Abstract
Hepatitis A virus (HAV) is a hepatotropic picornavirus that causes acute liver disease worldwide. Here, we report on the identification of a novel hepatovirus tentatively named Marmota Himalayana hepatovirus (MHHAV) in wild woodchucks (Marmota Himalayana) in China. The genomic and molecular characterization of MHHAV indicated that it is most closely related genetically to HAV. MHHAV has wide tissue distribution but shows tropism for the liver. The virus is morphologically and structurally similar to HAV. The pattern of its codon usage bias is also consistent with that of HAV. Phylogenetic analysis indicated that MHHAV groups with known HAVs but forms an independent branch and represents a new species in the genus Hepatovirus within the family Picornaviridae. Antigenic site analysis suggested MHHAV has a new antigenic property to other HAVs. Further evolutionary analysis of MHHAV and primate HAVs led to a most recent common ancestor estimate of 1,000 years ago, while the common ancestor of all HAV-related viruses including phopivirus can be traced back to 1800 years ago. The discovery of MHHAV may provide new insights into the origin and evolution of HAV and a model system with which to explore the pathogenesis of HAV infection.
Similar content being viewed by others
Introduction
Hepatitis A virus (HAV), a causative agent of water- and food-borne hepatitis, is the only reported member of the genus Hepatovirus within the family Picornaviridae1. HAV is widely distributed in humans and primates and has caused hepatitis A epidemics and outbreaks worldwide, particularly in developing countries2,3. It has a single-stranded, positive-sense RNA genome of 7,500 base pairs (nt) with a single open reading frame (ORF) flanked by 5′ and 3′ untranslated regions (UTRs). The polyprotein of HAV is cleaved co- and post-translationally into 11 proteins: 4 structural proteins, VP1–VP4 (P1 region) that comprise the viral capsid and 7 non-structural proteins, 2A–2C (P2 region) and 3A–3D (P3 region)4.
The first comparative study of HAV strains in primates demonstrated significant inter-strain heterogeneity5. In the mid-1980s, various HAVs were isolated from hepatitis outbreaks of diverse origin6,7,8,9. Based on analysis of the 900 nt of the complete VP1 protein, HAV is grouped into six genotypes (I-VI), among which, genotypes I, II and III are of human origin, while IV, V and VI are of simian origin10. Despite its considerable genetic variability, HAV exhibits low antigenic variability. Only a single serotype has been described11 and the only naturally antigenic variants are HAV strains collected from Old World monkeys12. Furthermore, studies using monoclonal antibodies have suggested the presence of a limited number of antigenic epitopes of VP3 and VP1 clustered at the surface of the virus13. A recent study using a structure-based predictive method suggested that additional epitopes might exist in VP2 and VP314.
HAV differs greatly from other picornaviruses in many respects1 and has specific molecular mechanisms for viral adaptation, including deoptimization of codon usage, Treg shut-off and ‘membrane hijacking’15. Furthermore, the clinical spectrum of disease caused by HAV varies considerably, from asymptomatic cases to mild and transient or severe hepatitis16. The tropism of HAV for the liver also remains unresolved17. Animal models used for HAV most commonly are primates18,19,20,21,22. No closely related homolog virus has been identified in animals other than primates (human and non-human primates) except for a hepatovirus (phopivirus) was very recently discovered in seals23. The restricted host range and lack of homologous viruses in other species has hampered the understanding of HAV pathogenesis and immune response. A renewed effort is needed to better characterize animal HAV-related viruses and to develop new animal models for hepatitis A virus infection24.
Here, we report on the discovery of a novel Hepatovirus from wild woodchuck Marmota Himalayana, provisionally named Marmota Himalayana hepatovirus (MHHAV) which may represent a new species and serotype in the genus Hepatovirus within the family Picornaviridae.
Results
MiSeq high-throughput sequencing
Two unique reads with lengths of 2,738 nt (contig1) and 3,324 nt (contig2) were assembled from the initial sequencing data. Blastn showed that contig1 had the best hit to simian HAV, with 75% nt identity, while contig2 had the best hit to human HAV, with 70% nt identity. Contigs1 and 2 were confirmed using specific primers.
Genomic characterization
Based on the sequences of contigs1 and 2, specific primers were designed to generate overlapping polymerase chain reaction (PCR) products. Two full-length genome sequences showing 99% nt identity from two different samples (ID 2 and 3) were acquired and deposited into GenBank under accession numbers KT229611 and KT229612. The MHHAV genome comprises 7,566 bp (excluding the polyadenylated tail), with a 713 nt 5′ UTR, an ORF of 6,756 nt (encoding a potential polyprotein precursor of 2,252 amino acids [aa]), followed by a 100 nt 3′ UTR and a poly (A) tail. The base content of the MHHAV genome is 31.39% A, 13.30% C, 20.41% G and 34.89% U, with a pyrimidine content of 48.19%, which is similar to the known HAVs (simian HAV prototype, 49.21%; human HAV prototype, 48.95%; phopivirus prototype, 46.83%). Sequence analysis revealed that the polyprotein of MHHAV showed 67% aa identity to that of HAV and 58% aa identity to that of phopivirus. Furthermore, MHHAV displays a strong codon bias that is complementary to that of the woodchuck (Table 1). The rare codons of MHHAV are very abundant in woodchuck codons (Table 2).
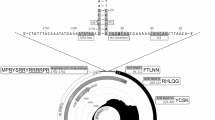
A hypothetical cleavage map of the MHHAV polyprotein was derived by alignment with other HAVs (Fig. 1a). Only 3 cleavage sites are conserved in MHHAV, primate HAV and phopivirus (VP2–VP3, 3B–3C, 3C–3D) (Fig. 1b). The P1 region of MHHAV is 2,400 nt in length, 27, 15 and 90 nt longer than those of the prototypic human HAV, simian HAV and phopivirus, respectively. Interestingly, compared to previously known HAVs an additional 18 nt sequence was detected in the VP1 region (positions 3,009–3,026 nt, sequence: TCTTCCTCTAGGAGAACA, coding for six aa “SSSRRT”); whereas only 15 nt was found in the recently reported phopivirus (TCCTCTAGGAGAACA). An RGD motif was found in the middle region of VP3 in MHHAV.
MHHAV genome organization and cleavage sites.
(a). Structure map of the MHHAV genome. P1, encoding the viral structural proteins VP4-VP2-VP3-VP1; P2 and P3 are nonstructural proteins, of which, P2 contains the 2A–2C regions and P3 the 3A–3D regions. (b). Amino acid sequences of MHHAV, the prototypic human and simian HAVs and phopivirus adjacent to the predicted protease cleavage sites (10 aa on each side are shown). The amino acids in red indicated by an arrow represent cleavage sites.
The P2 regions of the prototypic human and simian HAVs are 1,893 nt in length, but it is 1,896 nt in MHHAV and 1,932 nt in phopivirus; highly conserved aa motifs, GXXGXGKT (G1250KRGGGKS) and D1301DIGQ, in the nt-binding domain of the putative picornavirus NTPase and helicase, were also found in the 2C protein of MHHAV. The P3 region of MHHAV is 2,460 nt in length, which is 48 and 51 nt longer than those of the prototypic human and simian HAV, respectively, but 15 nt shorter than that of the phopivirus. The 3D region includes the KDELR, YGDD and FLKR motifs of the RNA-dependent RNA polymerase.
Antigenic site analysis
HAV has a conformation-dependent immunodominant neutralization site. Residues S102, V171, A176 and K221 of VP1 as well as Q70, S71, E74 and 102–121 of VP3 have been implicated in neutralizing epitopes; residues 71 and 198 of VP2 as well as residues 89–96 of VP3 may harbor other epitopes14. Sequence alignment showed that all the aa in the antigenic sites were different between MHHAV and HAV prototype Fig. S1a) with the exception of S102 of VP1 and T71 of VP2. In addition, only two aa (A70 of VP3 and T71 of VP2) were identical between MHHAV and simian prototype. Furthermore, three aa are the same between MHHAV and the recently reported phopivirus, T74 of VP3 as well as T71 and P198 of VP2. The detailed alignment of the MHHAV capsid protein with counterparts was shown in Fig. S2. The antigenic sites in the MHHAV model are presented in both cartoon (Fig. S1a) and surface (Fig. S1c) forms.
Secondary RNA structure of the 5′ UTR
The 5′ UTR of MHHAV is 20 nt shorter than that of human HAV (734 nt), 45 nt and 73 nt longer than those of simian HAV (669 nt) and phopivirus (648 nt), respectively. It shares 56.72%, 54.48% and 41.73% nucleotide identity with human HAV, simian HAV and phopivirus, respectively. The predicted secondary structure of MHHAV (Fig. 2a) shows that MHHAV 5′ UTR contains five major structural domains (including six stem-loops) labeled from I to V beginning at the 5′ terminus of the 5′ UTR and lacks the first domain found in human HAV. The first domain (stem-loop Ia and Ib) of MHHAV corresponds to domain II (stem-loop) of human HAV. The predicted secondary structure of simian HAV 5′ UTR (Fig. S3) is similar to that of human HAV but lacks the first domain and stem-loop Ia. The stem-loop Ia and Ib of domain I, present in human HAV domain II, may form a pseudoknot; however, the pseudoknot in domain V of human and simian HAVs was not found in MHHAV.
The segment from 79 to 120 nt of the MHHAV 5′ UTR represents a polypyrimidine tract that has been suggested not to be involved in the formation of conserved helical structures25. In the 5′ UTR secondary structure of MHHAV, stem-loop III is a long multi-loop cloverleaf structure that corresponds in position and shape to the previously designated stem-loop IV (in primate HAVs and phopivirus), is an internal ribosome entry site (IRES). The IRESes in MHHAV, primate HAV and phopivirus have highly conserved base paired regions governing internal translation initiation and belong to IRESes of type III.
Two important motifs near the 3′ border of the picornavirus IRES were also found in the MHHAV 5′ UTR. The first motif is an UUUCC sequence (box A) within the second pyrimidine-rich tract; the second motif is an AUG triplet (box B) that functions as an initiation codon in HAV and phopivirus26. Similar to human and simian HAVs a putative cis-acting RNA replication element (cre) is located near the 5′ end of the 3D Pol-coding sequence of MHHAV. It contains a top loop of 18 nt with a stem segment of 34 nt, interrupted by four internal loops and two 1 nt bulges (Fig. 2b). However, in spite of the 68% nt identity between the cres of MHHAV and phopivirus, the similar region in phopivirus includes three loops and a stem segment (Fig. S4).
Detection of MHHAV in wild woodchucks
Sixteen (16.16%) (ID1-16) of 99 enteric lysates from wild woodchucks were positive for MHHAV RNA by RT-PCR, among which seven animals (ID1-7) were chosen for the collection of blood, liver, spleen, lung and trachea samples. All sample types were MHHAV RNA-positive, with the highest virus load in the liver and the lowest in the trachea. T-test showed the viral loads in liver and other tissues were statistically significant (blood, p = 0.000; spleen, p = 0.022; lung, p = 0.000; trachea, p = 0.000). Two woodchucks (ID 17 and 18) had MHHAV RNA in their tissues and blood, but not in their feces (Fig. 3a). The sequences of the complete VP1 region amplified from these positive samples were determined. The 34 VP1 sequences showed 99% nt identity. Sequences within an individual woodchuck showed 100% nucleotide identity. All sequences were deposited into GenBank under accession numbers KT229577-KT229610. Furthermore, negative-stranded MHHAV RNA was detected only in the seven MHHAV-positive livers, while it is not detected in the spleen, lung, trachea, blood and feces samples (Fig. 3b).
Positive-strand and negative-strand viral RNA copy number determined by real-time PCR.
(a) Numbers of positive-strand viral RNA copies in feces, blood and other tissues. The highest viral RNA load was in the liver and the lowest in the trachea. T-test showed the viral loads in liver and other tissues were statistically significant (blood, p = 0.000; spleen, p = 0.022; lung, p = 0.000; trachea, p = 0.000). (b) Negative-strand RNA in the liver. The amplification curves of the negative-strand viral RNA were not observed in all collected tissues except for the liver. Note: “ID” stands for “identifier”.
Phylogenetic and evolutionary analysis
Recombination analysis showed no evidence of inter-genotype recombination among primate HAVs, phopivirus and MHHAV (Fig. S5). Phylogenetic analysis suggested that MHHAV forms a distinct lineage compared to previous reported HAVs by the neighbor-joining method and 1,000 bootstrap replications. In contrast, the distance between phopivirus and previously reported HAVs is much closer (Fig. 4).
Phylogenetic analysis of sequences of MHHAV: (a) VP1 region (nucleotide); (b) polyprotein (amino acid).
The tree was constructed using the neighbor-joining method by MEGA ver. 5 with 1,000 bootstrap replicates. The virus in this study is indicated by the red “•.” Bootstrap values are shown on the branches. Results showed that MHHAV formed a distinct lineage to the known primate HAVs and phopivirus.
Under the best-fit model, the mean substitution rate was 8.62 × 10−4 substitutions per site per year (ssy), with a 95% HPD of 6.96 × 10−3–7.03 × 10−3. The time of the most recent common ancestor of MHHAV and the primate HAV isolates was estimated to be around 1,000 years ago. In contrast, the common ancestor of MHHAV and phopivirus was estimated to be 1800 years ago (Fig. 5).
The Bayesian Markov Chain Monte Carlo (MCMC) tree of the VP1 regions of MHHAV and the known primate HAVs and phopivirus.
Horizontal branches are drawn to scale of estimated year of divergence, with tip times reflecting sampling data (year). The estimated time for the most recent common ancestors of the major nodes of the lineages are shown. IA, IB, IIIB: subgenotypes of HAV genotypes I and III.
Electron microscopy
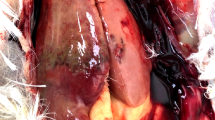
MHHAV was visualized by negative-staining electron microscopy, which revealed the presence of spherical, non-enveloped virus particles of ~27 nm in diameter, morphologically similar to HAV. Chloroform-purified virions were only rarely detected in feces, but were readily apparent following incubation with a polyclonal antibody (Fig. 6).
Discussion
HAV continues to be a source of morbidity and mortality despite the availability of an effective vaccine15,27. Recently a new HAV-related hepatovirus known as phopivirus was reported in seals23. Here, we report on the identification of a novel hepatovirus, tentatively named MHHAV, from wild woodchuck Marmota Himalayana. The purified virus particles from the fecal sample are morphologically similar to HAV virions by negative-staining electron microscopy. The complete genome and VP1 capsid protein of MHHAV was best hit to simian HAV, with 67% nt and 75% aa identities, respectively. Based on the classification of enteroviruses, in which a VP1 sequence identity of 70 to 85% is defined as a heterologous serotype, it is suggested that MHHAV may be a new serotype of HAV. Phylogenetic analyses of the polyprotein and VP1 protein indicate that MHHAV forms a separate branch from previous HAVs and may represent a new species in the genus Hepatovirus within the family Picornaviridae.
Although MHHAV has a genome organization identical to that of previously reported HAVs, the length of MHHAV polyprotein is different to those of the prototypic human HAV, simian HAV and phopivirus, respectively. Furthermore, the aa sequences of the predicted cleavage sites of MHHAV differ substantially from these viruses with only three cleavage sites (VP2/VP3, 3B/3C and 3C/3D) conserved. The common motifs in the non-structural proteins of picornaviruses, such as the NTPase and helicase motifs, were also found in MHHAV. In most picornaviruses, RGD motif is located near the C terminus of VP128, however, this motif was found in the middle of the VP3 region in MHHAV, primate HAVs and phopivirus. The RGD motif is conserved in picornaviruses and functions in recognizing and attaching to host cells or enabling cell-to-cell and cell-to-matrix interactions29,30. It is inferred from differences in the positions of RGD motifs that HAV interactions with host cell-surface integrins differ from those of other picornaviruses14. Furthermore, hepatovirus differ from other picornaviruses in that they rarely use codons most often preferred by their hosts23,31. MHHAV appears to follow HAV and phopivirus in codon usage. We speculate that this strategy may minimize direct competition of hepatovirus with host cell systems and enable persistence31.
The antigenic sites of HAVs have been characterized by several research teams13,14,32. Sequence alignment showed that these antigenic sites differed between MHHAV, primate HAVs and phopivirus. Further sequence alignment of the whole genome of MHHAV, primate HAVs and phopivirus revealed an 18 nt and 15nt insertion encoding six aa ([S]SSRRT) at the C terminus of VP1, with three or two potential O-glycosylation sites (glycosylation site prediction website: http://www.cbs.dtu.dk/services/NetNGlyc/). As glycosylation is essential for antigen processing and presentation and VP1 is a major antigenic protein, we speculate that these sequence differences may indicate antigenic differences between MHHAV and other HAVs. However, the real antigenic characteristics should be determined by a neutralization assay when an in vitro cell culture system for these viruses has been developed.
The predicted secondary structure of the 5′ UTRs of MHHAV, human HAV,, simian HAV and phopivirus is very similar. The most important structure in the 5′ UTR is the IRES, which directs internal initiation of translation25. The IRESes of primate HAVs, MHHAV and phopivirus exhibit evolutionarily conserved secondary structure including a long multi-loop cloverleaf structure23, which has been grouped into type III. This is different from IRESes of poliovirus and human rhinovirus (type I), encephalomyocarditis virus and foot-and-mouth disease virus (type II) and HCV like IRES (type IV)33,34. A conserved RNA structure, the 110 nt HAV cre element located near the 5′ end of the 3DPol region, is present in both human and simian HAVs as well as in the distantly related avian encephalomyelitis virus35, but the similar cre element was not present in the corresponding region in phopivirus. However, this element was also found in MHHAV, as was the AAACA/G motif that serves as the template for uridylylation of VPg by a slide-back mechanism35. These findings suggest that MHHAV might have replication mechanisms and tissue tropism similar to those of other known HAVs, but the shape and position of cre element in phopivirus were still unknown. Further studies are needed to address the subtle difference in the secondary structure of the 5′ UTR among MHHAV, HAV and phopivirus.
HAV is highly transmissible and HAV infection is acquired primarily by the fecal-oral route. The poor sanitary condition can cause HAV outbreak locally. In the present study, 16 out of 99 (16.16%) wild woodchucks were detected to carry the MHHAV. Furthermore 34 MHHAV VP1 sequences from different animals in this study shared 99% nt identity and sequences in the same woodchuck showed 100% nt identity. All these indicated that an MHHAV outbreak might happen in wild woodchucks at the time we collected the samples. As anticipated for an HAV replication in the liver36, negative-sense RNA complementary to the positive-sense genomic MHHAV RNA was only detected in the liver, however, though with the highest RNA viral load in the livers, MHHAV distributed widely in different organs in the wild woodchucks, which was in accordance with the previous studies that HAV (phopivirus included) can also be detected in extrahepatic organs in the hosts22,23,37,38. This phenomenon may be partly explained by the fact that the HAV cellular receptor 1 was expressed wildly in different organ, such as liver, spleen, kidney and testis39, so these organs can capture HAV virons. However, they cannot support the virus replication by some specific reasons. These findings suggest that the liver is the target organ of MHHAV infection and replication in wild woodchucks. Although its hepatotropism and ability to cause disease remain to be determined, the presence of MHHAV in the liver of wild woodchuck Marmota Himalayana is reminiscent of human HAV infection.
The phopivirus reported in seals in USA23 has similar geomic organization, codon usage bias and hepatic tropism to HAV and MHHAV. Phylogenetic analyses in this study indicated that the phopivirus is more distant to the previously reported HAVs than MHHAV. Evolutionary analysis suggested that phopivirus has a common evolutionary ancestry with HAVs and MHHAV. The estimated substitution rate of these viruses was 8.62 × 10−4 ssy, similar to a French study based on VP1 sequences from primate genotype IA HAVs (9.76 × 10−4 ssy)40. This substitution rate is lower than those found in other picornaviruses41,42. However, the common ancestor of MHHAV, primate HAVs and phopivirus was older than that of MHHAV and primate HAVs, indicating that the diversity and evolutionary pathway of HAV are far more complex than previously thought.
Human hepatotropic viruses or related viruses that infect wild woodchuck include woodchuck hepatitis virus (WHV) and hepatitis delta virus (HDV)43,44. Natural infection with WHV results in liver disease similar to that induced by HBV in humans45. Woodchuck Marmota monax is commonly used as an animal model for hepatitis B virus (HBV) infection46,47. There are currently no non-primate models of HAV infection. Guinea pigs can be infected by HAV but do not develop signs of disease or seroconvert38. The discovery of MHHAV in woodchuck may facilitate the development of a new tractable animal model of human HAV infection and thus provide further insights into the evolution and pathogenesis of, HAVs.
Materials and Methods
Specimens and high-throughput sequencing
Ninety-nine wild woodchucks were caught from Haixi, Qinghai Province in 2013. Enteric lysates of all the animals and the liver, spleen, lung and trachea specimens of some animals were collected after exsanguination. The samples were transported on dry ice and stored at −80 °C at the China Center for Disease Control (CDC). After dilution (1:5 ratio, wt/vol) and filtration (0.45 μm and 0.22 μm membranes), total nucleic acid was extracted from 99 enteric lysates, followed by cDNA synthesis. Random PCR amplification was performed on each sample using primers with different barcodes. The PCR products were pooled for sequencing on the Illumina MiSeq platform (Illumina, San Diego. CA). Metagenomic profiling of the shotgun datasets was carried out using the customized informatics pipeline VirusSeeker to computationally identify viral sequences48. The study protocol was approved by the Ethics Committee of the China CDC and was performed according to Chinese ethics laws and regulations. Furthermore, the methods were carried out in accordance with the approved guidelines.
Full-length genomic amplification
To determine the full-length genomic sequence of MHHAV, primers were initially designed based on contigs obtained by miseq high-throughput sequencing. Further synthesis was based on newly amplified MHHAV sequences. Long fragments (1500–3000 bp in length) were amplified for final confirmation. All PCR amplifications were performed using ExTaq DNA polymerase. The extreme 5′ and 3′ ends of the genome were determined using a SMART RACE cDNA Amplification Kit (Clontech, US) and Genome Walking Kit (Takara, Japan). Sequences were assembled and manually edited to produce the final sequence of the viral genome. Codon usage was assessed both for the MHHAV and woodchuck. For woodchuck, codon usage tables were obtained from a database based on genomes in NCBI GenBank (http://www.kazusa.or.jp). For MHHAV, codon usage frequencies were determined by the Cusp program (http://emboss.sourceforge.net/apps/cvs/emboss/apps/cusp.html).
Detection of MHHAV and amplification of complete VP1 region
The presence of the MHHAV in wild woodchucks was confirmed by using the primers (forward: 5′-GATCCACAATATCCAGTTTGGG-3′, reverse: 5′- CATGGTGTGCTACATTACTAGG-3′) targeting 658nt region spanning the VP2/VP3-coding junction of the virus using RT-PCR. Complete VP1 genome sequences were amplified with the primers listed below: forward: 5′-CTTTGAAGCAGGCAACTACTGGACC-3′, reverse: 5′- AAGAGATAGGTTCCCCTGCTTGTGT-3′.The reaction mixture included 20 pmol of each primer and 2.5 U of ExTaq DNA polymerase (Takara Bio). After 5 min at 94 °C, 35 cycles of amplification (94 °C for 30 s, 59 °C for 30 s and 72 °C for 1 min) were performed, followed by a 7-min extension at 72 °C. Products were resolved on a 1.5% agarose gel and purified (QIAquick PCR purification kit, QIAGEN). Nucleotide sequences were determined using the Big-Dye terminator cycle sequencing kit and the ABI Prism 310 Genetic Analyzer (Applied Biosystems Inc.). Sequences were determined and analyzed using the software package DNAStar.
Quantitative PCR
Viral RNA copies of MHHAV were quantified by real-time PCR. A forward primer (MHHAVF: 5′- GTCCTCTTTAAGGCACTCAT -3′), a reverse primer (MHHAVR:5′-TGGGTCAGTCCATCTGGCAAG-3′) and a probe (MHHAV Probe: 5′-FAM- CATCTTCATTTCCCTGGCTCTCACC-MGB -3′) were designed from sequences in the 5′ UTR region of the virus. The reaction condition involved 50 °C for 30 min, 95 °C for 10 min and 40 cycles of 95 °C for 15 s and 58 °C for 30 s.
Negative-strand MHHAV RNA testing
A tagged primer system was used to detect the negative-strand RNA. Negative-strand cDNA was generated with Tag MHHAV primer (5′ - CCTCCGCTGCCATCTGATTGCGTCCTCTTTAAGGCACTCAT - 3′, targeting 5′ UTR of MHHAV), performed at 65 °C for 5 min, 25 °C 10 min, 50 °C 50 min and 72 °C 15 min. Then quantitative PCR was established to quantify the negative-strand RNA, a forward primer (TagF: 5′- CCTCCCGATCATCTGGTTGC -3′), a reverse primer (UTRR:5′-TGGGTCAGTCCATCTGGCAAG -3′) and a probe (NProbe: 5′- FAM-CATCTTCATTTCCCTGGCTCTCACC-MGB -3′) were used. The reaction condition involved 50 °C for 2 min, 95 °C for 10 min and 40 cycles of 95 °C for 15 s and 58 °C for 30 s.
Recombination and phylogenetic analysis
Complete genomes of the known HAVs were downloaded. To detect potential combination, aligned sequences were analyzed by using the Boots canning method and the neighbor-joining algorithm was run with 100 pseudo replicates implemented in Simplot software. Phylogenetic tree were performed using nucleic acid sequences of VP1 and polyprotein by the Neighbor-joining method and subsequently subjected to bootstrap analysis with 1000 replicates. Tree figures were produced using MEGA software (version 5).
Evolutionary analysis
To precisely estimate MHHAV, phopivirus and HAV substitution, a Bayesian Markov chain Monte Carlo (MCMC) approach was implemented in the BEAST package (v 1.8.2, available from http://beast.bio.ed.ac.uk/downloads). The jModelTest software 2.1.7 was used to identify the optimal evolutionary, Akaike Information Criterion and hierarchical likelihood ratio test suggested that the GTR (general time reversible) + Γ (gamma distributed rate variation) model best fitted the sequences in this study. Different population dynamic models were used (constant size, exponential growth, logistic growth, expansion growth and Bayesian skyline). The MCMC analysis was performed with 50 million generations and sampled every 1000 generations with 10% burnin. The results were computed and analyzed using Tracer 1.6. The effective sample size values for the estimated parameters in the MCMC analysis were greater than 200. Statistical uncertainty in the data was reflected in the 95% highest probability density values (HPD).
RNA structure prediction of the 5′ UTR
The secondary structure of the 5′ UTR RNA of MHHAV was predicted using consecutive fragments of the complete nucleotide sequence of the 5′ UTR of MHHAV and a thermodynamic folding energy minimization algorithm with RNA structure software (version 5.3); the graph was integrated using RnaViz (version 2.0.3).
Structure prediction of MHHAV
Based on a previously solved HAV particle structure (PDB: 4QPG)14, the structure of MHHAV was modeled using Phyre49 (http://www.sbg.bio.ic.ac.uk/phyre2/html/page.cgi?id=index).
Virus purification
Stool samples were diluted to 20% suspensions in phosphate-buffered saline (PBS). Beads and chloroform were added to the suspensions, followed by 20 min centrifugation at 1,500 × g. Supernatant was collected and then subjected to a single ultracentrifugation step through a discontinuous sucrose/glycerol density gradient for HAV purification, as described previously28.
Electron microscopy
Fifty-microliter volumes of chloroform-purified MHHAV (1 × 107 copies/ml) in PBS were examined directly by negative staining with 1% phosphotungstic acid (pH 6.8). Chloroform-purified MHHAV (450 μl) was incubated with a 1:10 dilution of 50 μl MHHAV polyclonal antibody (BALB/c mice immunized subcutaneously with purified MHHAV) at 37 °C for 1 h. Following centrifugation at 23,000 rpm for 1 h, the sediment was resuspended in 50 μl PBS and the suspension subjected to negative staining. Grids were examined using a transmission electron microscope (TECNAI 12, FEI, Blackwood, NJ).
Statistical Analysis
The statistical significance of viral load means between the liver and other tissues was assessed using the Student’s t test and the statistical analyses were performed using SPSS 16.0.
Additional Information
How to cite this article: Yu, J.- et al. A novel hepatovirus identified in wild woodchuck Marmota himalayana. Sci. Rep. 6, 22361; doi: 10.1038/srep22361 (2016).
References
Koff, R. S. . Hepatitis A. Lancet 351, 1643–1649, doi: 10.1016/S0140-6736(98)01304-X (1998).
Hepatitis A vaccines. Wkly Epidemiol Rec 75, 38–44 (2000).
Jacobsen, K. H. & Wiersma, S. T. Hepatitis A virus seroprevalence by age and world region, 1990 and 2005. Vaccine 28, 6653–6657 (2010).
Totsuka, A. & Moritsugu, Y. Hepatitis A virus proteins. Intervirology 42, 63–68 (1999).
Weitz, M. & Siegl, G. Variation among hepatitis A virus strains. I. Genomic variation detected by T1 oligonucleotide mapping. Virus Research 4, 53–67 (1985).
Baroudy, B. M. et al. Sequence analysis of hepatitis A virus cDNA coding for capsid proteins and RNA polymerase. Proc Natl Acad Sci USA 82, 2143–2147 (1985).
Linemeyer, D. L. et al. Molecular cloning and partial sequencing of hepatitis A viral cDNA. J Virol 54, 247–255 (1985).
Najarian, R. et al. Primary structure and gene organization of human hepatitis A virus. Proc Natl Acad Sci USA 82, 2627–2631 (1985).
Ticehurst, J. R. et al. Molecular cloning and characterization of hepatitis A virus cDNA. Proc Natl Acad Sci USA 80, 5885–5889 (1983).
Costa-Mattioli, M. et al. Genetic variability of hepatitis A virus. J Gen Virol 84, 3191–3201 (2003).
Desbois, D. et al. Epidemiology and genetic characterization of hepatitis A virus genotype IIA. J Clin Microbiol 48, 3306–3315 (2010).
Nainan, O. V., Margolis, H. S., Robertson, B. H., Balayan, M. & Brinton, M. A. Sequence analysis of a new hepatitis A virus naturally infecting cynomolgus macaques (Macaca fascicularis). J Gen Virol 72 (Pt 7), 1685–1689 (1991).
Ping, L. H. & Lemon, S. M. Antigenic structure of human hepatitis A virus defined by analysis of escape mutants selected against murine monoclonal antibodies. J Virol 66, 2208–2216 (1992).
Wang, X. et al. Hepatitis A virus and the origins of picornaviruses. Nature 517, 85–88 (2015).
Vaughan, G. et al. Hepatitis A virus: host interactions, molecular epidemiology and evolution. Infect Genet Evol 21, 227–243 (2014).
Shapiro, C. N. & Margolis, H. S. Worldwide epidemiology of hepatitis A virus infection. J Hepatol 18 Suppl 2, S11–14 (1993).
Dotzauer, A. et al. Hepatitis A virus-specific immunoglobulin A mediates infection of hepatocytes with hepatitis A virus via the asialoglycoprotein receptor. J Virol 74, 10950–10957 (2000).
Dienstag, J. L., Popper, H. & Purcell, R. H. The pathology of viral hepatitis types A and B in chimpanzees. A comparison. Am J Pathol 85, 131–148 (1976).
Popper, H., Dienstag, J. L., Feinstone, S. M., Alter, H. J. & Purcell, R. H. The pathology of viral hepatitis in chimpanzees. Virchows Arch A Pathol Anat Histol 387, 91–106 (1980).
LeDuc, J. W. et al. Experimental infection of the New World owl monkey (Aotus trivirgatus) with hepatitis A virus. Infect Immun 40, 766–772 (1983).
Pinto, M. A. et al. Experimental hepatitis A virus (HAV) infection in Callithrix jacchus: early detection of HAV antigen and viral fate. Exp Toxicol Pathol 53, 413–420 (2002).
Baptista, M. L., Marchevsky, R. S., Oliveira, A. V., Yoshida, C. F. & Schatzmayr, H. G. Histopathological and immunohistochemical studies of hepatitis A virus infection in marmoset Callithrix jacchus. Exp Toxicol Pathol 45, 7–13 (1993).
Anthony, S. J. et al. Discovery of a Novel Hepatovirus (Phopivirus of Seals) Related to Human Hepatitis A Virus. MBio 6, doi: 10.1128/mBio.01180-15 (2015).
Walker, C. M., Feng, Z. & Lemon, S. M. Reassessing immune control of hepatitis A virus. Curr Opin Virol 11, 7–13 (2015).
EDWI N A. BROWN, S. P. D., ROBERT, W. JANSEN, STANLEY M. LEMON. The 5′ nontranslated region of hepatitis A virus RNA secondary structure and elements required for translation in vitro. J Virol, 65, 5828–5838 (1991).
EdwinA B, A. J. Z. In Vitro Characterization of an Internal Ribosomal Entry Site (IRES) Present within the 5′ Nontranslated Region of Hepatitis A Virus RNA: Comparison with the IRES of Encephalomyocarditis Virus. J Virol 68, 1066–1074 (1994).
Deinhardt, F. Prevention of viral hepatitis A: past, present and future. Vaccine 10 Suppl 1, S10–14 (1992).
Bishop, N. E., Hugo, D. L., Borovec, S. V. & Anderson, D. A. Rapid and efficient purification of hepatitis A virus from cell culture. J Virol Methods 47, 203–216 (1994).
Stanway, G. et al. Molecular and biological characteristics of echovirus 22, a representative of a new picornavirus group. J Virol 68, 8232–8238 (1994).
Hynes, R. O. Integrins: versatility, modulation and signaling in cell adhesion. Cell 69, 11–25 (1992).
Sanchez, G., Bosch, A. & Pinto, R. M. Genome variability and capsid structural constraints of hepatitis a virus. J Virol 77, 452–459 (2003).
Nainan, O. V., Brinton, M. A. & Margolis, H. S. Identification of amino acids located in the antibody binding sites of human hepatitis A virus. Virology 191, 984–987 (1992).
Redondo, N. et al. Translation directed by hepatitis A virus IRES in the absence of active eIF4F complex and eIF2. PLoS One 7, e52065, doi: 10.1371/journal.pone.0052065 (2012).
Pacheco, A. & Martinez-Salas, E. Insights into the biology of IRES elements through riboproteomic approaches. J Biomed Biotechnol 2010, 458927, doi: 10.1155/2010/458927 (2010).
Yang, Y., Yi, M., Evans, D. J., Simmonds, P. & Lemon, S. M. Identification of a conserved RNA replication element (cre) within the 3Dpol-coding sequence of hepatoviruses. J Virol 82, 10118–10128 (2008).
de Paula, V. S. et al. Kinetics of hepatitis A virus replication in vivo and in vitro using negative-strand quantitative PCR. Eur J Clin Microbiol Infect Dis 28, 1167–1176 (2009).
Song, Y. J. et al. Experimental evidence of hepatitis A virus infection in pigs. J Med Virol, doi: 10.1002/jmv.24386 (2015).
Hornei, B. et al. Experimental hepatitis A virus infection in guinea pigs. J Med Virol 64, 402–409 (2001).
Feigelstock, D., Thompson, P., Mattoo, P., Zhang, Y. & Kaplan, G. G. The human homolog of HAVcr-1 codes for a hepatitis A virus cellular receptor. J Virol 72, 6621–6628 (1998).
Moratorio, G. et al. Bayesian coalescent inference of hepatitis A virus populations: evolutionary rates and patterns. J Gen Virol 88, 3039–3042 (2007).
Brown, B. A., Oberste, M. S., Alexander, J. P. Jr., Kennett, M. L. & Pallansch, M. A. Molecular epidemiology and evolution of enterovirus 71 strains isolated from 1970 to 1998. J Virol 73, 9969–9975 (1999).
Gavrilin, G. V., Cherkasova, E. A., Lipskaya, G. Y., Kew, O. M. & Agol, V. I. Evolution of circulating wild poliovirus and of vaccine-derived poliovirus in an immunodeficient patient: a unifying model. J Virol 74, 7381–7390 (2000).
Freitas, N. et al. Hepatitis delta virus infects the cells of hepadnavirus-induced hepatocellular carcinoma in woodchucks. Hepatology 56, 76–85 (2012).
Moreno-Cugnon, L. et al. Characterization and genotyping of the DRB1 gene of the major histocompatibility complex (MHC) in the Marmota monax, animal model of hepatitis B. Mol Immunol 63, 505–512 (2015).
Yamazoe, M., Nakai, S., Ogasawara, N. & Yoshikawa, H. Integration of woodchuck hepatitis virus (WHV) DNA at two chromosomal sites (Vk and gag-like) in a hepatocellular carcinoma. Gene 100, 139–146 (1991).
Summers, J., Smolec, J. M. & Snyder, R. A virus similar to human hepatitis B virus associated with hepatitis and hepatoma in woodchucks. Proc Natl Acad Sci USA 75, 4533–4537 (1978).
Wong, D. C., Shih, J. W., Purcell, R. H., Gerin, J. L. & London, W. T. Natural and experimental infection of woodchucks with woodchuck hepatitis virus, as measured by new, specific assays for woodchuck surface antigen and antibody. J Clin Microbiol 15, 484–490 (1982).
Finkbeiner, S. R. et al. Metagenomic analysis of human diarrhea: viral detection and discovery. PLoS Pathog 4, e1000011, doi: 10.1371/journal.ppat.1000011 (2008).
Kelley, L. A., Mezulis, S., Yates, C. M., Wass, M. N. & Sternberg, M. J. E. The Phyre2 web portal for protein modeling, prediction and analysis. Nat. Protocols 10, 845–858 (2015).
Acknowledgements
We thank David Wang and Guo-yan Zhao from the Department of Pathology and Immunology, Washington University School of Medicine for bioinformatics analysis; Jing-dong Song, Xiao-hui Zou and Jian-guo Qu from National Institute for Viral Disease Control and Prevention for electron microscopy; and Xiao-zhong Peng and Fei Ye from Institute of Basic Medical Sciences, Chinese Academy of Medical Sciences and Peking Union Medical College for RNA secondary structure prediction. This work was supported by the National Natural Science Foundation of China (Grant No. 81290345) and by the China Mega-Project for Infectious Disease (Grant No. 2013ZX10004).
Author information
Authors and Affiliations
Contributions
Z.J.D., J.G.X. and J.M.Y. designed research; L.L.L., J.M.Y., C.Y.Z., Y.Y.A., H.C.G., Z.P.X., G.C.X. and L.L.P. performed research; J.M.Y., L.L.L., X.M.S., Z.J.D. and W.I.L. analyzed data; S.L. collected the sample and contributed new reagents; J.M.Y., Z.J.D. and W.I.L. wrote the paper. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Yu, Jm., Li, Ll., Zhang, Cy. et al. A novel hepatovirus identified in wild woodchuck Marmota himalayana. Sci Rep 6, 22361 (2016). https://doi.org/10.1038/srep22361
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep22361
This article is cited by
-
Hepatitis A virus infection
Nature Reviews Disease Primers (2023)
-
A novel picornavirus identified in wild Macaca mulatta in China
Archives of Virology (2020)
-
Marmota himalayana in the Qinghai–Tibetan plateau as a special host for bi-segmented and unsegmented picobirnaviruses
Emerging Microbes & Infections (2018)
-
A new subtype of eastern tick-borne encephalitis virus discovered in Qinghai-Tibet Plateau, China
Emerging Microbes & Infections (2018)
-
Genomic Characterization of a Novel Hepatovirus from Great Roundleaf Bats in China
Virologica Sinica (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.