Abstract
This study is the first to evaluate the effects of five seaweeds (Ulva sp., Laminaria ochroleuca, Saccharina latissima, Gigartina sp., and Gracilaria vermiculophylla) on gas and methane production and ruminal fermentation parameters when incubated in vitro with two substrates (meadow hay and corn silage) for 24 h. Seaweeds led to lower gas production, with Gigartina sp. presenting the lowest value. When incubated with meadow hay, Ulva sp., Gigartina sp. and G. vermiculophylla decreased methane production, but with corn silage, methane production was only decreased by G. vermiculophylla. With meadow hay, L. ochroleuca and S. latissima promoted similar methane production as the control, but with corn silage, L. ochroleuca increased it. With the exception of S. latissima, all seaweeds promoted similar levels of total volatile fatty acid production. The highest proportion of acetic acid was produced with Ulva sp., G. vermiculophylla, and S. latissima; the highest proportion of butyric acid with the control and L. ochroleuca; and the highest proportion of iso-valeric acid with Gigartina sp. These results reveal the potential of seaweeds to mitigate ruminal methane production and the importance of the basal diet. To efficiently use seaweeds as feed ingredients with nutritional and environmental benefits, more research is required to determine the mechanisms underlying seaweed and substrate interactions.
Similar content being viewed by others
Introduction
Dietary nutrients are fermented in the rumen by a complex microbial population, producing volatile fatty acids (VFA), hydrogen, and carbon dioxide as the main fermentation products. Methane production results from the reduction of carbon dioxide with hydrogen by archaea, a group of methanogens frequently associated with ciliated protozoa1. Enteric methane production prevents increases in hydrogen pressure, which could inhibit the normal functioning of microbial enzymes and impair rumen fermentation2. Methane is a potent greenhouse gas and may represent a loss of 2–15% of the gross energy (GE) in the feed, depending on the diet3. Therefore, enteric methane mitigation may have a positive impact on feed utilization, diet digestibility, and, ultimately, livestock productivity4.
Seaweeds might be a natural alternative for the mitigation of greenhouse gas emissions by ruminants. Seaweeds have been used to feed livestock from time immemorial in coastal regions during periods of feed scarcity5. Renewed interest has emerged during recent decades in the use of seaweeds as feed ingredients due to their richness in organic minerals, complex carbohydrates, proteins and low-molecular-weight nitrogenous compounds, lipids, vitamins, volatile compounds, pigments6 and bioactive substances with broad biological activities7. Based on availability and market cost, seaweeds have been evaluated as a prebiotic promoter8 or a feed ingredient9 at low or high inclusion rates, respectively. In this context, due to the chemical diversity and complexity of polysaccharides, which may account for 25–75% of algae dry weight10, ruminants seem to be the most suitable animals to be fed on seaweeds. The intricate rumen ecosystem might provide the ruminant the ability to use seaweeds by breaking down the complex polysaccharides. Additionally, some seaweeds and seaweed extracts effectively reduce ruminal methane production in vitro (e.g., Asparagopsis taxiformis)11,12,13,14,15, although a broad range of results have been reported and with variable effects on rumen fermentation.
Notwithstanding the potential effect of the basal diet on the effectiveness of the compound used to mitigate methane emissions16, earlier research did not evaluate seaweed effects across more than one feed type. Therefore, the objective of this study was to determine the effects of seaweeds naturally occurring at the Atlantic and Mediterranean coasts (Laminaria ochroleuca and Gigartina sp.) or produced in an integrated multi-trophic aquaculture (IMTA) system (Ulva sp., Saccharina latissima and Gracilaria vermiculophylla) on the in vitro ruminal fermentation parameters, total gas production and methane production for two feed substrates (meadow hay and corn silage). As far as we know, this is the first report on the effects of these seaweeds on in vitro rumen fermentation across different substrates.
Results
Chemical composition
The chemical composition of the base substrates and the five seaweed species is presented in Table 1. The meadow hay and corn silage presented 723 and 493 g kg−1 dry matter (DM), and 565 and 377 g kg−1 neutral detergent fibre (NDF, DM basis), respectively. The chemical composition of the studied seaweeds showed a wide variation, particularly with respect to ash and NDF contents, which respectively ranged from 171 g kg−1 DM in S. latissima to 348 g kg−1 DM in Gigartina sp. and 71.1 g kg−1 DM in S. latissima to 335 g kg−1 DM in Ulva sp. Laminaria ochroleuca and Ulva sp. presented the highest acid detergent lignin (ADL) contents. Overall, seaweeds were poor sources of lipids, the highest content being found in S. latissima (7.87 g kg−1 DM), with GE ranging from 9.51 MJ kg−1 DM in Gigartina sp. to 12.8 MJ kg−1 DM in S. latissima. The three seaweeds cultivated in an IMTA system (Ulva sp., S. latissima, and G. vermiculophylla) presented the highest crude protein (CP) content.
Total gas and methane production
Total gas and methane production were strongly affected by the basal substrate (meadow hay or corn silage) and inoculum (adapted to 0% or 5% sunflower oil) used in in vitro incubations and by seaweed inclusion (Tables 2 and 3).
While the use of corn silage as a basal substrate increased total gas production (P < 0.001) after 24 h incubation, gas production was decreased by oil-adapted inoculum (P < 0.001; Table 2). Overall, seaweeds led to 17% less gas production than the control (101 mL g−1 DM vs. 83.1 mL g−1 DM, respectively; Table 3). Among seaweeds, the red algae Gigartina sp. had the lowest gas production of all species, producing a total of 67.5 mL g−1 DM after a 24 h incubation, while the others promoted similar gas production (respectively, 82.5, 86.2, 89.5, and 89.9 mL g−1 DM for G. vermiculophylla, Ulva sp., L. ochroleuca and S. latissima) that was still lower than the control (101 mL g−1 DM).
Similar to total gas production, methane production was increased by corn silage (P < 0.001) and by oil-unadapted inoculum (P < 0.001; Table 2). Seaweeds strongly affected methane production (P < 0.001), a reduction in methanogenesis being observed with Ulva sp., Gigartina sp., and G. vermiculophylla in comparison with the control, L. ochroleuca and S. latissima (Table 3). No significant relationships were observed between gas or methane production and the chemical composition of the studied seaweeds, except that ash tended to be negatively correlated with gas production (r = −0.847; P = 0.070).
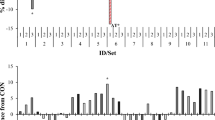
A significant interaction between substrate and seaweed was observed for methane production (Fig. 1a). When incubated with meadow hay, Ulva sp., Gigartina sp. and G. vermiculophylla decreased methane production to 55, 44 and 59% of the control, respectively. However, when these same seaweeds were incubated with corn silage, only G. vermiculophylla decreased methane production, to 63% of the control. Laminaria ochroleuca, and S. latissima promoted similar methane production as the control when incubated with meadow hay, but when incubated with corn silage, L. ochroleuca increased methane production to 148% of the control.
Effects of seaweed*substrate interaction on methane production [mL g−1 DM (a)], N-NH3 [mg g−1 DM (b)], total volatile fatty acid (VFA) production [mmol g−1 DM (c)], the iso-valeric proportion [% (d)], and the acetic:propionic acid ratio (e) after 24 h of in vitro incubation. Meadow hay (□), corn silage (■). Mean values with different superscript letters were significantly different (P < 0.05).
Fermentation pH and ammonia-N production
Fermentation pH was strongly affected by basal substrate (P < 0.001; Table 2) and tended to be affected by inoculum (P = 0.056; Table 2), while seaweed had no effect (P = 0.306; Table 3). Incubation of meadow hay led to a higher fermentation pH compared to corn silage, and oil-adapted inoculum tended to decrease pH.
Ammonia-N production (mg g−1 DM) was unaffected by substrate (P = 0.406) but was higher (P < 0.001) in oil-unadapted inoculum than inoculum adapted to 5% oil supplementation (Table 2). Overall, ammonia-N (NH3-N) production was increased by nearly 30% with seaweed inclusion compared to the control (4.42 vs. 3.43 mg g−1 DM, respectively; P < 0.001; Table 3). Among seaweeds, the brown L. ochroleuca and S. latissima had similar NH3-N production as the control (3.52 and 3.86 mg g−1 DM, respectively), and the red algae Gigartina sp. had the highest NH3-N production (6.07 mg g−1 DM; Table 3).
A significant interaction between substrate and seaweed was found for NH3-N production (Fig. 1b). When incubated with meadow hay, the red seaweeds increased NH3-N production by 1.5- to 2-fold. When corn silage was the substrate, Ulva sp. and G. vermiculophylla decreased NH3-N production by 1.4- and 1.6-fold.
Volatile fatty acid production and profile
Volatile fatty acid production and proportion were affected (P < 0.001) by basal substrate and inoculum, except valeric acid that was only affected by inoculum (P < 0.001; Table 2). Corn silage decreased the acetic acid proportion and the acetic:propionic ratio and increased all individual and total VFA concentrations (mmol g−1 DM; P < 0.001; Table 3). Oil-adapted inoculum increased the proportion of propionic, iso-butyric, and iso-valeric acids and total VFA but decreased the proportions of acetic, butyric, valeric and caproic acids and the acetic:propionic ratio (P < 0.05; Table 2). Total VFA production (mmol g−1 DM) was higher with S. latissima than with any other seaweed (P < 0.05; Table 3), although it did not differ from the control. Total VFA production of all the other seaweeds was also similar to the control. The interaction between substrate and seaweed showed that L. ochroleuca, Gigartina sp. and G. vermiculophylla tended (P = 0.092) to decrease total VFA production when incubated with corn silage (Fig. 1c).
The proportion of acetic acid was highest with Ulva sp., G. vermiculophylla, and S. latissima (63.4, 63.4, and 64.3%, respectively), and the lowest proportion was in the control (61.3%), which was not different from the remaining seaweeds (L. ochroleuca and Gigartina sp.). Conversely, the butyric acid proportion was highest in the control and L. ochroleuca and lowest with Ulva sp., S. latissima, Gigartina sp., and G. vermiculophylla. A tendency for a decrease in the propionic acid proportion with seaweeds was observed (P = 0.086), whereas the proportion of iso-valeric was highest and was significantly different from the control with Gigartina sp. When incubated with meadow hay, L. ochroleuca tended to decrease the iso-valeric proportion, while Gigartina sp. tended to increase the proportion of this VFA when incubated with corn silage (P = 0.068 for substrate and seaweed interaction; Fig. 1d).
The basal substrate did not alter the acetic:propionic ratio in the control, which was similar to that observed with the inclusion of seaweeds with corn silage, whereas meadow hay increased the ratio (Fig. 1e). The acetic:propionic ratio increased by 9% with seaweed inclusion compared to the control (P < 0.001; Table 4); no differences were observed among seaweed species. Additionally, the acetic:propionic ratio was affected by the interaction between substrate and seaweed (Fig. 1e) and between seaweed and inoculum (Fig. 2). Unadapted inoculum led to a similar and higher acetic:propionic ratio in the control and with seaweed inclusion, regardless of the species, compared to oil-adapted inoculum, the lowest ratio being detected in the control incubated with adapted inoculum (Fig. 2).
Hydrogen balance
Hydrogen generated and consumed, percentage recovery and fermentation efficiency were significantly affected (P < 0.001; Table 2) by substrate and inoculum, but not by seaweed inclusion (P > 0.05; Table 3). Corn silage generated and consumed more hydrogen than meadow hay, the percentage of recovery being 36.6% and the fermentation efficiency 75.4%. Similarly, oil-adapted inoculum generated and consumed more hydrogen, but the recovery and the fermentation efficiencies were lower than those observed with unadapted inoculum.
Discussion
The effects of five seaweed species (green, brown and red) either highly available on the Atlantic and Mediterranean coasts or produced in an IMTA system were evaluated in short-term in vitro rumen fermentation batches incubated at a high inclusion level (25% DM basis) with two different substrates (75% DM basis). In the IMTA system, the by-products (wastes) from one species (fish) are recycled to become inputs (fertilizers, food) for another (e.g., algae), resulting in the additional production of a marketable product with little or no additional input costs, a decrease in waste outputs from overall farming activities, and more environmentally sustainable farming. In this context, we evaluated the effects of seaweed inclusion and its interactions with basal diet on gas and methane production, VFA, hydrogen balance and NH3-N.
Feeding strategies to decrease gas emissions, particularly methane emissions, from livestock have focused on the manipulation of ruminal microbial populations and metabolism through the nutritional and biochemical properties of feeds. However, the decrease in methane production must be achieved with no or minimal adverse effects on overall rumen fermentation. In the present study, despite corn silage having decreased the fermentation pH below 6.0, which inhibits methanogen growth17, it increased gas and methane production, reflecting a greater extent of fermentation of this substrate relative to meadow hay. Rumen inoculum from cows adapted to a diet supplemented with 5% sunflower oil led to a decrease in gas and methane production. Dietary lipid supplementation constitutes a nutritional strategy to mitigate methane emissions, with differences in methanogenesis depending on the type of fat and its availability in the rumen18. Fatty acids may inhibit methane production by direct toxic effects on ruminal microorganisms and protozoa19 and indirectly on protozoa-associated methanogens20. Therefore, the decrease in methanogenesis might be attributed to a reduced abundance of archaea due to protozoan inhibition. Seaweeds have been evaluated for their effects on ruminal methane production. Although only a limited variety of seaweeds were assessed, some have shown a great potential to decrease methanogenesis (e.g., Asparagopsis taxiformis)13, while others have high nutritional value but lower antimethanogenic potential (Chondrus cripus, Laminaria longicruris, and Fucus vesiculosus15; Spirogyra and Derbesia12; and Caulerpa taxifolia and Tarong polyculture11). In our study, all seaweeds reduced total gas production (mL g−1 DM) after 24 h incubation, with Gigartina sp. promoting the greatest decrease, although chemical parameters were unable to explain this effect. Methane production (mL g−1 DM) was affected differently by different seaweeds. Green and red algae reduced methanogenesis, with G. vermiculophylla and Gigartina sp. having the most noticeable effects (38.2 and 35.8% reduction, respectively); brown seaweeds had no effect compared to the control. Red and brown algae exert more marked effects on methane production than green algae11,12,13. Indeed, the red Asparagopsis has potent antimethanogenic properties (more than 99% decrease in methane production) in vitro at 1% or 2% inclusion levels (organic matter, OM, basis)13,14. This effect on methanogenesis reduction has been suggested to be associated with seaweed secondary metabolites12,13,14,15. Indeed, seaweeds have developed a complexity and diversity of secondary compounds as a defence mechanism for survival in a highly competitive environment21. Red seaweeds are particularly rich with more than 1500 secondary metabolites of all classes, particularly halogenated compounds with bromine or chlorine22 that inhibit the methyl transfer reactions essential for methanogenesis23. Brown algae also possess a wealth of secondary metabolites (more than 1100 reported), in particular phlorotannins (polyphenols exclusive to brown algae), which exert an anti-microbial action, particularly on the widespread rumen cellulolytic bacterium Fibrobacter succinogenes24. Conversely, green seaweeds have the least variety of secondary metabolites, with fewer than 300 compounds found25. In addition to seaweed individual effects, an interaction between seaweeds and basal substrate was observed for methane production, with Ulva sp. and Gigartina sp. only decreasing methane production when incubated with corn silage and G. vermiculophylla decreasing methane production independently of the substrate used. Indeed, some studies suggest that regardless of the compound used to decrease methane emissions, the basal diet fed to the animal plays an important role in the effectiveness of the compound. For instance, the supplementation of Oedogonium (0.2 g OM basis) to different basal substrates (1 g OM basis) has been found to decrease methane at different rates, by nearly 40%11, 30%12 or 15%13, when Rhodes grass (107 g kg−1 CP, 672 g kg−1 NDF, DM basis), Finders grass (27.5 g kg−1 CP, 746 g kg−1 non-structural carbohydrates, DM basis) or Rhodes grass hay (66.9 g kg−1 CP, 766 g kg−1 carbohydrates, DM basis), respectively, was used as basal substrate. Machado et al.13 suggested that differences in the substrate used across different studies may have contributed to the variable in vitro antimethanogenic effect of Oedogonium sp., as high-protein substrates lead to lower gas and methane productions than low-quality fibrous substrates. Additionally, Machmuller et al.26 found that the decrease in methane production observed with myristic acid doubled when sheep consumed a concentrate (mean CP 167 g kg−1 DM) compared to when it consumed a forage-based diet (mean CP 139 g kg−1 DM). Conversely, O’Brien et al.16 found that the effectiveness of lauric, oleic, linoleic and linolenic acids and bromoethanesulfonate in reducing methane production was more pronounced when incubated with grass silage and barley grain (116 g CP kg−1 DM) than with perennial ryegrass (161 g CP kg−1 DM). In the present study, corn silage presented a CP content 39% higher than meadow hay, which could have partly contributed to the results observed. However, comparing our results to those in the literature is difficult because, to our knowledge, this is the first time that these five seaweeds have been studied and screened with contrasting substrates.
Volatile fatty acids are produced through the fermentation of dietary OM by the complex microbial ecosystem in the rumen. Volatile fatty acids are energy sources for maintenance and growth, propionic acid being a primary glycogenic precursor, butyric acid a lipogenic precursor of longer-chain fatty acids, and acetic acid a primary precursor of short- and medium-chain fatty acids27. The amount, type and rate of fermentation of dietary carbohydrates affect both the total amounts and proportions of individual VFAs formed and, ultimately, the amount of methane produced. In our study, corn silage increased total VFA production and decreased pH, reflecting again a higher extent of fermentation of this substrate relative to meadow hay. Additionally, corn silage (with 300 g kg−1 DM of starch) promoted higher propionic acid and lower acetic acid proportions than meadow hay. Diets rich in starch can decrease rumen pH, thus reducing fibre digestibility28 and promoting propionic acid production, whilst roughage-based diets promote acetic acid production29. Rumen inoculum from cows adapted to a diet supplemented with 5% sunflower oil led to a decrease in the acetic acid proportion and an increase in total VFA production and the propionic acid proportion. Acetic acid and butyric acid promote methane production, whilst propionic acid production can be considered a competitive pathway for hydrogen use in the rumen30. Methane production also decreased with oil-adapted rumen inoculum, suggesting that to compensate for the disruption of electron flow to methanogenesis, the rumen microbial population disposed of excess reducing equivalents by increasing the production of more reduced VFA, thus decreasing acetic acid production3,31. Fatty acids have a strong inhibitory effect on protozoa and cellulolytic bacteria19, while propionic acid-producing Gram-negative bacteria are not significantly inhibited32. A reduction in methane production thus shifts fermentation towards propionic acid production33. A slight decrease in the valeric acid proportion accompanying the decrease in methane production suggests a small shift in the fermentation pattern already seen in previous research with the addition of short-chain fatty acids34. Overall, seaweeds decreased methanogenesis, but for practical application, this reduction should have no or minimal negative effects on fermentation parameters, including the production of VFA.
In this study, the reduced fermentation suggested by the decreased total gas and methane production was not supported by total VFA production, which was unaffected by seaweed inclusion. With the non-significant differences between seaweed inclusion and the control on total VFA production, the reason for reduced gas and methane production remains unclear. The presence of bioactive compounds and the ability of the different classes of rumen microbes to efficiently use polysaccharides from the cell walls of the different seaweeds might contribute to explaining these results. Unlike terrestrial plants, seaweeds have complex polysaccharides in the cell wall structure, which greatly differ among seaweed classes and species35. Green algae are rich in soluble ulvans of the family of sulphated polysaccharides36. Conversely, brown algae are rich in laminarin, mannitol, alginic acid, fucoidans and, in only minor amounts, cellulose37, while carrageenans38, agars, and porphyran39 are the major matrix polysaccharides of red algae. Although ulvans are potentially hydrolysable to bioactive oligosaccharides40, ulvan lyases have only been isolated in marine environments41 and in Proteobacteria species found in soil42. Similarly, the hydrolysis of red algal galactans requires a range of enzymes predominantly encoded in the genomes of marine microbes but that are less frequent or even absent in the bacteria that hydrolyse polysaccharides from land plants43. Brown seaweed carbohydrates are hydrolysed by the rumen microbial population, producing methane and acetic acid44 (though to different extents). Indeed, Orpin et al.45 found that 13% of the culturable bacteria from seaweed-fed sheep grew on alginate, 71% on laminarin, 13% on fucoidan, and 99% on mannitol, whilst the percentages obtained from pasture-fed animals were significantly lower (2%, 32%, 0% and 0%, respectively). Differences in the ability to hydrolyse mannitol between seaweed-fed or grass-fed animals were also observed in other studies46.
The effects of seaweeds on total VFA production depended on the substrate used. The combination of L. ochroleuca, Gigartina sp., and G. vermiculophylla with corn silage had a negative effect on total VFA production, indicating that the type and quality of substrates influence the extent of the adverse effects on in vitro fermentation. Horn and Østgaard47 studied the anaerobic digestion of alginate from Laminaria hyperborea and reported that the production of extracellular polymer-degrading enzymes such as alginate lyase could be suppressed in the presence of easily usable alternative substrates. This could partly explain the decrease in total VFA production when L. ochroleuca was incubated with corn silage, as extra glucose can lead to a diauxic development with glucose as the preferred substrate and delayed initiation of alginate lyase activity47.
Seaweeds differently affected individual VFA proportions; only seaweeds from an IMTA system significantly increased the acetic acid proportion when compared to the control. No differences among seaweeds were observed regarding the propionic acid proportion, suggesting that reducing equivalents were not redirected to the production of this reduced fermentation product, not even with Gigartina sp., which promoted the lowest methane production. A significant interaction was observed for the acetic acid:propionic acid ratio, with seaweeds having a cumulative effect when incubated with meadow hay or with the oil-adapted inoculum. With the exception of L. ochroleuca, all other seaweeds decreased the butyric acid proportion, suggesting that the reducing equivalents spared from methanogenesis were not consumed during the formation of this fermentation product. With red seaweeds, some reducing equivalents may have been consumed via increased proportions of iso-valeric acid. The consumption of reducing equivalents might also have occurred during anabolic processes (e.g., cell growth, extracellular polysaccharide production)48 or through the reduction of carbon dioxide to acetic acid via acetogenesis49.
Methanogens are the main users of hydrogen within the rumen. The inhibition of methanogenesis can lead to the accumulation of excess reducing equivalents that can increase intracellular NADH/NAD, thus reducing overall fermentation efficiency by limiting the availability of oxidized cofactors required for glycolysis31 or leading to an increase in propionic acid or NH3-N production2. In this study, corn silage and oil-unadapted inoculum led to a high percentage of hydrogen recovery and fermentation efficiency. Although seaweed inclusion did not significantly affect hydrogen balance or fermentation efficiency, it increased NH3-N production, suggesting that reducing equivalents spared from methane production did not accumulate as reduced NADH. However, the ammonia accumulation must be interpreted with caution, as free ammonia may be both produced and assimilated by the rumen microbial population and can also reflect the higher CP content in seaweeds than in substrates.
In conclusion, this study demonstrated that Ulva sp., L. ochroleuca, S. latissima, Gigartina sp., and G. vermiculophylla strongly reduced methanogenesis when incubated at 25%, with Gigartina sp. promoting the lowest methane production. The studied seaweeds did not have any detrimental effects on in vitro rumen fermentation when compared to the control. However, the effects on methane and total VFA production depended on the substrate used. All seaweeds decreased methane production when incubated with meadow hay, but only G. vermiculophylla decreased it when incubated with corn silage. The combination of L. ochroleuca, Gigartina sp., and G. vermiculophylla with corn silage had a negative effect on total VFA production. The results suggest that seaweeds have the potential to be used as a feed ingredient in animal diets at relatively high levels as sources of macro-nutrients and bioactive compounds, with a beneficial effect on reduced methane emissions; however, the basal diet given to the animal must be considered. Moreover, longer-duration studies are required, particularly in vivo, to confirm the effectiveness of seaweeds as natural manipulators of ruminal methanogenesis and to screen them with different basal diets.
Methods
Seaweeds and basal substrates
Green macroalgae (Ulva sp.), brown macroalgae (L. ochroleuca, S. latissima) and red macroalgae (Gigartina sp., G. vermiculophylla) were studied. Seaweeds were harvested off the north coast of Portugal or produced in an IMTA system50 as described in Table 4. After collection, seaweed biomass was rinsed in freshwater to remove epiphytes, detritus, and sand and was transported to the laboratory and dried in a forced-air oven at 65 °C until a constant weight was achieved.
Meadow hay and corn silage were used as basal substrates in the in vitro incubations, after being dried for 48 h in a forced-air oven at 65 °C. Dried seaweeds, meadow hay and corn silage samples were ground to pass through a 1-mm screen and were stored at room temperature until incubation.
Ground (1 mm) samples of seaweeds and substrates used in the in vitro incubations were analysed for DM51, ash (ID 942.05)51, ether extract (EE; ID 920.39)51, Kjeldahl N (ID 954.01)51, NDF (with α-amylase and without sodium sulphite), acid detergent fibre (ADF) and ADL52,53. Crude protein was determined as Kjeldahl N × 6.25 for substrates and Kjeldahl N × 5.0 for seaweeds54. Neutral detergent fibre and ADL were expressed exclusive of residual ash. The starch content of corn silage was determined on finely ground samples with a 0.5-mm screen55. The GE of seaweeds and substrates were determined in an adiabatic bomb calorimeter (Werke C2000, IKA, Staufen, Germany). All chemical analyses were run in duplicate. Non-starch polysaccharides (NSP) were calculated by the difference between DM and the sum of ash, EE, CP, NDF and starch.
Rumen inoculum and diet
Rumen contents were obtained from two adult Holstein cows, dry and not pregnant, fitted with a rumen cannula (10 cm diameter; Bar Diamond Inc., Parma, ID). Cows were housed at the Vairão Agricultural Campus of Abel Salazar Biomedical Sciences Institute, University of Porto (Vila do Conde, Portugal) and were handled in strict accordance with good animal practice as defined by national authorities and the European Union Directive 2010/63/EU. The experimental animal procedures were approved by the Local Animal Ethics Committee of ICBAS-UP, licensed by the Portuguese Directorate-General of Food and Veterinary Medicine (Direção Geral de Alimentação e Veterinária) of the Ministry for Agriculture and Sea (Ministério da Agricultura e do Mar, permit #FT2014DGV 046412 ICB), and conducted by trained scientists following FELASA category C recommendations. All methods and procedures were performed following the established guidelines from these institutions.
A single total mixed ration (TMR) was used to feed the cows supplemented with 0% or with 5% sunflower oil (Fula Puro Girassol, Sovena, Algés, Portugal). The TMR comprised 14 kg corn silage (the same used as substrate in the in vitro incubations; Table 1), 3 kg wheat straw (37 g kg−1 CP, 811 g kg−1, NDF, DM basis), and 2 kg commercial concentrate for dry cows (230 g kg−1 CP, 294 g kg−1 NDF, 187 g kg−1 starch, DM basis). Cows were fed twice a day, at 0930 and 1730 h, the daily amount of feed being offered equally in both meals. Animals had continuous access to fresh drinking water. After a two-week adaptation period to the diet, rumen contents were collected from the four quadrants of the rumen of each cow and placed in a 4 L pre-warmed (39 °C) thermal jug. At the laboratory, each ruminal digesta was homogenized, strained through 4 layers of linen cloth, and maintained at 39 °C under O2-free CO2. The length of time between collection of rumen contents and incubation never exceeded 60 min. After rumen inocula collection, the diet was exchanged between cows and another two-week adaptation period began for a new collection of ruminal contents.
Rumen in vitro incubations
The effects of seaweed supplementation to meadow hay and corn silage on the ruminal fermentation parameters, total gas production and methane production were evaluated in short-term (24 h) batch incubations. To each basal diet, one of the five seaweeds was supplemented at 0% (control) and 25% of the total incubated DM. One part of strained ruminal fluid was diluted anaerobically into four parts of the medium described by Marten and Barnes56 and mixed at 39 °C under O2-free CO2. Twenty five millilitres of the buffered ruminal fluid was dispensed anaerobically into 125 mL serum bottles (Sigma-Aldrich Inc., St. Louis, MO) containing 250 mg DM of each experimental treatment, sealed with butyl rubber stoppers (Sigma-Aldrich Inc., St. Louis, MO), and incubated in a water bath at 39 °C. Fermentations were stopped after 24 h by cooling the bottles in an ice-slurry bath at 4 °C. Experimental treatments were incubated in duplicate per inoculum and per incubation, and batch incubations were replicated in two separate runs.
Incubation media sampling and analysis
Bottles were warmed to 25 °C, and head-space gas volume was measured with a pressure transducer (Bailey & Mackey Ltd., Birmingham, UK) as described by Theodorou et al.57. The composition of the head-space gas was determined in 0.5 mL samples collected with a gas-tight syringe (SGE international PTY Ltd, Australia) by gas chromatography, using a GC-4000A (East & West Analytical Instruments, Inc, Beijing, China) equipped with a Shincarbon ST 100/120 micropacked column (Restek Corporation, Bellefonte, PA) and a thermal conductivity detector. The temperature was held at 100 °C in the injector, 180 °C in the detector and in the bridge, and 60 °C in the oven. Helium was used as the carrier gas at a flow rate of 23 mL min−1. The analyses were performed in duplicate. An external standard with known composition (60% CO2, 25% N2, 10% CH4 and 5% H2; Air Liquide, Lda., Algés, Portugal) was used to identify and quantify gas peaks. Methane production was calculated according to Lopez and Newbold58, using CO2 as reference element of the gas mixture.
The pH of each bottle was measured immediately after gas sampling. Fermentation media contents were sub-sampled for the analysis of VFA and NH3-N. For VFA analyses, 0.25 mL of 25% ortho-phosphoric acid solution with internal standard (16 mM 3-methyl valeric acid; Sigma-Aldrich Inc., St. Louis, MO) was added to 1 mL of fermentation medium in a microcentrifuge tube, mixed and centrifuged at 19,800 × g at 4 °C for 15 min. The supernatant was filtered through a 25 mm polyethersulfone syringe filter (0.45 μm pore size; VWR International - Material de Laboratório, Lda., Carnaxide, Portugal) and stored at 4 °C until chromatographic analysis. Volatile fatty acids were analysed by gas chromatography using a Shimadzu GC-2010 Plus (Shimadzu Corporation, Kyoto, Japan) equipped with a capillary column (HP-FFAP, 30 m × 0.25 mm × 0.25 μm; Agilent Technologies, Santa Clara, CA), and a flame ionization detector. Injector and detector temperatures were held at 260 °C. The oven temperature started at 80 °C for 1 min, increased at 20 °C min−1 to 120 °C, then increased at 6 °C min−1 to 205 °C and finally increased at 20 °C min−1 to 240 °C. Helium was used as a carrier gas at a flow rate of 0.86 mL min−1. The injection volume was 1 μL and the split of 50:1. Volatile fatty acids were quantified with the internal standard (3-methyl valeric acid) and identified by comparison of retention times with a standard (Volatile Free Acid Mix, Sigma-Aldrich Inc., St. Louis, MO).
For NH3-N analysis, 5 mL of fermentation medium was added to 5 mL of 0.2 N HCl solution and steam-distilled (Vapodest 40, distiller unit, C. Gerhardt GmbH & Co. KG, Germany). N content was determined through titration (ID 954.01)51.
The reducing equivalents generated (expressed as μmol H2 mL−1 fermentation media) were estimated as 2 equiv. acetic acid + 1 equiv. propionic acid + 4 equiv. butyric acid + 2 equiv. valeric acid + 2 equiv. iso-valeric acid. The reducing equivalents (μmol H2 mL−1 fermentation media) consumed were estimated as 2 equiv. propionic acid + 2 equiv. butyric acid + 1 equiv. valeric acid + 4 equiv. methane59. Fermentation efficiency was calculated as (0.62 acetic acid + 1.09 propionic acid + 0.78 butyric acid)/(acetic acid + propionic acid + butyric acid) × 100 and is based on the heats of combustion of glucose in the respective VFA60.
Statistical analysis
All data were analysed using the MIXED procedure of the SAS software program (2002; version 9.1, SAS Institute Inc., Carry, NC). The statistical model included the fixed effect of seaweed, substrate, rumen inoculum, and all the interactions between main effects, the random effect of the trial, and the random residual error. Effects were considered significant when P ≤ 0.05 and a trend when 0.05 < P ≤ 0.10. When the interactions had a non-significant effect or tendency (P > 0.10), they were removed from the model. The linear relationships between the chemical composition of the seaweeds and gas and methane production were evaluated using the REG procedure of the SAS software program (2002; version 9.1, SAS Institute Inc., Carry, NC).
Additional Information
How to cite this article: Maia, M. R. G. et al. The Potential Role of Seaweeds in the Natural Manipulation of Rumen Fermentation and Methane Production. Sci. Rep. 6, 32321; doi: 10.1038/srep32321 (2016).
References
Williams, A. G. & Coleman, G. S. In The Rumen Microbial Ecosystem (eds Hobson, P. N. & Stewart, C. S. ), 73–139 (Springer Netherlands, 1997).
Morgavi, D. P., Forano, E., Martin, C. & Newbold, C. J. Microbial ecosystem and methanogenesis in ruminants. animal 4, 1024–1036 (2010).
Van Nevel, C. J. & Demeyer, D. I. Control of rumen methanogenesis. Environ. Monit. Assess. 42, 73–97 (1996).
Patra, A. K. Enteric methane mitigation technologies for ruminant livestock: a synthesis of current research and future directions. Environ. Monit. Assess. 184, 1929–1952 (2011).
Balasse, M., Tresset, A., Dobney, K. & Ambrose, S. H. The use of isotope ratios to test for seaweed eating in sheep. J. Zool. 266, 283–291 (2005).
Makkar, H. P. S. et al. Seaweeds for livestock diets: A review. Anim. Feed Sci. Technol. 212, 1–17 (2016).
Kumar, C. S., Ganesan, P., Suresh, P. V. & Bhaskar, N. Seaweeds as a source of nutritionally beneficial compounds - A review. J. Food Sci. Tech. Mys. 45, 1–13 (2008).
Ramnani, P. et al. In vitro fermentation and prebiotic potential of novel low molecular weight polysaccharides derived from agar and alginate seaweeds. Anaerobe 18, 1–6 (2012).
Machado, L., Kinley, R. D., Magnusson, M., Nys, R. & Tomkins, N. W. The potential of macroalgae for beef production systems in Northern Australia. J. Appl. Phycol. 27, 2001–2005 (2015).
Jiménez-Escrig, A. & Sánchez-Muniz, F. J. Dietary fibre from edible seaweeds: Chemical structure, physicochemical properties and effects on cholesterol metabolism. Nutr. Res. 20, 585–598 (2000).
Dubois, B. et al. Effect of Tropical Algae as Additives on Rumen in vitro Gas Production and Fermentation Characteristics Am. J. Plant Sci. 4, 34–43 (2013).
Machado, L., Magnusson, M., Paul, N. A., Nys, R. & Tomkins, N. Effects of marine and freshwater macroalgae on in vitro total gas and methane production. PLoS One 9, e85289 (2014).
Machado, L. et al. Dose-response effects of Asparagopsis taxiformis and Oedogonium sp. on in vitro fermentation and methane production. J. Appl. Phycol. 28, 1443–1452 (2015).
Kinley, R. D., de Nys, R., Vucko, M. J., Machado, L. & Tomkins, N. W. The red macroalgae Asparagopsis taxiformis is a potent natural antimethanogenic that reduces methane production during in vitro fermentation with rumen fluid. Anim. Prod. Sci. 56, 282–289 (2016).
Kinley, R. D. & Fredeen, A. H. In vitro evaluation of feeding North Atlantic stormtoss seaweeds on ruminal digestion. J. Appl. Phycol. 27, 2387–2393 (2015).
O’Brien, M., Navarro-Villa, A., Purcell, P. J., Boland, T. M. & O’Kiely, P. Reducing in vitro rumen methanogenesis for two contrasting diets using a series of inclusion rates of different additives. Anim. Prod. Sci. 54, 141–157 (2014).
Van Kessel, J. A. S. & Russell, J. B. The effect of pH on ruminal methanogenesis. FEMS Microbiol. Ecol. 20, 205–210 (1996).
Boadi, D., Benchaar, C., Chiquette, J. & Massé, D. Mitigation strategies to reduce enteric methane emissions from dairy cows: Update review. Can. J. Anim. Sci. 84, 319–335 (2004).
Zhang, C. M. et al. Effect of octadeca carbon fatty acids on microbial fermentation, methanogenesis and microbial flora in vitro . Anim. Feed Sci. Technol. 146, 259–269 (2008).
Newbold, C. J., Lassalas, B. & Jouany, J. P. The importance of methanogens associated with ciliate protozoa in ruminal methane production in vitro . Lett. Appl. Microbiol. 21, 230–234 (1995).
Paul, N. A., Nys, R. & Steinberg, P. D. Seaweed–herbivore interactions at a small scale: direct tests of feeding deterrence by filamentous algae. Mar. Ecol. Prog. Ser. 323, 1–9 (2006).
Blunt, J. W. et al. Marine natural products. Nat. Prod. Rep. 24, 31–86 (2007).
Liu, H., Wang, J., Wang, A. & Chen, J. Chemical inhibitors of methanogenesis and putative applications. Appl. Microbiol. Biotechnol. 89, 1333–1340 (2011).
Wang, Y., Alexander, T. W. & McAllister, T. A. In vitro effects of phlorotannins from Ascophyllum nodosum (brown seaweed) on rumen bacterial populations and fermentation. J. Sci. Food Agric. 89, 2252–2260 (2009).
Maschek, J. A. & Baker, B. J. In Algal Chemical Ecology (ed Amsler, C. D. ), 1–24 (Springer-Verlag Berlin Heidelberg, 2008).
Machmuller, A., Machmuller, A., Soliva, C. R. & Kreuzer, M. Methane-suppressing effect of myristic acid in sheep as affected by dietary calcium and forage proportion. Br. J. Nutr. 90, 529–540 (2003).
Bergman, E. N. Energy contributions of volatile fatty acids from the gastrointestinal tract in various species. Physiol. Rev. 70, 567–590 (1990).
Hoover, W. H. Chemical factors involved in ruminal fiber digestion. J. Dairy Sci. 69, 2255–2261 (1986).
Johnson, K. A. & Johnson, D. E. Methane emissions from cattle. J. Anim. Sci. 73, 2483–2492 (1995).
Moss, A. R., Jouany, J.-P. & Newbold, J. Methane production by ruminants: its contribution to global warming. Ann. Zootech. 49, 231–253 (2000).
Miller, T. L. I. E., Ferdinand Enke Verlag, Berlin, pp. 317–331. in Ruminant physiology: digestion, metabolism, growth and reproduction (ed Engelhardt, W. V., Leonhard-Marek, S., Breves, G., Giesecke, D. ), 317–331 (Ferdinand Enke Verlag, 1995).
Van Nevel, C. J. & Demeyer, D. I. In The Rumen Ecosystem (ed Hobson, P. N. ), 387–443 (Elsevier Science Publishers Ltd, 1988).
McAllister, T. A., Cheng, K. J., Okine, E. K. & Mathison, G. W. Dietary, environmental and microbiological aspects of methane production in ruminants. Can. J. Anim. Sci. 76, 231–243 (1996).
Zelenák, I., Jalc, D., Kmet, V. & Siroka, P. Influence of diet and yeast supplement on in vitro ruminal characteristics. Anim. Feed Sci. Technol. 49, 211–221 (1994).
Pereira, L., Amado, A. M., Critchley, A. T., van de Velde, F. & Ribeiro-Claro, P. J. A. Identification of selected seaweed polysaccharides (phycocolloids) by vibrational spectroscopy (FTIR-ATR and FT-Raman). Food Hydrocoll. 23, 1903–1909 (2009).
Domozych, D. S. et al. The Cell Walls of Green Algae: A Journey through Evolution and Diversity. Front. Plant Sci. 3, 82 (2012).
Sterner, M. & Edlund, U. Multicomponent fractionation of Saccharina latissima brown algae using chelating salt solutions. J. Appl. Phycol., 1–14 (2016).
Michel, G., Nyval-Collen, P., Barbeyron, T., Czjzek, M. & Helbert, W. Bioconversion of red seaweed galactans: a focus on bacterial agarases and carrageenases. Appl. Microbiol. Biotechnol. 71, 23–33 (2006).
Correc, G., Hehemann, J.-H., Czjzek, M. & Helbert, W. Structural analysis of the degradation products of porphyran digested by Zobellia galactanivorans [beta]-porphyranase A. Carbohyd. Polym. 83, 277–283 (2011).
Andrieux, C. et al. Ulva lactuca is poorly fermented but alters bacterial metabolism in rats inoculated with human fecal flora from methane and non-methane producers. J. Sci. Food Agric. 77, 25–30 (1998).
Barbeyron, T. et al. Persicivirga ulvanivorans sp. nov., a marine member of the family Flavobacteriaceae that degrades ulvan from green algae. Int. J. Syst. Evol. Microbiol. 61, 1899–1905 (2011).
Collén, P. N., Sassi, J.-F., Rogniaux, H., Marfaing, H. & Helbert, W. Ulvan lyases isolated from the Flavobacteria Persicivirga ulvanivorans are the first members of a new polysaccharide lyase family. J. Biol. Chem. 286, 42063–42071 (2011).
Hehemann, J. H. et al. Transfer of carbohydrate-active enzymes from marine bacteria to Japanese gut microbiota. Nature 464, 908–912 (2010).
Williams, A. G., Withers, S. & Sutherland, A. D. The potential of bacteria isolated from ruminal contents of seaweed-eating North Ronaldsay sheep to hydrolyse seaweed components and produce methane by anaerobic digestion in vitro . Microb. Biotechnol. 6, 45–52 (2013).
Orpin, C. G., Greenwood, Y., Hall, F. J. & Paterson, I. W. The rumen microbiology of seaweed digestion in Orkney sheep. J. Appl. Bacteriol. 58, 585–596 (1985).
Ahmed, S., Minuti, A. & Bani, P. In vitro rumen fermentation characteristics of some naturally occurring and synthetic sugars. Ital. J. Anim. Sci. 12, 359–365 (2013).
Horn, S. J. & Østgaard, K. Alginate lyase activity and acidogenesis during fermentation of Laminaria hyperborea . J. Appl. Phycol. 13, 143–152 (2001).
Russell, J. B. Strategies that ruminal bacteria use to handle excess carbohydrate. J. Anim. Sci. 76, 1955–1963 (1998).
Anderson, R. C. et al. Effect of nitroethane, dimethyl-2-nitroglutarate and 2-nitro-methyl-propionic acid on ruminal methane production and hydrogen balance in vitro . Biores. Technol. 101, 5345–5349 (2010).
Domingues, B., Abreu, M. & Sousa-Pinto, I. On the bioremediation efficiency of Mastocarpus stellatus (Stackhouse) Guiry, in an integrated multi-trophic aquaculture system. J. Appl. Phycol. 27, 1289–1295 (2015).
AOAC. Official Methods of Analysis. 15th ed. Association of Official Analytical Chemists, Arlington, Vo. (1990).
Van Soest, P. J., Robertson, J. B. & Lewis, B. A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 74, 3583–3597 (1991).
Robertson, J. B. & Van Soest, P. J. In The Analysis of Dietary Fiber in Food (eds James, W. P. T. & Theander, O. ) 123–158 (Marcel Dekker Inc., 1981).
Angell, A. R., Mata, L., Nys, R. & Paul, N. A. The protein content of seaweeds: a universal nitrogen-to-protein conversion factor of five. J. Appl. Phycol. 28, 511–524 (2016).
Salomonsson, A. C., Theander, O. & Westerlund, E. Chemical characterization of some Swedish cereal whole meal and bran fractions. Swed. J. Agr. Res. 14, 111–117 (1984).
Marten, G. C. & Barnes, R. F. Prediction of energy digestibility of forages with in vitro rumen fermentation and fungal enzyme systems. Standardization of analytical methodology for feeds: Proceedings of a workshop held in Ottawa, Canada. 12–14 March 1979. Ottawa, Ont. IDRC. (1980).
Theodorou, M. K., Williams, B. A., Dhanoa, M. S., McAllan, A. B. & France, J. A simple gas production method using a pressure transducer to determine the fermentation kinetics of ruminant feeds. Anim. Feed Sci. Technol. 48, 185–197 (1994).
Lopez, S. & Newbold, C. J. In Measuring Methane Production From Ruminants (eds HarinderP S., Makkar & PhilipE, Vercoe ) Ch. 1, 1–13 (Springer, Netherlands, 2007).
Demeyer, D. I. In Rumen Microbial Metabolism and Ruminant Digestion (ed Jouany, J. P. ) 217–237 (INRA Editions, 1991).
Chalupa, W. Manipulating rumen fermentation. J. Anim. Sci. 46, 585–599 (1977).
Acknowledgements
The authors are grateful for financial support from the European Union (FEDER funds through COMPETE) and National Funds (Fundação para a Ciência e a Tecnologia, FCT) through projects EXPL/CVT-NUT/0286/2013 - FCOMP-01-0124-FEDER-041111 and UID/QUI/50006/2013 - POCI/01/0145/FEDER/007265 (LAQV). M.R.G.M. and H.M.O were also supported by FCT through a post-doctoral grant (SFRH/BPD/70176/2010 and SFRH/BPD/75065/2010, respectively). The authors thank Sílvia Azevedo (ICBAS-UP) for the valuable technical assistance, Cláudio Azevedo and Pedro Maia for assistance with animal care, Ana Raquel Rodrigues for feeding the animals throughout the experiment, Isabel Sousa-Pinto and Isabel Azevedo (CIIMAR, Faculdade de Ciências – UP) for providing the seaweeds, and Paulo Cortez (ICBAS-UP) for fistulating the cows.
Author information
Authors and Affiliations
Contributions
M.R.G.M., A.R.J.C. and A.J.M.F. conceived the experiments. M.R.G.M. and A.R.J.C. conducted the experiments. M.R.G.M., A.R.J.C. and H.M.O. analysed the results. C.M. was responsible for the veterinary care of the cows. M.R.G.M. and A.R.J.C. wrote the manuscript with the contributions of A.J.M.F. and H.M.O.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Maia, M., Fonseca, A., Oliveira, H. et al. The Potential Role of Seaweeds in the Natural Manipulation of Rumen Fermentation and Methane Production. Sci Rep 6, 32321 (2016). https://doi.org/10.1038/srep32321
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep32321
This article is cited by
-
Anti-methanogenic potential of seaweeds and seaweed-derived compounds in ruminant feed: current perspectives, risks and future prospects
Journal of Animal Science and Biotechnology (2023)
-
Rumen microbial degradation of bromoform from red seaweed (Asparagopsis taxiformis) and the impact on rumen fermentation and methanogenic archaea
Journal of Animal Science and Biotechnology (2023)
-
Screening macroalgae for mitigation of enteric methane in vitro
Scientific Reports (2023)
-
Ruminant contribution to enteric methane emissions and possible mitigation strategies in the Southern Africa Development Community region
Mitigation and Adaptation Strategies for Global Change (2022)
-
Integrating the Carboxylate Platform into a Red Seaweed Biorefinery
Applied Biochemistry and Biotechnology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.



 ), 0% oil inoculum (
), 0% oil inoculum ( ). Mean values with different superscript letters were significantly different (P < 0.05).
). Mean values with different superscript letters were significantly different (P < 0.05).