig. 1). The design proposed here combines the following advantages: (1) the percarboxylatopillar[5]arenes is the most used anionic pillar[5]arene, which can be obtained easily and efficiently; (2) differing from the α-cyclodextrin, either trans- and cis- azobenzene derivatives G could be included in 2C-WP5A with similar affinity, which could result in the only reversible change of the nanoparticle’s size without disassembling; (3) the size change by photo/thermal-stimulation would result in the reversible switching the transparency of solution from turbid to clear.
Results and Discussion
Host-Guest Complexation For 2C-WP5A and trans-G
The host-guest complexation of 2C-WP5A and trans-G was first investigated by 1H NMR experiment in D2O at 25 °C (Fig. 2b). Signals for the protons on trans-G were significantly shifted upfield in the presence of 1 equiv. 2C-WP5A. This result suggested that the formation of the threaded structure 2C-WP5A trans-G, in which these protons of the guest molecule were shielded by the aromatic rings in 2C-WP5A. The Job’s plot was employed to find the binding stoichiometry of 2C-WP5A
trans-G, in which these protons of the guest molecule were shielded by the aromatic rings in 2C-WP5A. The Job’s plot was employed to find the binding stoichiometry of 2C-WP5A trans-G, the top point of the curve was appeared at the molar fraction Xguest = 0.5 (see Supplementary Fig. S12), indicating the 1:1 binding stoichiometry between 2C-WP5A and G.
trans-G, the top point of the curve was appeared at the molar fraction Xguest = 0.5 (see Supplementary Fig. S12), indicating the 1:1 binding stoichiometry between 2C-WP5A and G.
Partial 1H NMR spectra (D2O, 25 °C, 400 MHz) of (a) trans-G (1.6 mM); (b) trans-G (1.6 mM) and 2C-WP5A (1.6 mM); (c) trans-G (1.6 mM) and 2C-WP5A (1.6 mM) after irradiation at 365 nm for 10 min; (d) trans-G (1.6 mM) after irradiation at 365 nm for 10 min; (e) trans-G (1.60 mM) and 2C-WP5A (1.6 mM) after further heating at 80 °C for 1 h.
Interestingly, comparing to the chemical shift of the protons on the pyridinium unit, the signals of the protons on the azobenzene unit changed more obviously. The complexation-induced chemical shifts (Δδ) followed the order (see Supplementary Fig. S13): Hh > Hg > Hi > Hd > He = Hc > Hf > Hb > Ha. These shifts of methyl and both aromatic rings of the azobenzene unit were shielded by the pillar[5]arene, and the ring A is more shielded than others, which indicated that the aromatic rings of 2C-WP5A were located at the azobenzene unit of trans-G especially at ring A, instead of pyridinium unit. Although we failed to get the single crystal of the 2C-WP5A trans-G, the 2D ROESY NMR experiment (see Supplementary Fig. S14) also supported our speculation of the binding mode. A 2D ROESY spectrum of an equimolar (5 mM) mixture of trans-G and 2C-WP5A in D2O was obtained to confirm the peak assignments. NOE correlation signals were observed between protons Hg, He and Hf on the azobenzene unit of trans-G and side chain protons H3 and phenyl moieties protons H2 on 2C-WP5A. The results showed that azobenzene unit was indeed threaded in the cavity of anionic 2C-WP5A, which was consistent with the above-mentioned 1H NMR results. To further confirm this result, we synthesized another guest molecule G′ with 1,4-diazabicyclo[2.2.2]octane group. The additional experiment results (see Supplementary Figs S15 and S16) also supported our conclusion that the azobenzene unit was positioned inside the cavity of 2C-WP5A. Recently, an inclusion complex of azo dye and cationic pillar[5]arenes was reported by Stoikov, which is also confirmed that azobenzene unit threaded in the cavity of pillar[5]arene45.
trans-G, the 2D ROESY NMR experiment (see Supplementary Fig. S14) also supported our speculation of the binding mode. A 2D ROESY spectrum of an equimolar (5 mM) mixture of trans-G and 2C-WP5A in D2O was obtained to confirm the peak assignments. NOE correlation signals were observed between protons Hg, He and Hf on the azobenzene unit of trans-G and side chain protons H3 and phenyl moieties protons H2 on 2C-WP5A. The results showed that azobenzene unit was indeed threaded in the cavity of anionic 2C-WP5A, which was consistent with the above-mentioned 1H NMR results. To further confirm this result, we synthesized another guest molecule G′ with 1,4-diazabicyclo[2.2.2]octane group. The additional experiment results (see Supplementary Figs S15 and S16) also supported our conclusion that the azobenzene unit was positioned inside the cavity of 2C-WP5A. Recently, an inclusion complex of azo dye and cationic pillar[5]arenes was reported by Stoikov, which is also confirmed that azobenzene unit threaded in the cavity of pillar[5]arene45.
The photo/thermal responsive properties of G
To further investigation of the photo-responsive properties of the host-guest complex and supramolecular assembly of 2C-WP5A and G, we firstly examined the photoisomerization behavior of G by UV/vis (see Supplementary Fig. S17) and 1H NMR (Fig. 2d). The UV/vis spectrum of trans-G has a strong absorption maximum at 351 nm, which corresponds to the π-π* transition of the trans form. When the solution of trans-G was irradiated at 365 nm for 2 min, the main absorption band bleached greatly. Meanwhile, slightly increase in a new band at 432 nm, which is ascribed to the n-π* transition of the cis form of the azobenzene. The changes in the UV/vis spectrum indicated that the photoisomerization of G from the trans to cis conformation. Moreover, the trans/cis photoisomerization of G can be quantitatively calculated by integration of the signals in the 1H NMR spectrum. After irradiation with UV light at 365 nm for 10 min to ensure the fully transformed, a series of new signals appeared which is corresponding to the cis form, and the ratio of the trans:cis reached up to 10:90 (Fig. 2d). It is remarkable that G has not only photo-responsive but also thermal-responsive performances46. The continuous heating of the irradiated solution at 80 °C for one hour resulted in the regeneration of original protons signals of G, suggesting the recovery of trans form of G.
The photo/thermal responsive properties of 2C-WP5A G
G
After irradiation the solution of 1:1 2C-WP5A trans-G with UV light at 365 nm for 10 min, some new signals appeared as shown in Fig. 2c. In comparison to the signals of individual G at the same experimental condition (Fig. 2d), we observed that the chemical shift of proton Hi* shifted upfield from 3.75 ppm to 1.71 ppm, and the peaks of the protons related to the azobenzene altered slightly (he*, hf*, hg* and hh* Δδ = 0.15, 0.06, −0.13 and −0.92 ppm, respectively). Those changes indicated that the ring B was deshielded by aromatic ring of pillararene, and the methyl group was more shielded than others, which implied the most of azobenzene unit was threaded out of pillararene for 2C-WP5A
trans-G with UV light at 365 nm for 10 min, some new signals appeared as shown in Fig. 2c. In comparison to the signals of individual G at the same experimental condition (Fig. 2d), we observed that the chemical shift of proton Hi* shifted upfield from 3.75 ppm to 1.71 ppm, and the peaks of the protons related to the azobenzene altered slightly (he*, hf*, hg* and hh* Δδ = 0.15, 0.06, −0.13 and −0.92 ppm, respectively). Those changes indicated that the ring B was deshielded by aromatic ring of pillararene, and the methyl group was more shielded than others, which implied the most of azobenzene unit was threaded out of pillararene for 2C-WP5A cis-G. The complexion of 2C-WP5A
cis-G. The complexion of 2C-WP5A G exhibited photo-responsive and thermal-responsive as well, the reversible switchable process of the inclusion complex between 2C-WP5A and G could be modulated upon irradiation with UV at 365 nm and heating at 80 °C for one hour (Fig. 2e).
G exhibited photo-responsive and thermal-responsive as well, the reversible switchable process of the inclusion complex between 2C-WP5A and G could be modulated upon irradiation with UV at 365 nm and heating at 80 °C for one hour (Fig. 2e).
To further understand the interaction between 2C-WP5A and trans/cis- G, we used isothermal titration calorimetry (ITC) to explore the thermodynamics of the host-guest inclusion complex (Fig. 3). The Ka values and thermodynamic parameters are listed in Table 1, and we are surprised to find that there was no obvious difference between the Ka-cis and the Ka-trans. The hydrophobic interaction is characterized by positive value of ΔH45,47,48, which suggests that the hydrophobic interaction is the main force in the complexation. Meanwhile, 2C-WP5A and G immobilized their conformation during the complexation, resulted in a great negative entropy change which offset the positive entropy change aroused by solvent liberation49. This great unfavorable entropy make us easily understand why pillar[5]arene cannot bind with azobenzene in organic solution although its size is similar to α-cyclodextrin.
(a) Microcalorimetric titration of 2C-WP5A with trans-G in water at 25 °C; (b) Microcalorimetric titration of 2C-WP5A with trans-G/cis-G (molar ratio 10/90) in water at 25 °C. “Net” heat effect obtained by subtracting the heat of dilution from the heat of reaction, which was analyzed by computer simulation using the “one set of binding sites” model.
Construct supramolecular self-assembly
Recently, we have developed a novel strategy for the construction of supramolecular assemblies by means of host induced aggregating (HIA: a small amount of macrocyclic host could promote the aggregation of guest molecules by lowering the critical aggregating concentration (CAC) and even regulating the morphology of the aggregates)50,51,52,53,54,55,56,57. So after successful establishing the recognition motif between 2C-WP5A and G in aqueous solution, we were trying to explore the constructing supramolecular aggregates based on 2C-WP5A and trans-G. G could be induced by 2C-WP5A to form large assembly via HIA by monitoring the dependence of the optical transmittance at 600 nm on the concentration of G (There is no absorbance of G at 600 nm Fig. 4a). It is noteworthy that in the absence of 2C-WP5A, the optical transmittance of trans-G at 600 nm showed no appreciable change as the concentration increased form 0.8 mM to 2.0 mM (see Supplementary Fig. S18). It is requisite to find the best molar ratio between 2C-WP5A and G for fabricating the supramolecular assembly. As shown in Fig. 4b, the optical transmittance at 600 nm of the trans-G solution first decreased sharply upon the gradually added 2C-WP5A until reaching the minimum and then gradually increased as the amount of 2C-WP5A continued to increase. By monitoring the change of the transmittance at the wavelength of 600 nm in the process, the best molar ratio of trans-G and 2C-WP5A can be determined at 1:0.06. Redundant amount of 2C-WP5A leads to the formation of a simple inclusion complex and disaggregation of the aggregates, accompanied with the rising of the optical transmittance. This phenomenon highlighted the important role of HIA in the formation of the supramolecular assembly. In fact, only a small amount of macrocyclic host (6%) could promote the aggregation of guest molecules. In order to prove the prominent effect of the macrocyclic structure of host, a control experiment showed that no assembly appeared when replacing 2C-WP5A with its fragment benzene-1,4-dioxyacetic acid ammonium salt (DAAS) under comparable conditions (see Supplementary Fig. S19), indicating that the cyclic structure of 2C-WP5A is vital in inducing the supramolecular assembly. Furthermore, in order to make comparison with HIA, we fixed the concentration of 2C-WP5A at 1.6 mM, gradually added trans-G and controlled the experiment procedure same to the HIA. As shown in Fig. 4c,d, the transmittance the 2C-WP5A is not changed upon the addition of G from 0 to 1.0 eq. So for induced aggregating, the poly-charged additive is necessary such as the macrocyclic host molecules. Overall, there are three main points in HIA: (1) strong binding affinity between host and guest; (2) charge interaction between host and guest; (3) the macrocyclic structure of host molecule.
(a) Optical transmittance of aqueous solutions of 2C-WP5A at different concentrations in the presence of trans-G (1.6 mM) at 25 °C. (b) Dependence of optical transmittance at 600 nm on 2C-WP5A concentration in the presence of trans-G (1.6 mM) at 25 °C. (c) Optical transmittance of aqueous solutions of trans-G at different concentrations in the presence of 2C-WP5A (1.6 mM) at 25 °C. (d) Dependence of optical transmittance at 600 nm on trans-G concentration in the presence of 2C-WP5A (1.6 mM) at 25 °C.
Dynamic light scattering (DLS) and transmission electron microscopy (TEM) are used to characterize the size and morphology of 2C-WP5A+G assembly. As shown in Fig. 5a, DLS data showed that 2C-WP5A+trans-G formed well-defined aggregates with a narrow size distribution, giving an average diameter about 848 nm at a scattering angle of 90°. Meanwhile, the TEM image (Fig. 5c) also certificated the formation of spherical nanoparticles with diameter about 800 nm consistent with the DLS data. Furthermore, after irradiation the solution with UV light at 365 nm for 10 min, the solution became clear and the size of the aggregates formed by 2C-WP5A+cis-G changed into smaller nanoparticles with diameter about 253 nm (Fig. 5b,d). Afterwards, upon heating the solution at 80 °C for one hour, the size of the aggregates returned to the original status. So it is incontrovertible that the isomerization of G by photo/thermal stimulation has significant influence on the size and morphology of the supramolecular nanoparticles assembly.
DLS data: (a) the 2C-WP5A+trans-G assembly at 25 °C. Inset: Photograph of the solution of the 2C-WP5A+trans-G assembly. (b) After irradiation with UV light at 365 nm of (a) for 10 min. Inset: Photograph of the solution of the 2C-WP5A+G assembly after irradiation with UV light at 365 nm for 10 min. (c) TEM image of (a). (d) TEM image of (b).
Zeta potential measurement was further performed to identify the supramolecular assembly surface charged distribution (see Supplementary Figs S20 and S21), both of the 2C-WP5A+trans/cis G assembly giving an average negative zeta potential to maintain the stability of the assembly. Based on the above experimental results and analysis, we postulate that the formation of the supramolecular assembly occurred in two steps. Firstly, the 2C-WP5A and G instantaneously formed a host-guest complex. Subsequently, excess guest molecules arranged around the host-guest complex due to electrostatic effect with redundant carboxylate anion of 2C-WP5A. According to the experimental results of the zeta potential, the 2C-WP5A was on the surfaces of the nanoparticles. Based on the above description, the poly-anionic structure of 2C-WP5A is very important in the formation of the supramolecular nanoparticles assembly.
It is also required to make a thorough inquiry about the repetitiveness of the photo/thermal driven size switching of the supramolecular assembly. Size switching process was determined by monitoring the optical transmittance of 2C-WP5A+G solution at 600 nm (Fig. 6) and DLS experiment (see Supplementary Fig. S22). The presence of the large aggregates of the solution was evident from the decrease in transmittance, which was corresponding to the formation of spherical nanoparticles with diameter of 800 nm from DLS and TEM results. After irradiation with UV light at 365 nm for 10 min, the solution turned clear and transparent with nearly 100% transmittance. By continuous heating at 80 °C for one hour, the solution reverted to the initial turbidity state. Significantly, the reversible transition between turbidity and clarity could repeat several times, indicating that the good photo/thermal responsiveness of the supramolecular assembly.
Conclusions
In conclusion, a novel recognition motif between anionic pillar[5]arene (2C-WP5A) and azobenzene derivative G has been developed, and both of the trans- and cis- G have the similar affinity with 2C-WP5A. The complexation process was well studied and confirmed by 1H NMR, 2D ROESY, and ITC experiment. The recognition motif would greatly expand the application in the area of construction of stimuli-responsive supramolecular assembly based on anionic pillar[5]arene. Furthermore, we utilized this novel recognition motif to construct supramolecular assembly nanoparticles which could reversibly switch the size from ~800 nm to ~250 nm by photo/thermal stimulation, presenting the turbid-to-clear switch of the solution state with excellent reversibility. To the best of our knowledge, this is the first example of supramolecular aggregate based anionic pillar[5]arene and azobenzene derivative. The high affinity of anionic pillar[5]arene with azobenzene derivative is expected to have great potential applications in further fabrication of more sophisticated stimuli-responsive supramolecular system.
Methods
Materials
2C-WP5A was prepared according to the published procedures58. All reagents were commercially available and used without further purification, unless otherwise noted. Solvent were dried according to procedures described in the literature. Column chromatography was performed on silica gel (200–300 mesh).
Measurements
All experiments were performed in deionized water at 25 °C unless noted otherwise.
NMR Spectra
1H NMR and 13C NMR spectra were recorded on a Bruker AVANCE AV400 (400 and 100 MHz). Signal positions were reported in part per million (ppm) relative to the residual solvent peaks or to the peak of Si(CH3)4 used as an internal standard with the abbreviations s, d, t, q, and m, denoting singlet, doublet, triplet, quartlet and multiplet, respectively. The residual 1H peak of deuterated solvent appeared at 4.79 ppm in D2O, at 7.26 ppm in CDCl3. All coupling constants J are quoted in Hz.
High resolution mass spectra (HRMS)
HRMS were performed on an Agilent 6520 Q-TOF LC/MS with ESI ionization.
DLS Measurement
The sample solution was filtered through a 450 nm Millipore filter into a clean scintillation vial and then was examined using a laser light scattering spectrometer equipped with a digital correlator at 640 nm at a scattering angle of 90°.
UV/Vis spectra
UV/Vis spectra were recorded in a quartz cell (light path 1 mm) on a Thermo Scientific EVOLUTION 300 spectrophotometer equipped with a HAAKE SC 100 temperature controller to keep the temperature at 25 °C.
Optical transmittance Measurement
The optical transmittance of the aqueous solution was measured in a quartz cell (light path 10 mm) on a Shimadzu UV-3600 spectrophotometer equipped with a PTC-348WI temperature controller.
TEM Measurement
The sample for TEM measurement was prepared by dropping the solution onto a copper grid. The grid was then air-dried. The samples were examined by a high-resolution TEM (Tecnai G2 F20 microscope, FEI) equipped with a CCD camera (Orius 832, Gatan) operating at an accelerating voltage of 200 kV.
ITC experiment
The ITC experiment was carried out at 25 °C in aqueous solution. In a typical experiment, the solution of 2C-WP5A in a 0.250 mL syringe was sequentially injected with stirring at 300 rpm into a solution of G in the sample cell (1.4227 mL). A control experiment to determine the heat of dilution was carried out by performing the same number of injections with the same concentration of host compound into ultrapure water. The dilution enthalpies determined in control experiments were subtracted from the enthalpies measured in the titration experiments to obtain the net reaction heat. All thermodynamic parameters reported in this work were obtained by using the “one set of binding sites” model. Two titration experiments were independently performed to give the averaged values with standard error (see Supplementary Fig. S23).
Zeta (ζ) Potential Measurement
Zeta potential values were determined at 25 °C on a Brookhaven ZetaPALS (Brookhaven Instrument, USA). The instrument utilizes phase analysis light scattering to provide an average over multiple particles.
Additional Information
How to cite this article: Zhang, C.-C. et al. Size Switchable Supramolecular Nanoparticle Based on Azobenzene Derivative within Anionic Pillar[5]arene. Sci. Rep. 6, 37014; doi: 10.1038/srep37014 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Arvizo, R. R. et al. Intrinsic therapeutic applications of noble metal nanoparticles: past, present and future. Chem. Soc. Rev. 41, 2943–2970 (2012).
Wang, W., Zhang, M. J. & Chu, L. Y. Functional polymeric microparticles engineered from controllable microfluidic emulsions. Acc. Chem. Res. 47, 373–384 (2013).
Wang, T., LaMontagne, D., Lynch, J., Zhuang, J. & Cao, Y. C. Colloidal superparticles from nanoparticle assembly. Chem. Soc. Rev. 42, 2804–2823 (2013).
Klajn, R., Bishop, K. J. & Grzybowski, B. A. Light-controlled self-assembly of reversible and irreversible nanoparticle suprastructures. Proc Natl Acad of Sci USA 104, 10305–10309 (2007).
He, H., Feng, M., Chen, Q., Zhang, X. & Zhan, H. Light-induced reversible self-assembly of gold nanoparticles surface-immobilized with coumarin ligands. Angew. Chem. Int. Ed. 128, 948–952 (2016).
Merino, E. Synthesis of azobenzenes: the coloured pieces of molecular materials. Chem. Soc. Rev. 40, 3835–3853 (2011).
Dong, M., Babalhavaeji, A., Samanta, S., Beharry, A. A. & Woolley, G. A. Red-shifting azobenzene photoswitches for in vivo use. Acc. Chem. Res. 48, 2662–2670 (2015).
Turansky, R., Konopka, M., Doltsinis, N. L., Stich, I. & Marx, D. Switching of functionalized azobenzene suspended between gold tips by mechanochemical, photochemical, and opto-mechanical means. Phys. Chem. Chem. Phys. 12, 13922–13932 (2010).
Henzl, J., Mehlhorn, M., Gawronski, H., Rieder, K. H. & Morgenstern, K. Reversible cis-trans isomerization of a single azobenzene molecule. Angew. Chem. Int. Ed. 45, 603–606 (2006).
Tong, X., Pelletier, M., Lasia, A. & Zhao, Y. Fast cis-trans isomerization of an azobenzene derivative in liquids and liquid crystals under a low electric field. Angew. Chem. Int. Ed. 47, 3596–3599 (2008).
Muraoka, T., Kinbara, K. & Aida, T. Mechanical twisting of a guest by a photoresponsive host. Nature 440, 512–515 (2006).
Ichimura, K., Oh, S. K. & Nakagawa, M. Light-driven motion of liquids on a photoresponsive surface. Science 288, 1624–1626 (2000).
Kumar, A. S. et al. Reversible photo-switching of single azobenzene molecules in controlled nanoscale environments. Nano Lett. 8, 1644–1648 (2008).
Alvaro, M., Benitez, M., Das, D., Garcia, H. & Peris, E. Reversible porosity changes in photoresponsive azobenzene-containing periodic mesoporous silicas. Chem. Mater. 17, 4958–4964 (2005).
Fujiwara, M. & Imura, T. Photo Induced Membrane Separation for Water Purification and Desalination Using Azobenzene Modified Anodized Alumina Membranes. ACS Nano 9, 5705–5712 (2015).
Hagen, R. & Bieringer, T. Photoaddressable polymers for optical data storage. Adv. Mater. 13, 1805–1810 (2001).
Oliveira, O. N. Jr., Dos Santos, D. S. Jr., Balogh, D. T., Zucolotto, V. & Mendonca, C. R. Optical storage and surface-relief gratings in azobenzene-containing nanostructured films. Adv. Colloid interface Sci. 116, 179–192 (2005).
Lee, K. M., Koerner, H., Vaia, R. A., Bunning, T. J. & White, T. J. Light-activated shape memory of glassy, azobenzene liquid crystalline polymer networks. Soft Matter 7, 4318–4324 (2011).
Schut, M. et al. Photocontrol of Cell Adhesion Processes: Model studies with cyclic azobenzene-RGD peptides. Chem. Biol. 10, 487–490 (2003).
Tang, X. & Dmochowski, I. J. Regulating gene expression with light-activated oligonucleotides. Mol. BioSyst. 3, 100–110 (2007).
Zhang, F. et al. Structure-based approach to the photocontrol of protein folding. J. Am. Chem. Soc. 131, 2283–2289 (2009).
Yagai, S. & Kitamura, A. Recent advances in photoresponsive supramolecular self-assemblies. Chem. Soc. Rev. 37, 1520–1529 (2008).
Natali, M. & Giordani, S. Molecular switches as photocontrollable “smart” receptors. Chem. Soc. Rev. 41, 4010–4029 (2012).
Takashima, Y. et al. Expansion-contraction of photoresponsive artificial muscle regulated by host-guest interactions. Nat. Commun. 3, 1270, 10.1038/ncomms2280 (2012).
Mazzier, D. et al. Photoresponsive supramolecular architectures based on polypeptide hybrids. Macromolecules 47, 7272–7283 (2014).
Nachtigall, O., Kordel, C., Urner, L. H. & Haag, R. Photoresponsive switches at surfaces based on supramolecular functionalization with azobenzene-oligoglycerol conjugates. Angew. Chem. Int. Ed. 53, 9669–9673 (2014).
Shinkai, S., Nakaji, T., Ogawa, T., Shigematsu, K. & Manabe, O. Photoresponsive crown ethers. 2. Photocontrol of Ion extraction and ion transport by a bis(crown ether) with a butterfly-like motion. J. Am. Chem. Soc. 103, 111–115 (1991).
Yamaguchi, H. et al. Photoswitchable gel assembly based on molecular recognition. Nat. commun. 3, 603, 10.1038/ncomms1617 (2012).
Iwaso, K., Takashima, Y. & Harada, A. Fast response dry-type artificial molecular muscles with [c2]daisy chains. Nat. chem. 8, 625–632 (2016).
Stoffelen, C., Voskuhl, J., Jonkheijm, P. & Huskens, J. Dual stimuli-responsive self-assembled supramolecular nanoparticles. Angew. Chem. Int. Ed. 53, 3400–3404 (2014).
Ogoshi, T., Kanai, S., Fujinami, S., Yamagishi, T. A. & Nakamoto, Y. para-Bridged symmetrical pillar[5]arenes: their Lewis acid catalyzed synthesis and host-guest property. J. Am. Chem. Soc. 130, 5022–5023 (2008).
Cragg, P. J. & Sharma, K. Pillar[5]arenes: fascinating cyclophanes with a bright future. Chem. Soc. Rev. 41, 597–607 (2012).
Xue, M., Yang, Y., Chi, X., Zhang, Z. & Huang, F. Pillararenes, a new class of macrocycles for supramolecular chemistry. Acc. Chem. Res. 45, 1294–1308 (2012).
Strutt, N. L., Zhang, H., Schneebeli, S. T. & Stoddart, J. F. Functionalizing pillar[n]arenes. Acc. Chem. Res. 47, 2631–2642 (2014).
Li, S. H., Zhang, H. Y., Xu, X. & Liu, Y. Mechanically selflocked chiral gemini-catenanes. Nat. commun. 6, 7590, 10.1038/ncomms8590 (2015).
Ogoshi, T., Yamagishi, T. A. & Nakamoto, Y. Pillar-shaped macrocyclic hosts pillar[n]arenes: new key players for supramolecular chemistry. Chem. Rev. 116, 7937–8002 (2016).
Yu, G. et al. Pillar[6]arene-based photoresponsive host-guest complexation. J. Am. Chem. Soc. 134, 8711–8717 (2012).
Chi, X., Ji, X., Xia, D. & Huang, F. A dual-responsive supra-amphiphilic polypseudorotaxane constructed from a water-soluble pillar[7]arene and an azobenzene-containing random copolymer. J. Am. Chem. Soc. 137, 1440–1443 (2015).
Ogoshi, T., Kida, K. & Yamagishi, T. A. Photoreversible switching of the lower critical solution temperature in a photoresponsive host-guest system of pillar[6]arene with triethylene oxide substituents and an azobenzene derivative. J. Am. Chem. Soc. 134, 20146–20150 (2012).
Xia, D., Yu, G., Li, J. & Huang, F. Photo-responsive self-assembly based on a water-soluble pillar[6]arene and an azobenzene-containing amphiphile in water. Chem. Commun. 50, 3606–3608 (2014).
Hu, X. Y. et al. Dual photo- and pH-responsive supramolecular nanocarriers based on water-soluble pillar[6]arene and different azobenzene derivatives for intracellular anticancer drug delivery. Chem. Eur. J. 21, 1208–1220 (2015).
Qin, S., Xiong, S., Han, Y., Hu, X.-Y. & Wang, L. Controllable fabrication of various supramolecular nanostructures based on nonamphiphilic azobenzene derivatives and pillar[6]arene. Chin. J. Chem. 33, 107–111 (2015).
Harada, A., Hashidzume, A., Yamaguchi, H. & Takashima, Y. Polymeric rotaxanes. Chem. Rev. 109, 5974–6023 (2009).
Ogoshi, T., Yamafuji, D., Aoki, T. & Yamagishi, T. A. Photoreversible transformation between seconds and hours time-scales: threading of pillar[5]arene onto the azobenzene-end of a viologen derivative. J. Org. Chem. 76, 9497–9503 (2011).
Yakimova, L. S. et al. Highly selective binding of methyl orange dye by cationic water-soluble pillar[5]arenes. Org. Biomol. Chem. 14, 4233–4238 (2016).
Cheng, H. B., Zhang, Y. M., Xu, C. & Liu, Y. Photoresponsive supramolecular complexes as efficient DNA regulator. Sci. Rep. 4, 4210, 10.1038/srep04210 (2014).
Zhang, C. F., Li, S. H., Zhang, C. C. & Liu, Y. Molecular binding behaviors and thermodynamics of ferrocenyl dimethylaminium derivatives by anionic pillar[5]arene. Org. Biomol. Chem. 13, 10808–10812 (2015).
Fu, J. & Schlenoff, J. B. Driving forces for oppositely charged polyion association in aqueous solutions: enthalpic, entropic, but not electrostatic. J. Am. Chem. Soc. 138, 980–990 (2016).
Zheng, P. J., Wang, C., Hu, X., Tam, K. C. & Li, L. Supramolecular complexes of azocellulose and α-cyclodextrin: isothermal titration calorimetric and spectroscopic studies. Macromolecules 38, 2859–2864 (2005).
Guo, D. S., Chen, K., Zhang, H. Q. & Liu, Y. Nano-supramolecular assemblies constructed from water-soluble bis(calix[5]arenes) with porphyrins and their photoinduced electron transfer properties. Chem. Asian J. 4, 436–445 (2009).
Guo, D. S., Jiang, B. P., Wang, X. & Liu,Y. Calixarene-induced aggregation of perylene bisimides. Org. Biomol. Chem. 10, 720–723 (2012).
Guo, D. S., Wang, K., Wang, Y. X. & Liu, Y. Cholinesterase-responsive supramolecular vesicle. J. Am. Chem. Soc. 134, 10244–10250 (2012).
Cao, Y., Wang, Y., Guo, D. & Liu, Y. p-Sulfonatocalix[4]arene-induced amphiphilic aggregation of fluorocarbon surfactant. Sci. China Chem. 57, 371–378 (2013).
Guo, D. S. & Liu, Y. Supramolecular chemistry of p-sulfonatocalix[n]arenes and its biological applications. Acc. Chem. Res. 47, 1925–1934 (2014).
Wang, Y. X., Zhang, Y. M. & Liu, Y. Photolysis of an amphiphilic assembly by calixarene-induced aggregation. J. Am. Chem. Soc. 137, 4543–4549 (2015).
Wang, Y. X. & Liu, Y. Supramolecular assemblies based on p-sulfonatocalixarenes and their functions. Acta Chim. Sinica 73, 984–991 (2015).
Shi, R. J., Chen, Y., Hou, X. F. & Liu, Y. Effect of head/tail groups on molecular induced aggregation of polycationic cyclodextrin towards anionic surfactants. RSC Adv. 6, 15175–15179 (2016).
Ogoshi, T., Hashizume, M., Yamagishi, T. A. & Nakamoto, Y. Synthesis, conformational and host-guest properties of water-soluble pillar[5]arene. Chem. Commun. 46, 3708–3710 (2010).
Acknowledgements
We thank the National Natural Science Foundation of China (21432004 and 91527301) and the China Postdoctoral Science Foundation (2016M591380) for financial support.
Author information
Authors and Affiliations
Contributions
C.-C.Z. and S.-H.L. performed the experiments and analyzed the nanoparticle. S.-H.L. and C.-F.Z. synthesized the 2C-WP5A. Y.L. supervised the work and edited the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhang, CC., Li, SH., Zhang, CF. et al. Size Switchable Supramolecular Nanoparticle Based on Azobenzene Derivative within Anionic Pillar[5]arene. Sci Rep 6, 37014 (2016). https://doi.org/10.1038/srep37014
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37014
This article is cited by
-
Unraveling the binding nature of hexane with quinone functionalized pillar[5]quinone: a computational study
Journal of Inclusion Phenomena and Macrocyclic Chemistry (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.




 G
G


