Abstract
For ex-situ co-doping methods, sintering at high temperatures enables rapid diffusion of Sn4+ and Be2+ dopants into hematite (α–Fe2O3) lattices, without altering the nanorod morphology or damaging their crystallinity. Sn/Be co-doping results in a remarkable enhancement in photocurrent (1.7 mA/cm2) compared to pristine α–Fe2O3 (0.7 mA/cm2), and Sn4+ mono-doped α-Fe2O3 photoanodes (1.0 mA/cm2). From first-principles calculations, we found that Sn4+ doping induced a shallow donor level below the conduction band minimum, which does not contribute to increase electrical conductivity and photocurrent because of its localized nature. Additionally, Sn4+-doping induce local micro-strain and a decreased Fe-O bond ordering. When Be2+ was co-doped with Sn4+-doped α–Fe2O3 photoanodes, the conduction band recovered its original state, without localized impurities peaks, also a reduction in micro-strain and increased Fe-O bond ordering is observed. Also the sequence in which the ex-situ co-doping is carried out is very crucial, as Be/Sn co-doping sequence induces many under-coordinated O atoms resulting in a higher micro-strain and lower charge separation efficiency resulting undesired electron recombination. Here, we perform a detailed systematic characterization using XRD, FESEM, XPS and comprehensive electrochemical and photoelectrochemical studies, along with sophisticated synchrotron diffraction studies and extended X-ray absorption fine structure.
Similar content being viewed by others
Introduction
Improvement of the electrical conductivity of semiconductor metal oxides is one of the most profound challenges in the development of high performance photoanodes for photoelectrochemical (PEC) water splitting1,2. Various semiconducting metal oxides like TiO2 3, WO3 4, CdWO4 5 and α-Fe2O3 6 have been studied for PEC water splitting. Hematite (α–Fe2O3) is considered an ideal metal-oxide semiconductor photoanode for PEC applications, owing to its stability, suitable band gap (2.2 eV), low cost and non-toxic nature7. However, pristine α–Fe2O3 exhibits poor performance due to short hole diffusion lengths (2–4 nm) and low electron mobility8. Doping of α–Fe2O3 photoanodes has been extensively investigated to improve its photoelectrochemical properties9. The introduction of mono-dopants (such as Sn10, Ti11, Si12, Pt13, Zr14, Ge15,16, Cr17 and Zn18) has been used to enhance the PEC performance of α–Fe2O3 photoanodes. Electron-donor dopants introduce electrons into neighboring Fe3+ sites and reduce Fe3+ to Fe2+ 19,20. Since the electrical conductivity of α-Fe2O3 follows the polaron hopping mechanism7, the newly formed Fe2+ sites can improve electrical conductivity with electron donor dopants such as Si21 and Sn10. The cationic elemental doping enhances conductivity as a result of increased donor concentration and improved charge transfer8. Sn is an effective dopant for α–Fe2O3 photoanodes, with an ionic radius and Pauling electronegativity similar to those of Fe ions22. Introduction of co-dopants generally improves the photoelectrochemical properties of α–Fe2O3 photoanodes. Recently, many research groups have reported that co-doping may significantly improve the PEC performance of α–Fe2O3 photoanodes through various mechanisms23,24,25,26,27,28. For example, the ionic radius difference between Fe3+, Si4+ and Ti4+ increases the donor concentration for Si and Ti co-doped α–Fe2O3 24. N and Zn co-doped α–Fe2O3 possesses a higher concentration of acceptors, and exhibits improved photo-response with Zn doping and lower dark current with N doping26. Zn and Ti co-doped α–Fe2O3 enhances PEC device performance through increased electrical conductivity and improved charge transport properties28. When electron donor dopants such as Si4+, Ti4+ and Sn4+ are introduced into the α-Fe2O3 lattice, Fe3+ in the α-Fe2O3 lattice is replaced by the respective 4+ dopant cations19,29. Generally, in-situ doping methods alter the crystallinity and morphology of α–Fe2O3, which may have a substantial impact on PEC device performance24,26,27,28,30.
Here we discuss a simple ex-situ co-doping method, employing Sn and Be as dopants in a lattice of α–Fe2O3 nanorods. For the ex-situ co-doping method, Sn and Be were deposited onto α–Fe2O3 nanorods by dip-coating, followed by high temperature sintering (800 °C for 10 min). High temperature sintering and an ultrafast heating rate enable rapid dopant diffusion into the α–Fe2O3 lattice, and such brief 800 °C sintering minimizes morphology and crystallinity changes in the α–Fe2O3 nanorods. Introduction of Sn4+ mono-dopants into α-Fe2O3 photoanodes enhances their PEC properties by improving electrical conductivity and reducing transport resistance (by improving the photoanode/electrolyte interface)30. However, the Sn4+ mono-doping induces various undesirable changes in the α-Fe2O3 photoanodes, including a change in conduction band position and an increase in micro-strain compared to pristine photoanodes. However, the introduction of a secondary dopant, such as Be2+, along with Sn4+ dramatically enhances the PEC properties of α-Fe2O3 photoanodes from 0.7 mA/cm2–1.7 mA/cm2 at 1.23 VRHE. Here we present a detailed, systematic characterization of the role of Be2+ as an effective co-dopant using XRD, FESEM, XPS and detailed electrochemical and photoelectrochemical studies. Furthermore, we performed density functional theory (DFT) calculations to understand the doping effects induced on the structural and electronic properties. Structural information obtained by EXAFS and micro-strain analysis from the synchrotron XRD studies give a clearer picture of the micro- and macro- changes in the doped photoanodes. To the best of our knowledge, this is the first experimental demonstration of Sn4+ and Be2+ co-doping of α–Fe2O3 photoanodes employing an ex-situ co-doping method.
Results & Discussion
The Sn4+ and Be2+ ex-situ co-doping method for α–Fe2O3 photoanodes is illustrated in the Fig. 1. First, β-FeOOH nanorods, average length of 400 nm, was grown on FTO substrates by a hydrothermal method followed by 550 °C sintering in order to promote a phase transition from β–FeOOH to pure α–Fe2O3. Afterwards, Sn4+ and Be2+ co-doping on α–Fe2O3 nanorods were carried out using a simple ex-situ doping method. The α–Fe2O3 nanorods were dip-coated with the Sn-precursor, dried by air blowing (leaving a thin layer of Sn precursor), then dipped into Be-precursor and dried once again. The Sn/Be-coated α–Fe2O3 nanorods were finally sintered at 800 °C for 10 min, promoting the effective diffusion of Sn4+ and Be2+ dopants into the α–Fe2O3 lattice.
Figure S1 illustrates the XRD patterns of pristine, Sn-doped, Be-doped, and Sn/Be-doped α–Fe2O3 photoanodes. With the exception of FTO substrate peaks, all other peaks can be indexed to α–Fe2O3 (JCPDS card #33–0664). Aside from those of α–Fe2O3, no diffraction peaks of Sn, Be or other impurity phases were observed. All pristine and doped α–Fe2O3 photoanodes displayed similar diffractograms with a predominant (110) diffraction peak10,31. Figure 2 illustrates the FESEM images comparing pristine, Sn-doped, Be-doped, and co-doped α–Fe2O3 photoanodes. Both pristine and doped α–Fe2O3 photoanodes were sintered at 800 °C and show very similar nanorod morphology, with diameters of 30–50 nm and lengths of approximately 400 nm, roughly vertical to FTO substrates13. From the UV-vis spectroscopy measurements (Fig. S2), we determined that doping did not produce intermediate band levels within the bandgap of α–Fe2O3, since doping did not alter the bandgap of α–Fe2O3 photoanodes. Our ex-situ, sequential co-doping method has several advantages over in-situ or other physical vapor deposition methods that have been reported previously. Our ex-situ doping method involves rapid dopant diffusion at high temperature, which minimizes crystallinity and morphology changes in α–Fe2O3 photoanodes32,33,34. In order to confirm the incorporation of Sn4+ and Be2+ dopants into the α–Fe2O3 nanorods, X-ray photoelectron spectroscopy (XPS) analyses were performed on co-doped α–Fe2O3 photoanodes. Figure S3 displays the XPS spectra of the full survey scan, Fe 2p, Sn 3d and Be 1s regions obtained from co-doped α–Fe2O3 nanorods. Fe 2p spectra revealed that iron existed predominately as Fe3+, with binding energies for Fe2p3/2 and Fe2p1/2 levels located at 710.9 and 723.9 eV, respectively. These peaks are consistent with Fe3+ ions in α-Fe2O3 35. We also observed two XPS peaks for Sn 3d at 486.3 and 494.1 eV, corresponding to binding energies of Sn3d5/2 and Sn3d3/2 respectively36. Similarly, the presence of Be2+ dopants was supported by peaks around 113.5 eV, evidence of Be incorporation into the α–Fe2O3 photoanodes37. XPS (Table S1) and ICP analysis (Table S2) confirmed that Sn4+ and Be2+ were successfully co-doped into the α-Fe2O3 lattice with our ex-situ co-doping method.
Figure 3(a) illustrates the PEC performance of pristine, Sn-, Be-doped and Sn/Be co-doped α–Fe2O3 photoanodes. The photocurrent density of Sn-doped and Sn/Be co-doped α–Fe2O3 photoanodes was dramatically improved compared to pristine α–Fe2O3 photoanodes. A photocurrent density of 0.75 mA/cm2 was observed at 1.23 VRHE for pristine α–Fe2O3 photoanodes, and increased dramatically to 1 mA/cm2 for Sn-doped α–Fe2O3 photoanodes. The maximum photocurrent density, 1.7 mA/cm2, was exhibited by a Sn/Be co-doped photoanode with a dopant concentration of 4% and 6%, respectively, this photocurrent density is two times larger than that of pristine α–Fe2O3 photoanodes (see Fig. 3a and S4), proving that the incorporation of Sn4+ dopants into the α-Fe2O3 lattices enhances PEC device performance. We observed similar results for Sn4+ mono-doped α-Fe2O3 photoanodes with improved performance compared to pristine α-Fe2O3 30. Surprisingly, incorporation of Be2+ as a mono-dopant does not enhance the photocurrent of pristine α-Fe2O3 photoanodes. However, when Be2+ is co-doped with Sn4+, there is a synergetic increase in photocurrent when compared with pristine and Sn4+ mono-doped α-Fe2O3 photoanodes. With Sn as a mono-dopant, Fe3+ ions will be replaced by the dopant Sn4+ ions, thereby increasing the electron carrier concentration and enhancing electron transport properties, in turn increasing PEC device performance.
From transient photocurrent measurements (Fig. 3b) at a constant potential (1.1 VRHE), the current decay (Id) (difference between initial current (Ii) and final current (If); Id = Ii − If) decreased from 0.12–0.07 mA/cm2 for co-doped photoanodes compared to pristine α–Fe2O3 photoanodes. IPCE analyses of doped and pristine α–Fe2O3 photoanodes were performed at various wavelengths, as shown in Fig. S5. The Sn-Be co-doped α–Fe2O3 photoanodes had the highest IPCE values (29% at 340 nm) at 1.4 VRHE. Both Sn-doped α–Fe2O3 photoanodes (25% at 340 nm) and pristine α–Fe2O3 photoanodes (17% at 340 nm) exhibited reduced IPCE values. In comparison to pristine α–Fe2O3 photoanodes, the co-doped α–Fe2O3 photoanodes exhibited excellent photochemical stability and photochemical response (Fig. S6). These IPCE results were consistent with the difference in photocurrent densities observed in pristine and doped α–Fe2O3 photoanodes. Sn4+ donor dopants in α-Fe2O3 lattices introduce electrons to Fe3+ sites, reducing Fe3+ to Fe2+ 1920. These Fe2+ sites can thus improve the electrical conductivity of α-Fe2O3 photoanodes via the polaron hopping mechanism10. The substitutional Sn4+ dopant ions induce an extra positive charge on the α-Fe2O3 lattice which can be compensated by reducing Fe3+ to Fe2+ (preserving charge neutrality)38. In order to attain charge neutrality at the doping interface, it is necessary to incorporate another stable 2+ cation, such as Be2+ dopants23.
To further understand the effect of Sn/Be co-doping on the charge transfer kinetics of α–Fe2O3 photoanodes, EIS measurements were taken, as shown in Fig. 4a. Nyquist plots were obtained for all samples under standard illumination conditions, at an applied potential of 1.23 VRHE. In the Nyquist plots and the equivalent circuit, RS is the series resistance, which includes mainly the sheet resistance of the FTO substrate and RCT1 and CPE1 elements that characterize the charge transfer resistance and the double layer capacitance at the FTO/α–Fe2O3 interface, respectively. RCT2 and CPE2 characterize the charge transport resistance and double layer capacitance of α–Fe2O3 and the α–Fe2O3/electrolyte interface, respectively39. As shown in the fitted results (Table S4), RCT1 decreased from 133.1 to 75.1 to 48.3 , while RCT2 decreased from 198.7–113.8 Ohms and finally to 87.3 Ohms for pristine, Sn-doped and co-doped α–Fe2O3 photoanodes, respectively. Both the Sn-doped and co-doped α–Fe2O3 photoanodes exhibited reduced electron transport resistance compared to pristine α–Fe2O3 photoanodes, consistent with the enhanced electrical conductivity upon Sn4+ mono-doping and Sn/Be co-doping. Sn/Be co-doped samples showed the lowest electron transport resistance and the highest capacitance values, in agreement with PEC and conductivity data. Hall Effect measurements were performed to determine the conductivity of pristine, Sn-doped and Sn/Be co-doped α–Fe2O3 photoanodes (Table S3). Conductivity values of co-doped α–Fe2O3 photoanodes were two times larger to those of pristine α–Fe2O3 photoanodes and slightly larger than those of Sn-doped α–Fe2O3 photoanodes, indicating that Be2+ co-doping along with Sn4+-dopant effectively increases conductivity. Figure 4(b) shows the Mott-Schottky plots of pristine and doped α–Fe2O3 photoanodes. Donor concentrations of photoanodes were calculated from the slopes of Mott-Schottky plots, yielding values of 1.21 × 1019cm−3, 7.64 × 1019cm−3 and 8.03 × 1019cm−3 for pristine, Sn-doped and Sn/Be co-doped α–Fe2O3 photoanodes, respectively. As discussed earlier, when Sn4+ is introduced into the α-Fe2O3 lattice, Fe3+ in the α-Fe2O3 lattice is replaced by Sn4+ donor dopants adjacent to Fe3+ sites. The Fe3+ ions are reduced to Fe2+ to preserve charge neutrality, and the newly formed Fe2+ sites result in improved electrical conductivity of α-Fe2O3 photoanodes. However, the two charge neutrality levels will have a residual mismatch, which can be easily overcome by co-doping with Be2+ dopants.
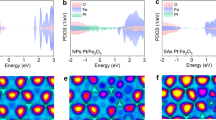
In order to analyze the doping effect on the electronic structure of α-Fe2O3, we carried out a series of first-principles calculations using density functional theory (DFT). We built four different systems, an undoped-, Be-, Sn- and Be/Sn-doped α-Fe2O3, with a dopant concentration corresponding to 3.3, 6.6 and 8.3 At%. These concentrations were selected to study diverse scenarios where either Sn or Be are predominant dopants, or when an equal concentration is found. We first focus on the systems doped at 3.3 At%. The optimized atomic structures and calculated density of states (DOS) of Be-, Sn-, and Sn/Be-doped α-Fe2O3 photoanodes are shown in Fig. 5(b–d), and compared to those of pure α-Fe2O3, Fig. 5(a). We observe that the undoped α-Fe2O3 exhibit an electronic band gap equal to 2.2 eV, in good agreement with the experimental value of 2.6 eV40. It is also well-known that the conduction band edge of α-Fe2O3 is highly localized, leading to a heavy electron-effective mass and, therefore, a very low electron conductivity41.
The optimized structure of (a) pristine, doped with (b) Be, (c) Sn, and (d) Be-Sn co-doped hexagonal α-Fe2O3. The GGA + U calculated PDOS with Gaussian broadening of 0.1 eV for (e) Be-Sn co-doped and doped with (f) Sn and (g) Be compared with (h) pure α-Fe2O3.(i) respective oxygen coordination states for pristine, Be-,Sn-, and Sn-Be-α-Fe2O3 photoanodes. The energy levels in different structures are aligned comparing the deep lying oxygen 2s orbital and the Fermi levels are shown with vertical (orange) arrows.
When Be is used to replace two Fe atoms, Be-doped α-Fe2O3, a sharp, partially-filled states appears on the valence band maximum (VBM) of α-Fe2O3, indicated by an arrow in Fig. 5g. The position of the peak with respect to the VBM is mainly determined by the O 2p orbitals. As a result, the Fermi level for Be-doped α-Fe2O3 lies on the top of the valence band, thus inducing a p-type doping. Additionally, the Be2+ doping induce significant structural changes on the lattice structure of α-Fe2O3. From Fig. 5b we observe that the coordination number of Be2+ dopants decreases to 3 (6 for Fe3+ on undoped systems), via the breakage of several Be-O bonds along the Be-O-Be path. Therefore, the dangling bonds of oxygen atom form strongly localized impurity states around the Fermi level acting as scattering points for electron transport. The remaining Be-O bonds decrease in length up to 1.63 Å which indicates a strong interaction, and as a consequence, the Be-Be distance increase up to 4.12 Å, compared to 2.86 Å for the undoped case, leaving a small cavity between them, as seen in Fig. 5b. Thus, the Be dopant induces a clear red shift in the band gap transition by a 0.22 eV upshift in valence band edge of Be-doped α-Fe2O3 photoanodes compared to pristine α-Fe2O3 photoanodes. On the other hand, the introduction of Sn-dopant atoms apparently does not induce a significant lattice distortion on the atomic structure of α-Fe2O3, where the Sn atoms only move from the ideal position by 0.06 Å, see Fig. 5c, and the distance between two Sn atoms is 2.98 Å, just 0.12 Å larger than the undoped case. However, Sn-doping actually induce a larger strain, accompanied with a very shallow donor level below the conduction band minimum (CBM), as indicated by the DOS in Fig. 5f, inducing an n-type doping, contrary to the Be-doped system. The band gap of Sn-doped α-Fe2O3 photoanodes shrinks by ~0.40 eV compared to pristine α-Fe2O3 photoanodes, and the Fermi level moves to a higher energy level, eventually laying on the conduction band edge. In addition, the concentration of free electrons is increased due to the new donor level42,43, resulting on enhanced photoactivity when compared with Be-doped and pristine α-Fe2O3. Despite of the increased charge carrier density, the newly created impurity levels might still act as recombination centers for electron-hole pairs, therefore, removing those localized states is a key step towards a photoanode with improved PEC performance. In this case, by introducing simultaneously Sn/Be onto α-Fe2O3 photoanodes, the structural deformation and lattice strain is less pronounced than the Be- or Sn-doped α-Fe2O3 cases, as shown in Fig. 5d. From Fig. 5e, we observe that the electronic structure is very similar to that of pristine α-Fe2O3 photoanodes, similar results are obtained for dopant concentration of 6.6 At% (See Fig. S7). However, this is not the case when the Sn and Be concentrations are dissimilar, as explained below. Interestingly, both localized impurity peaks at VBM observed in Be-doping, and the shallow n-type defect state beneath the CBM observed in Sn-doping are compensated on the Sn/Be co-doped α-Fe2O3 case, thus minimizing the presence of recombination centers. Additionally, the CBM of Sn/Be co-doped α-Fe2O3 exhibit less localized features suggesting a smaller effective electron mass, and hence, improved electron conductivity when compared with the pristine α-Fe2O3, similar effects has been observed for Ti/N co-doped hematite44. As a result, the charge carrier density is greatly improved, and due to fewer recombination sites, the Sn/Be co-doped α-Fe2O will exhibit enhanced photoactivity, in agreement with our experimental observations. On the other hand, doped α-Fe2O3 systems with dissimilar concentration of Sn and Be exhibit an intermediate behaviour when compared with a single doped (Sn or Be) and 1:1-Sn/Be co-doped samples. For example, we observed that a co-doped α-Fe2O3 with 5 and 3 At% of Sn and Be, respectively, (5Sn/3.3Be-co-doped Fe2O3) exhibit n-type characteristics with a reduced band gap (1.85 eV) when compared with non-doped α-Fe2O3. The excess of Sn dopant generates intermediate peaks in the band gap (See Fig. S7b), however, the density of localized states is significantly lower than those single-doped systems with just Sn or Be dopants (See Fig. S7c).
Bader charge analysis45 shows that Fe atom lose ~1.7 electrons in pure Fe2O3, whereas the charge states of Sn and Be dopants were calculated to be Sn+2.2 and Be+1.4 in Sn-Be co-doped α-Fe2O3 photoanodes, respectively. From the above results, Be dopants substituting in Fe sites should be regarded as the p-type dopant, whereas Sn should be regarded as the n-type dopant, in agreement with previous experimental reports10,43. The carrier mobility is significantly affected on Be-doped α-Fe2O3 photoanodes due to the localized nature of dopant-induced states at the top of the valence band. Similarly, in the case of Sn-doping, the Coulomb scattering induced by charged defect (donor) sites results in reduced carrier mobility.
Finally, by investigating the formation energy, the relative difficulty to incorporate Sn or Be dopants into the α-Fe2O3 lattice was evaluated. The formation energy of Sn/Be co-doped α-Fe2O3 (0.44 eV) is considerably smaller than that of Be- (2.47 eV) or Sn- (4.26 eV) doped α-Fe2O3 photoanodes. Since the optimized structures are obtained by relaxing only the atomic coordinates (keeping the crystal structure unchanged), large formation energies imply not only low dopability, but also reduced structural stability. Because of the large formation energy, Be or Sn-mono dopant induces large structural distortion and breaks the crystallinity of α-Fe2O3, which results in low electrical conductivity in spite of high carrier concentration from dopants. In addition, we also performed geometric optimization with a variable cell scheme to determine the change in lattice parameter, and hence the induced strain. The magnitudes of the induced strain measured along the (220) crystal plane are ~1% for both Sn-, and Be- doped systems, but just ~0.2% for Sn/Be-doped, see Fig. S8, these results fits remarkably well with the synchrotron XRD profiles as explained below.
Figure 6(a) shows the synchrotron XRD profiles of the pristine, Be-doped, Sn-doped, and Sn/Be co-doped α–Fe2O3 photoanodes. With the exception of FTO substrate peaks (denoted as ‘F’, JCPDS 41–1445), all peaks can be indexed to the α–Fe2O3 phase (denoted as ‘H’, JCPDS 33–0664). Aside from those of α–Fe2O3, no diffraction peaks of Sn, Be or other impurity phases were observed, indicating that the doping does not significantly disturb the crystal structure of α–Fe2O3. The pristine and doped α–Fe2O3 photoanodes displayed similar XRD patterns, with a predominant diffraction peak at the (110) plane as well as the (220) plane10,31. In order to evaluate the effect of doping on the average crystallite size and micro strain, the Williamson-Hall method was utilized by the equation (1) 46.

where β is the integral breadth of the peak from the (hkl) plane, θ is the Bragg angle, D is the average crystallite size, and εμ is the micro-strain. These parameters are summarized in Table 1. Interestingly, Sn or Be single-dopant doping increases the micro strain on the photoanodes by more than 35%, however, it reduced by Sn/Be co-doping effect, in good agreement with our theoretical observations. Otherwise, Be/Sn co-doping sequence results in the highest micro-strain. Meanwhile, a clear angle shift of the hematite (220) peak was observed, as shown in Fig. 6b. Both the amount and direction of angle shift were dependent on doping sequence. As a result, the lattice strain, expressed by  , where ddoped (dpris) is d-spacing of the hematite (220) plane for the pristine sample, was obtained and also summarized in Table 1. The micro-strain versus lattice strain was depicted in Fig. 6c. Both strains were minimized in the Sn/Be co-doped photoanode sample, as indicated by our DFT calculations. However, the Be/Sn co-doping sequence accumulated the highest lattice strain and micro-strain, most probable because Be will first occupy the most preferable and large sites, resulting in larger number of under-coordinated O atoms, and also leaving smaller or unsuitable doping sites for Sn in Fig. 5i. Under-coordinated O atoms induced as a result of Be/Sn co-doping sequence might act as potential recombination centers and hence contributes to undesired electron recombination. On the other hand, when Sn is introduced first, Sn will occupy those larger sites reducing the under-coordinated atoms, and leaving the smaller ones to Be, resulting in a more efficient doping. Thus the sequence of ex-situ co-doping is very important. In addition, Sn/Be co-doped photoanodes showed the highest (220) peak intensity. Otherwise, Be/Sn co-doped photoanodes exhibited the lowest intensity, indicating a lower crystalline order might be related a larger lattice distortion.
, where ddoped (dpris) is d-spacing of the hematite (220) plane for the pristine sample, was obtained and also summarized in Table 1. The micro-strain versus lattice strain was depicted in Fig. 6c. Both strains were minimized in the Sn/Be co-doped photoanode sample, as indicated by our DFT calculations. However, the Be/Sn co-doping sequence accumulated the highest lattice strain and micro-strain, most probable because Be will first occupy the most preferable and large sites, resulting in larger number of under-coordinated O atoms, and also leaving smaller or unsuitable doping sites for Sn in Fig. 5i. Under-coordinated O atoms induced as a result of Be/Sn co-doping sequence might act as potential recombination centers and hence contributes to undesired electron recombination. On the other hand, when Sn is introduced first, Sn will occupy those larger sites reducing the under-coordinated atoms, and leaving the smaller ones to Be, resulting in a more efficient doping. Thus the sequence of ex-situ co-doping is very important. In addition, Sn/Be co-doped photoanodes showed the highest (220) peak intensity. Otherwise, Be/Sn co-doped photoanodes exhibited the lowest intensity, indicating a lower crystalline order might be related a larger lattice distortion.
XAFS is an element-specific and bulk-local structure-determining probe. Figure 7 displays X-absorption near-edge structure (XANES) spectra and Fourier-transformed spectra of extended X-ray absorption fine structure (EXAFS) functions for Fe K-edges of doped α-Fe2O3 photoanodes. The XANES spectra for the samples were exactly the same as those of reference α-Fe2O3. The pre-edge peak denoting a quadrupole transition of 1s → 3d were observed at 7,115 eV, and the absorption rising feature and energy positions were the same. Doped α-Fe2O3 photoanodes exhibited two peaks in the Fourier transforms of EXAFS functions; the first peak A at 0.8–2.0 Å is due to the nearest Fe-L (L=O or Be) bond, while the second peak B at 2.1–3.9 Å is the contribution from Fe-M (M=Fe or Sn) and Fe-O bonds at a greater distance. Compared with reference α-Fe2O3 in powder, the samples exhibited increased intensities in both peaks, indicating enhanced orderings of the respective bonds in the films on substrates. It is a noteworthy observation that Be mono-doped and Sn/Be co-doped photoanodes had the highest intensity for the peak A. In the case of pristine samples, the Sn diffused from the FTO substrates had a detrimental effect on Fe-O bonds. While the Fe-O bond for Sn-doped sample was affected by both Sn diffused from FTO and dopant on calcination, stabilization of Be2+ dopant in Sn4+ in the co-doped sample would have had a positive influence on the nearest Fe-O bond, resulting in improved bond ordering. The enhancing effect in the structural ordering by Be doping is also confirmed as the highest intensity for Be mono-doped Fe2O3. Beryllium is doped into the position of oxygen in hematite lattice and thus, the scattering on Fe atom from neighboring Be will be much weaker than that from neighboring O unless the structural ordering is considered. On the other hand, for the peak B, both co-doped and mono-doped samples had increased intensities compared with pristine α-Fe2O3. This effect is not due to enhanced bond ordering, but rather to strong backscattering from Sn in those samples. When both Fe and Sn (types of metal scatterers) are present in the samples, Sn overwhelms the scattering from the lighter Fe atom47.
Pristine α-Fe2O3 photoanodes have low electron conductivity due to heavier electron-effective masses. When α-Fe2O3 photoanodes were doped with Sn, the Sn4+ dopants introduced a shallow donor level below the conduction band minimum of α-Fe2O3, which enhanced electron conductivity and PEC properties of Sn-doped α-Fe2O3 photoanodes. However, Sn4+ dopants induced a distortion in α-Fe2O3 structure, which was further confirmed with an increase in micro-strain and lower bond-ordering. The observed defects can be effectively overcome by co-doping with Be2+. The shallow, n-type defects introduced by Sn4+ doping can be overcome with Sn/Be co-doping, resulting in further enhancement of electrical conductivity. This is done by improving charge carrier density while mobility remains unchanged, leading to improved bond-ordering, reduced micro-strain and further enhancement in photocurrent (1.7 mA/cm2) with minimal transport resistance for Sn-Be co-doped α-Fe2O3 photoanodes.
Conclusion
In summary, α-Fe2O3 photoanodes sequentially co-doped with Sn4+ and Be2+ were investigated as means for efficient PEC water splitting. The photoactivity of α-Fe2O3 was remarkably improved by Sn4+ and Be2+ co-doping. Maximum photocurrent density was exhibited by the Sn(4%)-Be(6%) co-doped α–Fe2O3 photoanode (1.7 mA/cm2), with a photocurrent density two times larger than pristine α–Fe2O3 photoanodes. Both the Sn-doped and Sn/Be co-doped α–Fe2O3 photoanodes exhibited lower electron transport resistances compared to undoped samples, consistent with the enhanced electrical conductivity upon Sn4+ mono-doping and Sn-Be co-doping. From DFT calculations, the localized impurity peak at valance band maximum in Be doping and the shallow n-type defect state beneath the conduction band minimum in Sn doping are effectively balanced by Sn/Be co-doping. Sn4+ dopants introduced undesired band shifts and increased micro-strain in α-Fe2O3 photoanodes. This issue was resolved by employing Be2+ as a co-dopant, and ultimately confirmed with DFT, EXAFS and synchrotron XRD studies.
Experimental Section
α-Fe2O3 nanorods on FTO glass were prepared using a simple hydrothermal method, as reported by Vayssieres et al.48. In typical fashion, a piece of cleaned FTO glass was placed within a vial containing a solution consisting of 0.4 g FeCl3•6H2O and 0.85 g NaNO3 at pH 1.5 (adjusted by HCl)29. The hydrothermal reaction was conducted at 100 °C for 6 h. After cooling to room temperature, the FTO glass was rinsed several times with distilled water and dried at 60 °C. Annealing at 550 °C for 4 h was carried out for the phase transition from β–FeOOH to pure α–Fe2O3 49. The ex-situ Sn doping was carried out using a simple dipping method to treat the α-Fe2O3 photoanodes with a Sn precursor solution30. A similar procedure was followed for Be doping using BeSO4 as a Be precursor. Different concentrations of SnCl4 dissolved in ethanol and BeSO4 dissolved in deionized water were used to prepare the Sn and Be precursor solutions, respectively. After dipping, the photoanodes were allowed to dry in air at room temperature. The surface-treated samples were then subjected to high temperature sintering. This high temperature sintering (800 °C for 10 min) is believed to be important for activating the α-Fe2O3 photoanodes by enhancing electron transfer between α-Fe2O3 and conductive substrates50,51.
X-ray diffraction (XRD) patterns of all samples were collected using an X-ray diffractometer (Rigaku RINT 2500) with CuKα radiation. The surface morphology of the samples was analyzed using field emission scanning electron microscopy (FESEM, JEOL JSM 700F). X-ray absorption fine structure (XAFS) experiments were carried out on the 7D beamline of the Pohang Accelerator Laboratory (PLS-II, 3.0GeV). Synchrotron radiation was monochromatized using a Si (111) double crystal monochromator. At room temperature, the spectra for the Fe K-edge (E0 = 7112 eV) were taken in fluorescence mode. The incident beam was detuned by 30% for the Fe K-edge in order to minimize contamination of higher harmonics. The intensity of the incident beam was monitored using a He-filled IC SPEC ionization chamber. The fluorescence signal from the sample was measured with a passivated implanted planar silicon (PIPS) detector. During the measurements, helium was continuously pumped into the sample chamber to minimize fluorescence signals for spectra with elevated signal-to-noise ratios. AHENA in the IFEFFIT suite of software programs was used to analyze the data for the local-structure study of Fe in doped α-Fe2O3 photoanodes52. Pristine, Be-doped, Sn-doped, and co-doped α–Fe2O3 photoanodes were investigated by XRD measurement with the 5A beamline of the Pohang Light Source II (PLS-II) in Korea. The XRD data was obtained with conventional theta/two-theta scans, at a wavelength of 0.1072 nm. All photoelectrochemical measurements were carried out in 1 M NaOH (pH = 13.8) using a potentiostat (Ivium CompactStat) with a Pt coil as the counter electrode and Ag/AgCl as the reference electrode. Photocurrent-potential (J-V) curves were swept at 50 mVs−1 from −0.7 to +0.7 V compared to Ag/AgCl. To measure incident photon-to-current conversion efficiencies (IPCE), a 300 W Xe lamp (Newport, 6258) was coupled to a grating monochromator (Newport, 74125) operating in the wavelength range of 330–600 nm, and incident light intensity was measured with a UV silicon detector (Newport, 71675). The photoelectrode was biased at +0.6 or +1.0 V (compared to Ag/AgCl) during all IPCE measurements. Impedance spectroscopy measurements were performed using an impedance analyzer (Ivumstat). Impedance spectra were measured over a frequency range of 1 × 10−2 to 3 × 106Hz at 25 °C under open circuit conditions, with amplitude of 10 mV and under a bias illumination of 100 mWcm−2. For ICP analysis, 39.34 mL of conc. HCl (37%) solution was taken in a 100 mL Teflon beaker. 10 mL of conc. HNO3 (70%) solution was slowly added to conc.HCl solution. Photoanodes were dipped in aqua regia solution up to film dissolution. In each condition 10 samples were dissolved with aqua regia solution. Atomic ratio of elements in as-prepared hematite samples were measured by inductively coupled plasma mass spectrometry (ICP-MS; Agilent 2500a, Santa Clara, CA, USA).
Computational Details
Ab initio calculations were performed within the framework of the density functional theory (DFT)53 using the generalized gradient approximation and the model of Perdew, Burke and Ernzerhof54 as the exchange-correlation term. The electronic structure was solved using Vanderbilt ultrasoft pseudopotentials55. An kinetic energy cutoff for wave functions was set to 40 Ry and 360 Ry for the charge density. A Marzari-Vanderbilt56 smearing of 0.01 Ry was used to aid convergence. The integration of the Brillouin zone was carried out using 3 × 3 × 3 Monkhorst-Pack grid57. The DFT computations were performed using the Quantum Espresso (QE) code58 .We also compared our results with the VASP code59,60 using a cut-off energy of 500 eV, the rest of the variables were kept similar to those used in QE. In order to take into account the strong on-site Coulomb interaction present in d orbitals of Fe, we adopted the LSDA+U formalism as described by Dudarev, et al.61. An effective Hubbard correction term, Ueff, of 4.5 eV was used to properly describe the electronic structure of α–Fe2O3 62,63. A super cell oriented towards the [220] crystal direction containing 60 atoms was used as a model, where the doping was performed on two contiguous Fe atoms with different spin orientations, and thus maintaining an antiferromagnetic configuration64. These two contiguous Fe atoms were replaced by Be, Sn, and a co-doped Be/Sn system was also investigated. Finally, the ion positions were geometrically optimized by conjugate gradient minimization.
Additional Information
How to cite this article: Annamalai, A. et al. Sn/Be Sequentially co-doped Hematite Photoanodes for Enhanced Photoelectrochemical Water Oxidation: Effect of Be2+ as co-dopant. Sci. Rep. 6, 23183; doi: 10.1038/srep23183 (2016).
References
Gratzel, M. Photoelectrochemical cells. Nature 414, 338–344 (2001).
Li, X. et al. Engineering heterogeneous semiconductors for solar water splitting. J. Mater. Chem. A 3, 2485–2534 (2015).
Mali, M. G., An, S., Liou, M., Al-Deyab, S. S. & Yoon, S. S. Photoelectrochemical solar water splitting using electrospun TiO2 nanofibers. Appl. Surf. Sci. 328, 109–114 (2015).
Singh, T., Mueller, R., Singh, J. & Mathur, S. Tailoring surface states in WO3 photoanodes for efficient photoelectrochemical water splitting. Appl. Surf. Sci. 347, 448–453 (2015).
Hu, W. et al. Synthesis and photoelectrochemical properties of CdWO4 and CdS/CdWO4 nanostructures. Appl. Surf. Sci. 327, 140–148 (2015).
Liu, Y., Yu, Y.-X. & Zhang, W.-D. Photoelectrochemical study on charge transfer properties of nanostructured Fe2O3 modified by g-C3N4 . Int. J. Hydrogen Energy. 39, 9105–9113 (2014).
Li, Z. S., Luo, W. J., Zhang, M. L., Feng, J. Y. & Zou, Z. G. Photoelectrochemical cells for solar hydrogen production: current state of promising photoelectrodes, methods to improve their properties, and outlook. Energy Environ. Sci. 6, 347–370 (2013).
Sivula, K., Le Formal, F. & Gratzel, M. Solar water splitting: Progress using hematite (α-Fe2O3) photoelectrodes. Chemsuschem 4, 432–449 (2011).
Liu, C., Dasgupta, N. P. & Yang, P. D. Semiconductor nanowires for artificial photosynthesis. Chem. Mater. 26, 415–422 (2014).
Ling, Y. C., Wang, G. M., Wheeler, D. A., Zhang, J. Z. & Li, Y. Sn-doped hematite nanostructures for photoelectrochemical water splitting. Nano Lett. 11, 2119–2125 (2011).
Shen, S. H. et al. Physical and photoelectrochemical characterization of Ti-doped hematite photoanodes prepared by solution growth. J. Mater. Chem. A 1, 14498–14506 (2013).
Lukowski, M. A. & Jin, S. Improved synthesis and electrical properties of Si-Doped α-Fe2O3 Nanowires. J. Phys. Chem. C 115, 12388–12395 (2011).
Kim, J. Y. et al. Single-crystalline, wormlike hematite photoanodes for efficient solar water splitting. Sci.Rep. 3, 2681 (2013).
Sun, X. F., Hu, C., Hu, X. X., Qu, J. H. & Yang, M. Characterization and adsorption performance of Zr-doped akaganeite for efficient arsenic removal. J. Chem. Technol Biotechnol. 88, 629–635 (2013).
Liu, J. et al. Highly oriented Ge-doped hematite nanosheet arrays for photoelectrochemical water oxidation. Nano Energy 9, 282–290 (2014).
Liu, J. et al. Ge-doped hematite nanosheets with tunable doping level, structure and improved photoelectrochemical performance. Nano Energy 2, 328–336 (2013).
Shen, S. et al. Effect of Cr doping on the photoelectrochemical performance of hematite nanorod photoanodes. Nano Energy 1, 732–741 (2012).
Xi, L. F. et al. Surface treatment of hematite photoanodes with zinc acetate for water oxidation. Nanoscale 4, 4430–4433 (2012).
Velev, J., Bandyopadhyay, A., Butler, W. H. & Sarker, S. Electronic and magnetic structure of transition-metal-doped α-hematite. Phys. Rev. B 71, 205208 (2005).
Yang, T. Y. et al. A new hematite photoanode doping strategy for solar water splitting: oxygen vacancy generation. Phys. Chem. Chem Phys. 15, 2117–2124 (2013).
Cesar, I., Kay, A., Martinez, J. A. G. & Gratzel, M. Translucent thin film Fe2O3 photoanodes for efficient water splitting by sunlight: Nanostructure-directing effect of Si-doping. J. Am. Chem. Soc. 128, 4582–4583 (2006).
Wang, W. W. & Yao, J. L. Effect of metal ions (Sn and Zn) on the thermal property of akaganeite nanorods. Mater. Res. Bull. 47, 1762–1767 (2012).
Jang, J. S., Lee, J., Ye, H., Fan, F. R. F. & Bard, A. J. Rapid screening of effective dopants for Fe2O3 photocatalysts with scanning electrochemical microscopy and investigation of their photoelectrochemical properties. J. Phys. Chem. C 113, 6719–6724 (2009).
Zhang, M. L., Luo, W. J., Li, Z. S., Yu, T. & Zou, Z. G. Improved photoelectrochemical responses of Si and Ti codoped α-Fe2O3 photoanode films. Appl. Phys. Lett. 97, 042105 (2010).
Wang, L., Lee, C. Y. & Schmuki, P. Ti and Sn co-doped anodic α-Fe2O3 films for efficient water splitting. Electrochem. Commun. 30, 21–25 (2013).
Morikawa, T., Arai, T. & Motohiro, T. Photoactivity of p-Type α-Fe2O3 induced by anionic/cationic codoping of N and Zn. Appl. Phys. Express 6, 041201 (2013).
Miseki, Y., Majima, Y., Gunji, T. & Sayama, K. Codoping effect of Sr and Ti for α-Fe2O3 photocatalyst on water oxidation utilizing IO3 − as a reversible redox ion under visible light. Chem. Lett. 43, 1560–1562 (2014).
Mirbagheri, N. et al. Visible light driven photoelectrochemical water oxidation by Zn- and Ti-doped hematite nanostructures. Acs Catal. 4, 2006–2015 (2014).
Annamalai, A. et al. Bifunctional TiO2 underlayer for α-Fe2O3 nanorod based photoelectrochemical cells: enhanced interface and Ti4+ doping. J. Mater. Chem. A 3, 5007–5013 (2015).
Annamalai, A. et al. Fabrication of superior α-Fe2O3 nanorod photoanodes through ex-situ Sn-doping for solar water splitting. Sol. Energ. Mat. Sol. Cells 144, 247–255 (2016).
Kay, A., Cesar, I. & Gratzel, M. New benchmark for water photooxidation by nanostructured α-Fe2O3 films. J. Am. Chem. Soc. 128, 15714–15721 (2006).
Cho, I. S. et al. Codoping titanium dioxide nanowires with tungsten and carbon for enhanced photoelectrochemical performance. Nat. Commun. 4, 1723 (2013).
Franking, R. et al. Facile post-growth doping of nanostructured hematite photoanodes for enhanced photoelectrochemical water oxidation. Energy Environ. Sci. 6, 500–512 (2013).
Wang, L. et al. Enhancing the Water Splitting Efficiency of Sn-Doped Hematite Nanoflakes by Flame Annealing. Chem. Eur. J. 20, 77–82 (2014).
Mcintyre, N. S. & Zetaruk, D. G. X-Ray Photoelectron Spectroscopic Studies of Iron-Oxides. Anal. Chem. 49, 1521–1529 (1977).
Xi, L. F. et al. A novel strategy for surface treatment on hematite photoanode for efficient water oxidation. Chem. Sci. 4, 164–169 (2013).
Su, L. X. et al. Solar-blind wurtzite MgZnO alloy films stabilized by Be doping. J. Phys. D: Appl. Phys. 46, 245103 (2013).
Berry, F. J., Bohórquez, A. & Moore, E. A. Rationalisation of defect structure of tin- and titanium-doped α-Fe2O3 using interatomic potential calculations. Solid State Commun. 109, 207–211 (1998).
Lopes, T., Andrade, L., Ribeiro, H. A. & Mendes, A. Characterization of photoelectrochemical cells for water splitting by electrochemical impedance spectroscopy. Intl. J. Hydrogen Energy. 35, 11601–11608 (2010).
Zimmermann, R. et al. Electronic structure of 3d-transition-metal oxides: on-site Coulomb repulsion versus covalency. J. Phys.Condens. Matter 11, 1657 (1999).
Huda, M. N., Walsh, A., Yan, Y. F., Wei, S. H. & Al-Jassim, M. M. Electronic, structural, and magnetic effects of 3d transition metals in hematite. J. Appl. Phys. 107, 123712 (2010).
Liao, P., Toroker, M. C. & Carter, E. A. Electron transport in pure and doped hematite. Nano Lett. 11, 1775–1781 (2011).
Muta, H., Kurosaki, K., Uno, M. & Yamanaka, S. Thermoelectric properties of Ti- and Sn-doped α-Fe2O3 . J. Alloys Compd. 335, 200–202 (2002).
Pan, H., Meng, X., Liu, D., Li, S. & Qin, G. (Ti/Zr,N) codoped hematite for enhancing the photoelectrochemical activity of water splitting. Phys. Chem. Chem. Phys. 17, 22179–22186 (2015).
Henkelman, G., Arnaldsson, A. & Jonsson, H. A fast and robust algorithm for Bader decomposition of charge density. Comput. Mater. Sci. 36, 354–360 (2006).
Williamson, G. K. & Hall, W. H. X-Ray line broadening from filed aluminium and wolfram. Acta Metall. 1, 22–31 (1953).
Sayers, D. E. & Bunker, B. A. X-ray Absorption: Principles, Applications, Techniques of EXAFS, SEXAFS and XANES (eds Koningsberger, D. C. & Prins, R. ) 211–235 (Wiley-Interscience, 1988).
Vayssieres, L., Beermann, N., Lindquist, S. E. & Hagfeldt, A. Controlled aqueous chemical growth of oriented three-dimensional crystalline nanorod arrays: Application to iron(III) oxides. Chem. Mater. 13, 233–235 (2001).
Annamalai, A. et al. Role of Graphene Oxide as a Sacrificial Interlayer for Enhanced Photoelectrochemical Water Oxidation of Hematite Nanorods. J. Phys. Chem. C 119, 19996–20002 (2015).
Sivula, K. et al. Photoelectrochemical Water Splitting with Mesoporous Hematite Prepared by a Solution-Based Colloidal Approach. J. Am. Chem. Soc. 132, 7436–7444 (2010).
Annamalai, A. et al. Activation of hematite photoanodes for solar water Splitting: Effect of FTO deformation. J. Phys. Chem. C 119, 3810–3817 (2015).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
Kohn, W. & Sham, L. J. Self-Consistent equations including exchange and correlation effects. Phy. Rev. 140, 1133–1138 (1965).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Vanderbilt, D. Soft Self-Consistent Pseudopotentials in a Generalized Eigenvalue Formalism. Phys. Rev. B 41, 7892–7895 (1990).
Marzari, N., Vanderbilt, D., De Vita, A. & Payne, M. C. Thermal contraction and disordering of the Al(110) surface. Phys. Rev. Lett. 82, 3296–3299 (1999).
Monkhorst, H. J. & Pack, J. D. Special points for brillouin-zone integrations. Phys. Rev. B 13, 5188–5192 (1976).
Giannozzi, P. et al. Quantum Espresso: a modular and open-source software project for quantum simulations of materials. J. Phys.:Condens. Matter 21, 395502 (2009).
Kresse, G. & Furthmuller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Furthmuller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Dudarev, S. L., Botton, G. A., Savrasov, S. Y., Humphreys, C. J. & Sutton, A. P. Electron-energy-loss spectra and the structural stability of nickel oxide: An LSDA+U study. Phys. Rev. B 57, 1505–1509 (1998).
Wrobel, R. J., Becker, S. & Weiss, H. Influence of subsurface oxygen in the catalytic CO oxidation on Pd(111). J. Phys. Chem. C 119, 5386–5394 (2015).
Sandratskii, L. M., Uhl, M. & Kübler, J. Band theory for electronic and magnetic properties of α-Fe2O3 . J. Phys. Condens. Matter 8, 983 (1996).
Meng, X. Y. et al. Enhanced photoelectrochemical activity for Cu and Ti doped hematite: The first principles calculations. Appl. Phys. Lett. 9, 112104 (2011).
Acknowledgements
This research was supported by the Basic Science Research Programs through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2012R1A6A3A04038530), and Korea Ministry of Environment (MOE) as Public Technology Program based on Environmental policy (2014000160001).
Author information
Authors and Affiliations
Contributions
A.A. and J.S.J. planned the project. A.A. managed and performed all the detailed experiments, material synthesis and electrochemical measurements. A.S. performed additional experiments for ICP-MS and XPS during the revision. H.H.L. and S.Y.L performed synchrotron x-ray diffraction studies, while S.H.C. conducted the EXAFS analysis. E.G.E., J.P. and K.J.K. assisted with DFT calculations. J.S.J. supervised the project. All the authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Annamalai, A., Lee, H., Choi, S. et al. Sn/Be Sequentially co-doped Hematite Photoanodes for Enhanced Photoelectrochemical Water Oxidation: Effect of Be2+ as co-dopant. Sci Rep 6, 23183 (2016). https://doi.org/10.1038/srep23183
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep23183
This article is cited by
-
Gradient tantalum-doped hematite homojunction photoanode improves both photocurrents and turn-on voltage for solar water splitting
Nature Communications (2020)
-
A Synergistic Effect of Surfactant and ZrO2 Underlayer on Photocurrent Enhancement and Cathodic Shift of Nanoporous Fe2O3 Photoanode
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.