Abstract
Tree species identity and tree genotypes contribute to the shaping of soil microbial communities. However, knowledge about how these two factors influence soil ecosystem functions is still lacking. Furthermore, in forest ecosystems tree genotypes co-occur and interact with each other, thus the effects of tree genotypic diversity on soil ecosystem functions merit attention. Here we investigated the effects of tree species, tree genotypes and genotypic diversity levels, alongside soil physicochemical properties, on the overall and specific soil enzyme activity patterns. Our results indicate that tree species identity, tree genotypes and genotypic diversity level have significant influences on overall and specific soil enzyme activity patterns. These three factors influence soil enzyme patterns partly through effects on soil physicochemical properties and substrate quality. Variance partitioning showed that tree species identity, genotypic diversity level, pH and water content all together explained ~30% variations in the overall patterns of soil enzymes. However, we also found that the responses of soil ecosystem functions to tree genotypes and genotypic diversity are complex, being dependent on tree species identity and controlled by multiple factors. Our study highlights the important of inter- and intra-specific variations in tree species in shaping soil ecosystem functions in a subtropical forest.
Similar content being viewed by others
Introduction
Individual tree genotypes and genotypic diversity play crucial roles in shaping the structure of communities of organisms in forests by mediating covariance among various associated functional groups such as microorganisms, lichens and invertebrates1. As have been shown in the cases of Populus angustifolia and Populus balsamifera, different tree genotypes can support specific soil microbe, leaf pathogen, foliar fungi, twig endophyte, lichen and arthropod communities2,3,4,5,6,7. Such tree genotype-specific organism communities have been reported to be heritable and consistent across different years4. However, the extent of this community-shaping effect mediated by tree genotype varies according to the species of tree, the environmental conditions, and the associated functional group under consideration, as was shown for the root-microbiome of different genotypes of Populus deltoides8. Most previous studies have analyzed the effects of the genotypes of individual trees on the composition of dependent communities; it is still unknown how tree genotypes and genotypic diversity affect ecosystem functions1,2,3,7.
Measuring enzyme activities in the soil remains the most direct way to investigate soil ecosystem functions9,10, and soil enzyme activities are often used as indicators of soil health and stability11,12,13. In soils, microorganisms produce various enzymes that decompose plant litter and organic compounds; however changes in the structure of the microbial community are not always linked with changes in enzyme activities and the associated ecosystem functions10. This uncoupling between structure and function can be explained by both functional redundancies within microbial communities and the absence of any quantitative link between microbial growth and functions9,10.
Enzyme activities in forest soil are affected by both biotic and abiotic factors14. Among the biotic factors, the composition of tree species, tree species identity and microbial community have been found to be significant predictors of soil enzyme activities15,16,17,18,19. Of the abiotic factors, soil physicochemical properties and substrate quality have often been reported as significantly affecting soil enzyme activities14,16. Biotic and abiotic factors in forest soils are closely linked to the identity of the dominant tree species (interspecific variation), which occupy a pivotal function in forest ecosystems 16,17,20. In particular, feedback between the roots of dominant trees and their associated soil microbial community governs soil enzyme activities21 and consequently has a major impact on ecosystem functions16,21. In addition to tree species identity and diversity, genetic variation within a tree species (intraspecific variation), i.e. the identity22,23 and diversity24 of tree genotypes, has been shown to significantly influence growth, performance and the responses of trees to different environmental parameters. Because of this, factors influenced by tree genotype, e.g. litter quality and root exudate profiles, are also expected to impact on the rhizosphere microbial community and the associated enzymatic activities22.
Soil is the largest pool of organic carbon in terrestrial ecosystem25,26. Organic carbon as well as other organic compounds are distributed to the soil through plant litter and root exudates13. These organic compounds consist of both readily available substrates such as sugars, starch, organic and amino acids, and larger complex substrates such as cellulose, hemi-cellulose and lignin which can be decomposed and transformed to low-molecular-mass compounds by a variety of microbial extracellular enzymes in the soil13,27,28. The low-molecular-mass compounds can then be available in soil for microbial and plant assimilations29. In this context, soil extracellular enzymes produced by microorganisms play vital roles in the biogeochemical reactions of organic matter decomposition, regulate global carbon and nutrient cycles and thus maintain soil functions in terrestrial ecosystem30. Extracellular soil activities are reported to be related to amounts of organic matter inputs, organic carbon, total nitrogen and microbial activity. For examples low and high amounts of leaf litter in the study forest plots show tendencies to decrease and increase enzyme activities (and also organic carbon, total nitrogen and microbial activity), respectively31,32. These shifts in enzyme activity patterns could thus, reflect changes in relative nutrient limitations with altered organic matter inputs as well as indicate the nutrient status and soil microbial activity of the study area31,32.
Although soil enzyme activities are useful indicator for soil health and quality, there are some limitations and cautions that should be considered30. These include, (i) inability of currently used enzyme assays to distinguish between the contributions of different fractions of enzyme activities (i.e. enzymes absorbed by inorganic or organic colloids, enzyme activities due to abiotic transformation and intracellular enzymes in viable cells), (ii) to investigate the microbial functional diversity, measuring a range of enzymes that reflect the same soil ecosystem function of interest is needed otherwise, it is important to identify and measure the key enzyme catalyzing the rate-limiting step of the overall metabolic pathway, (iii) results from specific and overall pattern (i.e. by means of multivariate analysis) of enzyme activities are useful information as the result from overall pattern alone may hinder the specific enzyme activity, and (iv) enzyme activities are highly variable across space and time13,30.
Research has generally focused on enzymes important for the acquisition of macronutrients such as carbon (C), nitrogen (N) and phosphorus (P)9,10,16,33. In this study we used β-glucosidase and xylosidase to represent enzymes of C acquisition10. β-glucosidase catalyzes the hydrolysis of glycosidic bonds in β-D-glucosides and oligosaccharides, whereas xylosidase is important for the degradation of xylooligomers into xylose27. N-acetylglucosaminidase (NAG) and acid phosphatase are representative of N and P acquisition respectively10. NAG catalyzes the hydrolysis of N-acetyl-beta-D-glucosaminide residues in chitin-derived oligomers, while acid phosphatase catalyzes the hydrolysis of phosphoric (mono) ester bonds (i.e. it mineralizes organic phosphorus into phosphate) under acidic conditions27. In addition, we investigated two oxidative enzymes (phenol oxidase and general peroxidase) important in lignin degradation and humus formation, which are important processes in soil C stabilization15.
In this study we carried out an experiment in a recently established platform on genetic diversity in forest trees within the Biodiversity-Ecosystem Functioning (BEF) China project34. Given the fact that the contribution made by tree genotypes to soil microbial communities and their enzymatic activity may depend on the identity of the tree species concerned, we investigated soil enzyme profiles in the root zone of different genotypes of four broadleaf tree species (Alniphyllum fortunei, Cinnamomum camphora, Daphniphyllum oldhamii and Idesia polycarpa) planted at different levels of genetic diversity (Tables S1 and S2).
The major objectives of this study were to investigate (i) the effects of tree species identity, tree genotype and genotypic diversity level on the overall patterns of soil enzyme activity, and (ii) the specific responses of different enzymatic activities important for C, N and P cycling and acquisition to tree species identity, tree genotype and genotypic diversity level. We hypothesized that tree species identity, tree genotype, and genotypic diversity have significant effects on the overall and specific patterns of soil enzyme activity. We assume that interspecific variation has a greater influence on soil physicochemical properties compared to the intraspecific variation. As the four tree species (as compared to the four tree genotypes) used in this experiment differ greatly in leaf litter chemical composition, we expect tree species identity to have more influence than tree genotypes on patterns of soil enzyme activity. Due to plant-microbe and microbe-microbe interactions we hypothesized that there is less competition among soil microbes in a mono-genotypic treatment compared with a multi-genotypic treatment35 and thus, we expected higher enzyme activities in the mono-genotypic treatment.
Results
Overall patterns of soil enzyme activities: interplay between tree species identity, genotype and genotypic diversity level
Tree species identities, tree genotype and genotypic diversity significantly affected the overall patterns of soil enzyme activity (P < 0.01; Fig. 1). They were also correlated with soil physicochemical properties (water content, pH) and substrate quality (C:N ratio) (Fig. 1; Table 1). Distance-based redundancy analysis (dbRDA) showed that tree species identity, genotypic diversity level, soil pH and soil water content were most influential for the overall patterns of soil enzymes (P < 0.05) and were retained for variance partitioning analysis (Table 2).Variance partitioning showed that enzyme activities were explained by plant (tree species identity = 12%; genotypic diversity level = 2%) and soil (pH = 7%; water content = 1%) related factors and the shared fraction between these factors account for 8% (tree species identity and pH = 4%; tree species identity and water content = 4%) (Fig. 2). Pairwise comparisons among tree species showed that the overall enzyme patterns associated with different tree species were significantly different (P = 0.048–0.006; Fig. 1).
Non-metric multidimensional scaling (NMDS) ordination of activities of six soil enzymes under four tree species.
Significant factors (P < 0.05) correlated with overall patterns of enzyme activities are shown. C_N = soil C: N ratio; Water = soil water content. The effects of tree species identity (A = Alniphyllum fortunei; C = Cinnamomum camphora; D = Daphniphyllum oldhamii and I = Idesia polycarpa), tree genotypes (S = seed family) and genotypic diversity analyzed using PERMANOVA are shown on the top left. Pair-wise comparisons of overall patterns of enzyme activities between different tree species are shown on the top right.
In each of the four tree species, overall patterns of soil enzyme activities responded differently to genotype and soil physicochemical and substrate quality. Only genotypic diversity level significantly correlated with the observed patterns of soil enzymes across all tree species (Table 1). Soil pH significantly correlated with the pattern of soil enzymes in three out of the four species (Table 1). For Alniphyllum, level of genotypic diversity, together with tree genotype, soil physicochemical properties (water content, pH) and substrate quality (C content, N content, C: N ratio) shaped the overall enzyme pattern (Table 1, Fig. 3). The effect of tree genotype was not significant for the other three species. In Cinnamomum, enzyme patterns correlated with genotypic diversity level and soil physicochemical properties (water content, pH) and substrate quality (C: N ratio) (Table 1, Fig. 3). In Daphniphyllum, however, the pattern was correlated solely with genotypic diversity level and in Idesia there were correlations with genotypic diversity level and soil pH (Table 1, Fig. 3). Permutational Multivariate Analysis of Variance (PERMANOVA) confirmed that tree genotype significantly affected overall soil enzyme activities in Alniphyllum, while genotypic diversity level played a significant role in Alniphyllum, Cinnamomum and Daphniphyllum but its effect was only marginally significant in Idesia (P = 0.052; Fig. 3).
Non-metric multidimensional scaling (NMDS) ordination of activities of six soil enzymes in each of four tree species.
Significant factors (P < 0.05) correlated with overall patterns of enzyme activities are shown. C_N = soil C: N ratio; Water = soil water content. The effects of tree genotypes (S = seed family) and genotypic diversity analyzed using PERMANOVA are shown on the top left.
Effect of tree species identity on specific enzyme activities
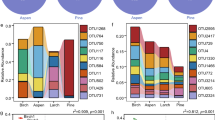
Tree species identity significantly influenced most of the enzyme activities measured, the exception being xylosidase (Fig. 4). The activities of the two hydrolytic enzymes related to C acquisition responded differently to tree species identity. β-glucosidase activity was highest in soil close to Daphniphyllum and lower for the other three tree species, while xylosidase activity was similar for all four species (P > 0.05). NAG activity was highest for Cinnamomum and Daphniphyllum and lowest for Idesia. Acid phosphatase activity was (27–48%) higher in Cinnamomum than in the other three tree species. Phenol oxidase activity was highest for Daphniphyllum followed by Idesia and lowest for Alniphyllum and Cinnamomum. Peroxidase activity was also lowest in the case of Alniphyllum and Cinnamomum and highest in Idesia. Taking the overall results for all specific enzyme activities, we found that different tree species significantly impact on C, N and P related enzymes in different ways.
Enzyme activities (mean ± SE) in soil samples collected under four different tree species (A = Alniphyllum fortunei; C = Cinnamomum camphora; D = Daphniphyllum oldhamii and I = Idesia polycarpa).
Different letters indicate significant differences (P < 0.05) between species according to split-plot ANOVA and an LSD test.
Responses of specific enzymes to genotype and genotypic diversity levels of the different tree species
Responses of the different soil enzyme activities to tree genotype and genotypic diversity level varied among the four tree species. In all but one species, tree genotype and genotypic diversity level showed significant effects on some enzymes (Figs 5 and 6). In Idesia, however, none of the six soil enzymes measured responded to tree genotype or genetic diversity (P > 0.05; Figs 5 and 6).
Hydrolytic enzyme activities (mean ± SE) in soil samples collected under four different genotypes (SA = seed family from Alniphyllum fortune (A); SC = seed family from Cinnamomum camphora (C); SD = seed family from Daphniphyllum oldhamii (D) and SI = seed family from Idesia polycarpa (D)) and two genotypic diversity levels (Mo = mono-genotypic treatment; Mu = multi-genotypic treatment).
Different letters indicate significant differences (P < 0.05) between different genotypes or genotypic diversity levels according to split-plot ANOVA and an LSD test (the latter only for different genotypes).
Oxidative enzyme activities (mean ± SE) in soil samples collected under four different genotypes (SA = seed family from Alniphyllum fortune (A); SC = seed family from Cinnamomum camphora (C); SD = seed family from Daphniphyllum oldhamii (D) and SI = seed family from Idesia polycarpa (I)) and two genotypic diversity levels (Mo = mono-genotypic treatment; Mu = multi-genotypic treatment).
Different letters indicate significant differences (P < 0.05) between different genotypes or genotypic diversity levels according to split-plot ANOVA and LSD test (the latter only for different genotypes).
In Alniphyllum, tree genotypic diversity level had greater effects than tree genotype, and it affected the activity of all enzymes except phenol oxidase (Figs 5 and 6). Multi-genotypic treatment resulted in lower enzyme activities than mono-genotypic treatment in all cases. Tree genotypes, however, significantly influenced peroxidase activity (Fig. 6).
In Cinnamomum, genotypic diversity level also exerted a greater effect than genotype in terms of its effect on most of the hydrolytic enzymes measured, the exception being NAG. However, the responses to genotypic diversity level for the two C acquisition enzymes differed from those of Alniphyllum. β-glucosidase and xylosidase activities were significantly higher in multi-genotypic than in mono-genotypic treatments (Fig. 5). Similar to the findings for Alniphyllum, acid phosphatase was higher in mono-genotypic than multi-genotypic treatments. Tree genotype, however, only significantly affected acid phosphatase activities (Fig. 5). The two oxidative enzymes measured were not affected by either genotypic diversity level or tree genotype.
In Daphniphyllum, tree genotype had a greater effect than tree genotypic diversity level on hydrolytic enzyme activities, in contrast to the findings for Alniphyllum and Cinnamomum. Xylosidase and acid phosphatase activities were significantly and solely influenced by tree genotype. On the other hand, phenol oxidase and peroxidase activities were mainly affected by tree genotypic diversity level; multi-genotypic treatment gave significantly lower enzyme activities in both cases (P < 0.01; Fig. 6).
Discussion
Some recent studies have highlighted the effects of tree species and tree genotypes in shaping the associated soil microbial communities1,35. These results indicate the importance of inter- and intraspecific variations among host species in defining the structure of the microbial community in the soil. To complement these studies, we investigated the effects of tree species and tree genotype on soil enzyme activities, which are among the direct indicators of soil ecosystem functions. Since in real forest ecosystems different tree genotypes co-occur and interact, we also investigated the effects of genotypic diversity level on microbe-mediated soil ecosystem functions36. Our results highlight the importance of inter- and intra-specific variation, and especially of tree genotypic diversity, in shaping soil microbe-driven ecosystem functions across the subtropical experimental forests analyzed here.
We found a major effect of tree species identity (interspecific variation) on the overall, and most of the specific, patterns of soil enzyme activity. This is consistent with a recent study on the effects of tree species in a tropical montane forest16, which reported effects on overall soil enzyme activities and on two of the three specific enzymes (acid phosphatase and β-D-glucosidase) measured, mediated through effects on soil physicochemical and microbial properties. Similarly we also found that the most important factors affecting overall soil enzyme activities were tree species identity, followed by soil physicochemical properties (pH and water content). In total, tree species identity in combination with soil water content, pH and the shared fraction between tree species identity and pH and tree species identity and water content, explain 93% of the explainable variances. This finding may reflect the fact that in forest ecosystems, tree species can affect soil physicochemical properties and substrate quality by altering the quantity and quality of leaf litter input and also via root activities15,16,17.
The four tree species that we studied comprised both evergreen (Cinnamomum and Daphniphyllum) and deciduous (Alniphyllum and Idesia) species, thus the quantities and patterns of leaf litter fall varied greatly within this experimental design37. The physical (i.e. toughness) and chemical (C, N and phenolics and tannin concentrations) traits shown by leaf litter from these species also differed greatly38. Alniphyllum fortunei exhibits low leaf toughness with very high phenolic and tannin contents, whereas Daphniphyllum and Cinnamomum have tougher leaves with low levels of phenolics and tannins (Table S3). Litter quality, especially tannin concentration, has been reported to be responsible for differences in both soil microbial community and enzyme activities16. Specifically, Dacrydium (high tannin content) and Lithocarpus (low tannin content) exhibited significantly different overall enzyme activity patterns, and the soil under Dacrydium also had significantly higher acid phosphatase and β-D-glucosidase activities. Our results support the conclusion that tannin concentrations in leaf litter may affect soil enzyme activity underneath the tree, as Alniphyllum fortunei (high tannin) showed differences in overall enzyme activity that were significant compared with Daphniphyllum and Cinnamomum (both low-tannin species) respectively.
As in the case of species identity effects, the effects of intraspecific variations among tree genotypes on soil enzyme activities may be explained partly by genotype-specific influences on soil physicochemical properties and substrate quality. Particularly in the cases of Alniphyllum, Cinnamomum and Idesia tree genotypes and/or tree genotypic diversity were influential, along with soil physicochemical properties and/or substrate quality, in shaping overall enzyme activity patterns. Different tree genotypes can affect soil physicochemical properties and substrate quality by exerting different specific effects on litter chemical composition and root morphology, which subsequently affect soil microbial community structure35,39,40,41 and lead to unique soil microbial communities35. Interestingly, in Daphniphyllum, we found that genotypic diversity affected the overall soil enzyme activity pattern independently of the effects of the soil physicochemical properties and substrate quality that we measured. Specific enzyme activities were also affected by either tree genotype or tree genotypic diversity. Our results may thus indicate the existence of different host species-related drivers and mechanisms, and the findings highlight the effects of tree genotype and genotypic diversity in shaping soil enzyme activity patterns. Furthermore, the degrees of how different tree genotypes perform and adapt to the environmental parameters (including their feedback on the belowground microbial community) are not equal across different tree species, which might explain why we detected the effects of tree genotype or tree genotypic diversity level on soil enzyme activity patterns only in some cases (Table S2). Functional redundancy within microbial community may also explain our results as the microbial communities under different tree genotypes may be different but composed of communities that could produce similar enzymes9,10. In this study the level of genotypic diversity was found to be one of the most important predictors of overall soil enzyme pattern. This was also the case when each tree species was considered separately, as genotypic diversity level was significantly correlated with overall enzyme patterns in all tree species used in this experiment. In Alniphyllum, the activities of almost all specific soil enzymes were significantly higher in the mono-genotypic treatment than in multi-genotypic treatments. Similar results were found for acid phosphatase in the case of Cinnamomum camphora, and for Daphniphyllum oldhamii for all oxidative enzymes. This may have resulted from the combined effects of plant-microbe and microbe-microbe interactions as we have hypothesized35. As individual tree genotypes may be associated with unique soil microbial communities, competition among different soil microbial communities may be stronger under multi-genotypic treatment than under mono-genotypic treatment1,35. Where there is competition, high energy and resource use can directly and negatively impact the production of microbial enzymes42,43, resulting in the pattern that we observed.
In summary, we provide empirical evidence indicating that tree species identity and tree genotypes (inter- and intra-specific variation), as well as genotypic diversity levels, significantly influence soil ecosystem functions derived from microbial communities. These three factors influence the pattern of soil enzyme activity partly through their effects on soil physicochemical properties and substrate quality. In this ecosystem, both plant and soil related factors influence the pattern of soil microbe derived enzymes: tree species identity >soil pH >shared fractions [(between tree species identity and soil pH) and (tree species identity and soil water content)] >genotypic diversity level >soil water content. Furthermore, our results support the importance of intraspecific interaction (i.e. competition among different tree genotypes of a single tree species) in shaping the overall pattern of microbial enzymes in soils. An investigation of the soil and root-associated microbial communities of these tree species and tree genotypes across genotypic diversity levels using next generation sequencing tools is needed in order to provide a taxonomic and mechanistic understanding of the ways in which these factors affect plant-microbe and microbe-microbe interactions, and therefore microbially-mediated soil ecosystem functions (e.g. nutrient cycling, soil C pools) and developing forest management strategies in subtropical forests.
Methods
Study site
This experiment was conducted at the Biodiversity-Ecosystem Functioning (BEF)-China experimental platform located near Xingangshan village, Jiangxi Province (29.08–29.11 N, 117.90–117.93 E), P.R. China34,44. The climate of the study area is characterized as subtropical summer monsoon. The annual mean temperature and precipitation are 17.4 °C and 1635 mm, respectively. The experimental site was previously used as a Cunninghamia lanceolata plantation. In 2008, this plantation was clear-cut and the BEF-China experimental platform was established. Trees were planted in plots, with 400 tree individuals per plot (25.8 m × 25.8 m; planted in 20 rows of 20 tree individuals each), at a planting distance of 1.29 m, and the platform included a total of 24 tree species on 261 plots in order to investigate biodiversity effects on ecosystem functions34. Within this framework, the genetic diversity experiment represents a factorial combination of species diversity (1 or 4 species) and genetic diversity (1 or 4 maternal seed families per species) to specifically address the effects of genetic identity and genetic diversity. Henceforth we refer to a maternal seed family as a “genotype”, since members of a seed family share one parent in common and are on average more closely related than different seed families. A detailed description of the genetic diversity experiment has been given elsewhere34,45. Four tree species (Alniphyllum fortunei (Hemsley) Makino, Cinnamomum camphora (L.) Presl. Daphniphyllum oldhamii (Hemsley) Rosenthal and Idesia polycarpa Maximowicz.) were used in this experiment. For the present study, we used a subset of the genetic diversity experimental plots and investigated species-monocultures of the four species with either one (mono-genotypic treatment) or four (multi-genotypic treatment) seed families per species. Due to the large number of available monocultures of species and seed families, we used ¼ plots of 10 × 10 individuals for monocultures. No specific permissions were required for these locations and activities. The field studies did not involve endangered or protected species.
Experimental design and sampling
Soil sampling was carried out in October 2014. Soil in the root zones of individual trees of four tree species, with four genotypes and at two genotypic diversity levels per species with four replicates each, was collected using an auger of diameter 8 cm and depth 10 cm. All soil samples were collected within one week in order to minimize the impact of weather variations. All plots were located at the same study site (site B), thus minimizing variation in soil physicochemical properties and quality. Within each plot we selected trees at minimum distances of 3 to 5 m apart to ensure independence among different soil samples. This distance is much larger than the spatial autocorrelation of soil enzymes measured in this study, which is within a range of a few square centimeters13 or up to 1 to 2 m46. Four soil cores were collected in four directions at 0.50–0.60 m from the base of each selected tree within the canopy projection area and pooled to produce one sample. We followed the roots from the tree base to ensure that we obtained soil samples influenced by the selected trees. The pooled samples were immediately homogenized and sieved (2 mm) in the field to remove stones, roots, macrofauna, and litter. Between samples the sieve was cleaned with a brush, sprayed with 70% ethanol and dried. After sieving, two subsamples (30 g) were immediately frozen in dry ice and transported to a laboratory near the study site. One subsample was stored at −20 °C until required for transport in dry ice to Germany for enzyme analysis. The other subsample was freeze-dried for soil physicochemical analyses. Further details of sampling design, characteristics of selected trees and soil physicochemical and substrate quality are shown in supporting information (Tables S1 and S2).
Enzyme activities
Hydrolytic enzyme analyses were done for β-glucosidase (EC 3.2.1.21), xylosidase (EC 3.2.1.37), N-acetylglucosaminidase (EC 3.1.6.1) and phosphatase (EC 3.1.3.2) using 4-methylumbelliferone (MUB) as substrate as described elsewhere27,47. Briefly, soil slurries (sample suspensions) were prepared by adding 0.5 g soil to 50 ml of 50 mM sodium acetate buffer (pH 5.5) followed by homogenization for 5 min in a bath sonicator. Substrate solutions were prepared as follows: 150 (4-MUB-β-glucopyranoside, 4-MUB-β-D-xyloside and 4-MUB-phosphate) or 200 (4-MUB-N-acetyl-β-glucosaminide) μmol/L. Substrate blank wells received 50 μl substrate solution and 200 μl acetate buffer. Assay wells received 50 μl substrate solution and 200 μl soil slurry. Quench coefficient and emission coefficient wells received 50 μl of a MUB dilution series (concentrations (μmol/L) were: 2.5, 1.25, 0.625, 0.16) and 200 μl soil slurry or 200 μl acetate buffer. Homogenate blanks received 200 μl soil slurry and 50 μl acetate buffer. Plate blanks received 250 μl acetate buffer. There were eight replicate wells for each substrate blank, assay, quench standard, emission standard, homogenate blank and plate blank. The microplates were incubated in the dark at room temperature for 60 min. To stop the reaction, a 10 μl aliquot of 1.0 M NaOH was added to each well approximately 1 minute before reading the plates. Fluorescence was measured using a microplate fluorometer with 360 nm excitation and 465 nm emission filters. After correcting for blanks, emission and quenching, activities were expressed in units of nmol hr−1 g dry soil−1. Oxidative enzyme analyses were done for phenol oxidase (EC 1.10.3.2) and general peroxidase (EC 1.11.1.7) using 3, 3′, 5, 5′-tetramethylbenzidine (TMB) as substrate48. The protocol was slightly modified after pre-testing27. Briefly, sample suspensions were prepared as described above. Substrate solutions (TMB) were prepared in sodium citrate buffer with (peroxidase) or without (phenol oxidase) hydrogen peroxide at a concentration of 0.283 μmol/L. Substrate blank wells received 50 μl substrate solution and 200 μl 50 mM sodium acetate buffer. Assay wells received 50 μl substrate solution and 200 μl soil suspension. Homogenate blanks received 200 μl soil suspension and 50 μl sodium acetate buffer. Plate blanks received 250 μl sodium acetate buffer. This assay was also done with eight replicate wells. The microplates were incubated in the dark at room temperature for 20 min. To stop the reaction, a 30 μl aliquot of 1.5 M sulfuric acid was added to each well, and then plates were centrifuged at 2000 rpm for 5 min. Supernatant (200 μl) from each well (avoiding soil particles) was pipetted into a clean clear plate. The absorbance was measured using a microplate fluorometer with 260 nm excitation and 465 nm emission filters.
Soil physicochemical property and quality analyses
Water content was calculated as the difference between initial and freeze-dried soil weight. Total C and N and C: N ratio were determined by dry combustion at 1000 °C with an Elementar Vario EL III (Hanau, Germany) elemental analyzer (DIN/ISO 10694 (Aug. 1996)). Soil pH was determined based on 10 g homogenized soil. Water content and pH were classed as soil physicochemical properties. The C: N ratio was used as a soil quality factor.
Statistical analysis
The effects of tree species identity, tree genotype and genotypic diversity level on overall soil enzyme patterns for the six enzymes were analyzed using Permutational Multivariate Analysis of Variance (PERMANOVA) based on Euclidean distances implemented in PAST49. Statistical significances were based on 999 permutations and Bonferroni-corrected P values were applied in all cases when more than two groups were compared with PERMANOVA. Non-metric multidimensional scaling (NMDS) ordination based on Euclidean distances with the envfit function of the vegan package in R was used to investigate and visualize correlations among different factors influencing overall soil enzyme activities50. All significant factors (P < 0.05) were plotted in the respective NMDS ordinations. We determine the individual factors that were most influential for the overall pattern of soil enzyme activity using distance-based redundancy analysis (dbRDA), based on Euclidean and Bray–Curtis distances with the function capscale in vegan51. We tested for collinearity between all factors using Spearman’s rank correlation (ρ) before carrying out dbRDA. When two or more factors were correlated (ρ > 0.70 or ρ < −0.70 and P < 0.01), only one factor was retained for further analysis. In this case, only total soil C and total soil N were highly correlated (ρ = 0.86, P < 0.01), thus only N was kept for dbRDA. Four factors including tree species identity, genotypic diversity level, soil pH and soil water content were significant based on dbRDA and were selected for variance partitioning analysis. Variance partitioning analysis was performed using the varpart function in the vegan package in R to take into account the influence of four significant factors based on dbRDA (including both soil and plant related factors) on overall enzyme activity pattern51,52,53. We used the same enzyme activity matrix and factors as used for NMDS analysis. We also test whether shared fraction of these four factors explains the variance of soil enzyme activity53,54,55. The effects of tree species identity on specific enzyme activities were analyzed (as between plot factor) using split-plot ANOVA simultaneously accounting for tree genotype (within plot factor) and genotypic diversity level (between plot factor) (IBM SPSS Statistics 22, New York, NY, USA). Effects of tree genotype (within plot factor) and genotypic diversity level (between plot factors) were also analyzed for each tree species using a split-plot ANOVA. All data sets were tested for normality and the equality of group variances using a JB test and the Levene statistic56 and log10 transformed when necessary. Fisher’s Least Significant Difference (LSD) post hoc test was applied when more than two treatments were tested (IBM SPSS Statistics 22, New York, NY, USA).
Additional Information
How to cite this article: Purahong, W. et al. Tree species, tree genotypes and tree genotypic diversity levels affect microbe-mediated soil ecosystem functions in a subtropical forest. Sci. Rep. 6, 36672; doi: 10.1038/srep36672 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Lamit, L. J. et al. Tree genotype mediates covariance among communities from microbes to lichens and arthropods. J. Ecol. 103, 840–850 (2015).
Lamit, L. J. et al. Tree genotype and genetically based growth traits structure twig endophyte communities. Am. J. Bot. 101, 467–478 (2014).
Lamit, L. J. et al. Genotype variation in bark texture drives lichen community assembly across multiple environments. Ecology 96, 960–971 (2014).
Keith, A. R., Bailey, J. K. & Whitham, T. G. A genetic basis to community repeatability and stability. Ecology 91, 3398–3406 (2010).
Whitham, T. G. et al. Community specificity: life and afterlife effects of genes. Trends Plant Sci. 17, 271–281 (2012).
Busby, P. E., Newcombe, G., Dirzo, R. & Whitham, T. G. Differentiating genetic and environmental drivers of plant–pathogen community interactions. J. Ecol. 102, 1300–1309 (2014).
Bálint, M. et al. Host Genotype Shapes the Foliar Fungal Microbiome of Balsam Poplar (Populus balsamifera). PLoS ONE 8, e53987 (2013).
Shakya, M. et al. A Multifactor Analysis of Fungal and Bacterial Community Structure in the Root Microbiome of Mature Populus deltoides Trees. PLoS ONE 8, e76382 (2013).
Frossard, A., Gerull, L., Mutz, M. & Gessner, M. O. Disconnect of microbial structure and function: enzyme activities and bacterial communities in nascent stream corridors. ISME J. 6, 680–691 (2012).
Purahong, W. et al. Uncoupling of microbial community structure and function in decomposing litter across beech forest ecosystems in Central Europe. Sci. Rep. 4 (2014).
Acosta-Martínez, V. & Tabatabai, M. A. Enzyme activities in a limed agricultural soil. Biol. Fertil. Soils 31, 85–91 (2000).
Burns, R. G. & Dick, R. P. Enzymes in the Environment: Activity, Ecology, and Applications. (CRC Press, 2002).
Baldrian, P. Distribution of Extracellular Enzymes in Soils: Spatial Heterogeneity and Determining Factors at Various Scales. Soil Sci. Soc. Am. J. 78, 11–18 (2014).
Brockett, B. F. T., Prescott, C. E. & Grayston, S. J. Soil moisture is the major factor influencing microbial community structure and enzyme activities across seven biogeoclimatic zones in western Canada. Soil Biol. Biochem. 44, 9–20 (2012).
Weand, M. P., Arthur, M. A., Lovett, G. M., McCulley, R. L. & Weathers, K. C. Effects of tree species and N additions on forest floor microbial communities and extracellular enzyme activities. Soil Biol. Biochem. 42, 2161–2173 (2010).
Ushio, M., Kitayama, K. & Balser, T. C. Tree species effects on soil enzyme activities through effects on soil physicochemical and microbial properties in a tropical montane forest on Mt. Kinabalu, Borneo. Pedobiologia 53, 227–233 (2010).
Binkley, D. & Giardina, C. Why Do Tree Species Affect Soils? The Warp and Woof of Tree-Soil Interactions. Biogeochemistry 42, 89–106 (1998).
Grierson, P. F. & Adams, M. A. Plant species affect acid phosphatase, ergosterol and microbial P in a Jarrah (Eucalyptus marginata Donn ex Sm.) forest in south-western Australia. Soil Biol. Biochem. 32, 1817–1827 (2000).
Fang, S., Liu, D., Tian, Y., Deng, S. & Shang, X. Tree Species Composition Influences Enzyme Activities and Microbial Biomass in the Rhizosphere: A Rhizobox Approach. PLoS ONE 8, e61461 (2013).
Binkley, D. & Valentine, D. Fifty-year biogeochemical effects of green ash, white pine, and Norway spruce in a replicated experiment. For. Ecol. Manag. 40, 13–25 (1991).
Gianfreda, L. Enzymes of importance to rhizosphere processes. J. Soil Sci. Plant Nutr. 15, 283–306 (2015).
Whitham, T. G. et al. Community and ecosystem genetics: a consequence of the extended phenotype. Ecology 84, 559–573 (2003).
Aspinwall, M. J., King, J. S. & McKeand, S. E. Productivity differences among loblolly pine genotypes are independent of individual-tree biomass partitioning and growth efficiency. Trees 27, 533–545 (2012).
Latutrie, M., Mérian, P., Picq, S., Bergeron, Y. & Tremblay, F. The effects of genetic diversity, climate and defoliation events on trembling aspen growth performance across Canada. Tree Genet. Genomes 11, 1–14 (2015).
Jobbágy, E. G. & Jackson, R. B. The Vertical Distribution of Soil Organic Carbon and Its Relation to Climate and Vegetation. Ecol. Appl. 10, 423–436 (2000).
Post, W. M., Emanuel, W. R., Zinke, P. J. & Stangenberger, A. G. Soil carbon pools and world life zones. Nature 298, 156–159 (1982).
German, D. P. et al. Optimization of hydrolytic and oxidative enzyme methods for ecosystem studies. Soil Biol. Biochem. 43, 1387–1397 (2011).
Purahong, W. et al. Influence of Different Forest System Management Practices on Leaf Litter Decomposition Rates, Nutrient Dynamics and the Activity of Ligninolytic Enzymes: A Case Study from Central European Forests. PLoS ONE 9, e93700 (2014).
Allison, S. D. & Vitousek, P. M. Responses of extracellular enzymes to simple and complex nutrient inputs. Soil Biol. Biochem. 37, 937–944 (2005).
Nannipieri, P., Kandeler, E. & Ruggiero, P. In Enzymes in the Environment (eds R. G. Burns & R. Dick ) 1–33 (Marcel Dekker, 2002).
Sun, X.-L., Zhao, J., You, Y.-M. & Jianxin Sun, O. Soil microbial responses to forest floor litter manipulation and nitrogen addition in a mixed-wood forest of northern China. Sci. Rep. 6, (2016).
Weintraub, S. R., Wieder, W. R., Cleveland, C. C. & Townsend, A. R. Organic matter inputs shift soil enzyme activity and allocation patterns in a wet tropical forest. Biogeochemistry 114, 313–326 (2012).
Wang, Q., Xiao, F., He, T. & Wang, S. Responses of labile soil organic carbon and enzyme activity in mineral soils to forest conversion in the subtropics. Ann. For. Sci. 70, 579–587 (2013).
Bruelheide, H. et al. Designing forest biodiversity experiments: general considerations illustrated by a new large experiment in subtropical China. Methods Ecol. Evol. 5, 74–89 (2014).
Schweitzer, J. A. et al. Plant–Soil–Microorganism Interactions: Heritable Relationship Between Plant Genotype and Associated Soil Microorganisms. Ecology 89, 773–781 (2008).
Ehlers, B. K., Damgaard, C. F. & Laroche, F. Intraspecific genetic variation and species coexistence in plant communities. Biol. Lett. 12, 20150853 (2016).
Staelens, J. et al. Aporte, descomposición de hojarasca y mineralización de nitrógeno en bosques siempreverdes de antiguo crecimiento y bosques secundarios deciduos, centro-sur de Chile. Rev. Chil. Hist. Nat. 84, 125–141 (2011).
Eichenberg, D., Purschke, O., Ristok, C., Wessjohann, L. & Bruelheide, H. Trade-offs between physical and chemical carbon-based leaf defence: of intraspecific variation and trait evolution. J. Ecol. 103, 1667–1679 (2015).
Schweitzer, J. A. et al. The interaction of plant genotype and herbivory decelerate leaf litter decomposition and alter nutrient dynamics. Oikos 110, 133–145 (2005).
O’reilly-Wapstra, J. M., Mcarthur, C. & Potts, B. M. Linking plant genotype, plant defensive chemistry and mammal browsing in a Eucalyptus species. Funct. Ecol. 18, 677–684 (2004).
Kondo, M. et al. In Roots: The Dynamic Interface between Plants and the Earth (ed. Abe, J. ) 189–200 (Springer: Netherlands,, 2003).
Hoppe, B. et al. Linking molecular deadwood-inhabiting fungal diversity and community dynamics to ecosystem functions and processes in Central European forests. Fungal Divers. 77, 367–379, doi: 10.1007/s13225-015-0341-x (2016).
Fukami, T. et al. Assembly history dictates ecosystem functioning: evidence from wood decomposer communities. Ecol. Lett. 13, 675–684 (2010).
Yang, X. et al. Establishment success in a forest biodiversity and ecosystem functioning experiment in subtropical China (BEF-China). Eur. J. For. Res. 132, 593–606 (2013).
Hahn, C., Niklaus, P., Fischer, M. & Durka, W. The effects of species and genetic diversity on ecosystem functioning: a study in experimental tree communities in subtropical China. (in prep).
Luis, P. et al. Patchiness and spatial distribution of laccase genes of ectomycorrhizal, saprotrophic, and unknown basidiomycetes in the upper horizons of a mixed forest cambisol. Microb. Ecol. 50, 570–579 (2005).
Sinsabaugh, R. L. et al. Soil microbial activity in a Liquidambar plantation unresponsive to CO2-driven increases in primary production. Appl. Soil Ecol. 24, 263–271 (2003).
Johnsen, A. R. & Jacobsen, O. S. A quick and sensitive method for the quantification of peroxidase activity of organic surface soil from forests. Soil Biol. Biochem. 40, 814–821 (2008).
Hammer, Ø., Harper, D. A. T. & Ryan, P. D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 4, 9 (2001).
Oksanen, J. Multivariate analysis of ecological communities in R: vegan tutorial. URL: http://cran.r-project.org. (2013).
Wu, Y. T. et al. Forest Age and Plant Species Composition Determine the Soil Fungal Community Composition in a Chinese Subtropical Forest. PLoS ONE 8, e66829 (2013).
Oksanen, J. et al. Vegan: Community Ecology Package. R-package version 2, 4–0 (2016).
Borcard, D., Legendre, P. & Drapeau, P. Partialling out the Spatial Component of Ecological Variation. Ecology 73, 1045–1055 (1992).
Peres-Neto, P. R., Legendre, P., Dray, S. & Borcard, D. Variation Partitioning of Species Data Matrices: Estimation and Comparison of Fractions. Ecology 87, 2614–2625 (2006).
Pei, Z. et al. Soil and tree species traits both shape soil microbial communities during early growth of Chinese subtropical forests. Soil Biol. Biochem. 96, 180–190 (2016).
Purahong, W. et al. Changes within a single land-use category alter microbial diversity and community structure: Molecular evidence from wood-inhabiting fungi in forest ecosystems. J. Environ. Manage. 139, 109–119 (2014).
Acknowledgements
Our work was funded in part by grants from the DFG-German Research Foundation (WU 614/1–3; BU941/12–2). We thank Keping Ma, Zhiqin Pei, Xiaojuan Liu, Yang Bo, Chen Lin and the entire team of BEF-China for their support. We would also like to thank Christoph Hahn for compiling the tree data and Katalee Jariyavidyanont and Antje Thondorf for their help with measuring enzyme activity and soil physicochemical property. We thank the Sino-German Centre for funding writing summer schools.
Author information
Authors and Affiliations
Contributions
T.W., W.D., M.F., F.B. and W.P. conceived and designed the experiments. W.P. performed the field experiments. W.P., S.D. and R.S. performed the laboratory work. W.P. and T.W. analysed the data, interpreted the results and wrote the manuscript. T.W. and F.B. obtained funding. W.P., T.W., W.D., F.B., S.D. and R.S. contributed to revision of the manuscript and gave approval for submission.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Purahong, W., Durka, W., Fischer, M. et al. Tree species, tree genotypes and tree genotypic diversity levels affect microbe-mediated soil ecosystem functions in a subtropical forest. Sci Rep 6, 36672 (2016). https://doi.org/10.1038/srep36672
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep36672
This article is cited by
-
Enhancing Tree Performance Through Species Mixing: Review of a Quarter-Century of TreeDivNet Experiments Reveals Research Gaps and Practical Insights
Current Forestry Reports (2024)
-
Adaptive mechanisms of Ardisia crenata var. bicolor along an elevational gradient on Gaoligong Mountain, Southwest China
Journal of Mountain Science (2023)
-
Plant Traits and Phylogeny Predict Soil Carbon and Nutrient Cycling in Mediterranean Mixed Forests
Ecosystems (2023)
-
Life in the Wheat Litter: Effects of Future Climate on Microbiome and Function During the Early Phase of Decomposition
Microbial Ecology (2022)
-
Fungal community succession on decomposing leaf litter across five phylogenetically related tree species in a subtropical forest
Fungal Diversity (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.