Abstract
Brain responses to facial attractiveness induced by facial proportions are investigated by using functional magnetic resonance imaging (fMRI), in 41 young adults (22 males and 19 females). The subjects underwent fMRI while they were presented with computer-generated, yet realistic face images, which had varying facial proportions, but the same neutral facial expression, baldhead and skin tone, as stimuli. Statistical parametric mapping with parametric modulation was used to explore the brain regions with the response modulated by facial attractiveness ratings (ARs). The results showed significant linear effects of the ARs in the caudate nucleus and the orbitofrontal cortex for all of the subjects, and a non-linear response profile in the right amygdala for only the male subjects. Furthermore, canonical correlation analysis was used to learn the most relevant facial ratios that were best correlated with facial attractiveness. A regression model on the fMRI-derived facial ratio components demonstrated a strong linear relationship between the visually assessed mean ARs and the predictive ARs. Overall, this study provided, for the first time, direct neurophysiologic evidence of the effects of facial ratios on facial attractiveness and suggested that there are notable gender differences in perceiving facial attractiveness as induced by facial proportions.
Similar content being viewed by others
Introduction
Facial attractiveness is a facial attribute that conveys significant biological advantages as expressed in mating success1, earning potential2 and longevity3, across different cultures and age groups4. Facial attractiveness judgment even exists in infancy5. A large body of studies have found that facial attributes that contribute to attractiveness include averageness6,7,8,9, symmetry9,10, sexual dimorphism11,12, expression13, and skin texture14. Among these factors, averageness and symmetry were found to be important criteria. Attractiveness increases with an increasing level of averageness and symmetry, which can be understood as evolutionary pressures that operate against the extremes of the population15.
Apart from averageness and symmetry, the sizes of individual features significantly influence the perception of facial attractiveness. Previous studies that address measuring certain facial features or manipulating individual feature sizes have found that female faces are more attractive when the faces have certain features, such as large eyes, prominent cheekbones, thick lips, thin eyebrows and a small nose and chin16,17. It is believed that an average face is attractive, but not optimally attractive, because attractive composite faces can be made to be more attractive by changing some of the feature sizes to be different from the sample mean18. Moreover, enhanced masculine facial characteristics increase both perceived dominance and negative attributes (e.g., coldness or dishonesty), which suggests that humans have a selection pressure that limits sexual dimorphism and encourages neoteny11.
In addition, some of the other behavioral studies have also attempted to make clear whether the facial measurements should follow certain defined ratios for attractive faces19,20. For example, men with higher width-to-height ratios (fWHRs) of faces are positively associated with having self-perceived power21, perceived dominance, and attractiveness to women for short-term relations22,23. A recent study reported that there possibly exist golden ratios of the face’s vertical distance between the eyes and the mouth and of the horizontal distance between the eyes, although different faces have varying attractiveness24. In particular, Fan et al.25 investigated the effect of facial proportions on facial attractiveness using computer generated face images with controlled skin tone and expression, and they found a different ideal facial proportion for facial attractiveness.
Despite the substantial number of behavioral studies on the influence of facial proportions or features sizes on facial attractiveness, little is known of how human brains respond to facial attractiveness as induced by varying the facial proportions. Past neuroimaging studies using real faces have found that some emotion- and reward-related regions, which involve the orbitofrontal cortex (OFC), anterior cingulate cortex, nucleus accumbens, caudate, and amygdala, respond to facial attractiveness26,27,28,29. More recent meta-analyses have observed consistent activations to be associated with facial attractiveness across neuroimaging studies30,31, especially in putative reward circuitry, such as linearly increased responses in the medial orbitofrontal cortex and nucleus accumbens for attractive faces27, and non-linear responses in the amygdala for both attractive and unattractive faces26. However, there are multiple dimensional aspects of facial characteristics that influence facial attractiveness perception, such as facial expressions, hairstyles and skin tones. Hence, it is extremely difficult to disentangle the effect of an individual factor from the composite effects of these factors in natural face images, despite a recent trend in exploring the high-dimensional nature of neural representations that characterize social perception in natural and realistic environments32. In the present study, we therefore used computer-generated, yet realistic face images that had varying facial proportions as stimuli to investigate the brain responses to facial attractiveness as induced by facial proportions. This approach allowed us to explore only the effect of facial ratios on facial attractiveness using an event-related fMRI design, by manipulating the individual facial ratios and controlling the effects of other confounding factors, such as the hairstyle, skin texture and expression, to test whether some of the facial ratios or their combinations contribute to facial attractiveness.
Another objective of this study is to investigate whether there is any difference in the brain responses to facial attractiveness as induced by facial proportions between male and female viewers. Behavioral studies have indicated that men assign greater importance to physical attractiveness compared with women when evaluating a potential mate33. More specifically, sexual preferences for some facial features, such as lip size, mouth width, cheekbones height and chin size, could result in gender differences in the perception of facial attractiveness20,34. Furthermore, our recent behavioral studies have demonstrated gender differences in ranking facial physical attractiveness25. In fact, there has been some evidence of subject gender-by-attractiveness in judging face attractiveness, especially in evaluating opposite-sex faces26,33,35. For example, increased responses in some reward-encoding regions such as OFC35 and anterior cingulate26 were observed only for male participants. When viewing male faces, women showed stronger linear effects, while men showed stronger nonlinear effects, etc. These findings suggest that the reward value of facial attractiveness is more pronounce in men than that in women, and support sex differences in mate selection in that men identify attractiveness as a stronger motivation35. Inspired by these previous findings, we therefore speculate that there exist gender differences between the neural responses of male and female brains, which underlie the behavioral discrepancy between genders in the judgment of facial geometric attractiveness.
Methods
Face images as stimuli
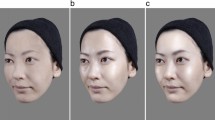
All of the face images that were used as stimuli in this study were selected from our previous study25. All of them were created by altering certain key facial dimensions using computer software from a so called “Original Face” (See Fig. 1), which was obtained by averaging the features of the facial images of some arbitrarily chosen famous oriental ladies (Japanese, Korean and Chinese) that were available on the internet. Furthermore, the faces were hairless to eliminate the effects of hairstyles, and they were controlled to have the same neutral facial expression and skin tone. Finally, we created sufficient but realistic variations in the facial dimensions and ratios of the facial images, by applying gradual alteration to the “Original Face” with nine different approaches (See Fig. 2 and also refer to ref. 25 for details of the method). During each approaches, the maximum possible alteration of the targeted dimension(s) without being overly unrealistic was determined by a preliminary visual assessment. Then, the change in each step of the alterations was calculated by dividing the maximum possible dimensional alternation by 14. This strategy resulted in 14 progressive facial images in each approach. The above nine approaches created wide variations in the length and width of the face, the length of the nose, and the positions of the eyes, while keeping other features such as the size of the eyes, the width of the nose, and the size and shape of the mouth unchanged. Furthermore, based on the “original face” and the selected 18 facial images created by the above 9 approaches, the mouth width, eye fissure width, nose width and lip height were altered in a pre-defined proportion by the orthogonal experimental design25. Using this method, a total of 432 face images were created for further study.
The alterations of the “Original Face” to generate the other facial images.
(A) demonstrates nine different approaches (a–i) of changing the facial ratios (refer to Fan et al.,25 for detail). (B) Examples of facial images generated for this study.
The above approach of manipulating individual feature sizes provides a simple but efficient way in which we can search the entire space of face shapes starting from the mean face of a cohort, with minimization of the sample size and preservation of the symmetry. Unlike previous studies that intentionally exaggerate masculine or feminine facial characteristics11, variations in the facial proportions or feature sizes were created by a stepwise procedure (e.g., −10%, −5%, + 5% and + 10% for the mouth width), regardless of the sexual dimorphism dimension. Moreover, the levels of proportional changes for all of the facial ratios were optimally determined, to have a wider range of variation but without creating too unrealistic images. These strategies for face image generation facilitate the investigation of only the effect of facial ratios on facial attractiveness as independent variables.
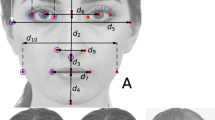
All of the face images have variable geometric morphometric parameters that consist of morphological and/or functional points as well as contours of the eyes, nose and mouth. Based on the previous literature36, we further identified 29 landmarks (See Fig. 1) for each sample image using computer software, and we generated the 21 ratios listed in Table 1 by measuring the vertical or horizontal distances between these landmarks. The measures of the 21 ratios compose the low-level features that result in facial attractiveness for an individual face image. Furthermore, for each face, its distance from the ‘original face’ was calculated by the Euclidean distance between its ratio features and the ratio features of the ‘original face’.
Human Subjects
fMRI data were collected from 41 healthy subjects (22 males) with normal or corrected-to-normal vision who gave written informed consent to participate in this study. All of the subjects were revealed to have non-homosexual preferences by using a debriefing questionnaire. There was no significant difference between the ages of the males and females (age: 23.57 ± 1.10, p = 0.745). All of the participants were in good health and had no past history of psychiatric or neurological diseases. Only 36 subjects (19 males) were available for further analysis after head motion inspection (see the Data Preprocessing section). This study was conducted according to the principles in the Declaration of Helsinki and approved by the Ethics Committee of the Third Xiangya Hospital of Central South University, Changsha, China.
Experimental paradigm
Each human subject underwent an explicit judgment task of facial attractiveness on the face stimulus. Face images (800 × 600 pixels; subtending 23 × 18° of visual angle) were presented in the center of the SVGA display, using an MR-compatible liquid crystal display goggle operating at a resolution of 800 × 600 pixels, at 60 Hz. The stimuli series were generated with the E-Prime software (http://www.pstnet.com/eprime.cfm). The subjects were scanned in four runs, and each run involved 108 face presentation trials in which a face image was presented for 1 second followed by a fixation cross for a further 2 seconds, and the subjects were instructed to maintain fixation. In each run, 54 null event trials, in which a fixation cross was presented for 3 seconds, were randomly interspersed with the face presentation trials. These face images (432 faces in total) were selected once for all of the subjects, and were randomly presented during the scanning. The scanner was in the acquisition mode for 15 seconds before each series to achieve steady-state transverse magnetization. Following the presentation of each face, the subjects were instructed to judge the attractiveness by pressing one of four buttons with their right hands (denoting “highly attractive” and “attractive”) and their left hands (denoting “unattractive” and “highly unattractive”) on a response keypad, respectively. Their response time from the presentation of a face to the pressing of a button was also recorded.
fMRI data
Data were collected on a 3.0T MRI scanner (Philips, MR Ingenia, the Netherland) at the Third Xiangya Hospital of Central South University, Hunan Province, China. To reduce their head motion, the participants’ heads were fixed using foam pads with a standard birdcage head coil. The functional images were collected using a gradient-echo T2*-weight echo-planar imaging (EPI) sequence with the following parameters: repetition time (TR) = 3000 ms, echo time (TE) = 40 ms, field of view (FOV) = 240 × 240 mm, flip angle (FA) = 90°, slice thickness = 2 mm, gap = 1 mm, and matrix = 88 × 86. The volumes consisted of 36 slices angled at −30° to the horizontal, which could improve the signal quality in the ventral prefrontal cortex and amygdala26. Each session lasted 8 min and 15 seconds, and 165 volumes were obtained. Finally, T1-weighted anatomical images were acquired for each subject for detailed anatomical information.
Debriefing
The participants undertook one debriefing task outside the scanner immediately after the experiments. They were asked to rate all of the faces in term of attractiveness, using a computerized visual analogue scale. The scale was marked with the extremes of “highly unattractive” and “highly attractive”, with the mid-point marked. The ratings were scaled between 1 and 9, with 9 representing ratings of the highest attractiveness. The mean attractiveness rating (AR) for each image was obtained by averaging the ARs over all of the subjects.
Data preprocessing
Imaging data were preprocessed and analyzed using SPM8 (Wellcome Department of Imaging Neuroscience, London; http://www.fil.ion.ucl.ac.uk/spm). For each participant, the first three volumes of scanned data were discarded because of magnetic saturation effects. The remaining volumes were realigned to the first image for correcting the head motion, and a mean functional image was correspondingly obtained. Three male and two female participants whose head motion exceeded 1.8 mm were discarded, and the residual 36 participants had less than 1 mm of translation in the x, y, or z axis and 2° of rotation in each axis. The images were then normalized to an MNI echo planar imaging template with an affine registration followed by a nonlinear transformation, using a voxel size of 3 × 3 × 3 mm. Finally, a 6-mm FWHM Gaussian kernel was used to smooth the data.
Statistical parametric mapping analysis
The functional images were analyzed statistically using a two-stage procedure to explore the brain responses while judging the attractiveness of faces that had varying facial proportions. In the first stage, we estimated the effect sizes for each regressor in the design matrix within the framework of the generalized linear model, with parameter modulation for each subject. To identify the brain regions that showed a linear or nonlinear effect of attractiveness ratings, we used a parametric approach37 that allows one to characterize brain responses as a linear combination of (basis) functions of the experimental parameter (e.g., f(x) = a + bx + cx2, where x is the subject-specific attractiveness ratings). We used a first-order and a second-order polynomial expansion, respectively, as the basis functions in which each polynomial term represents an interaction between the attractiveness ratings and a boxcar stimulus function box. The standard boxcar function (i.e., zeroth order) modes the difference between the face presentation and the silent baseline condition, irrespective of the face attractiveness. The linear (i.e., lin = r · box) term and the quadratic (i.e., sec = r2 · box) term account for the linear and second-order nonlinear changes in the blood oxygen level dependent (BOLD) signal relative to the parameter of facial attractiveness ratings, respectively. Prior to model fitting, these terms (as parametric modulations in SPM8) were convolved with a hemodynamic response function.
In the design matrix of the first level model, the covariates of interest include the linear term and the quadratic term, both of which model all of the trials on one regressor but includes a linear or second-order nonlinear parametric modulation of this regressor by using subject-specific ratings for each image. A linear (f(x) = a + b · AR) and a quadratic nonlinear polynomial function (f(x) = a + b · AR + c · AR2) of attractiveness ratings (ARs) were used to estimate the regions that had linear and nonlinear effects of facial attractiveness, respectively. In the linear expansion model, the remaining columns (confounds or covariates of no interest) include the zeroth-order term (modeling the main effect of face presentation irrespective of attractiveness), a constant term, regressors that pertain to the reaction time for each trial and six head movement parameters. However, in the quadratic expansion model, the linear term of attractiveness ratings was also moved into the covariates of no interest. The obtained effects of the linear term and the second-order term across the population of subjects were submitted to the second stage of analysis, in which the significance of the linear and nonlinear effects was tested by using random effects analyses with ANOVA models in SPM8.
In the second level analysis, men and women were treated as a single group, and a one-sample t-test was used to evaluate the linear or second-order nonlinear effects of the facial attractiveness irrespective of the subject gender. Additionally, for estimating the effect of the subject gender-by-attractiveness, the males and females were also treated as separate groups and a two-sample t-test (with contrasts of 1 for the males and −1 for the females) was applied to investigate the gender difference in the brain responses to facial attractiveness. All of the activation was reported by using the more liberal threshold of p < 0.001 (uncorrected) to explore potential regions that respond to perceiving the attractive faces. We also used small volume corrections (SVCs, 12 mm radius) with p < 0.05 as the significance threshold to indicate whether the activation within the regions of interest (ROIs) can survive for multiple comparisons over small regions of interest. To show the modulation effect of the attractiveness ratings on the brain responses for each subject, we also computed the average BOLD signals for all of the images within an individual attractiveness group as defined by the button that the subject pressed. A bar figure was used to indicate the relative percent signal change of the BOLD signal for different attractiveness-level face images.
Learning the most relevant face ratios from the fMRI responses
Next, we attempted to identify the most relevant low-level feature (face ratio) sets with facial attractiveness, based on the fMRI scans. First, we constructed the fMRI-derived semantic feature sets that were relevant to facial attractiveness, which comprise the neural response of all of the ROIs that significantly linearly responded to the facial attractiveness ratings. The data were then normalized to represent percentage signal changes with respect to the temporal mean of each run. The brain volumes that corresponded to each image trial were extracted from the time series with a lag of 4 s to account for the delay in the hemodynamic response. Using the statistical parameter model, we identified eight brain ROIs that had linear effects of facial attractiveness, including bilateral OFC, caudate, mOFC and postcentral gyrus (See the Results section and Table 2). The signals at the peak within the ROIs were concatenated for the representation of fMRI-derived high-level semantic features that were relevant to the face attractiveness.
Second, we performed canonical correlation analysis (CCA) to extract the most correlated variables from the fMRI-derived semantic features sets and the facial ratio feature sets. CCA can obtain the correlation structure of two sets of latent variables, which represent a set of independent variables and a set of dependent variables38. Assuming two sets of variables, which are denoted

CCA is used to extract the correlated modes between vectors X and Y, by seeking a set of transformation vector pairs, Ai ∈ RP×1 and Bi ∈ Bq×1, in such a way that the linear correlation of the canonical variates ui ∈ Rn×1 and vi ∈ Rn×1 is maximized:

where symbol i denotes the i-th pair of transformation vectors, Ai and Bi, which result in the i-th pair of variates Ui and Vi. The correlation between Ui and Vi is

where CXY is the cross-covariance matrix of X and Y, while CXX and CXY are auto-covariance matrix. By solving the maximization problem of Eq. (3), we obtain the ascending ordered correlation values {ρ1, ρ2, …,ρr} with r ≤ min(p, q), and the corresponding transformation matrices and
and  , in such a way that the elements within
, in such a way that the elements within  and
and  are orthogonal, and the correlation between Ui and Vi is maximized. Thus, the predictability between Ui and Vi is maximized.
are orthogonal, and the correlation between Ui and Vi is maximized. Thus, the predictability between Ui and Vi is maximized.
Here, we considered X to be the fMRI-derived high-level semantic feature vector (the averaged response across all of the subjects to each face image) and Y to be the low-level feature vector (the facial ratios of each face image). With CCA, we learned the most relevant components of the face ratios to the face attractiveness, and the corresponding transform model is represented by the transform matrix B. Note that all of the input data were Z-score normalized prior to the CCA.
Predicting facial attractiveness based on fMRI-derived face ratios
We obtained six components of the face ratios that are the most relevant to face attractiveness via the CCA (see the Results for details). Next, we used the following transformed Gaussian formula to capture the main trend in each of the six components:

where  is the transformed value with the Box-Cox transformation function
is the transformed value with the Box-Cox transformation function

where cm is an arbitrary value that is chosen to ensure that  . The values of ai, di, ui, σi and ri can be determined by applying nonlinear regression.
. The values of ai, di, ui, σi and ri can be determined by applying nonlinear regression.
We constructed the following prediction model of the attractiveness ratings by assuming that the overall facial attractiveness is the linear combination of the contributions of each of the six components:

where the coefficients b0 and bi(i = 1, …, 6) can be obtained by multiple linear regression based on the least-squares model.
Results
Linear effect of attractiveness ratings
We used a linear term of the mean facial attractiveness ratings with a one-sample t-test to evaluate the linear changes in the BOLD signal relative to the facial attractiveness. The results are listed in Table 2. Figure 3 shows a significant positive linear effect of the average attractiveness ratings (higher attractiveness ratings associated with linear increases in the BOLD signals) with the significance level of p < 0.05 (SVC; cluster size >5 voxels) in the caudate nucleus (Fig. 3A), inferior frontal gyrus (Fig. 3B) and right postcentral gyrus, as well as a significant negative linear effect in the left postcentral gyrus.
Linear effects of the attractiveness ratings for all of the subjects.
(A) demonstrates the linear effect of the attractiveness ratings in the caudate nucleus (left panel) and the relative change in the BOLD signals across four different groups (right panel), with group 1 representing the highly unattractive faces and 4 denoting the highly attractive faces. (B) The orbitofrontal cortex (OFC) shows significant positive linear effects of attractiveness ratings (left panel). In other words, more attractive faces evoke stronger activation in the OFC (right panel). Here, p < 0.001 uncorrected is used as the threshold for display. The stimuli were divided into four groups for each subject based upon which button was pressed. Then, the BOLD responses are averaged for each cohort of faces. Because there was no baseline condition in the experimental design, only the relative values of the response with respect to the mean activity over each subject were calculated. The units are the % signal change, and the error bars represent the standard error of the mean over the subjects.
Non-linear effects of attractiveness ratings
A second-order term in the polynomial expansion of facial ARs was used to test whether the addition of a quadratic term accounts for the change in the BOLD signal relative to the subject-specific attractiveness ratings. Only males showed significant nonlinear effects in the right superior frontal gyrus, right superior parietal gyrus, right angular gyrus (BA40) and right precuneus (BA7) (see Table 3 for details). The direct comparison between the males and females demonstrated the significant interaction effects between subject gender and attractiveness ratings in the left cuneus, superior occipital gyrus, angular gyrus, and right amygdala (p < 0.015, SVC), as listed in Table 4. Notably, a significant positive second-order relationship between the BOLD activation and the facial attractiveness was observed in the right amygdala (Fig. 4).
Decoding attractiveness ratings from responses to facial images
We considered the predictor X to be the fMRI-derived feature vector that consists of the BOLD responses within the eight ROIs and the predictant Y to be the 21 facial ratios. Next, we used CCA to extract the most relevant components of the facial ratios with respect to the facial attractiveness. We obtained eight components of the facial ratios that are represented by the columns in the transformation matrix B. Furthermore, the two columns were removed due to their low correlation values (p > 0.001). The residual six columns represent the most relevant components of facial ratios to facial attractiveness, which are listed in Table 5. From the correlations between the six components and the ratios, it can be learned that some of the ratios have higher weights in determining the facial attractiveness. For example, the first and sixth components are largely related to the nose width and the interocular distance. The second and fourth components are largely related to the ratios of mideye distance and the interocular distance to the nose width, and so on. In particular, multiple components that involve the components 1, 3, 5 and 6 indicate that the ratio of the ear length to the interocular distance and the ratio of the lips-chin distance to the interocular distance play important roles in factors that influence the facial attractiveness, which is largely in line with the results from behavioral experiments25. Overall, the ear length, nose size, interocular distance and lip-chin distance are of more importance in determining the facial attractiveness compared with the other feature sizes. We have also visualized the effects of these components by averaging the 20 faces that have the highest and lowest scores on them (see supplemental Fig. S1).
Each component is plotted against the attractiveness ratings in Fig. 5. By applying nonlinear regression, we obtained the coefficients of model (4) and listed them in Table 6. Figure 6 plots the mean attractiveness ratings of the 432 facial images against the predicted attractiveness ratings using a regression equation (6). It is evident that the predicted attractiveness ratings are significantly linearly related with the visually assessed mean attractiveness ratings (R2 = 0.58, p < 0.001).
Discussion
The present study aims to explore how human brains perceive the attractiveness of faces that have varying facial proportions while excluding the influence of facial expression, the preference of hairstyle and the skin tone. The results revealed that distributed cortical regions, including the caudate nucleus, OFC, and the amygdala responded to facial attractiveness as induced by variant proportions. Furthermore, the correlation analysis between the brain response intensity that was measured by fMRI and the low-level features of the facial ratios showed that there were obvious couplings between fMRI-derived semantics of facial attractiveness and specific components of facial ratios. These findings provide direct neurophysiologic evidence for the suggestion that facial proportions significantly influence the perception of facial attractiveness. Furthermore, activities in the amygdala exhibited significant interaction effects between the viewers’ genders and attractiveness ratings, which suggests a noticeable discrepancy between the male and female observers on perceiving the attractiveness of faces with morphological variations.
Regions with responses to facial attractiveness
Our results show significant linear effects of facial attractiveness in the postcentral gyrus, caudate nucleus, and bilateral inferior frontal gyrus. Furthermore, the precuneus, angular gyrus, and amygdala exhibit significant non-linear effects of the facial ARs for the males. These observations suggest that there exist distributed regions that are involved in evaluating the attractiveness of face images with variant proportions, even when eliminating the influence of facial expression, hairstyle and skin tone.
We observed a significant linear effect of attractiveness ratings in the caudate nucleus. It has been previously reported that the caudate nucleus was sensitive to the emotional content of stimulation, including the viewing of a loved romantic partner39, and the experience of beauty40,41. Furthermore, Bartels and Zeki have reported that the caudate nucleus and putamen are linked to both positive and negative emotion. Notably, the reported location where the caudate nucleus was activated is similar to the location of activity observed in previous studies about beauty and romantic love42. This evidence supports my suggestion that the appropriate ratios of faces induce a facial attractiveness experience, to evoke the activation of the caudate nucleus, which could be parametrically modulated by the strength of the experience. More evidence on the response of the caudate nucleus to facial attractiveness has been reported recently31,43.
Another key region that demonstrates significant linear effects of attractiveness ratings is the posterior orbitofrontal cortex (inferior frontal cortex). It has been observed that the orbitofrontal cortex (OFC) is involved in reward and has complex response patterns in the perception of attractive faces44. It have been observed that the medial and lateral OFC respond to facial attractiveness and unattractiveness, respectively28, which suggests that rewards and punishments are represented separately in the OFC. However, this response pattern was not replicated in another experiment26, which instead exhibited significant non-linear responses to attractiveness in a portion of the medial OFC. Our results showed no response to attractiveness in the medial OFC. In the lateral OFC, however, we observed more responses to high attractive faces than to low attractive faces. This opposite response pattern compared with the previous report in which the lateral OFC showed greater activation to unattractive relative to attractive faces28 is difficult to interpret, although other types of reward such as pleasant gustatory stimuli have been found to activate the lateral OFC45. One possible explanation is the use of realistic faces with varying expressions as stimuli in O’Doherty et al.28 which could introduce more dimensions of perceiving the facial attractiveness, such as sexually dimorphic facial features and emotions. These controversial results also suggest the OFC is involved in rewards in a rather complex manner.
Subject gender matters in the perception of facial proportions
As expected, we observed a positive non-linear response profile of facial attractiveness ratings in the right amygdala, which is in line with the previous finding of increased neural activity in the amygdala to both extremely attractive and extremely unattractive faces26,31, which supports the role of the amygdala in social and emotional perception. More importantly, the activation in the amygdala showed a significant gender-by-attractiveness interaction (Fig. 4). In other words, the males exhibited greater responses to highly attractive and highly unattractive faces compared with middle-rank faces in the amygdala, whereas the effect in the females fails to pass the test for statistical significance.
To our knowledge, there is no evidence that shows that only opposite-sex faces lead to nonlinear responses in the amygdala. In fact, women show a non-linear response in the right amygdala when viewing male or female faces, as do men26. For both men and women, however, looking at opposite-sex faces produces stronger activity in the amygdala than looking at same-sex faces at the both ends of the attractiveness continuum, although the difference between the subjects and face genders has not achieved the statistical significance level26. Nevertheless, a clear difference in how men and women rate female facial attractiveness has been observed46. Moreover, attractive female faces activate reward regions in men more than attractive males or unattractive faces of either gender27. In this paper, the original face was obtained by averaging the faces of a cohort of young women. This approach makes most of the facial images appear to be more feminine. Hence, we speculate that the subject-gender differences in the activation of the amygdala are likely due to an opposite-sex bias in facial attractiveness evaluation. In other words, for heterosexual individuals, opposite-sex faces could hold greater significance or reward value than same-sex faces33, which is supported by consistent evidence for opposite-sex biased neural responses in the amygdala27,47, OFC48 and additional reward-related structures such as the nucleus accumbens49 and ventral tegmental area27.
Furthermore, the observed gender differences in the activation of the amygdala suggest differential preference to facial proportions between the subject genders. Given that the response in the amygdala could code the emotional value26 and track the stimulus intensity31, the finding that the male subjects showed a more significant nonlinear response in the right amygdala than the female subjects, implies that men are likely to assign more importance than women to facial proportions or shape in the facial attractiveness experience. This finding is also supported by the behavior and neuroimaging evidence that compared with women, men would pay more attention to facial physical attractiveness in mate preferences33,35.
Attractive faces have ideal facial measurements
In this paper, we found that facial attractiveness induced by facial proportions evoked a neural response within some of the reward regions. Furthermore, the significant negative correlation between the attractiveness ratings and the distance of the face images away from the ‘original face’ (average face) was observed (see supplemental Fig. S2), i.e., a face was observed to be more attractive when its ratios were close to the average. This finding suggests that the observed modulated effects within some specific regions in the reward circuit are likely attributed to composite effects of facial ratios on the averageness of the facial shape and some of the sexual dimorphic features, which lead to a distinct perception of facial attractiveness. Indeed, a quantitative analysis of female faces by manipulating individual facial features has demonstrated that facial attractiveness benefitted from averageness either at a overall level (e.g., the face shape), or for many individual facial features (e.g., eyes, nose, mouth)16. Moreover, some of the sexually dimorphic features are not of average size, and in the contrast, were larger or smaller, which would enhance attractiveness16,18. For example, feminine traits such as small jaw and full lips for female faces and masculine traits such as thick brow ridges and a large jaw structure for male faces are suggested to enhance the ratings of facial attractiveness by opposite-sex subjects24,36.
Given that these responses can be modulated by subject-specific attractiveness ratings, we sought to identify the most relevant components of the facial ratios to facial attractiveness by correlating the low-level facial ratio features and the intensity of the neural activity. Based on these components, we learned a regression model that can effectively predict the mean attractiveness ratings of each face image. This approach suggested that the neural activity in the activated regions contains meaningful high-level semantic information about facial attractiveness, which further confirms the suggestion that facial ratios or proportions contribute to facial attractiveness. Similar methods have been applied to predicting the semantics of nouns that are represented by fMRI neural activation50 and bridging the low-level features of video stimuli and fMRI-derived high-level semantics for video classification51.
Our results also suggested that attractive faces have optimal facial ratios or proportions. The previous literature has reported that beautiful faces have facial measurements that are close to the golden ratio36. These studies, however, used realistic face images as experimental materials that are difficult to control the effects of variations in hairstyles, skin texture, skin color and facial expressions. The current study investigates only the effect of the facial ratios, by excluding the impact of these confounding factors. Our results indicate that some specific facial ratios play important roles in determining the facial attractiveness. This finding provides us additional neurophysiological evidence for the suggestion that attractive faces follow optimum facial proportions.
Limitations
Here, we used computer-generated artificial face images as stimuli instead of images of realistic human faces. However, it should be noted that the mechanism of viewing a synthetic face is somewhat different from that of viewing a real face. In some face-sensitive structures such as the fusiform gyrus, the response to human facial expressions of emotions is significantly stronger than that of viewing computer-generated faces52. Hence, a further validation on real faces might be necessary, although in such a study, it is a large challenge, if not impossible, to control the facial expression, hairstyle, and skin tone and texture in real face images. Another feasible avenue for addressing this issue is to utilize the data-driven approach to explore the high-dimensional nature of face beauty based on realistic face images. The data-driven approach does not manipulate the features and need not constrain some of the variants that are of no interest. However, due to the high-dimensional traits of the facial attractiveness space (e.g., sexual dimorphism, symmetry, averageness, expression, hairstyle, shape, skin tone), a large dataset and powerful data-driven algorithm are required to tackle a truly ‘holistic’ outcome of face attractiveness perception, in which different characteristics or dimensions interact in more complicated ways.
Conclusions
To conclude, the present study indicated that some specific brain regions, including the caudate nucleus, OFC, and amygdala, were involved in the perception of attractiveness of faces that have varying facial proportions. Furthermore, we found notable between-gender differences in the neural activity in the right amygdala, which suggests that there is a discrepancy between the females and males in the brain responses to faces that have different proportions. To our knowledge, these observations for the first time provided neurophysiologic evidence for the hypothesis that human faces with variable proportions have differential attractiveness, even excluding the influence of hairstyle and facial expression as well as skin tone and texture.
Additional Information
How to cite this article: Shen, H. et al. Brain responses to facial attractiveness induced by facial proportions: evidence from an fMRI study. Sci. Rep. 6, 35905; doi: 10.1038/srep35905 (2016).
References
Pashos, A. & Niemitz, C. Results of an explorative empirical study on human mating in Germany: Handsome men, not high-status men, succeed in courtship. Anthropologischer Anzeiger 331–341 (2003).
Frieze, I. H., Olson, J. E. & Russell, J. Attractiveness and Income for Men and Women in Management1. Journal of Applied Social Psychology 21, 1039–1057 (1991).
Henderson, J. J. & Anglin, J. M. Facial attractiveness predicts longevity. Evolution and Human Behavior 24, 351–356 (2003).
Bronstad, P. M. & Russell, R. Beauty is in the ‘we’of the beholder: Greater agreement on facial attractiveness among close relations. Perception 36, 1674–1681 (2007).
Roggman, L. A. & Casey, R. J. Infant preferences for attractive faces: Rudiments of a stereotype? Developmental Psychology 23, 363–369 (1987).
Little, A. C. & Hancock, P. J. The role of masculinity and distinctiveness in judgments of human male facial attractiveness. British Journal of Psychology 93, 451–464 (2002).
Rhodes, G. et al. Attractiveness of facial averageness and symmetry in non-Western cultures: In search of biologically based standards of beauty. Perception 30, 611–625 (2001).
Thornhill, R. & Gangestad, S. W. Facial attractiveness. Trends in cognitive sciences 3, 452–460 (1999).
Komori, M., Kawamura, S. & Ishihara, S. Averageness or symmetry: which is more important for facial attractiveness? Acta psychologica 131, 136 (2009).
Jones, B. et al. The relationship between shape symmetry and perceived skin condition in male facial attractiveness. Evolution and Human Behavior 25, 24–30 (2004).
Perrett, D. et al. Effects of sexual dimorphism on facial attractiveness. Nature 394, 884–887 (1998).
Penton-Voak, I. S. et al. Male facial attractiveness, perceived personality, and child-directed speech. Evolution and human behavior 28, 253–259 (2007).
Satterthwaite, T. D. et al. Frontolimbic responses to emotional face memory: The neural correlates of first impressions. Human brain mapping 30, 3748–3758 (2009).
Matts, P. J. In Journal of Investigative Dermatology Symposium Proceedings. 6–9 (Nature Publishing Group, 2008).
Symons, D. The evolution of human sexuality. Behavioral and Brain Sciences 3, 203–214 (1980).
Baudouin, J. Y. & Tiberghien, G. Symmetry, averageness, and feature size in the facial attractiveness of women. Acta Psychologica 117, 313–332 (2004).
Cunningham et al. “Their Ideas of Beauty Are, on the Whole, the Same as Ours”: Consistency and Variability in the Cross-Cultural Perception of Female Physical Attractiveness. Journal of Personality & Social Psychology 68, 261–279 (1995).
Perrett, D. I., May, K. A. & Yoshikawa, S. Facial shape and judgements of female attractiveness. Nature 368, 239–242 (1994).
Valenzano, D. R., Mennucci, A., Tartarelli, G. & Cellerino, A. Shape analysis of female facial attractiveness. Vision Research 46, 1282–1291 (2006).
Rhodes, G. The evolutionary psychology of facial beauty. Annu. Rev. Psychol. 57, 199–226 (2006).
Haselhuhn, M. P. & Wong, E. M. Bad to the bone: facial structure predicts unethical behaviour. Proceedings of the Royal Society B Biological Sciences 279, 571–576 (2012).
Valentine, K. A., Li, N. P., Penke, L. & Perrett, D. I. Judging a man by the width of his face: the role of facial ratios and dominance in mate choice at speed-dating events. Psychological Science 25, 806–811 (2014).
Carré, J. M. & Mccormick, C. M. In your face: facial metrics predict aggressive behaviour in the laboratory and in varsity and professional hockey players. Proceedings Biological Sciences 275, 2651–2656 (2008).
Pallett, P. M., Link, S. & Lee, K. New “golden” ratios for facial beauty. Vision research 50, 149–154 (2010).
Fan, J., Chau, K., Wan, X., Zhai, L. & Lau, E. Prediction of facial attractiveness from facial proportions. Pattern Recognition 45, 2326–2334 (2012).
Winston, J. S., O’Doherty, J., Kilner, J. M., Perrett, D. I. & Dolan, R. J. Brain systems for assessing facial attractiveness. Neuropsychologia 45, 195–206 (2007).
Aharon, I. et al. Beautiful faces have variable reward value: fMRI and behavioral evidence. Neuron 32, 537–551 (2001).
O’Doherty, J. et al. Beauty in a smile: the role of medial orbitofrontal cortex in facial attractiveness. Neuropsychologia 41, 147–155 (2003).
Ishai, A. Sex, beauty and the orbitofrontal cortex. International Journal of Psychophysiology 63, 181–185 (2007).
Bzdok, D. et al. ALE meta-analysis on facial judgments of trustworthiness and attractiveness. Brain Structure and Function 215, 209–223 (2011).
Mende-Siedlecki, P., Said, C. P. & Todorov, A. The social evaluation of faces: a meta-analysis of functional neuroimaging studies. Social Cognitive and Affective Neuroscience 8, 285–299 (2013).
Adolphs, R., Nummenmaa, L., Todorov, A. & Haxby, J. V. Data-driven approaches in the investigation of social perception. Phil. Trans. R. Soc. B 371, 20150367 (2016).
Hahn, A. C. & Perrett, D. I. Neural and behavioral responses to attractiveness in adult and infant faces. Neuroscience & Biobehavioral Reviews 46, 591–603 (2014).
Iaria, G., Fox, C. J., Waite, C. T., Aharon, I. & Barton, J. J. The contribution of the fusiform gyrus and superior temporal sulcus in processing facial attractiveness: Neuropsychological and neuroimaging evidence. Neuroscience 155, 409 (2008).
Cloutier, J., Heatherton, T. F., Whalen, P. J. & Kelley, W. M. Are attractive people rewarding? Sex differences in the neural substrates of facial attractiveness. Journal of Cognitive Neuroscience 20, 941–951 (2008).
Schmid, K., Marx, D. & Samal, A. Computation of a face attractiveness index based on neoclassical canons, symmetry, and golden ratios. Pattern Recognition 41, 2710–2717 (2008).
Büchel, C., Holmes, A., Rees, G. & Friston, K. Characterizing stimulus–response functions using nonlinear regressors in parametric fMRI experiments. Neuroimage 8, 140–148 (1998).
Hotelling, H. Relations between two sets of variates. Biometrika, 321–377 (1936).
Zeki, S. & Romaya, J. P. The brain reaction to viewing faces of opposite-and same-sex romantic partners. PloS One 5, e15802 (2010).
Vartanian, O. & Goel, V. Neuroanatomical correlates of aesthetic preference for paintings. Neuroreport 15, 893–897 (2004).
Carretié, L. et al. The striatum beyond reward: caudate responds intensely to unpleasant pictures. Neuroscience 164, 1615–1622 (2009).
Bartels, A. & Zeki, S. The neural basis of romantic love. Neuroreport 11, 3829–3834 (2000).
Liang, X., Zebrowitz, L. A. & Zhang, Y. Neural activation in the “reward circuit” shows a nonlinear response to facial attractiveness. Social neuroscience 5, 320–334 (2010).
Kim, H., Adolphs, R., O’Doherty, J. P. & Shimojo, S. Temporal isolation of neural processes underlying face preference decisions. Proceedings of the National Academy of Sciences 104, 18253–18258 (2007).
O’Doherty, J. P., Deichmann, R., Critchley, H. D. & Dolan, R. J. Neural responses during anticipation of a primary taste reward. Neuron 33, 815–826 (2002).
Nestor, M. S., Stillman, M. A. & Frisina, A. C. Subjective and Objective Facial Attractiveness: Ratings and Gender Differences in Objective Appraisals of Female Faces. The Journal of clinical and aesthetic dermatology 3, 31 (2010).
Fischer, H. et al. Sex-differential brain activation during exposure to female and male faces. Neuroreport 15, 235–238 (2004).
Spreckelmeyer, K. N., Rademacher, L., Paulus, F. M. & Gründer, G. Neural activation during anticipation of opposite-sex and same-sex faces in heterosexual men and women. Neuroimage 66c, 223–231 (2013).
Bray, S. & O’Doherty, J. Neural coding of reward-prediction error signals during classical conditioning with attractive faces. Journal of Neurophysiology 97, 3036–3045 (2007).
Mitchell, T. M. et al. Predicting human brain activity associated with the meanings of nouns. science 320, 1191–1195 (2008).
Hu, X. et al. Bridging the semantic gap via functional brain imaging. Multimedia, IEEE Transactions on 14, 314–325 (2012).
Moser, E. et al. Amygdala activation at 3T in response to human and avatar facial expressions of emotions. Journal of neuroscience methods 161, 126–133 (2007).
Acknowledgements
This work was supported by Hong Kong Polytechnic University through an Interfaculty project (Project No: G-YG13) and a Niche Area Project (Project No: J-BB6T) as well as the start-up fund of Cornell University’s College of Human Ecology, which were awarded to Prof. Jintu Fan. This work was also supported in part by the National Natural Science Foundation of China (61375111, 61375034), and the National Basic Research Program of China (2013CB329401).
Author information
Authors and Affiliations
Contributions
H.S., J.H., J.T.F. and D.W.H. designed the project and D.K.P.C. provided the stimulus images. J.S. and W.J. performed the experiment and collected the data. H.S., J.S. and L.-L.Z. analyzed the data. H.S. and J.S. prepared the figures and wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Shen, H., Chau, D., Su, J. et al. Brain responses to facial attractiveness induced by facial proportions: evidence from an fMRI study. Sci Rep 6, 35905 (2016). https://doi.org/10.1038/srep35905
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep35905
This article is cited by
-
Neural Correlates and Perceived Attractiveness of Male and Female Shoulder-to-Hip Ratio in Men and Women: An EEG Study
Archives of Sexual Behavior (2023)
-
The impact of repeated exposure on facial attractiveness: The role of cognition and emotion
Current Psychology (2023)
-
Assessing Facial Symmetry and Attractiveness using Augmented Reality
Pattern Analysis and Applications (2022)
-
Human brain activity reflecting facial attractiveness from skin reflection
Scientific Reports (2021)
-
The Ideal Lips: Lessons Learnt from the Literature
Aesthetic Plastic Surgery (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.