Abstract
Ecological communities comprise diverse species and their interactions. Notably, ecological and evolutionary studies have revealed that reciprocal interactions such as predator–prey, competition and mutualism, are key drivers of community dynamics. However, there is an argument that many species interactions are asymmetric, where one species unilaterally affects another species (amensalism or commensalism). This raises the unanswered question of what is the role of unilateral interactions in community dynamics. Here I use a theoretical approach to demonstrate that unilateral interactions greatly enhance community stability. The results suggested that amensalism and commensalism were more stabilizing than symmetrical interactions, such as competition and mutualism, but they were less stabilizing than an asymmetric antagonistic interaction. A mix of unilateral interactions increased stability. Furthermore, in communities with all interaction types, unilateral interactions tended to increase stability. This study suggests that unilateral interactions play a major role in maintaining communities, underlining the need to further investigate their roles in ecosystem dynamics.
Similar content being viewed by others
Introduction
In natural ecosystems, a diverse number of species interact and coexist with each other. Species interactions that have reciprocal effects on the interacting partners, such as antagonism (predation, herbivory, or parasitism), competition and mutualism, are the driving forces of population and community dynamics1. Recent studies have revealed the important roles of such diverse types of reciprocal interaction in community stability and ecosystem functioning2,3,4,5,6,7,8,9,10,11,12,13,14,15. However, potentially common interactions in natural communities are not reciprocal but unilateral (one species has a marked effect on only one other species). Nevertheless, it remains unclear how such unilateral interactions (amensalism and commensalism) affect the stability of community dynamics16.
Amensalism can be defined as an interaction in which one organism inflicts harm to another organism without receiving any costs or benefits. For example, the movement of large terrestrial vertebrates crushes grass and small terrestrial invertebrates16. Similarly, the bread mold Penicillium kills certain bacteria by producing penicillin. In contrast, commensalism can be defined as an intraspecific relationship in which one species (the commensal) obtains benefits such as food, shelter, or locomotion from another species (the host) without causing adverse effects. Commensalism often occurs between a larger host and a smaller commensal. The host is unaffected, whereas the commensal can receive great benefits. For example, remoras attach to sharks and other fishes and various biting lice, fleas and mosquitoes feed harmlessly on the skin of mammals. There are also many organisms that use trees and hermatypic corals as a habitat.
Many interactions are strongly asymmetric17,18,19,20,21,22 (one interaction strength in a pairwise interaction is strong and another is weak) and, therefore, are almost unilateral instead of reciprocal. Species interactions can be classified by a combination of interaction coefficient signs +, −, or 023. Reciprocal interactions include antagonism, competition and mutualism are defined as (+, −), (−, −) and (+, +), respectively. Unilateral interactions include amensalism and commensalism are defined as (−, 0) and (+, 0), respectively. Antagonism (+, −), such as prey–predator and host–parasite, may be amensalistic (0, −) or commensalistic (+, 0)17. Competition (−, −) also may be amensalistic18,19,20. An interaction that is mutualistic (+, +) in one ecological circumstance could be commensalistic in another circumstance21,22,24. The inherent diversity of unilateral interactions25 suggests that elucidating the role of unilateral interactions in community dynamics leads to an understanding of whole communities rather than parts of a community16.
The diversity of interaction signs is related to the asymmetry of interaction strengths. The interaction compass26 shows that interaction types classified by sign combinations transition by changing the interaction strengths. For example, if aij ≈ aji > 0 (where aij is the interaction coefficient), the interaction between species i and species j is mutualistic (+, +) and symmetric. If aji decreases so that aij > aji > 0, the interaction is still mutualistic but is also asymmetric (+, +). When aji = 0 (one species diminishes its resource supply to other species), the interaction is commensalistic (+, 0). When aji < 0 (one species overexploits the other species), the interaction changes to antagonism or parasitism (+, −). If aij ≈ aji < 0, the interaction is competition (−, −). Similarly, it can be highly asymmetric and shift to amensalism (0, −) if one species is the superior competitor. If aij > 0 > aji, the interaction is antagonism (+, −). It can be symmetric or asymmetric. For example, intraguild predation (+, −) may shift to mutual predation (−, −) if the predator-prey interaction is bi-directional. Predator-prey interaction (+, −) may be highly asymmetric and shift to amensalism (0, −) if the prey is nutritionally poor. Parasitism (+, −) may be also highly asymmetric and shift to commensalism (+, 0) if the parasite uses the host as a shelter.
Interaction networks in natural communities are comprised of such diverse interaction sign types and strengths. Unilateral interactions are intermediate cases when the effect of one species on another transitions from a positive effect to a negative effect and vice versa. In this context, revealing the effects of unilateral interactions in community dynamics facilitates our understanding of the roles of both interaction signs and strengths that are the key elements of natural communities.
A community dynamics model is presented in which reciprocal and unilateral interactions are combined to examine the role of unilateral interactions in community stability. Reciprocal interactions include antagonism, competition and mutualism, whereas unilateral interactions include amensalism and commensalism. By controlling the asymmetry of interaction strengths in communities with a single interaction type and the composition of unilateral interactions within a whole community with all interaction types, I reveal the effects of unilateral interactions on community stability following May’s approach1 (Methods).
Results
Consider a community where N species interact with each other with a probability C (connectance) through reciprocal interactions of antagonism, competition, or mutualism and through unilateral interactions of amensalism or commensalism. The proportion of unilateral interactions within a community is defined as pu (i.e., the proportion of reciprocal interactions is 1 − pu). The population dynamics of N species and the effect of unilateral interactions on the stability of population dynamics by systematically changing pu were evaluated (see Methods).
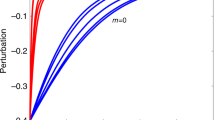
Consider communities with only reciprocal interactions (pu = 0). Congruent with a previous study27, communities with antagonism are more stable than those with mutualism or competition (Fig. 1). Here I introduce a parameter f that controls the asymmetry of interaction strengths (Methods). Controlling the asymmetry of interaction strengths demonstrates the effects of unilateral interactions on community stability (Fig. 1). In antagonistic communities, increasing the asymmetry of interaction strengths decreases stability. Extreme asymmetry leads to communities with unilateral interactions (amensalism and/or commensalism), resulting in less stability than communities with reciprocal interactions. In contrast, increasing the asymmetry of interaction strengths in competitive and mutualistic communities increases stability. These simulations are supported by a mathematical analysis (SI text). The results suggest that unilateral interactions have a more stabilizing effect than symmetrical reciprocal interactions such as competition and mutualism, but more destabilizing effects than asymmetric reciprocal interactions such as predator-prey or host-parasite interactions.
Effects of interaction strength asymmetry on stability of communities with reciprocal interactions (pu = 0).
(a) Antagonistic community, (b) Competitive or mutualistic community. Parameter values are N = 50, C = 0.2, s = 4.0 and σ = 0.5. The interaction strength asymmetry is controlled by asymmetry parameter f (see Methods). As f decreases, the asymmetry of interaction strengths increases (f = 0 is perfect asymmetry). In antagonistic community, the relative strengths of interaction coefficients of victims, exploiters or half of victims and exploiters are changed by multiplying the asymmetry parameter f by one interaction coefficient in each interaction pair.
Consider an extreme case where all species interactions are asymmetrical. The communities are then only comprised of unilateral interactions (pu = 1). Communities with only unilateral interactions (amensalism and commensalism) tend to need both unilateral interaction types for higher stability (Fig. 2). Communities skewed to either unilateral interaction type are unstable, whereas those with a moderate mix of amensalism and commensalism tend to be more stable. This unimodal pattern of stability is observed in intermediate levels of σ (that represents variation of parameters) (Fig. 2). A mathematical analysis shows that this unimodal pattern does not appear without parameter variations (SI text). Furthermore, the overall unimodal pattern observed remains qualitatively unchanged over a wide range of N and C (Fig. S1), regardless of the distribution from which the parameters are chosen (Fig. S2). This result suggests that mixing of unilateral interactions have a tendency to enhance community stability when all parameters have moderate variations.
Stability of communities with unilateral interactions (pu = 1) with varying proportion of commensalism pCo.
Colors indicate different values of σ, the standard deviation of normal distribution n(0, σ2) from which all parameter values were randomly chosen (Methods). Parameter values are N = 50, C = 0.2 and s = 4.0.
Do reciprocal interactions or unilateral interactions result in a more stable community? To answer this question, first consider a scenario in which the proportions of all interaction types are the same. In this case, unilateral interaction communities are more stable than reciprocal interaction communities (pu = 0 and pu = 1, Fig. 3). This simulation result is supported by a mathematical analysis (SI text). In addition, the result was upheld regardless of the proportions of each interaction type (Fig. 3). Unilateral interactions also compensate for unstable reciprocal interactions (Fig. 3). The stabilization due to unilateral interactions remains qualitatively unchanged over a wide range of N and C (Fig. S3), regardless of the distributions from which the parameters are chosen (Fig. S4).
Stability of hybrid communities with reciprocal and unilateral interactions with variable proportions of unilateral interactions pu.
(a) Effects of different community composition of the reciprocal interactions, balanced interactions, pa dominated, pm dominated and pc dominated. Black, blue, red and green dots indicate different proportions of reciprocal interactions, (pa, pm, pc) = (1/3, 1/3, 1/3), (0.7, 0.15, 0.15), (0.15, 0.7, 0.15) and (0.15, 0.15, 0.7), respectively. It was assumed that pCo = pAm. (b) Effects of different community composition of the unilateral interactions, balanced interactions, pAm dominated and pCo dominated. Black, purple and orange dots indicate different unilateral interaction proportions, pCo = 0.5, 0.1 and 0.9, respectively. It was assumed that pa = pm = pc. Parameter values are N = 50, C = 0.2, s = 4.0 and σ = 0.3.
What is the key factor that determines stability? The stability due to unilateral interactions is explained by the realized half connectance of reciprocal interactions (SI text). The zero interaction strengths in unilateral interactions have an effect to decrease the connectance by half, resulting in stabilization of communities as predicted by earlier theories1,27. Although the decrease in the statistical quantities such as the mean, variance and correlation of the distribution of interaction coefficients is also known to increase stability27, these effects on stability are not enough large to explain the effects of pu on stability (Figs S5 and S6).
Discussion
Ecological theory has emphasized the importance of reciprocal species interactions in community dynamics8,27. In fact, this study also shows that reciprocal interactions such as antagonism, competition and mutualism largely affect community stability. However, I also revealed that the often overlooked unilateral interactions, amensalism and commensalism, also affect community dynamics in three ways. First, unilateral interactions tend to have a stabilizing effect on community dynamics. Second, communities comprised of only unilateral interactions tend to be more stable than communities with mixed reciprocal interactions, as purely antagonistic communities are more stable than communities with only unilateral interactions. Third, unilateral interactions stabilize otherwise less stable communities with reciprocal interactions. These results suggest that unilateral interactions play a key role in ecosystem dynamics.
Two types of asymmetry in species interactions can have major roles in community stability. First one is the interaction sign asymmetry27. The communities with reciprocal interactions with symmetrical signs, mutualism (+, +) or competition (−, −), are inherently more unstable than those with asymmetrical signs, antagonism (+, −). Earlier studies have predicted that the community stability requires any special non-random interaction network structure22,28,29,30. However, even without such network structures, mutualism and competition can stabilize population dynamics by coexisting in the community15 or by the support of an inherently stable antagonism27. A balance of interaction signs within a community may maintain ecological communities.
Second type of asymmetry in species interactions is the interaction strength asymmetry22. To achieve community stability, one interaction coefficient must be smaller than the other and unilateral interactions are an extreme example of this type of asymmetry. This idea was demonstrated in a community with a single interaction type (e.g., mutualism); however, whether the asymmetry of interaction strengths is crucial for the maintenance of communities with other interaction types remains unresolved. The present results support the stabilizing effect of interaction asymmetry in communities with symmetrical interaction signs (mutualistic or competitive communities). In contrast, the interaction strength asymmetry has destabilizing effects in communities with asymmetrical interaction signs (antagonistic communities). If the interaction strengths of interacting partners are asymmetrical, the world is approximately comprised of unilateral interactions with different signs, (+, 0) and (−, 0). Even in such a case, different interaction types stabilize community dynamics by coexisting within a community. Generally, these augments suggest that either interaction signs or strengths is necessary for stabilizing communities.
The importance of asymmetric interaction signs and strengths has been independently shown in previous studies22,27. Unilateral interactions inevitably involve both types of asymmetry. In a community with unilateral or highly asymmetrical interactions, the asymmetry of interaction strengths and interaction signs (+ and −) leads to a balance in nature. In fact, analysis of field studies suggests that the balance of interaction signs +:− is equal to 1:1 25. In addition, previous theory predicts that unilateral interactions are more common than reciprocal interactions25, suggesting that natural ecosystems are stabilized by a balance of different interaction signs or unilateral interactions. Whether the predictions of a local stability analysis captures the natural environment remains open, although the present study strongly suggests the importance of unilateral interactions in community dynamics and ecosystem functioning.
Methods
Consider a community where N species may interact with each other through antagonism, competition, mutualism, or act through amensalism or commensalism. In the model, competition represents direct competition, such as interference competition. If a type I functional response is assumed, the population dynamics of species i can be described as follows:

where Xi is the abundance of species i, ri is the intrinsic rate of change in species i, si is density-dependent self-regulation and aij is the interaction coefficient between species i and j. A random network31 was utilized to only focus on the effects of interaction types on community stability and to respond to limited empirical data in the ecological network, including diverse interaction types. Connectance (C) was defined as the proportion of realized interaction links L in the possible maximum interaction links Lmax (=N(N − 1)/2) of a given network model (L = CLmax). The interaction coefficient, aij ( ), is determined as aij = eijAij in a mutualistic interaction, aij = −Aij in a competitive interaction, aij = −gijAij and aji = −aij/gij in an antagonistic interaction between the exploiter i and the resource j, aij = 0 and aji = −Aji in amensalism and aij = cijAij and aji = 0 in commensalism. In these equations, Aij is the encounter rate for interaction partners, eij, gij and cij are conversion efficiencies when species i utilizes species j in a mutualistic, antagonistic and commensalistic interaction, respectively. Although this biological assumption on interaction asymmetry was often not considered in community dynamics researches27,32, it is known to completely change a key prediction33. si is defined as si = sδi, where s controls the magnitude of self-regulations and δi is potential self-regulation. Parameters δi, eij, gij, cij, Aij and Xi* are the absolute values randomly chosen from a normal distribution n(0, σ2) with mean 0 and standard deviation σ (Figures in the supporting information describe the case used in a uniform distribution). The intrinsic rate of change, ri, is determined to hold dXi/dt = 0 after imposing an equilibrium density for each species, Xi*.
), is determined as aij = eijAij in a mutualistic interaction, aij = −Aij in a competitive interaction, aij = −gijAij and aji = −aij/gij in an antagonistic interaction between the exploiter i and the resource j, aij = 0 and aji = −Aji in amensalism and aij = cijAij and aji = 0 in commensalism. In these equations, Aij is the encounter rate for interaction partners, eij, gij and cij are conversion efficiencies when species i utilizes species j in a mutualistic, antagonistic and commensalistic interaction, respectively. Although this biological assumption on interaction asymmetry was often not considered in community dynamics researches27,32, it is known to completely change a key prediction33. si is defined as si = sδi, where s controls the magnitude of self-regulations and δi is potential self-regulation. Parameters δi, eij, gij, cij, Aij and Xi* are the absolute values randomly chosen from a normal distribution n(0, σ2) with mean 0 and standard deviation σ (Figures in the supporting information describe the case used in a uniform distribution). The intrinsic rate of change, ri, is determined to hold dXi/dt = 0 after imposing an equilibrium density for each species, Xi*.
Stability analysis was based on a Jacobian community matrix. The consequences of a small perturbation in the population dynamics equilibrium model governed by equation 1 were considered. The dynamics of small deviations, xi, away from the equilibrium point, Xi*, is given by  where
where  and J is the Jacobian matrix. The diagonal elements of J are represented by −siXi* and off-diagonal elements by aijXi*. Stability was defined as the probability of local equilibrium stability, which was estimated as the frequency of locally stable systems across 1000 sample communities34. Direct calculation of the dominant eigenvalue also supports the results in the main text (Figs S7–S9). In addition, I directly calculated the Jacobian Matrix following May’s approach27,32, because some mathematical analysis assumes this approach (SI text). However, this assumption does not affect the present results (Figs S10 and S11).
and J is the Jacobian matrix. The diagonal elements of J are represented by −siXi* and off-diagonal elements by aijXi*. Stability was defined as the probability of local equilibrium stability, which was estimated as the frequency of locally stable systems across 1000 sample communities34. Direct calculation of the dominant eigenvalue also supports the results in the main text (Figs S7–S9). In addition, I directly calculated the Jacobian Matrix following May’s approach27,32, because some mathematical analysis assumes this approach (SI text). However, this assumption does not affect the present results (Figs S10 and S11).
Consider a community where N species may interact with each other with a probability C (connectance) through reciprocal interactions (antagonism, competition, or mutualism) and through unilateral interactions (amensalism or commensalism). In communities with only reciprocal interactions, proportions of antagonistic, mutualistic and competitive interactions are defined as pa, pm and pc (= 1 − pa − pm), respectively. In communities with unilateral interactions, proportions of commensalistic and amensalistic interactions are defined as pCo and pAm (=1 − pCo), respectively. The proportion of unilateral interactions within a community is defined as pu (i.e., the proportion of reciprocal interactions is 1 − pu). The population dynamics of N species and the effect of unilateral interactions on population dynamics stability were evaluated by systematically changing pu.
The asymmetry of interaction strengths in communities with a single interaction type is controlled by changing the relative strengths of one interaction coefficient of all interacting pairs (Fig. 1). In antagonistic community, the relative strengths of interaction coefficients of victims, exploiters or half of victims and exploiters are changed by multiplying the asymmetry parameter f (0 < f < 1) by one interaction coefficient in each interaction pair. The same manner is applied to mutualistic and competitive communities.
Additional Information
How to cite this article: Mougi, A. The roles of amensalistic and commensalistic interactions in large ecological network stability. Sci. Rep. 6, 29929; doi: 10.1038/srep29929 (2016).
References
May, R. M. Qualitative stability in model ecosystems. Ecology 54, 638–641 (1973).
Thompson, J. N. Variation in interspecific interactions. Annu Rev Ecol Syst 19, 65–87 (1988).
Arditi, R., Michalski, J. & Hirzel, A. H. Rheagogies: modelling non-trophic effects in food webs. Ecol Comp 2, 249–258 (2005).
Lafferty, K., Dobson, A. P. & Kuris, A. M. Parasites dominate food web links. Proc Nat Acad Sci USA 103, 11211–11216 (2006).
Goudard, A. & Loreau, M. Non-trophic interactions, biodiversity and ecosystem functioning: an interaction web model. Am Nat 171, 91–106 (2008).
Merián, C. J., Bascompte, J., Jordano, P. & Krivan, V. Diversity in a complex ecological network with two interaction types. Oikos 118, 122–130 (2009).
Kéfi, S. et al. More than a meal… integrating non-feeding interactions into food webs. Ecol Lett 15, 291–300 (2012).
Mougi, A. & Kondoh, M. Diversity of interaction types and ecological community stability. Science 337, 349–351 (2012).
Mougi, A. & Kondoh, M. Adaptation in a hybrid world with multiple interaction types: a new mechanism for species coexistence. Ecol Res 29, 113–119 (2014).
Mougi, A. & Kondoh, M. Instability of a hybrid module of antagonistic and mutualistic interactions. Pop Ecol 56, 257–263 (2014).
Mougi, A. & Kondoh, M. Stability of competition-antagonism-mutualism hybrid community and the role of community network structure. J Theor Biol 360, 54–58 (2014).
Suweis, S., Grilli, J. & Maritan, A. Disentangling the effect of hybrid interactions and of the constant effort hypothesis on ecological community stability. Oikos 123, 525–532 (2014).
Allesina, S. & Tang, S. The stability-complexity relationship at age 40: a random matrix perspective. Pop Ecol 57, 63–75 (2015).
Kondoh, M. & Mougi, A. Interaction-type diversity hypothesis and interaction strength: the condition for the positive complexity-stability effect to arise. Pop Ecol 57, 21–27 (2015).
Coyte, K. Z., Schluter, J. & Foster, K. R. The ecology of the microbiome: Networks, competition and stability. Science 350, 663–666 (2015).
Abrams, P. A. On classifying interactions between populations. Oecologia 73, 272–281 (1987).
Williamson, M. The analysis of biological populations. (Edward Arnold: London, 1972).
Paine, R. T. Food webs: linkage, interaction strength and community infrastructure. J Anim Eol 49, 667–685 (1980).
Lawton, J. H. & Hassell, M. P. Asymmetrical competition in insects. Nature 289, 793–795 (1981).
Shoener, T. W. Field experiments on interspecific competition. Am Nat 122, 240–285 (1983).
Jordano, P. Patterns of mutualistic interactions in pollination and seed dispersal: connectance, dependence asymmetries and coevolution. Am Nat 129, 657–677 (1987).
Bascompte, J., Jordano, P. & Olesen, J. M. Asymmetric coevolutionary networks facilitate biodiversity maintenance. Science 312, 431–433 (2006)
Odum, E. P. Basic Ecology. (W. B. Saunders: Philadelphia, 1983).
Bronstein, J. L. Conditional outcomes in mutualistic interactions. Trends Ecol Evol 9, 214–217 (1994).
Dodds, W. K. Interspecific interactions: constructing a general neutral model for interaction type. Oikos 78, 377–383 (1997)
Holland, J. N. & DeAngelis, D. L. Consumer-resource theory predicts dynamic transitions between outcomes of interspecific interactions. Ecol Lett 12, 1357–1366 (2009).
Allesina, S. & Tang, S. Stability criteria for complex ecosystems. Nature 483, 205–208 (2012).
Rozdilsky, I. D. & Stone, L. Complexity can enhance stability in competitive systems. Ecol Lett 4, 397–400 (2001).
Okuyama, T. & Holland, J. N. Network structural properties mediate the stability of mutualistic communities. Ecol Lett 11, 208–216 (2008).
Allesina, S. & Levine, J. M. A competitive network theory of species diversity. Proc Nat Acad Sci USA 108, 5638–5642 (2011).
Erdős, P. & Rényi, A. On random graphs I. Publi Math 6, 290–297 (1959).
May, R. M. Will a large complex system be stable? Nature 238, 413–414 (1972).
Gellner, G. & McCann, K. S. Consistent role of weal and strong interactions in high- and low-diversity trophic food webs. Nat Commun 7, 11180 (2016).
Chen, X. & Cohen, J. E. Transient dynamics and food-web complexity in the Lotka-Volterra cascade model. Proc Roy Soc B 268, 869–877 (2001).
Acknowledgements
This study was supported by a Grant-in-Aid for Young Scientists (B) (#25840164, #16K18621) of the Japan Society for the Promotion of Science. A.M. is grateful for the valuable comments I received from anonymous referees and Y. Kodama.
Author information
Authors and Affiliations
Contributions
A.M. designed the study, analyzed the model and wrote the paper.
Ethics declarations
Competing interests
The author declares no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Mougi, A. The roles of amensalistic and commensalistic interactions in large ecological network stability. Sci Rep 6, 29929 (2016). https://doi.org/10.1038/srep29929
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep29929
This article is cited by
-
Ecological networks of an Antarctic ecosystem: a full description of non-trophic interactions
Marine Biology (2023)
-
Exploiting mosquito microbiome to combat the epidemic threat posed by Aedes aegypti
International Journal of Tropical Insect Science (2023)
-
Network analysis suggests changes in food web stability produced by bottom trawl fishery in Patagonia
Scientific Reports (2022)
-
Interspecies information systems
Requirements Engineering (2021)
-
Microbial formulation and growth of cereals, pulses, oilseeds and vegetable crops
Sustainable Environment Research (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.