Abstract
Undifilum oxytropis is a fungal endophyte of locoweeds. It produces swainsonine, which is the principal toxic ingredient of locoweeds. However, the genes, pathways and mechanisms of swainsonine biosynthesis are not known. In this study, the genome of U. oxytropis was firstly sequenced and assembled into a 70.05 megabases (Mb) draft genome, which encoded 11,057 protein-coding genes, and 54% of them were similar to current publicly available sequences. U. oxytropis genes were annotated and 164 putative genes were annotated into enzymes, such as Saccharopine dehydrogenase, Saccharopine oxidase, and Pyrroline-5-carboxylate reductase, hypothesized to be involved in the biosynthesis pathway of swainsonine. The genome sequence and gene annotation of U. oxytropis will provide new insights into functional analyses. The characterization of genes in swainsonine biosynthesis will greatly facilitate locoweed poisoning research and help direct locoism management.
Similar content being viewed by others
Introduction
Locoweeds are poisonous legumes that belong to the genera Oxytropis and Astragalus. After ingesting these toxic plants, livestock can develop diseases characterised by chronic dysfunction of the nervous system1,2,3. In China, the morbidity and death rate of livestock caused by locoweed poisoning have increased annually and led to large economic losses in the western rangelands of China in the last fifty years4.
The principal toxic ingredient of locoweeds is the indolizidine alkaloid swainsonine (SW)5,6,7,8,9,10. SW primarily inhibits the activities of Golgi apparatus α- mannosidase II (MAN2A1) and lysosome α- mannosidase (MAN2B1)11,12,13. Recent studies showed that SW treatment could reduce the expression of MAN2A1 and MAN2B1 in some animal tissues and cells14,15,16.
Swainsonine was shown to be produced by two unrelated fungi, Rhizoctonia leguminicola and Metarhizium anisopliae17,18. In 2009, a fungal endophyte, Undifilum oxytropis19, was reported to produce swainsonine in locoweeds20. The Undifilum is now categorized as Alternaria sect. Undifilum21,22, which belong to Pleosporaceae in the phylum Ascomycota. Undifilum species have been found to be associated with swainsonine-containing Astragalus and Oxytropis species in China and North America19,23,24. In these plants, swainsonine levels were correlated positively with U. oxytropis content25, which provided original evidence that fungal genetics might play an important role in swainsonine production20. The metabolic pathway that leads to the formation of swainsonine has been studied in Metarhizium, and R. leguminocola26,27,28, in which the swainsonine metabolic pathway appears to be a branch of the lysine biodegradation pathway26.
However, the biosynthetic pathway of swainsonine in U. oxytropis is still not clear. With the development of next-generation DNA sequencing, annotation of the genome of U. oxytropis can provide important data on genes involved in swainsonine biosynthesis. In this study, we firstly sequenced and assembled the genome of U. oxytropis that produced swainsonine. We made full annotations with the predicted genes in this genome. With integrated gene prediction and annotation, we illuminated the biosynthesis and predicted key enzymes that may regulate SW in U. oxytropis. This study will further contribute to the understanding of the biosynthesis pathway and metabolic mechanism of swainsonine, and provide an important reference for prevention and treatment of locoweed poisoning in livestock.
Results
Genome sequencing and analysis
After filtering low quality and adapter contamination reads, the genome of U. oxytropis was sequenced by whole genome shotgun sequencing strategy and produced 3,950 Mb clean data (Table S1). The assembly was performed by SOAP de novo genome assembler29, which generated 9,757 contigs with N50 of 24,851 base pairs (bp) and then assembled into 6367 scaffolds with N50 of 33,191 bp. The lengths of scaffolds ranged from 1,000 bp to 345,265 bp (Table 1). Finally, we assembled a 70.05 Mb draft genome sequence for U. oxytropis. The expected genome size was 79.71 Mb as determined by k-mer length (Figures S1, S2, S3), so these scaffolds covered 87.88% of the whole genome. The G + C content of the U. oxytropis genome was 40.37% (Figure S4). The features of the assembled genome sequences are shown in Table 1.
The analysis of genome component
We identified 11,057 protein-coding genes with a total length of 18,400,467 bp, accounting for 26.27% of the genome by combining several different gene predictors, (Table S2). The gene number was equal to number of coding sequence (CDS), The total length of exons was also equal to total length of CDS (Table S2). The gene density was 157.8 genes/per Mbp and the average size of protein coding genes was 1,664.15 bp. Genes contained small exons (average 505.17 bp) and introns (average 149.64 bp). There were an average of 2.77 exons in one gene, which was similar to that found for other Ascomycetes30. U. oxytropis was predicted to have 11,057 protein coding genes, which was similar to the coding capacity of other Ascomycetes30. In addition, in the U. oxytropis genome, genes of <2000 bp account for 81%, gene length distribution 0 ~ 1000 bp account for 41.6% of the gene length, 1000 ~ 2000 bp account for 39.4% of the gene length, and >2000 bp account for 19% (Figure S5).
Gene function annotation
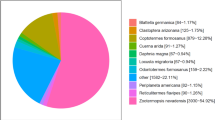
We mapped our predicted proteins to Gene Ontology (GO) using homology search, 5,731 (51.8%) of which were assigned to GO terms, including 10,933, 2,354 and 5,470 genes that mapped to the molecular function, cellular component, and biological process categories, respectively. In molecular function, “metabolic process”, “binding” and “catalytic activity” were the top three categories, which matched 3,383, 3,370 and 3,159 genes, respectively. Within cellular component, “cell” and “cell part” were the top 2 categories, which matched 1,173 and 1,173 genes. In biological process, “metabolic process” and “cellular process” were the top 2 categories, which matched 3,098 and 2,453 genes (Fig. 1). Based on this, we screened 164 genes and assigned GO terms; the highest number of genes were found to be involved in metabolic process and catalytic activity (Fig. 2). Meanwhile, we also assigned 5,965 proteins according to the Kyoto Encyclopedia of Genes and Genomes (KEGG) database. The KEGG function classification is shown in Fig. 3, in which “Xenobiotics Biodegradation and Metabolism”, “Translation” and “replication and repair” were the top 3 categories, followed by “Amino Acid Metabolism” and “Carbohydrate Metabolism”, which included up to 1,065, 944, 912, 791 and 780 predicted U. oxytropis genes, respectively. As a result, about 54% of predicted genes were similar to sequences in public databases and only 5,092 genes were not similar to current public sequences, some of which might be Undifilum specific genes.
The pathway of swainsonine synthesis
Swainsonine is of great importance in U. oxytropis because of its significant roles in immune regulation and anticancer activity31,32,33,34. Swainsonine can induce toxicosis in animals that consume the alkaloid. Therefore, it is very important to understand the biosynthesis of swainsonine and identify the key genes associated its biosynthesis. According to “Tropane, piperidine and pyridine alkaloid biosynthesis” pathway in KEGG (map00960), we speculated the putative swainsonine biosynthesis pathway in U. oxytropis (Fig. 4). We knew that swainsonine is a final product of L-lysine degradation, from L-Lysine to Saccharopine and L-2-aminoadipate 6-semialdehyde, the two intermediates can be obtained from L-lysine degradation (KEGG map00310), from the genome annotation results, and we found that 5 genes was annotated to a key enzyme, Saccharopine dehydrogenase (SDH, EC:1.5.1.-) of this pathway. In L-Lysine degradation process, from L-2-aminoadipate 6-semialdehyde to ∆1-piperideine-6-carboxylic acid (P6C), 1 gene was annotated to Saccharopine oxidase (FAP2, EC 1.5.3.1), furthermore, we found 2 genes annotated to pyrroline-5-carboxylate reductase (P5CR, EC:1.5.1.2) from P6C to L-pipecolate. However, we also found that many more genes that were annotated to two enzymes, polyketide synthase (PKS, EC: 2.3.-.-) and cytochrome P450 (P450, EC: 1.14.-.-), from L-pipecolate to swainsonine, the former was annotated to 79 genes, and the latter was 77 genes. To sum up, we found that some genes can be annotated to key enzymes, which were connected with swainsonine biosynthesis pathway. The important genes information annotated to some enzymes were shown in Table 2.
Enzymes involved in this pathway are: SDH: saccharopine dehydrogenase, [EC:1.5.1.7], K00290, [EC:1.5.1.9], K00292, [EC:1.5.1.10], K00293; FAP2: saccharopine oxidase, [EC: 1.5.3.1], K00301; P5CR: pyrroline-5-carboxylate reductase, [EC:1.5.1.2], K00286; PKS: polyketide synthase, [EC: 2.3.-.-]; P450: cytochrome P450, [EC: 1.14.-.-].
Discussion
Swainsonine, as the active principle in locoweeds, is a water-soluble indole alkaloid and alpha-mannosidase inhibitor which blocks Golgi oligosaccharide processing. It has gained attention widely, because it represents a new class of compounds that could inhibit tumor growth and metastasis34. Swainsonine has also aroused interest because of its immunostimulatory properties and possible use in cancer chemotherapy31,32,33,34,35,36. However, swainsonine may have detrimental effects, especially on animals such as goats and sheep, which can be poisoned after ingesting swainsonine-containing locoweeds. Recent findings have indicated that swainsonine is produced by the slow growing endophytic fungus, Undifilum oxytropis19,20. However, little is known about the biosynthesis of swainsonine by U. oxytropis. In order to gain further understanding into the biosynthesis of swainsonine, genome sequencing and annotation of U. oxytropis are crucial. Here we sequenced the genome sequence of U. oxytropis by Solexa technology. We assembled the sequences into 6,367 scaffolds in 70.05 Mb sequences represented about 87.88% of the whole genome and annotated 11,057 gene models at genome level. We compared the genome sequencing and assemble of U. oxytropis with another swainsonine-producing fungus, Metarhizium anisopliae, and we found the genome of U. oxytropis is different from the genome of M. anisopliae, which is a small genome size (39.04 Mb), the higher G + C content (51.49%) and similar genes number (10,582 bp)30. U. oxytropis was also different from the related, but non-swainsonine produce Altermaria arborescens. The Altermaria arborescens genome had a genome size of 34 Mb, predicted 9,167 genes with an average length of 1.8 Kb and gene density of 3.7 Kb/gene and GC content of 51.34%37. Li et al. (2012) analyzed the proteome of U. oxytropis and categorized 52 proteins. Our results were similar to their findings. Within molecular function, our top two categories were binding, where Li et al. found 49% of the proteins were related to binding and catalytic activity, where 10% of the proteins were given that category by Li et al.38. In our work, within the biological process area, the largest number of genes were metabolic process which corresponds with 34% of the proteins within the biological process.
Currently, studies on swainsonine biosynthesis have mostly been performed on Rhizoctonia leguminicola, renamed as Slafractonia leguminicola39 and Metarhizium anisopliae26,40,41. However the detailed biosynthesis pathway is still unclear. The pathway for swainsonine biosynthesis has been partially characterized in R. leguminicola and M. anisopliae26. In the partially characterized pathway, L-pipecolic acid was shown to serve as a precursor for the production of swainsonine. In fungi like Metarhizium and Rhizoctonia, pipecolic acid is formed by the catabolism of L-lysine that in turn allows for the formation of the alkaloid, swainsonine26,28.
In the “Tropane, piperidine and pyridine alkaloid biosynthesis” pathway from KEGG map database (map00960), swainsonine is one of the final products in lysine metabolism. In the Undifilum oxytropis genome, we found some putative genes that were annotated to lysine degradation pathway. Thus, at the genome level, we confirmed that some key enzymes that could be involved in swainsonine biosynthesis are present in the fungus. Previous studies have established two basic routes for converting lysine into pipecolic acid, which are P2C pathway and P6C pathway42. The important intermediates are ∆1-piperideine-2-carboxylic acid (P2C) and ∆1-piperideine-6-carboxylic acid (P6C)43. The P6C pathway has been intensively studied in R. leguminicola, because the synthesis of pipecolic acid via this route represents the initial steps in the production of two toxic octahydroindolzine alkaloids, slaframine, and swainsonine44. Another experimental result implied that ∆1-piperideine-6-carboxylate, not ∆1-piperideine-2-carboxylate, was involved in the conversion of lysine to pipecolic acid, and this conclusion was fully supported by relevant proton NMR studies27. Subsequently, a chain of reactions have been established through which L-lysine was converted to saccharopine, which was in turn converted to P6C through oxidative cleavage. The latter was then readily reduced to pipecolic acid. A previously unrecognized flavin enzyme, Saccharopine oxidase was identified, which oxidatively cleaves saccharopine to yield P6C. Since saccharopine is a major metabolite in lysine degradation in R. leguminicola, saccharopine oxidase apparently functions to shunt saccharopine into secondary metabolism pathway to supply precursor (pipecolic acid) for slaframine and swainsonine production28. Several of these proteins were identified from proteomics work from R. leguminicola, including L-pipecolate oxidase, L-aminoadipate semialdehyde dehydrogenase, and saccharopine dehydrogenase. In addition, six cytochrome P450 and two PKS were identified38.
Fujii et al.45 found that E. coli pyrroline-5-carboxylate (P5C) reductase (EC 1.5.1.2) (encoded by proC gene) acted efficiently with Flavobacterium lutescens LAT to convert L-lysine into L-pipecolic acid45. It is noteworthy that P5C reductase is present in almost all organisms46. It is possible that in the microorganisms that produce L-pipecolic acid via P6C pathway, the universally conserved P5C reductase is actually responsible, at least in part, for the reduction of P6C into L-pipecolic acid45. Although little is known about the biosynthetic pathway of swainsonine in the endophytic fungus Undifilum oxytropis, genome sequencing and function annotation in this fungus will open avenues for future research on control of loco-disease. According to the putative swainsonine biosynthesis pathway in U. oxytropis, we have gained an insight into swainsonine metabolism pathway and some key enzymes involved in this process such as SDH, FAP2, P5CR, PKS and cytochromes P450 enzymes. These enzymes play a very important role in swainsonine biosynthesis. In our results, we found that many genes might be involved in swainsonine biosynthesis in U. oxytropis. After predicting some key enzymes of swainsonine biosynthesis pathway in U. oxytropis, 5 putative SDH genes, 1 putative FAP2 gene, 2 putative P5CR genes, 79 putative PKS genes and 77 putative cytochromes P450 genes were found.
Previous studies reported that P2C and P6C pathways are two basic routes for converting lysine into pipecolic acid42. Moreover, combining “Tropane, piperidine and pyridine alkaloid biosynthesis” pathway, we knew that there were 3 enzymes that play an important role in this two pathways, that is L-lysine oxidase (EC1.4.3.14), ∆1-piperideine-2-carboxylic reductase (EC1.5.1.21) and L-lysine-6- dehydrogenase (EC1.4.1.18). However, no clear gene was annotated to these enzymes, which confirmed previous studies27. In P6C pathway, we found L-2-aminoadipate 6-semialdehyde, is key intermediate compound in this route, according to L-lysine degradation pathway, it is catalyzed as a product of saccharopine by SDH, and 4 genes were annotated to this enzyme, which indicated that SDH played a very important role in swainsonine biosynthesis in U. oxytropis. Meanwhile, ∆1-piperideine-6-carboxylic acid (P6C) is also the important intermediates in P6C pathway43. Fujii et al.45 thought P5CR is responsible for the reduction of P6C into pipecolic acid45, our experimental results agreed, and we found that 2 genes were annotated to this enzyme, which indicated P5CR could be a key enzyme in swainsonine biosynthesis.
Polyketides, the ubiquitous products of secondary metabolism in microorganisms, are made by a process resembling fatty acid biosynthesis that allows the suppression of reduction or dehydration reactions at specific biosynthetic steps, giving rise to a wide range of often medically useful products47. The polyketide synthases (PKS) are a large class of natural products, which are produced by bacteria, actinomyces, fungi and plants. These natural products play an important role in anti-infection, anti-fungus, anti-tumor and immunologic suppression. In our studies, we also found 79 genes were annotated to PKS in U. oxytropis genome. Since 1-indolizinone is a ketone compound, we speculated that L-pipecolic acid could be changed into 1-indolizinone through PKS. But, so far we have no evidence to support this speculation, and this needs be confirmed by future research.
Undifilum oxytropis showed rich P450 family (77 genes annotated to cytochromes P450), which is involved in the biotransformation of drugs, the bioconversion of xenobiotics, the metabolism of chemical carcinogens, the biosynthesis of physiologically important compounds such as steroids, fatty acids, eicosanoids, the conversion of alkanes, terpenes, and aromatic compounds as well as the degradation of herbicides and insecticides. There is also a broad versatility of reactions catalysed by cytochromes P450 such as carbon hydroxylation and aromatic hydroxylation48. In biosynthesis of alkaloids derived from ornithine, lysine and nicotinate (KEGG map01064), we found that 1-indolizidinone was hydroxylated into swainsonine. As a result, we speculated that cytochromes P450 hydroxylase could also play a very important role in swainsonine biosynthesis pathway.
In conclusion, in U. oxytropis genome research, we clarified the sizes and characteristics of this fungus, and screened some genes that were annotated to key enzymes, which are theoretically involved in swainsonine biosynthesis by using whole genome shotgun sequencing strategy. In future, genes knockout of some key enzymes will be carried out and obtain mutant of U. oxytropis, which do not produce swainsonine, and on this basis we will gain a new locoweed species that do not contain swainsonine, this will fundamentally resolve locoweeds poisoning of animals and then implement the comprehensive utilization and management for locoweeds of grasslands in western China.
Materials and Methods
Strains and culture conditions
Undifilum oxytropis (OK3UNF) was isolated from Oxytropis kansuensis, a locoweed widely distributed in Qianlian County of Qinghai province in western China (38°3.249N, 100°13.660E), and deposited at the Animal Toxicology Institute of Northwest A&F University (Yangling, Shaanxi, China). The hyphal tipped culture of U. oxytropis was stored in a tube at 4 °C prior to transfer to fresh PDA (Potato Dextrose Agar) media and culture at room temperature. After 10-14 days growth, the mycelium was collected and preserved at 4 °C for genomic DNA isolation and extraction.
DNA isolation, genome sequencing and assembly
Genomic DNA of U. oxytropis was isolated by an improved cetyl trimethylammonium bromide (CTAB) method49 and sequenced using a whole-genome shotgun strategy. All data were generated by paired-end sequencing of cloned inserts with insert size (500 bp) using an Illumina Hiseq2000 Sequencer at BGI-Shenzhen. After removing the low complexity, low quality, adapter and duplication contamination raw reads, the clean reads were assembled using the whole-genome de novo assembler SOAP de novo29,50.
Genome Annotation
Protein coding gene models were predicted using de novo prediction tools SNAP51, GeneMarks52 and Augustus53 and homology based gene prediction tool Genewise54 with the default parameters. The homology-based and de novo gene sets were merged to form a comprehensive, non-redundant reference gene set by Glean55. The functional annotation of predicted gene models were mainly based on homology to known annotated genes; BLAST was the primary tool in our analyses. We aligned all protein models by BLASTP to SwissProt56, NR, and PHI57, P45058, CAZy59, and mapped them by function with GO60, COGs61,62 and KEGG pathways63,64,65. Since each gene mapped to different database sequences, there could be multiple aligned results meeting the cut-off, so the annotations of the sequences with the best score were chosen to be the annotation of the gene in U. oxytropis.
Additional Information
How to cite this article: Lu, H. et al. The Genome of Undifilum oxytropis Provides Insights into Swainsonine Biosynthesis and Locoism. Sci. Rep. 6, 30760; doi: 10.1038/srep30760 (2016).
References
Marsh, C. D. The Locoweed Disease of the Plains. United States Department of Agriculture Bureau Animal Industry Bulletin, No. 112, Washington, DC (1909).
Gardiner, M. R., Linto, A. C. & Aplin, T. E. H. Toxicity of Swainsona canescens for sheep in western Australia. Aust J Agri Res 20, 87–97 (1969).
Lu, H. et al. Characterisation of locoweeds and their effect on livestock production in the western rangelands of China: a review. Rangeland J 36, 121–131 (2014).
Lu, H., Wang, S. S., Zhou, Q. W., Zhao, Y. N. & Zhao, B. Y. Damage and control of major poisonous plants in the western grasslands of China-a review. Rangeland J 34, 329–339 (2012).
Colegate, S. M., Dorling, P. R. & Huxtable, C. R. A spectroscopic investigation of swainsonine: an alpha-mannosidase inhibitor isolated from Swainsona canescens . Aust J Chem 32, 2257–2264 (1979).
Molyneux, R. J. & James, L. F. Loco intoxication: indolizidine alkaloids of spotted locoweed (Astragalus lentiginosus). Science 216, 190–191 (1982).
Tulsiani, D. R., Broquist, H. P., James, L. F. & Touster, O. The similar effects of swainsonine and locoweed on tissue glycosidases and oligosaccharides of the pig indicate that the alkaloid is the principal toxin responsible for the induction of locoism. Arch Biochem Biophys 232, 76–85 (1984).
Cao, G. R., Li, S. J., Duan, D. X., Zhao, X. W. & Russell, J. M. Isolation and identification of the toxic ingredients from Oxytropis ochrocephala . J Northwest A&F Univ (Nat. Sci. Edition) 17, l–7 (1989).
Tong, D. W., Cao, G. R. & Li, S. J. Isolation and identification of swainsonine from Oxytropis kansuensis Bunge. J Northwest A&F Univ (Nat Sci Edit) 29, 5–8 (2001).
Zhao, B. Y. The systematic analysis and toxic comparative pathology research of locoweed (Oxytropis Kansuensis) alkaloid composition. Ph.D thesis, Northwest A&F University, Yangling, Shaanxi, China (2001).
Dorling, P. R., Huxtable, C. R. & Colegate, S. M. Inhibition of lysosomal alpha-mannosidase by swainsonine, an indolizidine alkaloid isolated from Swaisona canescens . Biochem J 191, 649–651 (1980).
Tulsiani, D. R., Harris, T. P. & Touster, O. Swainsonine inhibits the biosynthesis of complex glycoproteins by inhibition of Golgi mannosidase II. J Biol Chem 257, 7936–7939 (1982).
Kang, M. & Elbein, A. Mechanism of inhibition of jack bean a-mannosidase by swainsonine. Plant Phys 71, 551–554 (1983).
Wang, S., Jia, Q. Z., Chen, G. Y., Zhang, L. & Ma, C. H. Study on correlation between serum biochemical indexes and Oxytropis glabra DC poisoning in Hetian sheep. J Agri 3, 40–43 (2013).
Lu, H., Wang, S. S., Wang, W. L., Zhang, L. & Zhao, B. Y. The effect of swainsonine in Oxytropis kansuensis on Golgi α-mannosidase II expression in the brain tissues of SD rats. J Agri Food Chem 62, 7407–7412 (2014).
Lu, H. et al. The effects of swainsonine on the activity and expression of a-mannosidase in BRL-3A cells. Toxicon 99, 44–50 (2015).
Schneider, M., Ungemach, F., Broquist, H. & Harris, T. (1S,2R,8R,8aR)-1,2,8 trihydroxyoctahydroindolizidne (swainsonine), an a-mannosidase inhibitor from Rhizoctonia leguminicola . Tetrahedron 39, 29–32 (1983).
Patrick, M., Adlard, M. W. & Keshavarz, T. Production of an indolizidine alkaloid, swainsonine by the filamentous fungus, Metarhizium anisopliae. Biotechnol Lett 15, 997–1000 (1993).
Pryor, B. M., Creamer, R., Shoemaker, R. A., McClain-Romero, J. & Hambleton, S. Undifilum, a new genus for endophytic Embellisia oxytropis and parasitic Helminthosporium bornmuelleri on legumes. Botany 87, 178–194 (2009).
Braun, K., Romero, J., Liddell, C. & Creamer, R. Production of swainsonine by fungal endophytes of locoweed. Mycol Res 107, 980–988 (2003).
Woudenberg, J. H. C., Groenewald, J. Z., Binder, M. & Crous, P. W. Alternaria redefined. Stud Mycol 75, 171–172 (2013).
Lawrence, D. P., Rotondo F. & Gannibal P. B. Biodiversity and taxonomy of the pleomorphic genus Alternaria . Mycol Prog 15, 3 (2016).
Yu, Y. T. et al. Swainsonine-producing fungal endophyte from major locoweed species in China. Toxicon 56, 330–338 (2010).
Baucom, D., Romero, M., Belfon, R. & Creamer, R. Two new species of Undifilum, the swainsonine producing fungal endophyte, from Astragalus species of locoweed in the United States. Botany 90, 866–875 (2012).
Cook, D. et al. Swainsonine concentrations and endophyte amounts of Undifilum oxytropis in different plant parts of Oxytropis sericea . J Chem Ecol 35, 1272–1278 (2009).
Sim, K. L. & Perry, D. Analysis of swainsonine and its early metabolic precursors in cultures of Metarhizium anisopliae . Glycoconj J 14, 661–668 (1997).
Wickwire, B. M., Harris, C. M., Harris, T. M. & Broquist, H. P. Pipecolic acid biosynthesis in Rhizoctonia leguminicola. I. The lysine, saccharopine, ∆1-piperideine-6-carboxylic acid pathway. J Biol Chem 265, 14742–14747 (1990a).
Wickwire, B. M., Wagner, C. & Broquist, H. P. Pipecolic acid biosynthesis in Rhizoctonia leguminicola. II. Saccharopine oxidase: a unique flavin enzyme involved in pipecolic acid biosynthesis. J Biol Chem 265, 14748–4753 (1990b).
Li, R. et al. De novo assembly of human genomes with massively parallel short read sequencing. Genome Res 20, 265–272 (2010).
Gao, Q. et al. Genome sequencing and comparative transcriptomics of the model entomopathogenic fungi Metarhizium anisopliae and M. acridum . Plos Genet 7, 1–18 (2011).
Goss, P. E., Baptiste, J., Fernandes, B., Baker, M. & Dennis, J. W. A phase I study if swainsonine in patients with advanced malignancies. Cancer Res 54, 1450–1457 (1994).
Watson, A. A., Fleet, G. W., Asano, N., Molyneux, R. J. & Nash, R. J. Polyhydroxylated alkaloids-natural occurrence and therapeutic applications. Phytochem 56, 265–295 (2001).
Oredipe, O. A. et al. Mice primed with swainsonine are protected against doxorubicin induced lethality. Cell Mol Biol 49, 1089–1099 (2003).
Sun, J. Y. et al. Suppressive effects of swainsonine on C6 glioma cell in vitro and in vivo . Phytomedicine 16, 1070–1074 (2009).
Hino, M. et al. Studies of an immunomodulator, swainsonine. I. Enhancement of immune response by swainsonine in vitro . J Antibiot 38, 926–935 (1985).
Dennis, J. W. Effects of Swainsonine and Polyinosinic: Polycytidylic Acid on Murine Tumor Cell Growth and Metastasis. Cancer Res 46, 5131–5136 (1986).
Hu, J. N. et al. Genomic characterization of the conditionally dispensable chromosome in Alternaria arborescens provides evidence for horizontal gene transfer. BMC Genomics 13, 171 (2012).
Li, H. L. et al. Proteomic analysis of the endophytic fungus Undifilum oxytropis . Afr J Biotechnol 46, 10484–10493 (2012).
Alhawatema, M. S., Sanogo, S., Baucom, D. L. & Creamer, R. A Search for the Phylogenetic Relationship of the Ascomycete Rhizoctonia leguminicola Using Genetic Analysis. Mycopathologia 179, 381–389 (2015).
Harris, C. M., Campbell, B. C., Molyneux, R. J. & Harris, T. M. Biosynthesis of swainsonine in the diablo locoweed (Astragalus oxyphysus). Tetrahedron Lett 29, 4815–4818 (1988a).
Harris, C. M., Schneider, M. J., Ungemach, F. S., Hill, J. E. & Harris, T. M. Biosynthesis of the toxic indolizidine alkaloids slaframine and swainsonine in Rhizoctonia leguminicola: metabolism of 1-hydroxyindolizidines. J Am Chem Soc 110, 940–949 (1988b).
He, M. Pipecolic acid in microbes: biosynthetic routes and enzymes. J Ind Microbiol Biotechnol 33, 401–407 (2006).
Gupta, R. N. & Spenser, I. D. Biosynthesis of the piperidine nucleus. The mode of incorporation of lysine into pipecolic acid and into piperidine alkaloids. J Biol Chem 244, 88–94 (1969).
Broquist, H. P. The indolizidine alkaoids, slaframine and swainsonine: contaminants in animal forages. Annu Rev Nutr 5, 391–409 (1985).
Fujii, T., Mukaihara, M., Agematu, H. & Tsunekawa, H. Biotransformation of L-lysine to L-pipecolic acid catalyzed by L-lysine 6-aminotransferase and pyrroline-5-carboxylate reductase. Biosci Biotech Bioch 66, 622–627 (2002).
Brandriss, M. C. & Falvey, D. A. Proline biosynthesis in Saccharomyces cerevisiae: analysis of the PRO3 gene, which encodes D1-pyrroline-5-carboxylate reductase. J Bacteriol 174, 3782–3788 (1992).
Kennedy, J. et al. Modulation of polyketide synthase activity by accessory proteins during lovastatin biosynthesis. Science 28, 1368–1372 (1999).
Bernhardt, R. Cytochromes P450 as versatile biocatalysts. J Biotechnol 124, 128–145 (2006).
Wu, F. H., Huang, D. Y., Huang, X. L., Zhou, X. & Cheng, W. J. Comparing Study on several Methods for DNA Extraction from endophytic fungi. Chin Agri Sci Bull 25, 62–64 (2009).
Li, R., Li, Y., Kristiansen, K. & Wang, J. SOAP: short oligonucleotide alignment program. Bioinformatics 24, 713–714 (2008).
Johnson, A. D. et al. SNAP: A web-based tool for identification and annotation of proxy SNPs using HapMap. Bioinformatics 24, 2938–2939 (2008).
Ter-Hovhannisyan, V., Lomsadze, A., Chernoff, Y. O. & Borodovsky, M. Gene prediction in novel fungal genomes using an abinitio algorithm with unsupervised training. Genome Res 18, 1979–1990 (2008).
Keller, O., Kollmar, M., Stanke, M. & Waack, S. A novel hybrid gene prediction method employing protein multiple sequence alignments. Bioinformatics 27, 757–763 (2011).
Birney, E., Clamp, M. & Durbin, R. GeneWise and Genomewise. Genome Res 14, 988–995 (2004).
Elsik, C. G. et al. Creating a honey bee consensus gene set. Genome Biol 8, R13 (2007).
Magrane, M. & Consortium, U. UniProt Knowledgebase: a hub of integrated protein data. Database (Oxford) bar009 (2011).
Vargas, W. A. et al. Plant defense mechanisms are activated during biotrophic and necrotrophic development of Colletotricum graminicola in maize. Plant Physiol 158, 1342–1358 (2012).
Fischer, M. et al. The Cytochrome P450 Engineering Database: a navigation and prediction tool for the cytochrome P450 protein family. Bioinformatics 23, 2015–2017 (2007).
Cantarel, B. L. et al. The Carbohydrate-Active EnZymes database (CAZy): an expert resource for Glycogenomics. Nucleic Acids Res 37, 233–238 (2009).
Ashburner, M. et al. Gene Ontology: tool for the unification of biology. Nat Genet 25, 25–29 (2000).
Tatusov, R. L., Koonin, E. V. & Lipman, D. J. A genomic perspective on protein families. Science 278, 631–637 (1997).
Tatusov, R. L. et al. The COG database: an updated version includes eukaryotes. BMC Bioinformatics 11, 41 (2003).
Kanehisa, M. A database for post-genome analysis. Trends Genet 13, 375–376 (1997).
Kanehisa, M., Goto, S., Kawashima, S., Okuno, Y. & Hattori, M. The KEGG resource for deciphering the genome. Nucleic Acids Res 32, D277–280 (2004).
Kanehisa, M. et al. From genomics to chemical genomics: new developments in KEGG. Nucleic Acids Res 34, D354–357 (2006).
Acknowledgements
This work was supported by the grants from the National Natural Science Foundation (No. 31201958) and the Special Fund for Agro-scientific Research in the Public Interest (No. 201203062) and the Key project of fundamental research Funds for the Central Universities (No. 2452016047).
Author information
Authors and Affiliations
Contributions
H.L. and B.Y.Z. conceived and designed the experiments; B.Y.Z. contributed reagents/materials; H.L., H.Y.Q., Z.H.R., S.W. and R.X.X. performed the experiments; H.L. analyzed the data and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Lu, H., Quan, H., Ren, Z. et al. The Genome of Undifilum oxytropis Provides Insights into Swainsonine Biosynthesis and Locoism. Sci Rep 6, 30760 (2016). https://doi.org/10.1038/srep30760
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep30760
This article is cited by
-
The hidden treasures in endophytic fungi: a comprehensive review on the diversity of fungal bioactive metabolites, usual analytical methodologies, and applications
Archives of Microbiology (2024)
-
swnk plays an important role in the biosynthesis of swainsonine in Metarhizium anisopliae
Biotechnology Letters (2023)
-
Transcriptome Profiles of Alternaria oxytropis Provides Insights into Swainsonine Biosynthesis
Scientific Reports (2019)
-
Application of Toxigenic Alternaria oxytropis to Soybeans and its Effect on Swainsonine Detection in Different Environments
Bulletin of Environmental Contamination and Toxicology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.