Abstract
Homotypic and heterotypic cell-to-cell fusion are key processes during development and tissue regeneration. Nevertheless, aberrant cell fusion can contribute to tumour initiation and metastasis. Additionally, a form of cell-in-cell structure called entosis has been observed in several human tumours. Here we investigate cell-to-cell interaction between mouse mesenchymal stem cells (MSCs) and embryonic stem cells (ESCs). MSCs represent an important source of adult stem cells since they have great potential for regenerative medicine, even though they are also involved in cancer progression. We report that MSCs can either fuse forming heterokaryons, or be invaded by ESCs through entosis. While entosis-derived hybrids never share their genomes and induce degradation of the target cell, fusion-derived hybrids can convert into synkaryons. Importantly we show that hetero-to-synkaryon transition occurs through cell division and not by nuclear membrane fusion. Additionally, we also observe that the ROCK-actin/myosin pathway is required for both fusion and entosis in ESCs but only for entosis in MSCs. Overall, we show that MSCs can undergo fusion or entosis in culture by generating distinct functional cellular entities. These two processes are profoundly different and their outcomes should be considered given the beneficial or possible detrimental effects of MSC-based therapeutic applications.
Similar content being viewed by others
Introduction
Cell-to-cell fusion is a highly regulated key process involved in development and tissue homeostasis1,2. In particular cell fusion is required for fertilization, macrophage-derived giant cells and skeletal muscle formation, bone development and syncytiotrophoblast generation. As an example, trophoblast cells have a remarkable fusion capability that allows the formation of the syncytiotrophoblast, which is indispensable for the blastocyst implantation3. Importantly in injured tissues bone marrow derived cells (BMDC) can fuse in vivo with differentiated cells and form hybrids with regenerative potential2. In fact, bone marrow-derived hybrids were found in many organs such as brain, retina, liver, muscle and gut where they participated in the reestablishment of tissue function4,5,6,7,8,9,10,11,12. Based on these premises, several cell therapy approaches using BM-transplantation have been carried out to regenerate different tissues13,14,15,16,17,18,19.
On the other hand, heterotypic cell fusion has also been associated to cancer development and metastasis formation. In particular, cancer cells can fuse with different cell types, including stromal, epithelial and endothelial cells generating genetically instable hybrids20,21,22. Additionally, it was shown that macrophages or bone marrow-derived cells behave as fusion partners in several types of tumours23,24,25,26,27.
Cell fusion is also an essential approach to study somatic cell reprogramming mechanisms28,29,30,31,32. Indeed, it has been extensively used in vitro to investigate the activity of several transcription factors and pathways for their role in the enhancement of the reprogramming process33,34,35.
Taking into account all these previous reports, despite cells can spontaneously fuse both in vitro and in vivo with low efficiency36,37,38,39, cell-to-cell fusion is a critical biological process, which warrants investigation.
Recent studies have reported and characterized another form of cell-cell interaction, named entosis, which has been found in a variety of human tumours and can either play a pro-tumorigenic or a tumour suppressor role40. Entosis is a form of cell-in-cell structure originated by the active invasion of one living cell into another. It is caused by the loss of cell-matrix adhesion and it is mediated by adherent junctions and by the activity of the Rho-ROCK-actin/myosin pathways41,42,43,44,45,46.
Here we found that mesenchymal stem cells (MSCs) can either fuse, thereby forming heterokaryons, or be invaded by mouse embryonic stem cells (mESCs) through entosis. Moreover, we found that the ROCK-actin/myosin pathway is necessary for both mESC fusion and entosis but only for entosis in the case of MSCs. Importantly we showed that, contrary to cytoplasmic membrane fusion, nuclear membranes appear not to fuse directly. Instead cell division, disassociation and reassembly of the nuclear envelope allow the mixing and redistribution of parental chromosomes to the daughter cells, therefore generating synkaryons. Finally, considering the importance of MSC-based therapeutic applications, we implemented a straightforward method to purify either entotic or fusion–derived hybrids. In the future our approach and observations could be extended to investigate the outcome of these two profoundly different processes in vivo.
Results
MSCs form fusion and entosis-derived hybrids with ESCs in culture
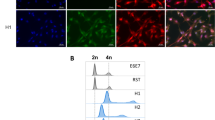
In order to investigate the mechanism of cell-to-cell fusion process we set up an in vitro system to identify cell lines that fuse more efficiently in culture. To this purpose a panel of either somatic, multipotent or pluripotent murine cell lines with a reported fusion capability33,47,48,49,50 were modified to constitutively express H2B tagged with either enhanced green fluorescent protein (H2B-eGFP) or monomeric red fluorescent protein (H2B-mRFP) (Fig. 1a,b and Supplementary Figure S1a). ESCs-mRFP were mixed in suspension for 45 min with either ESCs-eGFP, MSCs-eGFP, neural stem cells (NSCs)-eGFP or with hepatocarcinoma cells (Hepa-1–6)-eGFP, then cultured for 6 hrs and finally analysed by flow cytometry (Fig. 1c). When ESCs were mixed together with NSCs or with Hepa 1–6 or with themselves, we detected very few hybrids (eGFP+/mRFP+ cells) (Fig. 1d). Conversely, we identified almost 2% of double positive cells when ESCs were co-cultured with MSCs (Fig. 1d). Furthermore, confocal microscopy analysis on FACS-sorted eGFP+/mRFP+ cells confirmed the presence of two nuclei into a unique cytoplasm (Fig. 1e). Finally, cell cycle analysis performed by DAPI staining showed, as expected for hybrid cells, higher DNA amount as compared to the parental cell types (Supplementary Figure S1b). These data indicate that MSCs and ESCs spontaneously form hybrid cells already after 6 hrs of co-culture and with a higher efficiency compared to the other tested cell lines.
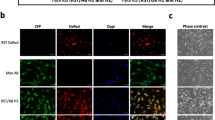
Mixed MSCs and ESCs form heterotypic hybrids in vitro.
(a) Experimental scheme representing the co-culture condition to identify the best fusogenic cell lines. (b) Representative fluorescence micrograph of different cell types transduced with human immunodeficiency lentiviral particles carrying HIV-H2B::mRFP or eGFP (scale bar 50 μm). (c,d) Representative FACS analysis and quantification of eGFP+/mRFP+ cells derived from the indicated co-cultured cells. Data are represented as means ± SE (number of independent experiments n = 5) and statistical significance is represented by unpaired t-Test ***P < 0,001. (e) Confocal images of FACS sorted eGFP+/mRFP+ cells derived from ESC-mRFP and MSC-eGFP co-cultured after being 45 min in suspension and 6 hrs in adhesion (scale bar 10 μm).
To better examine the nature of MSC/ESC-derived hybrids, we FACS-sorted the eGFP+/mRFP+ cells after 6 hrs of co-culture and the resulting hybrids were analysed by confocal microscopy after 24 hrs. Surprisingly we noticed two distinct phenotypes. First, as expected, the heterokaryons were characterised by the presence of two nuclei derived from the parental cells into the same cytoplasm where the eGFP-positive nucleus incorporated H2B-mRFP proteins from the fusion partner and vice versa (Fig. 2a, yellow arrowheads). In addition, beside fusion-derived heterokaryons, we observed MSC-like cells with punctate mRFP signal into the cytoplasm. These cells displayed a phenotype similar to previously reported entotic process that, to date has not been described in MSCs (Fig. 2a, white arrowheads).
ESCs spontaneously fuse with MSCs or undergo entosis.
(a) Representative confocal micrograph of FACS-sorted eGFP+/mRFP+ cells 24 hrs after the co-culture of ESCs-mRFP and MSCs-eGFP. Internalised and degraded ESCs-mRFP within MSCs-eGFP and heterokaryon cells are indicated by white and yellow arrowheads, respectively (scale bar 10 μm). (b) FACS-sorted eGFP+/mRFP+ cells were immunostained for ß-catenin to distinguish fused (upper panel) versus entotic (lower panel) cells (scale bar 10 μm).
Entosis occurs when a living cell invade another cell’s cytoplasm caused by the detachment from the extracellular matrix (ECM)41,42,43,44. To further investigate the two observed phenotypes, we analysed eGFP+/mRFP+-sorted cells by time-lapse microscopy. Interestingly, we noticed the presence of both cell fusion-derived hybrids and entosis-derived hybrids. Cell fusion-derived hybrids (heterokaryons) were characterized by the presence of 2 nuclei sharing the same cytoplasm that were capable to exchange H2B-eGFP or H2B-mRFP (Supplementary Movies S1 and S2). In contrast, in entosis-derived hybrids, the ESCs appeared to be inside a large vacuole, internalized into MSCs and histone exchange was never detected (Supplementary Movies S3 and S4). Remarkably, we noticed that MSC-like cells with punctate cytoplasmic mRFP resulted from the entotic process (Supplementary Movie S5), similarly to previous observations51,52. It has been reported that internalised cells after co-culture of mammary epithelial cells are initially alive and can eventually divide, be killed or released41. Similarly, we found that entosis of ESCs into MSCs could recapitulate these phenotypes (Supplementary Movies S6–S8).
To further confirm these observations, eGFP+/mRFP+ cells were analysed at earlier time point (16 hrs after sorting) by immunostaining against β-catenin to highlight the plasma membrane. Analysis by confocal microscopy demonstrated either the complete internalization of ESCs within MSCs or the fusion between the two different cell types generating thereby entosis-derived hybrids or fusion-derived hybrids (Fig. 2b). Moreover, this result was also confirmed by transmission electron microscopy (TEM) ultra-micrograph (Supplementary Figure S2a).
We performed a number of control experiments to exclude that MSC-like cells, which included punctate mRFP signal into the cytoplasm, could derive from a phagocytic response due to the differentiation of MSCs into macrophage-like cells in our co-culture condition. First, neither ESCs nor MSCs expressed the macrophage cell marker Mac-1 suggesting that none of these cell types differentiate into phagocytic cells in our culture conditions (Supplementary Figure S2b). Secondly, since cell opsonisation can enhances macrophage-mediated phagocytosis53, we co-cultured ESCs and MSCs after ESC opsonisation using an antibody against the ESC cellular marker SSEA-1. Even in this experimental condition we did not observe any increase in the amount of eGFP+/mRFP+ double positive cells confirming that MSCs did not differentiate into macrophage-like cells (Supplementary Figure S2c). Finally, MSCs and ESCs mixed together were examined for phosphatidylserine (PS) exposure with Annexin V protein. PS is an “eat-me” signal exposed on the outer leaflet of dying cell’s plasma membrane, which is recognised by phagocytic cells54. Annexin V+ cells were not detected after 6 hrs of co-culture suggesting that the PS pro-phagocytic signal is not involved in this process (Supplementary Figure S2d). Therefore, these data strongly suggest that phagocytic clearance does not explain the presence of punctate mRFP signal into the cytoplasm of MSC-like cell.
Overall, here we show that MSCs and ESCs can spontaneously form either heterokaryons or undergo entosis when cultured together.
Fused and entotic cells can be distinguished by surface markers
Since cell fusion and entosis give rise to functionally distinct hybrids, it is important to discriminate heterokaryons from entotic cells and study them separately. When MSCs and ESCs fuse together, the resulting hybrids are characterized by a mixed plasma membrane. On the contrary membrane/cytoplasm mixing does not occur in case of entosis. Therefore we reasoned that it is possible to discriminate fused from entotic cells using cell specific plasma membrane markers. In particular, ESCs express epithelial cadherin (E-cad) at the plasma membrane that, in contrast, it is not expressed in the MSCs (Supplementary Figure S3a,b). Given this specific feature, it is therefore possible to distinguish and isolate by FACS either fused (eGFP+/mRFP+/E-cad+) or entotic (eGFP+/mRFP+/E-cad−) cells based on the colour pattern (Fig. 3a and Supplementary Figure S3c). After 6 hrs of co-culture, approximately 0,25% of cells were eGFP+/mRFP+/E-cad+ and 1,75% were eGFP+/mRFP+/E-cad− (Fig. 3b), demonstrating that it is possible to quantify and enrich both entosis and fusion-derived hybrids after co-culture. To further confirm the efficacy of our E-cad-based purification system we analysed eGFP+/mRFP+ by imaging flow cytometry55. This method allowed us to simultaneously collect real-time images of each event in the flow stream during the FACS analysis. We observed that the heterokaryons were decorated by the E-cad ring on plasma membrane, while the entotic derived hybrids were negative for E-cad immunostaining (Supplementary Figure S3d). Of note, out of the fused cells we never observed synkaryons immediately after FACS-sorting.
Fused or entotic cells can be distinguished and quantified by E-cadherin staining.
(a) Confocal images of living FACS-sorted fused and entotic cells (scale bar 10 μm). (b) Quantification of fused versus entotic cells. Co-cultured cells were stained for E-cad to quantify fused (eGFP+/mRFP+/E-cad+) and entotic (eGFP+/mRFP+/E-cad−) cells. Data are represented as means ± SE (number of independent experiments n = 15). (c) Colony forming unit assay (CFU-F) of parental mESCs-mRFP, MSCs-eGFP, eGFP+/mRFP+/E-cad− cells and eGFP+/mRFP+/E-cad+ cells 6 days after sorting. Representative images of crystal violet stained colonies (upper images) and quantitative analysis of colony forming rate of mESCs-mRFP, MSCs-eGFP, eGFP+/mRFP+/E-cad− entosis-derived hybrids and eGFP+/mRFP+/E-cad+ fusion-derived hybrids (lower plots). Data are represented as means ± SE (number of independent experiments n = 4) and statistical significance is represented by unpaired t-Test *P < 0,05, **P < 0,01.
Finally, to assess whether cell fusion and entosis-derived hybrids could generate ESC-like and MSC-like colonies from single cells we performed a colony forming unit assay (CFU-F). We FACS-sorted entosis- (eGFP+/mRFP+/E-cad−), fusion-derived hybrids (eGFP+/mRFP+/E-cad+) after 6 hrs of co-culture and both ESCs-mRFP and MSCs-eGFP as controls. The different cell types were plated either in ESC or MSC media. We observed that MSCs-eGFP grew from single cells and formed colonies in both culturing media while ESCs-mRFP did not survive in MSC medium (Fig. 3c). In ESC-permissive growth conditions, both fusion and entosis-derived hybrids generated colonies from single cells. Importantly, fusion-derived hybrids (RFP+GFP+Ecad+ cells) formed ESC-like colonies, although in a reduced number if compared to the control ESCs-mRFP cells. In contrast, in MSC media, fusion-derived hybrids (RFP+GFP+Ecad+ cells), which retain ESC-like feature, did not survive while entosis-derived hybrids (RFP+GFP+Ecad− cells) formed MSC-like colonies with a growth rate comparable to the MSC control (Fig. 3c). Thus, heterokaryons as well as entosis-derived hybrids can proliferate in vitro after sorting and generate ESC- and MSC-like colonies respectively.
These data show that we developed a simple purification approach that allows the enrichment of either entotic or fusion-derived hybrids, which can thereby be studied separately.
Cytoskeleton components are essential for fusion and entosis
Entosis requires active actin polymerization, myosin II contraction and the activity of the Rho signalling pathway41,42,43,44,45,46. To investigate whether cell fusion requires the same machinery, mixed cells were treated either with cytochalasin D (cytD), an inhibitor of actin polymerization, or with the myosin II inhibitor blebbistatin. Inhibition of actin polymerization suppressed both entosis and fusion in a dosage dependent manner suggesting an active role for actin in both processes (Fig. 4a,d). Myosin II contraction inhibition has been shown to suppress entosis in epithelial breast cancer cells41. Surprisingly, mixed MSCs and ESCs treated with blebbistatin showed higher entotic capability when compared to the control but did not affect cell fusion (Fig. 4b,e). These results suggest that the cytoskeleton plays different functions in both processes in the two different cell types.
Cytoskeleton components are essential for fusion and entosis.
(a–c) Quantification of fused and entotic cells treated with an increasing concentration of cytochalasinD (cytD) (a) blebbistatin (b) and rock inhibitor Y-27632 (c). Data are represented as means ± SE (number of independent experiments n = 9) and statistical significance is represented by unpaired t-Test *P < 0,05, **P < 0,01, ***P < 0,001. (d–f) Representative FACS analysis of fused and entotic cells treated with an increasing concentration of cytochalasin D (cytD) (d) blebbistatin (e) and rock inhibitor Y-27632 (f). (g,h) Quantification and representative FACS analysis of fused and entotic cells after co-culture of ESCs and MSCs infected with lentiviruses carrying shCTR, shR1.1 or shR2.1. Data are represented as means ± SE (number of independent experiments n = 5) and statistical significance is represented by unpaired t-Test *P < 0,05, **P < 0,01, ***P < 0,001
To further investigate this phenotype we decided to examine Rho GTPases activity. In particular we analysed the role of Rho GTPases downstream effectors, the Rock proteins, which are both actin and myosin II regulators. We therefore assessed whether Rho signalling plays a role in either entosis or fusion of co-cultured MSCs/ESCs by using the Y-27632 inhibitor56. Similar to blebbistatin, Y-26732-treated cells exhibited an increased entotic capability whereas cell fusion was not affected (Fig. 4c,f). Of note, cytD, blebbistatin and Y-27632 treatment altered morphology of MSCs and ESCs, indicating that these drugs affected the cytoskeleton (Supplementary Figure S4a–c). Altogether, these data indicate that actin polymerization is essential for entosis as well as for fusion of ESCs with MSCs, whereas myosin II contraction is important only for cell internalization, i.e. for entosis, differently from what previously reported for epithelial breast cancer cells.
To further strengthen these observations, MSCs and ESCs were transduced with lentiviral vectors expressing small hairpin RNAs (shRNA) to knock-down either Rock1 or Rock2 (Supplementary Figure S4d). Based on the silencing efficiency we selected one shRNA per protein, shR1.1 and shR2.1 (Supplementary Figure S4d,e). Interestingly, Rock1 and Rock2 downregulation in ESCs resulted into an inhibitory effect of entosis (Fig. 4g,h). Silencing in ESCs of Rock2 inhibited cell fusion, which showed a tendency to decrease also upon Rock1 downregulation (Fig. 4g,h). This effect was similar to what observed with the cytD treatment (Fig. 4a). Instead, in MSCs only Rock1 silencing enhanced entosis, but not cell fusion (Fig. 4g,h), similar to blebbistatin and Y-27632 treatments (Fig. 4b,c). These data indicate that the Rho-Rock-actin/myosin pathway plays different roles in ESCs and MSCs. While the Rho pathway is specifically required in ESCs for both fusion and entosis processes, the downregulation of Rock1 in MSCs turned them into a more permissive partner for entosis but not for cell fusion.
To rule out the possibility that cytoskeleton perturbations could enhance cell doublets formation and that these aggregates could have been erroneously recognised as hybrids, we performed a cell-aggregate analysis based on a stringent gating strategy. In particular, we analysed the total amount of cell doublets after cytD, blebbistatin and Y-27632 treatment (Supplementary Figure S5a) and in Rock1/2 KD co-cultures (Supplementary Figure S5b). Importantly, no significant variation in the percentage of total cell doublets was observed in all the experimental conditions (Supplementary Figure S5c,d) indicating that cell doublet formation is not altered by drugs treatment or by the Rock1/2 silencing. Additionally, these observations are consistent with previous studies showing that Rock and myosin-based contraction of the actin cytoskeleton are required for cell-cell adhesion and maintenance of sarcoma cell doublets57,58. Overall, these data indicate that cytoskeleton rearrangement and the Rho-Rock pathway are important for both entosis and cell fusion. In particular, the Rho-Rock-actin/myosin pathway is specifically required in ESCs for both fusion and entosis processes, while this pathway turned MSCs into a more permissive partner for entosis but not for cell fusion.
Hetero to synkaryon transition requires cell division
Heterokaryons can convert into synkaryons during reprogramming of somatic cells in vivo and this might represent an important process for tissue regeneration. On the other hand, the possible aberrant chromosome segregation during hetero to synkaryon transition could ultimately result in the formation of hybrid cells characterized by genomic alterations. It is therefore important to study how synkaryons are generated. In addition, whether the transition from hetero to synkaryons in mammalian cells involves nuclear membrane fusion of the two parental nuclei or a different mechanism is still unclear.
To examine how the two distinct nuclei of MSCs and ESCs fuse to form synkaryon starting from living heterokaryons, MSCs-eGFP/ESCs-mRFP derived hybrids were FACS-sorted as described above (eGFP+/mRFP+/E-cad+) and analysed by time-lapse microscopy. As expected, the majority of the heterokaryons died quickly after sorting probably due to genomic instability as quantified in Fig. 5b (Supplementary Movie S9). Interestingly, among the surviving fusion-derived hybrids, the heterokaryons that underwent cell division mixed the two genomes to generate two-daughter synkaryon cells (Fig. 5a,b and Supplementary Movies S10 and S11). These data provide quantitative evidence that transition from hetero to synkaryons in MSC/ESC-derived hybrids occurs through cell division rather than from nuclear membrane fusion.
Hetero to synkaryon transition is mediated by mitosis.
(a) Snapshots of the time-lapse of ESCs and MSCs derived heterokaryon referred to movie S11 (scale bar 10 μm). FACS-sorted heterokaryons were plated and after 2 hrs the attached cells were processed for time-lapse imaging for additional 12 hrs. (b) Quantification of heterokaryon cells fate over approximately 12 hrs after sorting. Data represent the total number of heterokaryon analysed (heterokaryons analysed n = 55). (c) Schematic representation of the optimised experimental protocol to study heterokaryon or entotic cell fate.
Discussion
Cell-to-cell fusion is an essential process for development and tissue repair1,2. Beside these positive effects, aberrant segregation of chromosomes in the hybrid cells could result into cancer generation23. Entosis, a cell-in-cell invasion, has been firstly characterized in breast cancer cells41, and likewise cell fusion it was also recently seen to play a role during early development. Indeed, trophoblast cells not only fuse into a syncytia placental tissue but recent studies demonstrated that trophoblast cells can also remove uterine luminal epithelial cells through entosis, facilitating embryo implantation59.
In this report, we showed that MSCs and mESCs can spontaneously form either entotic or fusion-derived hybrids in co-culture, although with low efficiency. We developed a simple purification protocol based on surface markers that allowed the quantification and purification of both entotic and heterokaryon cells (Fig. 5c). Heterokaryons and entosis-derived hybrids can proliferate in vitro and generate ESC- and MSC-like colonies respectively. It will be interesting to analyse their ability to differentiate toward different lineages, although currently this is technically challenging due to the low number of hybrids obtained from sorting.
Notably, it has been demonstrated that different cell types, including MSCs, neuronal cells, endothelial cells and others, can exchange cytosolic elements such as vesicles and mitochondria through tunneling nanotubes (TNTs)60,61,62,63,64. Although we did not directly investigate the occurrence of TNTs, we cannot exclude this possibility in a limited number of cases.
Cytoskeleton rearrangement and the Rho-ROCK pathway are critical for both MSC differentiation and ESC self-renewal65,66. Here we showed that the Rho-Rock-actin/myosin pathway is important for both cell fusion and entosis in ESCs. On the contrary, its inhibition turned the MSCs into a more permissive partner for entosis but not for cell-fusion. These observations lead us to speculate that a reduction of MSC cortex tension might provoke the formation of a more fluid plasma membrane that facilitates MSC deformation and finally ESC invasion.
In this report we also aimed to study how synkaryons are generated. In yeast, during mating, the two haploid nuclei fuse their nuclear membrane to generate a diploid nucleus67. Similarly, in sea urchin fertilization sperm and egg pronuclear membrane fuse to mix the genomic material68. However, during mammalian fertilization sperm-derived and egg-derived chromosomes condense at the first mitotic prophase and mix on the metaphase equator initiating the first mitotic division69. Surprisingly, in the case of cell fusion we found that transition from hetero- to synkaryons occurs through cell division rather than by nuclear membrane fusion, whereas in the case of entosis internalized cells can be released, undergo cell division or be degraded, confirming earlier experimental observations41. We therefore demonstrated that heterokaryon cells are transient precursors that are indispensable to generate bona fide synkaryon daughter cells through mitosis-mediated mechanism. On the other hand, whether the efficiency of this process can be cell-type specific will be a matter of future investigation.
MSCs are an important adult stem cell resource with great potential for regenerative medicine. Therefore it is essential to fully investigate either the beneficial or detrimental effects of heterokaryons or entosis-derived hybrids in MSC-based therapeutic applications. Future studies will be needed to investigate how synkaryons divide and segregate their chromosomes to generate either stable or instable hybrids with regenerative or tumorigenic potential, respectively. Moreover it will be interesting to characterize whether the entotic activity of MSCs could contribute to either tumour specific suppression or progression.
Material and Methods
Co-cultures
At day 1 ESCs, NSCs, Hepa 1–6 and MSCs were plated respectively at 4*103 cell/cm2, 15*103 cell/cm2, 10*103 cell/cm2 and 10*103 cell/cm2. At day 3 each cell type was detached with cell dissociation buffer (GIBCO 13151–014) mixed in ESC medium in a 15 ml falcon tube, pelleted by centrifugation at 300 rcf for 5 min and the supernatant was aspirated. The pellet was re-suspended in two volumes of ESC medium, incubated at 37 °C for 45 min and plated in gelatine-coated dishes for other 6 hrs.
Time-lapse imaging
FACS-sorted cells were plate into Time-lapse gelatin (Millipore ES-006-B)-coated Thermo Scientific Nunc Lab-Tek chambered coverglass and images were acquired on Andor Revolution XD inverted microscope (Olympus). All imaging of living cells was performed in incubator chamber at 37 °C and 5% CO2.
Additional Information
How to cite this article: Sottile, F. et al. Mesenchymal stem cells generate distinct functional hybrids in vitro via cell fusion or entosis. Sci. Rep. 6, 36863; doi: 10.1038/srep36863 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Chen, E. H. & Olson, E. N. Unveiling the mechanisms of cell-cell fusion. Science 308, 369–373 (2005).
Lluis, F. & Cosma, M. P. Cell-fusion-mediated somatic-cell reprogramming: a mechanism for tissue regeneration. J Cell Physiol 223, 6–13 (2010).
Huppertz, B., Bartz, C. & Kokozidou, M. Trophoblast fusion: fusogenic proteins, syncytins and ADAMs, and other prerequisites for syncytial fusion. Micron 37, 509–517 (2006).
Nygren, J. M. et al. Myeloid and lymphoid contribution to non-haematopoietic lineages through irradiation-induced heterotypic cell fusion. Nat Cell Biol 10, 584–592 (2008).
Wang, X. et al. Cell fusion is the principal source of bone-marrow-derived hepatocytes. Nature 422, 897–901 (2003).
Alvarez-Dolado, M. et al. Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature 425, 968–973 (2003).
Weimann, J. M., Johansson, C. B., Trejo, A. & Blau, H. M. Stable reprogrammed heterokaryons form spontaneously in Purkinje neurons after bone marrow transplant. Nat Cell Biol 5, 959–966 (2003).
Weimann, J. M., Charlton, C. A., Brazelton, T. R., Hackman, R. C. & Blau, H. M. Contribution of transplanted bone marrow cells to Purkinje neurons in human adult brains. Proc Natl Acad Sci USA 100, 2088–2093 (2003).
Johansson, C. B. et al. Extensive fusion of haematopoietic cells with Purkinje neurons in response to chronic inflammation. Nat Cell Biol 10, 575–583 (2008).
Nern, C. et al. Fusion of hematopoietic cells with Purkinje neurons does not lead to stable heterokaryon formation under noninvasive conditions. J Neurosci 29, 3799–3807 (2009).
Ferrand, J. et al. Human bone marrow-derived stem cells acquire epithelial characteristics through fusion with gastrointestinal epithelial cells. PLoS One 6, e19569 (2011).
Rizvi, A. Z. et al. Bone marrow-derived cells fuse with normal and transformed intestinal stem cells. Proc Natl Acad Sci USA 103, 6321–6325 (2006).
Sanges, D. et al. Wnt/beta-catenin signaling triggers neuron reprogramming and regeneration in the mouse retina. Cell Rep 4, 271–286 (2013).
Nygren, J. M. et al. Bone marrow-derived hematopoietic cells generate cardiomyocytes at a low frequency through cell fusion, but not transdifferentiation. Nat Med 10, 494–501 (2004).
Vassilopoulos, G., Wang, P. R. & Russell, D. W. Transplanted bone marrow regenerates liver by cell fusion. Nature 422, 901–904 (2003).
Fukada, S. et al. Muscle regeneration by reconstitution with bone marrow or fetal liver cells from green fluorescent protein-gene transgenic mice. J Cell Sci 115, 1285–1293 (2002).
Willenbring, H. et al. Myelomonocytic cells are sufficient for therapeutic cell fusion in liver. Nat Med 10, 744–748 (2004).
Sanges, D. et al. Reprogramming Muller glia via in vivo cell fusion regenerates murine photoreceptors. J Clin Invest (2016).
Wassim Altarche-Xifro, U.d.V., Maria Isabel Muñoz-Martin, Analía Bortolozzi & Jordi Bové, Miquel Vila . Maria Pia Cosma Functional Rescue of Dopaminergic Neuron Loss in Parkinson’s Disease Mice After Transplantation of Hematopoietic Stem and Progenitor Cells. EBioMedicine 8, 83–95 (2016).
Jacobsen, B. M. et al. Spontaneous fusion with, and transformation of mouse stroma by, malignant human breast cancer epithelium. Cancer Res 66, 8274–8279 (2006).
Mortensen, K., Lichtenberg, J., Thomsen, P. D. & Larsson, L. I. Spontaneous fusion between cancer cells and endothelial cells. Cell Mol Life Sci 61, 2125–2131 (2004).
Dittmar, T. et al. Characterization of hybrid cells derived from spontaneous fusion events between breast epithelial cells exhibiting stem-like characteristics and breast cancer cells. Clin Exp Metastasis 28, 75–90 (2011).
Pawelek, J. M. & Chakraborty, A. K. Fusion of tumour cells with bone marrow-derived cells: a unifying explanation for metastasis. Nat Rev Cancer 8, 377–386 (2008).
Xu, M. H. et al. EMT and acquisition of stem cell-like properties are involved in spontaneous formation of tumorigenic hybrids between lung cancer and bone marrow-derived mesenchymal stem cells. PLoS One 9, e87893 (2014).
He, X. et al. Cell fusion between gastric epithelial cells and mesenchymal stem cells results in epithelial-to-mesenchymal transition and malignant transformation. BMC Cancer 15, 24 (2015).
Freeman, B. T., Jung, J. P. & Ogle, B. M. Single-cell RNA-seq reveals activation of unique gene groups as a consequence of stem cell-parenchymal cell fusion. Sci Rep 6, 23270 (2016).
Noubissi, F. K., Harkness, T., Alexander, C. M. & Ogle, B. M. Apoptosis-induced cancer cell fusion: a mechanism of breast cancer metastasis. FASEB J 29, 4036–4045 (2015).
Pereira, C. F. et al. ESCs require PRC2 to direct the successful reprogramming of differentiated cells toward pluripotency. Cell Stem Cell 6, 547–556 (2010).
Pereira, C. F. et al. Heterokaryon-based reprogramming of human B lymphocytes for pluripotency requires Oct4 but not Sox2. PLoS Genet 4, e1000170 (2008).
Han, D. W. et al. Pluripotential reprogramming of the somatic genome in hybrid cells occurs with the first cell cycle. Stem Cells 26, 445–454 (2008).
Cowan, C. A., Atienza, J., Melton, D. A. & Eggan, K. Nuclear reprogramming of somatic cells after fusion with human embryonic stem cells. Science 309, 1369–1373 (2005).
Palermo, A. et al. Nuclear reprogramming in heterokaryons is rapid, extensive, and bidirectional. FASEB J 23, 1431–1440 (2009).
Lluis, F., Pedone, E., Pepe, S. & Cosma, M. P. Periodic activation of Wnt/beta-catenin signaling enhances somatic cell reprogramming mediated by cell fusion. Cell Stem Cell 3, 493–507 (2008).
Lluis, F. et al. T-cell factor 3 (Tcf3) deletion increases somatic cell reprogramming by inducing epigenome modifications. Proc Natl Acad Sci USA 108, 11912–11917 (2011).
Foshay, K. M. et al. Embryonic stem cells induce pluripotency in somatic cell fusion through biphasic reprogramming. Mol Cell 46, 159–170 (2012).
Ying, Q. L., Nichols, J., Evans, E. P. & Smith, A. G. Changing potency by spontaneous fusion. Nature 416, 545–548 (2002).
Terada, N. et al. Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature 416, 542–545 (2002).
Lluis, F., Pedone, E., Pepe, S. & Cosma, M. P. The Wnt/beta-catenin signaling pathway tips the balance between apoptosis and reprograming of cell fusion hybrids. Stem Cells 28, 1940–1949 (2010).
Spees, J. L. et al. Differentiation, cell fusion, and nuclear fusion during ex vivo repair of epithelium by human adult stem cells from bone marrow stroma. Proc Natl Acad Sci USA 100, 2397–2402 (2003).
Krishna, S. & Overholtzer, M. Mechanisms and consequences of entosis. Cell Mol Life Sci (2016).
Overholtzer, M. et al. A nonapoptotic cell death process, entosis, that occurs by cell-in-cell invasion. Cell 131, 966–979 (2007).
Sun, Q., Cibas, E. S., Huang, H., Hodgson, L. & Overholtzer, M. Induction of entosis by epithelial cadherin expression. Cell Res 24, 1288–1298 (2014).
Ishikawa, F., Ushida, K., Mori, K. & Shibanuma, M. Loss of anchorage primarily induces non-apoptotic cell death in a human mammary epithelial cell line under atypical focal adhesion kinase signaling. Cell Death Dis 6, e1619 (2015).
Wang, M. et al. Impaired formation of homotypic cell-in-cell structures in human tumor cells lacking alpha-catenin expression. Sci Rep 5, 12223 (2015).
Wen, S., Shang, Z., Zhu, S., Chang, C. & Niu, Y. Androgen receptor enhances entosis, a non-apoptotic cell death, through modulation of Rho/ROCK pathway in prostate cancer cells. Prostate 73, 1306–1315 (2013).
Purvanov, V., Holst, M., Khan, J., Baarlink, C. & Grosse, R. G-protein-coupled receptor signaling and polarized actin dynamics drive cell-in-cell invasion. Elife 3 (2014).
Freeman, B. T., Kouris, N. A. & Ogle, B. M. Tracking fusion of human mesenchymal stem cells after transplantation to the heart. Stem Cells Transl Med 4, 685–694 (2015).
Bonde, S., Pedram, M., Stultz, R. & Zavazava, N. Cell fusion of bone marrow cells and somatic cell reprogramming by embryonic stem cells. FASEB J 24, 364–373 (2010).
Bae, J. S. et al. Bone marrow-derived mesenchymal stem cells promote neuronal networks with functional synaptic transmission after transplantation into mice with neurodegeneration. Stem Cells 25, 1307–1316 (2007).
Shadrin, I. Y., Yoon, W., Li, L., Shepherd, N. & Bursac, N. Rapid fusion between mesenchymal stem cells and cardiomyocytes yields electrically active, non-contractile hybrid cells. Sci Rep 5, 12043 (2015).
Florey, O., Kim, S. E., Sandoval, C. P., Haynes, C. M. & Overholtzer, M. Autophagy machinery mediates macroendocytic processing and entotic cell death by targeting single membranes. Nat Cell Biol 13, 1335–1343 (2011).
Krajcovic, M., Krishna, S., Akkari, L., Joyce, J. A. & Overholtzer, M. mTOR regulates phagosome and entotic vacuole fission. Mol Biol Cell 24, 3736–3745 (2013).
Sosale, N. G., Spinler, K. R., Alvey, C. & Discher, D. E. Macrophage engulfment of a cell or nanoparticle is regulated by unavoidable opsonization, a species-specific ‘Marker of Self’ CD47, and target physical properties. Curr Opin Immunol 35, 107–112 (2015).
Brumatti, G., Sheridan, C. & Martin, S. J. Expression and purification of recombinant annexin V for the detection of membrane alterations on apoptotic cells. Methods 44, 235–240 (2008).
Basiji, D. A., Ortyn, W. E., Liang, L., Venkatachalam, V. & Morrissey, P. Cellular image analysis and imaging by flow cytometry. Clin Lab Med 27, 653–670, viii (2007).
Riento, K. & Ridley, A. J. Rocks: multifunctional kinases in cell behaviour. Nat Rev Mol Cell Biol 4, 446–456 (2003).
Martinez-Rico, C., Pincet, F., Thiery, J. P. & Dufour, S. Integrins stimulate E-cadherin-mediated intercellular adhesion by regulating Src-kinase activation and actomyosin contractility. J Cell Sci 123, 712–722 (2010).
Chu, Y. S. et al. Force measurements in E-cadherin-mediated cell doublets reveal rapid adhesion strengthened by actin cytoskeleton remodeling through Rac and Cdc42. J Cell Biol 167, 1183–1194 (2004).
Li, Y., Sun, X. & Dey, S. K. Entosis allows timely elimination of the luminal epithelial barrier for embryo implantation. Cell Rep 11, 358–365 (2015).
Figeac, F. et al. Nanotubular crosstalk with distressed cardiomyocytes stimulates the paracrine repair function of mesenchymal stem cells. Stem Cells 32, 216–230 (2014).
Sinclair, K. A., Yerkovich, S. T., Hopkins, P. M. & Chambers, D. C. Characterization of intercellular communication and mitochondrial donation by mesenchymal stromal cells derived from the human lung. Stem Cell Res Ther 7, 91 (2016).
Spees, J. L., Olson, S. D., Whitney, M. J. & Prockop, D. J. Mitochondrial transfer between cells can rescue aerobic respiration. Proc Natl Acad Sci USA 103, 1283–1288 (2006).
Abounit, S. et al. Tunneling nanotubes spread fibrillar alpha-synuclein by intercellular trafficking of lysosomes. EMBO J 35, 2120–2138 (2016).
Wang, X., Veruki, M. L., Bukoreshtliev, N. V., Hartveit, E. & Gerdes, H. H. Animal cells connected by nanotubes can be electrically coupled through interposed gap-junction channels. Proc Natl Acad Sci USA 107, 17194–17199 (2010).
Mathieu, P. S. & Loboa, E. G. Cytoskeletal and focal adhesion influences on mesenchymal stem cell shape, mechanical properties, and differentiation down osteogenic, adipogenic, and chondrogenic pathways. Tissue Eng Part B Rev 18, 436–444 (2012).
Murray, P. et al. The self-renewal of mouse embryonic stem cells is regulated by cell-substratum adhesion and cell spreading. Int J Biochem Cell Biol 45, 2698–2705 (2013).
Melloy, P., Shen, S., White, E., McIntosh, J. R. & Rose, M. D. Nuclear fusion during yeast mating occurs by a three-step pathway. J Cell Biol 179, 659–670 (2007).
Holy, J. & Schatten, G. Spindle pole centrosomes of sea urchin embryos are partially composed of material recruited from maternal stores. Dev Biol 147, 343–353 (1991).
Simerly, C. et al. The paternal inheritance of the centrosome, the cell’s microtubule-organizing center, in humans, and the implications for infertility. Nat Med 1, 47–52 (1995).
Acknowledgements
We thank Pedro Carvalho, Maria Garcia-Parajo, Thomas Graf, Victoire Neguembor, Elisa Pedone and Jerome Solon for suggestions on the manuscript. Bryan Welm and Zena Werb for pHIV-H2BmRFP (Addgene plasmid # 18982) and Geoff Wahl for H2B-GFP (Addgene plasmid # 11680). We thank CRG flow cytometer and microscopy facilities, IDIBAPS flow cytometer facility and Centres Científics i Tecnològics de la Universitat de Barcelona (CCiTUB) for the electron microscopy. We are grateful for support from an ERC grant (242630-RERE to M.P.C.), Ministerio de Economia y Competitividad and FEDER funds (BFU2014-54717-P, and BFU2015-71984-ERC to M.P.C.), AGAUR grant (2014 SGR1137 to M.P.C.), the European Union’s Horizon 2020 research and innovation programme under grant agreement CellViewer No 686637 (to M.P.C.), La Caixa international PhD fellowship (to F.S.), People Programme Marie Curie Actions of the European Union’s Seventh Framework Programme (FP7/2007-2013/, n° 290123 to I.T.) and Ministerio de Ciencia e Innovacio´ FPI (to F.A.). We acknowledge support of the Spanish Ministry of Economy and Competitiveness, ‘Centro de Excelencia Severo Ochoa 2013–2017’.
Author information
Authors and Affiliations
Contributions
M.P.C. and F.S. designed the experiments and data analysis. F.S. performed experiments and data analysis. F.A. generated the constructs and I.T. contribute to cell line characterization. M.P.C. and F.S. wrote the manuscript. F.A. and I.T. revised the manuscript. M.P.C. supervised the project.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Sottile, F., Aulicino, F., Theka, I. et al. Mesenchymal stem cells generate distinct functional hybrids in vitro via cell fusion or entosis. Sci Rep 6, 36863 (2016). https://doi.org/10.1038/srep36863
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep36863
This article is cited by
-
Intrinsic signalling factors associated with cancer cell-cell fusion
Cell Communication and Signaling (2023)
-
Primary aldosteronism: molecular medicine meets public health
Nature Reviews Nephrology (2023)
-
Mechanisms underlying the protective effects of mesenchymal stem cell-based therapy
Cellular and Molecular Life Sciences (2020)
-
Enhanced metastatic capacity of breast cancer cells after interaction and hybrid formation with mesenchymal stroma/stem cells (MSC)
Cell Communication and Signaling (2018)
-
Mesenchymal Stem Cells: Miraculous Healers or Dormant Killers?
Stem Cell Reviews and Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.