Abstract
The MADS-box transcription factors play essential roles in various processes of plant growth and development. In the present study, phylogenetic analysis of 142 apple MADS-box proteins with that of other dicotyledonous species identified six putative Dormancy-Associated MADS-box (DAM) and four putative Flowering Locus C-like (FLC-like) proteins. In order to study the expression of apple MADS-box genes, RNA-seq analysis of 3 apical and 5 spur bud stages during dormancy, 6 flower stages and 7 fruit development stages was performed. The dramatic reduction in expression of two MdDAMs, MdMADS063 and MdMADS125 and two MdFLC-like genes, MdMADS135 and MdMADS136 during dormancy release suggests their role as flowering-repressors in apple. Apple orthologs of Arabidopsis genes, FLOWERING LOCUS T, FRIGIDA, SUPPRESSOR OF OVEREXPRESSION OF CONSTANS 1 and LEAFY exhibit similar expression patterns as reported in Arabidopsis, suggesting functional conservation in floral signal integration and meristem determination pathways. Gene ontology enrichment analysis of predicted targets of DAM revealed their involvement in regulation of reproductive processes and meristematic activities, indicating functional conservation of SVP orthologs (DAM) in apple. This study provides valuable insights into the functions of MADS-box proteins during apple phenology, which may help in devising strategies to improve important traits in apple.
Similar content being viewed by others
Introduction
In plants, MADS-box transcription factors (TFs) play variety of roles in various biological processes during growth and development. MADS-box gene family is one of the best studied gene families in plants and contributes majority of component to the well-known ABCDE model of flowering that depicts their roles in floral organ development. However, MADS-box genes were also found to be expressed in vegetative tissues, ovule, embryo, root, trichome and fruit, suggesting their diverse roles in plant development1.The MADS-box proteins contain a highly conserved 58 amino acid long DNA binding MADS domain. The term MADS domain was derived from four members of the family viz. MINICHROMOSOME MAINTENANCE1 (MCM1) from Saccharomyces cerevisiae, AGAMOUS (AG) from Arabidopsis, DEFICIENS (DEF) from Antirrhinum majus and SERUM RESPONSE FACTOR (SRF) from Homo sapiens1. Most of the studies on functional characterization of MADS-box genes have been done in model plant, Arabidopsis thaliana. However, studies on other flowering plant species like, Antirrhinum majus2, Solanum lycopersicum3 and Petunia hybrida4 also contributed significantly to reveal the diverse functions of MADS-box TFs. Although, MADS box TFs were originally found to be involved in determining floral organ identity, several recent studies showed their various important roles throughout the plant life cycle1.
In plants of Rosaceae family, particularly in apple (Malus x domestica), winter dormancy affects the flowering time by regulating dormancy release. The winter dormancy breaks only after the acquisition of sufficient chilling hours, which is genotype dependent5,6,7. In flowering plants, transition from vegetative phase to reproductive phase is a critical developmental process, which involves numerous molecular events. Role of many transcription factors (TFs) including those belonging to the MADS-box TF family has been shown in transition from vegetative to reproductive phase. One of these MADS-box TFs controlling flowering time is SHORT VEGETATIVE PHASE (SVP). The SVP mutant of Arabidopsis was shown to flower very early as compared to wild type, suggesting its role as a repressor of flowering8. The SVP was shown to repress flowering by regulating the expression of three flowering promoting genes namely; FLOWERING LOCUS T (FT ), TWIN SISTER OF FT (TSF ), and SUPPRESSOR OF OVEREXPRESSION OF CONSTANS 1 (SOC1). In Arabidopsis, the time of flowering in response to ambient temperature is also governed by SVP through repression of FT expression9,10. Another MADS-box gene, FLOWERING LOCUS C (FLC) was also shown to act as a key flowering repressor, which interacts with SVP and inhibit the flowering by repressing the expression of FT and SOC1 in different tissues1,11. The expression of SVP and FLC decreases gradually as plant becomes ready to flower12.
The dormancy insensitive ever growing peach mutant, which has mutation in EVG (EVERGREEN) locus, is the breakthrough in the dormancy studies in plants belonging to Rosaceae family, where deletion of tandem repeats of six orthologs of SVP was responsible for evergreen phenotype13. Therefore, these MADS-box genes were named as Dormancy Associated MADS-box (DAM) genes. The presence of homologs of DAM genes has also been reported in other Rosaceae plants like, raspberry, Japanese apricot and Japanese pears14,15,16. The expression pattern of DAM genes in various plants indicated role of low temperature in regulating their expression during dormancy period7,17,18. The ectopic expression of Japanese apricot DAM6 in poplar was able to induce growth cessation of bud in transgenic popular18. These studies clearly showed that DAM genes play an important role in vegetative to reproductive transition.
Despite the vast functions of MADS box TFs in flowering plants, only few apple MADS-box TFs have been characterized, so far19,20,21,22. However recently, identification and expression analysis of apple MADS-box gene family in root, stem, leaf, flower and five stages of fruit development was performed23. Whereas in present study, we show that comparative phylogenetic analysis of apple MADS-box family proteins with that of other dicotyledonous species identified six putative DAM genes and four putative FLC-like genes in apple. Moreover, expression profiling of apple MADS-box family was performed during the key phenological events, including various stages of dormancy, dormancy release, flower and fruit development using RNA-seq approach. In addition, we have also identified putative downstream targets for DAM TFs in apple genome using computational approaches. Our analysis provides a basic framework to uncover the biological role of MADS-box TFs in various developmental processes during cyclic phenological events in apple.
Results and Discussion
Annotation of MADS-box family in apple
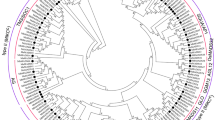
The “hmmsearch” using HMM profile of MADS-box domain and PfamScan, with stringent e-value 1e-04, resulted in identification of 142 putative MADS-box proteins in apple (Supplementary Table S1). The number of proteins in apple MADS-box family was found to be second highest after soybean with 163 members24. However, a recent study by Tian et al.23 reported 146 proteins in apple MADS-box family. This slight variation in the number of MADS-box proteins may be due to the difference in approach used to identify the MADS-box proteins. The comparison of MADS-box proteins identified in the present and previous studies is listed in Supplementary Table S2. Phylogenetic tree made using full-length MADS-box proteins of Arabidopsis and apple showed the presence of previously defined sub-families, Mα, Mβ and Mγ for type-I and MIKCC and MIKC* for type-II MdMADS proteins (Fig. 1). The three sub-families Mα, Mβ and Mγ contain 24, 12 and 20 proteins, respectively. Out of 86 type-II MdMADS, 79 were classified as MIKCC and 7 as MIKC*. In addition, two sub-groups named MIKCC-I and MIKCC-II each containing 9 MdMADS were found to form the apple-specific clade with no Arabidopsis ortholog. The distribution of MdMADS genes on 17 apple chromosomes was found to be non-random showing chromosomal bias with highest (13) MdMADS genes on chromosome 5 and least number (1) on chromosome 3 (Supplementary Fig. S1). Out of 142 MdMADS, 75 (approximately 53%) genes were found to be present in 29 clusters distributed on 15 chromosomes. Interestingly, 17 out of 29 gene clusters containing a total of 45 MdMADS genes were found to be solely comprised of MIKCC genes. Duplication analysis of MdMADS genes revealed the presence of 26.8%, 23.2%, 18.3% and 30.3% of segmental, tandem, dispersed and proximal duplication, respectively. In total, 18 type-I and 38 type-II MdMADS genes were found to be tandem (Supplementary Fig. S2) or segmental (Supplementary Fig. S3) duplicated. Therefore, expansion of type-II MdMADS genes might be due to the recent whole genome duplication during evolution25.
The multiple sequence alignment and construction of phylogenetic tree were performed with MEGA6.06 using neighbor-joining method with 1000 bootstrap replicates. The proteins were clustered and divided into five distinct sub-families. The MIKCC sub-family was further divided into 14 sub-groups. The two sub-groups MIKCC-I and MIKCC-II seem to be apple specific.
Phylogenetic relationship of MdMADS family with that of other plant species
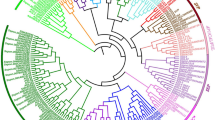
To examine the phylogenetic relationship of MdMADS proteins with that of Arabidopsis, peach, poplar, cucumber and grape, phylogentic tree was made that divided all these MADS-box proteins in to 16 distinct clades (A to P; Fig. 2). Interestingly, in all the clades, MADS-box proteins of members of Rosaceae family (apple and peach) were clustered in separate sub-clades. The members of each MADS-box protein sub-family (Mα, Mβ, Mγ, MIKCC and MIKC*) of different plant species were clustered in the same clade. For instance, members of Mα, Mγ and MIKC* sub-families were clustered in clade M, N and A, respectively. While, members of Mβ sub-family were clustered in clade O and P, and the remaining clades contain members of MIKCC sub-family (Fig. 2).
Orthologs of SVP were reported to play important roles in dormancy establishment and its maintenance and were characterized as Dormancy Associated MADS-box (DAM) TFs in poplar26, peach13 and leafy spurge17. From the phylogenetic analysis, the expansion of SVP clade was observed in apple with six orthologs. In previous studies, the tandem duplication of SVP genes was found to be responsible for their expansion in apricot27 and peach28. However, none of the apple orthologs of SVP was found to be tandem duplicated, suggesting that tandem duplication did not play any role in their expansion. In apple, four genes (MDP0000322567, MDP0000259294, MDP0000209705 and MDP0000527190) were previously annotated as DAM in Genome database for Rosaceae (https://www.rosaceae.org). However, out of these four DAM genes, only three were found to exhibit dormancy associated expression, in transgenic apple over-expressing peach CBF gene, which were then annotated as DAM1 (MDP0000322567), DAM2 (MDP0000259294) and DAM3 (MDP0000209705)29. However, the phylogenetic analysis of MdMADS with that of other dicotyledonous species resulted in identification of six putative DAM proteins in apple (Fig. 2). Although, only two MdMADS (MdMADS024; MDP0000209705 and MdMADS088: MDP0000233948) were found to be close orthologs of Arabidopsis SVP while, the remaining four (MdMADS061; MDP0000259294, MdMADS063; MDP0000527190, MdMADS124; MDP0000255146 and MdMADS125; MDP0000322567) were clustered in sub-clade along with the peach DAM proteins (Supplementary Fig. S4), therefore these six apple proteins were designated as MdDAMs. This observation suggests that DAMs are conserved in Rosaceae family during the course of evolution indicating the functional conservation of DAM proteins in peach and apple.
Similarly, the FLC was reported to be involved in vernalization in Arabidopsis to initiate flowering1. Phylogenetic analysis of MdMADS with their Arabidopsis counterparts did not identify any ortholog of Arabidopsis FLC and MAF in apple (Fig. 1). Similarly, no ortholog of FLC has been reported in apricot27 and cucumber30. However, FLC-like genes have been reported in other plants like poplar26, peach28 and grapevine31. Interestingly, the phylogenetic analysis of MdMADS with that of other dicotyledonous species indicates that four MdMADS (MdMADS053, MdMADS131, MdMADS135 and MdMADS136) were clustered in the clade H along with FLC-like proteins of peach and grape (PpeMADS08, VvFLC2); that suggests the presence of putative FLC-like MdMADS in apple (Fig. 2). Therefore, these four proteins were designated as FLC-like MdMADS. A recent transcriptome study in apple reported the presence of putative FLC-like gene (MDP0000207984), however it was not functionally characterized32. In the present study, that gene was not identified as MdMADS due to the absence of MADS domain.
Analysis of expression pattern of MdMADS genes in various developmental stages during apple phenological cycle
To examine the expression of MdMADS genes in various developmental stages of different tissue types, RNA-seq analysis of 21 samples representing 3 stages of apical bud, 5 stages of spur bud, 6 stages of flower development and 7 stages of fruit development (Supplementary Table S3) was performed. The paired-end sequencing generated 264,658,274 raw reads of which 17,227,734 low quality reads were filtered out. The de novo assembly of 247,430,540 high quality reads resulted in 64,669 contigs. Out of 142 MdMADS, 63 were found to be expressed in various tissues, while rest of the 79 genes exhibit either very low or no expression in any of the tissues.
Type-I MADS genes
In case of type-I MdMADS genes, only 11 out of 57 were found to be expressed in RNA seq data. The 3 genes MdMADS102, MdMADS110 and MdMADS137 of Mβ sub-family and two genes MdMADS077 and MdMADS103 of Mα sub-family were found to be ubiquitously expressed in all the samples analyzed (Fig. 3A). Recent studies showed involvement of type-I MADS-box genes in plant reproduction processes25,33,34. A member of Mγ sub-family in Arabidopsis, AGL80, which interacts with AGL61 of Mα sub-family was found to be involved in female gametophyte development1. In the present study, ortholog of AGL61, MdMADS103 was found to exhibit expression in all the tissues, while no close ortholog of AGL80 was found to be expressed. In addition, the genes of Mα and Mγ showed variable expression during fruit development (seed development along with the fruit development). Similarly genes of these sub-families were also found to be expressed during seed development in Arabidopsis35.
Type-II MADS genes
MIKC*
The expression of type-II MADS genes was found to be high in most of the tissue types, suggesting their role in diverse developmental processes in apple (Fig. 3B). The MIKC* genes in Arabidopsis have been found to control the pollen development36. Similarly in peach, the expression of PpeMADS20, PpeMADS36 and PpeMADS55 was found to be high in pollens during flower development28. The close orthologs of these genes in apple, MdMADS099, MdMADS48 and MdMADS59 also exhibit higher expression at later stages (FP and FF) of flower development (Fig. 3B). In contrary, Tian et al.23 did not find expression of these genes in flower tissue. This may be due to the difference in flower stage at which sample was collected.
Floral identity genes
Previous studies have shown the role of MIKCC genes as flower homeotic genes in Arabidopsis and other plants leading to establishment of ABCDE model of floral organ identity1. In Arabidopsis, the genes of AP1 clade, AP1 and CAL play role in petal and sepal identity with additional role of FUL in fruit development1. In the present study, close orthologs of AP1 and CAL in apple, MdMADS097 and MdMADS120 were found to exhibit high expression during flower and fruit development. Similar expression pattern of these genes was also observed by Tian et al.23. Previously, Cevik et al.22 reported the involvement of FUL-like gene in apple fruit development. Similarly in peach, FUL ortholog, PpeMADS37 was found to be expressed in fruit tissue28. Although, MdMADS050 and MdMADS113, close orthologs of FUL, were found to be expressed during initial stages of fruit development (FS2) in present study (Fig. 3B), while no expression of MdMADS050 was reported by Tian et al.23 in fruit tissue. Expression of genes of AP1 clade in flower as well as in fruit tissues might be due to fact that apple is not a true fruit and develops from the enlargement of basal part of sepal.
In Arabidopsis, the identity of petal and stamen is defined by AP3 and PI37. Similarly, the high expression of genes of AP3 clade, PpeMADS56 in peach28 and PmMADS12, PmMADS13 and PmMADS29 in apricot27 was also observed in flower tissue. In present study, two genes, MdMADS013 and MdMADS044 of AP3/PI clade were found to exhibit high expression during flower initiation and development (Fig. 3B), however, Tian et al.23 found expression of only MdMADS013 in flower tissue. The similar spatial expression of AP3/PI clade genes in apple, peach and apricot indicates their functional conservation in higher woody plants. Moreover, the Arabidopsis genes of AG clade perform C function and D function of homeotic genes. In apple, MdMADS027 a close ortholog of AG was found to be expressed in flower and initial stages of fruit development, while MdMADS075 an ortholog of SHP2 has expression in flower and fruit development stages (Fig. 3B), the similar expression patterns of these genes were observed by Tian et al.23 in flower and fruit tissues. Similar to apple, the members of AG clade were also observed to have high expression during fruit development in peach28 and apricot27. These finding suggest the possible role of AG class genes in development of pome and stone fruits.
Other MIKCC genes
Role of SEP gene orthologs has been established in tomato38 (TM29) and strawberry39 (FaMADS9) fruit development. While in Arabidopsis, the SEP genes (E class genes) are involved in multimeric protein complex formation with other homeotic genes and specify the stamen, carpel and petal identity40. In peach, the expression of four SEP genes was found restricted to fruit tissues28. However in apricot, three orthologs of SEP have higher expression in fruit and flower tissues27. Similarly, in the present study, six SEP orthologs (MdMADS049, MdMADS070, MdMADS112, MdMADS121, MdMADS123 and MdMADS127) have high expression in all the developmental stages from flower initiation to fruit development (Fig. 3B). A previous study in apple, also observed the expression of SEP ortholog during flower and fruit development19,23. In Arabidopsis, the functions of SEP genes were found to be largely similar to the functions of AP1 genes41. Interestingly, in the present study two orthologs of AP1 (MdMADS097 and MdMADS120) also have similar expression pattern to SEP orthologs (Fig. 3B), which indicates the conservation of functional redundancy of SEP orthologs in apple also.
The MIKCC genes are also known to be involved in processes other than the flower organ identity. The Arabidopsis AGL15 and AGL18 genes are expressed in embryo and act as additional flowering repressor in a redundant manner42. In contrast, the SOC1 gene regulates flowering time by preventing the premature differentiation of flower meristem and act as activator of flowering in vernalization and GA dependent pathway in Arabidopsis43. In apple, three members of AGL15 clade, MdMADS051, MdMADS108 and MdMADS109 were found to exhibit expression at later stages of flower development (time of embryo development), while in all other tissues their expression was negligible. The expression of seven members of SOC1 clade was high during the bud development stages, however MdMADS08 and MdMADS014 exhibit higher expression in flowering stages also (Fig. 3B), similarly, these genes were observed to be expressed in flower tissue by Tian et al.23. The AGL12, a sister gene of AG clade regulates root meristem growth in addition to promoting flowering in Arabidopsis44. In apple, AGL12 ortholog, MdMADS086 and AG ortholog MdMADS075 exhibit similar expression in flower and fruit tissues (Fig. 3B). Therefore, the AGL12 ortholog in apple might also have role in regulation of flowering. However, members of TM8 clade are not present in Arabidopsis28, similar to other dicotyledonous species, the interspecies phylogenetic analysis shows the presence of two members of TM8 clade in apple. The MdMADS045, a member of TM8 has high expression in flower development stages. While another TM8 member, MdMADS111 showed low expression during flower development (Fig. 3B). The similar expression of these genes was also observed by Tian et al.23, however, these genes were grouped into SOC1 clade based on phylogenetic analysis with Arabidopsis which lack members of TM8 clade. The low expression of genes belonging to TM8 clade in flower tissue has also been reported in grape31 and peach28. In Arabidopsis, the TT16 which is mainly expressed in ovule was shown to be involved in flavonoid biosynthesis in the seed coat45. In apple, we identified two members of TT16 clade (MdMADS031 and MdMADS139), however only MdMADS139 was expressed highly during fruit development.
FLC-like and DAM MADS-box TFs
The role of SVP and FLC as flowering time regulators has been extensively studied in Arabidopsis and a model for flowering time regulatory network has been hypothesized that depicts the inhibitory effect of FLC and SVP on flowering through different pathways1,43. In order to elucidate the functional conservation of this hypothetical model in apple, the expression analysis of SVP orthologs (DAM), FLC-like genes and other genes associated with floral regulation was performed. In Arabidopsis, FLC act as an important flowering repressor and its expression is epigenetically downregulated after prolonged cold exposure during vernalization process46. Moreover interaction between FLC and SVP has been found to repress the expression of FT gene, a flower promoting gene11. In addition, the FLC has also been suggested to be involved in flower development and juvenile to adult phase transition46. In apple, the putative FLC-like genes MdMADS135 and MdMADS136, identified through phylogenetic analysis (Fig. 2), exhibit increased expression with chilling acquisition during winter dormancy, while reduction in expression at the time of dormancy release (Figs 3B and 4A). Similar expression patterns of FLC in Arabidopsis46 and FLC-like gene in poplar47 have been reported. Therefore, we suggest that MdMADS135 and MdMADS136 may have similar role as negative regulator of flowering as defined in Arabidopsis and poplar. While another apple FLC-like gene, MdMADS053 was expressed in most of the tissue types. The similar expression of FLC-like gene, PpeMADS8 was also observed in peach28. In case of apple, FLC-like genes have shown high sequence divergence than that of Arabidopsis, which suggest their functional divergence. However, apple and poplar FLC-like genes exhibit dormancy associated expression. Moreover, FLC downregulates the expression of flower promoting genes SOC1 and FT gene in Arabidopsis1,11. In present study, the reciprocal expression of FLC-like genes and orthologs of SOC1 and FT was also observed during dormancy release and flower initiation (Fig. 4B,E). In addition, the expression of orthologs of FRIGIDA (FRI), an upstream positive regulator of FLC43, was found to be high during dormancy and reduced gradually with concomitant reduction in expression of FLC-like genes in apple (Fig. 4A). The similar expression patterns of Arabidopsis FLC and FLC-like genes of other dicotyledonous species indicate their functional conservation.
The SVP mutant of Arabidopsis flower earlier than the wild types, indicating its role in controlling vegetative to reproductive transition8. In peach, six tandemly duplicated orthologs of SVP, DORMANCY ASSOCIATED MADS-BOX (DAM) genes are involved in seasonal floral bud dormancy13. The phylogenetic analysis of MdMADS proteins with that of Arabidopsis and other dicotyledonous species also shows the presence of six SVP orthologs, DAM in apple (Fig. 2). The RNA-seq data showed that only four putative MdDAM genes (MdMADS024, MdMADS063, MdMADS088 and MdMADS125) were found to be expressed of which MdMADS063 and MdMADS125 showed reduction in expression along with the dormancy release (Fig. 3C). Similar to FLC, the SVP also acts as negative regulator of SOC1 and FT in Arabidopsis, therefore the expression of SOC1 and FT orthologs in apple was analyzed in relation to DAMs and FLC-like genes (Fig. 3G). The expression of MdDAM and orthologs of SOC1 and FT in apple was found to be reciprocal during dormancy to active phase transition (Fig. 4C,D). Similarly, reciprocal expression of DAM and FT genes was also reported in leafy spurge17. Repressive effect of PmDAM6 on expression of FT2 was also shown in transgenic poplar over-expressing PmDAM618. In addition, expression of DAM was induced with dormancy inducing signals and concomitantly decreased with dormancy release in raspberry14, pear16, apricot18,27, peach7 and leafy spurge17. Therefore, expression pattern of MdDAMs indicates the functional conservation of SVP orthologs in apple and other dicotyledonous species.
Since, the dormancy induction and its release in apple is controlled by low temperature5, the presence of low temperature responsive elements (C-repeat binding factor and low temperature responsive element) was also analyzed in promoter sequence of MdDAMs, which revealed presence of low temperature response elements (LTRE; Supplementary Table S4). The MdMADS063 and MdMADS125 have higher number of LTRE (two and seven, respectively) with in the 1.5Kb upstream region of transcription start site, which indicates their cold mediated regulation during dormancy period in apple. Similarly, presence of CBF in promoter regions of DAMs and their low temperature responsive expression was also reported in other plants5,17,18. Moreover, the expression of MdMADS063 and MdMADS125 was found to be induced in fruit tissues after which plant again undergoes next cycle of winter dormancy. Conclusively, the seasonal expression patterns of DAM genes in apple, peach, apricot, leafy spurge and poplar indicate that the orthologs of SVP have evolved to acquire the additional functions during seasonal dormancy in higher plant species beside their role in vegetative to reproductive transition and flower development, as reported in Arabidopsis. These results indicate the importance of FLC-like and MdDAM genes in regulating the dormancy. Therefore, we hypothesise that, FLC-like and MdDAM genes are the key regulators during dormant to active phase transition and their functions are largely conserved among higher dicotyledonous species. In addition, the expression of LEAFY (LFY), a downstream target of SOC1, was found to be increased along with SOC1 expression during dormancy release (Fig. 4I). Moreover, the photoperiodic pathway of flower initiation in Arabidopsis was found to be regulated by CONSTANS (CO), a non-MADS gene, however the expression pattern of CO orthologs in apple was not observed to be in accordance (Fig. 4H). This indicates the dormancy release in apple might not be under the control of photoperiod, which was also reported by Heide and Prestrud5. On the basis of present study and previous supporting literature, we hypothesized that the roles of apple orthologs of FLC, SVP, FRI, FT, SOC1 and LFY in dormancy and flower regulation are largely conserved, whereas CO does not seem to play role in photoperiodic floral signal integration (Fig. 5). However, functional characterization of these genes will reinforce the proposed hypothesis of winter dormancy and flowering time regulation.
The FPKM based expression of MdMADS genes obtained through RNA-seq was validated using qRT-PCR. The ten genes were randomly selected from various classes of type-II genes including FLC-like (MdMADS135), DAM (MdMADS024, MdMADS125), orthologs of SOC1 (MdMADS014), TM8 (MdMADS111), AP3/PI (MdMADS044 and MdMADS050), AGL15 (MdMADS051) SEP (MdMADS123) and gene of MIKCC-II clade (MdMADS126). Out of ten MdMADS, expression of six genes was observed in one or more of the tissues analyzed in present study. The relative expression analysis of DAM genes, FLC-like, orthologs of SOC1, TM8, SEP and AP3/PI was observed to be in accordance with FPKM based expression values (Supplementary Fig. S5).
In summary, the RNA-seq analysis revealed that expression of several MdMADS genes was modulated during dormancy to fruit ripening (Fig. 6). For instance, change in expression of DAM, FLC-like and orthologs of SOC1 appear to be necessary from dormant to active phase transition, while the orthologs of AG, SEP, AP3/PI, AGL12 and TM8 are associated with flower development and fruit initiation. In addition to these genes, ortholog of TT16 is also involved in fruit development. Thus, it is apparent that transitions of various developmental stages during apple phenology are associated with change in expression of MdMADS genes.
Identification and network analysis of putative targets of orthologs of SVP in apple
In order to further evaluate the functional conservation of SVP orthologs in apple, a computational approach was used to determine the putative targets of MdDAM. Recently, downstream targets of Arabidopsis SVP protein were identified using Chip-seq approach48; using the same Chip-seq data, we identified 214 and 261 SVP binding motifs in vegetative and reproductive phase data of Arabidopsis, respectively. In order to identify the putative targets of MdDAM, the pattern search of these identified motifs against apple genome revealed the presence of 22 and 23 unique motifs of reproductive phase and vegetative phase, respectively (Supplementary Table S5). The protein orthologs of predicted apple target genes were searched in Arabidopsis, and these Arabidopsis proteins were further considered to build the respective protein-protein interaction (PPI) networks viz., vegetative and reproductive phase PPI networks.
Analysis of topology of interaction network of putative downstream targets of MdDAM protein revealed the presence of hub proteins. Compared to the PPI network of vegetative phase, the node degree distribution (probability distribution of degrees) of reproductive phase network approximated a power-law (Supplementary Fig. S6). The degree distribution results indicate the presence of few highly connected proteins in reproductive phase (Fig. 7), while isolated entities (not as a giant cluster) are present in vegetative phase network; therefore, we limited our analysis only to reproductive phase PPI network.
The circles represent proteins, and the lines indicate an interaction between proteins. The highlighted Arabidopsis proteins represent five hub proteins viz, AT1G66410, AT5G23290, AT5G19820, AT3G19980 and AT4G14960. The interacting partners of each of the hub protein are also represented as zoomed-in view in squares.
A total of five hubs (Supplementary Table S6) were obtained, which constituted ~4% of total proteins in largest connected component (Fig. 7). Further, the clusters of interacting proteins were annotated for their ontological characterization using agriGO49, implementing hyper-geometric test. The GO terms with false discovery rate (FDR) corrected p-values less than 0.05 were considered as significantly enriched for biological process and molecular function categories. In Arabidopsis, Gregis et al.48 reported that SVP was found to be involved in regulation of reproduction and flowering related pathways. In the present study, the similar inference was also deduced from gene ontology analysis, where majority of putative interactors were found to be involved in major signalling pathways: hormone and other stimulus, negative regulation of flower development and reproductive process, regulation of root meristem and seed development; that suggests the regulation of flowering control and other meristematic activities by SVP ortholog in apple (DAM) also (Supplementary Table S7). In addition, this analysis provides new insights towards the functional conservation and importance of SVP orthologs (DAM) in woody plants.
Conclusions
Our analysis identified MADS-box TF family in apple genome, comprising of 142 proteins, which is second highest in number after soybean. The genome organization of apple MdMADS genes revealed that large number of MdMADS genes are present in clusters and significant expansion has taken place during recent whole genome duplication with equal contribution of segmental and tandem duplication. The phylogenetic analysis of MdMADS proteins with that of Arabidopsis and other dicotyledonous species resulted in identification of six DAM and four FLC-like proteins in apple. Using high-throughput Illumina RNA-seq approach, the expression of 63 MdMADS genes was detected in various developmental stages of different apple tissues. The expression profile of DAM and FLC-like genes and other genes (FRI, FT, LFY, SOC1 and CO) was discussed in the light of previous supporting literature that helped us to assign putative functions to these genes during dormancy and flower time regulation in apple. In addition, computational approach was used to evaluate the functional conservation of SVP orthologs (DAM) in apple by identification and characterization of their putative downstream targets. Our findings are largely consistent with the previous studies in Arabidopsis. Overall, the computational and experimental approaches illustrated the importance of various members of MADS-box TFs family in diverse developmental processes throughout the phenological events in apple.
Methods
Data retrieval
The whole apple genome sequence data set used for the identification and annotation of MADS box proteins was downloaded from Phytozome (www.phytozome.net) and Apple GFDB (http://www.applegene.org). The MADS-box protein sequences of Arabidopsis were retrieved from TAIR10 (http://www.arabidopsis.org), while poplar, peach, and grape MADS-box protein sequences were retrieved from Phytozome v10.3 (http://www.phytozome.net). The cucumber MADS-box protein sequences were downloaded from Cucumber Genome Database (http://cucumber.genomics.org).
Identification of MADS-box gene family in apple
The Hidden Markov Model (HMM) profile of MADS-box domain (PF00319) was retrieved from pfam v27.050 and employed to identify the putative MADS-box proteins in apple genome. For this, “hmmsearch” of HMMER V3.0 was performed, with e-value 1e-04. The identified putative MADS proteins were then subjected to PfamScan program to confirm the presence of MADS domain and to identify additional domains with parameters: -e_seq 1e-04 -e_dom 1e-04 -clan_overlap. The orthologs of apple MADS-box were identified in Arabidopsis thaliana using BLASTP search against Arabidopsis proteome from TAIR 10 (http://www.arabidopsis.org). The putative MADS-box proteins were further classified into regular and non-regular proteins based on the similarity in BLASTP51 against non-redundant database with cut-off criterion of >50% similarity and >50% query coverage.
Chromosomal mapping, nomenclature and gene duplication of MADS-box family
The coordinates of gene location on chromosome were extracted from general feature format (gff) file from phytozome and plotted on all 17 chromosomes using MapInspect program (http://www.plantbreeding.wur.nl/uk/software_mapinspect.html). Owing to the obscure nomenclature of apple MADS-box protein in NCBI and other databases, the identified putative apple MADS-box proteins were localized on apple chromosomes based on their genomic coordinates from 5′ to 3′ and numbered accordingly from top to bottom on 1 to 17 chromosomes along with suffix MdMADS to designate apple MADS-box protein (Supplementary Table S1). To analyze the duplication events in apple MADS-box gene family all-vs-all BLASTP51 was performed for 63,517 protein sequences with parameter: -evalue 1e-10, -outfmt 6, -num_threads 10 and -max_target_seqs 5. The BLASTP results were fed to MCScanX52 to identify the collinear gene pairs of MADS-box family in apple with default parameters.
Phylogenetic analysis
The full length protein sequences of representative MADS-box TFs of Arabidopsis, poplar, peach, grape and cucumber were used to find the evolutionary relationship among MADS-box candidates in different species. The sequences of identified MADS-box proteins were aligned using ClustalW2 with default parameters. The multiple sequence alignment generated was then used to construct the phylogenetic tree by employing MEGA version 6.06 using neighbor-joining method with 1000 bootstrap replicates53.
Identification of Putative targets of SVP like MADS protein in apple and their interaction network analysis
To identify the putative targets of orthologs of SVP (DAM) MADS-box proteins in apple, vegetative and reproductive phase chip-seq data of Arabidopsis SVP protein was used48. Further, all possible MADS-box nucleotide binding site patterns were extracted from AGRIS database (http://arabidopsis.med.ohio-state.edu). These nucleotide patterns were further searched against the Arabidopsis chromosomal coordinates identified through SVP chip-seq analysis in vegetative phase and reproductive phase separately; this was performed in order to limit the nucleotide patterns pertaining only to binding site of SVP protein. These identified patterns were searched against 2kb upstream sequence data of apple transcripts obtained from phytozome. The protein sequences of apple genes whose upstream region contains the SVP binding sites were extracted, and their orthologs were identified in Arabidopsis proteome dataset from TAIR 10. On the basis of BLASTP (e-value 1e-04), top hits were considered to build the protein-protein interaction (PPI) network.
We created two types of PPI networks: vegetative phase and reproductive phase PPI network. The networks were constructed by mapping Arabidopsis orthologs of apple candidate proteins onto the Arabidopsis thaliana Protein Interaction Network (AtPIN) interactome. AtPIN54 contains experimental and computational information pertaining to Arabidopsis PPIs. Proteins without any interaction partner were eliminated and the interactions that appeared more than once were simplified to single edges in the dataset. Degree distribution of both networks, viz. vegetative and reproductive was determined to compare their topological properties.
Plant material
The plant material was collected from apple orchard located at Palchan, Manali, Himachal Pradesh, India (32° 18′ 36″ N, 77° 10′ 40″ E) during the year 2012–2013 of complete phenological cycle of apple. The 3 and 5 stages of dormant apical and spur buds, respectively, during the winter from November, 2012 to February, 2013; 6 stages of flower development in the month of April, 2013 and 7 stages of fruit development from May, 2013 to September, 2013 were collected. The collected samples were immediately frozen in liquid nitrogen and stored at −80 °C till further analysis.
RNA-seq analysis
Total RNA was extracted from 100 mg of plant material using iRIS solution55. Total RNA was treated with RNase free DNase to eliminate genomic DNA contamination. The concentration of RNA was measured using NanoDrop UV-VIS spectrophotometer. The integrity of RNA was further checked on denaturing formaldehyde agarose gel. The RNA-Seq libraries were prepared using TruSeq RNA sample preparation kit v2 (Illumina Inc., USA) following manufacturer′s instructions. The libraries were quantified using Qubit dsDNA BR assay kit (Life technologies, USA) and insert size of libraries was determined using Agilent Bioanalyzer DNA 1000 chip (Agilent Technologies). For generating clusters, 10pM of each library was loaded onto the flow cell using TruSeq PE Cluster Kit v5 on cluster station (Illumina Inc., USA). The flow cell containing clonally amplified clusters was loaded onto the Genome Analyzer IIx (Illumina) and paired-end (PE) (2 × 72) sequencing was performed. The raw reads were quality filtered by means of NGS QC toolkit56 and the filtered high quality reads were then de novo assembled using CLC Genomics Wokbench 6.5 (http://www.clcbio.com). The assembled contigs were annotated by BLASTX search against non-redundant NCBI protein database with e-value cutoff 1e-05. The raw reads from Illumina GA IIx of all the analyzed samples were submitted as BioProject (PRJNA302879) to NCBI Sequence Read Archive under accession number SRP066478.
Quantitative real-time PCR (qRT-PCR) analysis
First strand cDNA was synthesized from 1 μg of total RNA in 20 μl reaction using RevertAid RNAse H minus cDNA synthesis kit as per manufacturer’s instructions (Fermentas Life Sciences, USA). For qRT-PCR analysis, primers (Supplementary Table S8) were designed using Primer Express software version 3.0.1 (Invitrogen). The qRT-PCR assays were performed as described earlier57. For data normalization, GAPDH was used as internal control gene at different developmental stages58. The relative expression value was expressed as fold change calculated using comparative delta-delta Ct method59.
Additional Information
How to cite this article: Kumar, G. et al. Comparative phylogenetic analysis and transcriptional profiling of MADS-box gene family identified DAM and FLC-like genes in apple (Malus x domestica). Sci. Rep. 6, 20695; doi: 10.1038/srep20695 (2016).
Accession codes
References
Smaczniak, C., Immink, R. G. H., Angenent, G. C. & Kaufmann, K. Developmental and evolutionary diversity of plant MADS-domain factors: insights from recent studies. Development 139, 3081–3098 (2012).
Masiero, S. et al. INCOMPOSITA: a MADS-box gene controlling prophyll development and floral meristem identity in Antirrhinum . Development 131, 5981–5990 (2004).
Nakano, T. et al. MACROCALYX and JOINTLESS Interact in the Transcriptional Regulation of Tomato Fruit Abscission Zone Development. Plant Physiol. 158, 439–450 (2012).
Vandenbussche, M. et al. Toward the analysis of the petunia MADS box gene family by reverse and forward transposon insertion mutagenesis approaches: B, C, and D floral organ identity functions require SEPALLATA-like MADS box genes in petunia. Plant Cell 15, 2680–2693 (2003).
Heide, O. M. & Prestrud, a K. Low temperature, but not photoperiod, controls growth cessation and dormancy induction and release in apple and pear. Tree Physiol. 25, 109–114 (2005).
Ruiz, D., Campoy, J. A. & Egea, J. Chilling and heat requirements of apricot cultivars for flowering. Environ. Exp. Bot. 61, 254–263 (2007).
Leida, C., Conesa, A., Llácer, G., Badenes, M. L. & Ríos, G. Histone modifications and expression of DAM6 gene in peach are modulated during bud dormancy release in a cultivar-dependent manner. New Phytol. 193, 67–80 (2012).
Hartmann, U. et al. Molecular cloning of SVP: A negative regulator of the floral transition in Arabidopsis. Plant J. 21, 351–360 (2000).
Tao, Z. et al. Genome-wide identification of SOC1 and SVP targets during the floral transition in Arabidopsis. Plant J. 70, 549–561 (2012).
Lee J. H. et al. Role of SVP in the control of flowering time by ambient temperature in Arabidopsis . Genes Dev 21, 397–402 (2007).
Li, D. et al. A Repressor Complex Governs the Integration of Flowering Signals in Arabidopsis . Dev. Cell 15, 110–120 (2008).
Gregis, V., Sessa, A., Dorca-Fornell, C. & Kater, M. M. The Arabidopsis floral meristem identity genes AP1, AGL24 and SVP directly repress class B and C floral homeotic genes. Plant J. 60, 626–637 (2009).
Bielenberg, D. G. et al. Sequencing and annotation of the evergrowing locus in peach [Prunus persica (L.) Batsch] reveals a cluster of six MADS-box transcription factors as candidate genes for regulation of terminal bud formation. Tree Genet. Genomes 4, 495–507 (2008).
Mazzitelli, L. et al. Co-ordinated gene expression during phases of dormancy release in raspberry (Rubus idaeus L.) buds. J. Exp. Bot. 58, 1035–1045 (2007).
Yamane, H. et al. Expressional regulation of PpDAM5 and PpDAM6, peach (Prunus persica) dormancy-associated MADS-box genes, by low temperature and dormancy-breaking reagent treatment. J. Exp. Bot. 62, 3481–3488 (2011).
Ubi, B. E. et al. Molecular cloning of dormancy-associated MADS-box gene homologs and their characterization during seasonal endodormancy transitional phases of Japanese Pear. J. Am. Soc. Hortic. Sci. 135, 174–182 (2010).
Horvath, D. P., Sung, S., Kim, D., Chao, W. & Anderson, J. Characterization, expression and function of DORMANCY ASSOCIATED MADS-BOX genes from leafy spurge. Plant Mol. Biol. 73, 169–179 (2010).
Sasaki, R. et al. Functional and Expressional Analyses of PmDAM Genes Associated with Endodormancy in Japanese Apricot. Plant Physiol. 157, 485–497 (2011).
Sung, S. K. & An, G. Molecular cloning and characterization of a MADS-box cDNA clone of the Fuji apple. Plant Cell Physiol. 38, 484–489 (1997).
Yao, J. L., Dong, Y. H., Kvarnheden, a & Morris, B. Seven MADS-box genes in apple are expressed in different parts of the fruit. J. Am. Soc. Hortic. Sci. 124, 8–13 (1999).
Sung, S. K., Yu, G. H., Nam, J., Jeong, D. H. & An, G. Developmentally regulated expression of two MADS-box genes, MdMADS3 and MdMADS4, in the morphogenesis of flower buds and fruits in apple. Planta 210, 519–528 (2000).
Cevik, V. et al. A FRUITFULL-like gene is associated with genetic variation for fruit flesh firmness in apple (Malus domestica Borkh.). Tree Genet. Genomes 6, 271–279 (2010).
Tian, Y. et al. Genome-wide identification and analysis of the MADS-box gene family in apple. Gene 25, 277–290 (2015).
Fan, C. M. et al. Genome-Wide Expression Analysis of Soybean MADS Genes Showing Potential Function in the Seed Development. PLoS One 8, e62288 (2013).
Velasco, R. et al. The genome of the domesticated apple (Malus × domestica Borkh.). Nature Genet 42, 833–839 (2010).
Leseberg, C. H., Li, A., Kang, H., Duvall, M. & Mao, L. Genome-wide analysis of the MADS-box gene family in Populus trichocarpa . Gene 378, 84–94 (2006).
Xu, Z. et al. Genome-wide identification, characterisation and expression analysis of the MADS-box gene family in Prunus mume . Mol. Genet. Genomics 289, 903–920 (2014).
Wells, C. E., Vendramin, E., Jimenez Tarodo, S., Verde, I. & Bielenberg, D. G. A genome-wide analysis of MADS-box genes in peach [Prunus persica (L.) Batsch]. BMC Plant Biol. 15, 41 (2015).
Wisniewski, M., Norelli, J. & Artlip, T. Overexpression of a peach CBF gene in apple: a model for understanding the integration of growth, dormancy, and cold hardiness in woody plants. Front. Plant Sci. 6, 85 (2015).
Hu, L., Liu, S. & Somers, D. J. Genome-wide analysis of the MADS-box gene family in cucumber. Genome 55, 245–256 (2012).
Diaz-Riquelme, J., Lijavetzky, D., Martínez-Zapater, J. M. & Carmona, M. J. Genome-wide analysis of MIKCC-type MADS box genes in grapevine. Plant Physiol. 149, 354–369 (2009).
Porto, D. D. et al. Transcription profiling of the chilling requirement for bud break in apples: a putative role for FLC-like genes. J. Exp. Bot. 66, 2659–72 (2015).
Parenicova, L. et al. Molecular and phylogenetic analyses of the complete MADS-box transcription factor family in Arabidopsis: new openings to the MADS world. Plant Cell 15, 1538–1551 (2003).
Masiero, S., Colombo, L., Grini, P. E., Schnittger, A. & Kater, M. M. The emerging importance of type I MADS box transcription factors for plant reproduction. Plant Cell 23, 865–872 (2011).
Day, R. C., Herridge, R. P., Ambrose, B. a & Macknight, R. C. . Transcriptome analysis of proliferating Arabidopsis endosperm reveals biological implications for the control of syncytial division, cytokinin signaling, and gene expression regulation. Plant Physiol. 148, 1964–1984 (2008).
Liu, Y. et al. Functional conservation of MIKC*-Type MADS box genes in Arabidopsis and rice pollen maturation. Plant Cell 25, 1288–303 (2013).
Kramer, E. M., Dorit, R. L. & Irish, V. F. Molecular evolution of genes controlling petal and stamen development: duplication and divergence within the APETALA3 and PISTILLATA MADS-box gene lineages. Genetics, 149, 765–83 (1998).
Vrebalov, J. et al. Fleshy fruit expansion and ripening are regulated by the Tomato SHATTERPROOF gene TAGL1 . Plant Cell 21, 3041–3062 (2009).
Seymour, G. B. et al. A SEPALLATA gene is involved in the development and ripening of strawberry (Fragaria x ananassa Duch.) fruit, a non-climacteric tissue*. J. Exp. Bot. 62, 1179–1188 (2011).
Ditta, G., Pinyopich, A., Robles, P., Pelaz, S. & Yanofsky, M. F. The SEP4 gene of Arabidopsis thaliana functions in floral organ and meristem identity. Curr. Biol. 14, 1935–1940 (2004).
Sun, W. et al. Functional and evolutionary analysis of the AP1/SEP/AGL6 superclade of MADS-box genes in the basal eudicot Epimedium sagittatum . Annals of Bot. 113, 653–668 (2014).
Adamczyk, B. J., Lehti-Shiu, M. D. & Fernandez, D. E. The MADS domain factors AGL15 and AGL18 act redundantly as repressors of the floral transition in Arabidopsis. Plant J. 50, 1007–1019 (2007).
Lee, J. & Lee, I. Regulation and function of SOC1, a flowering pathway integrator. J. Exp. Bot. 61, 2247–2254 (2010).
Tapia-López, R. et al. An AGAMOUS-related MADS-box gene, XAL1 (AGL12), regulates root meristem cell proliferation and flowering transition in Arabidopsis. Plant Physiol. 146, 1182–1192 (2008).
Nesi, N. et al. The TRANSPARENT TESTA16 locus encodes the ARABIDOPSIS BSISTER MADS domain protein and is required for proper development and pigmentation of the seed coat. Plant Cell 14, 2463–2479 (2002).
Deng, W. et al. FLOWERING LOCUS C (FLC) regulates development pathways throughout the life cycle of Arabidopsis. Proc. Natl. Acad. Sci. USA 108, 6680–6685 (2011).
Chen, K.-Y. Type II MADS-Box genes associated with poplar apical bud development and dormancy. Dissertation, PAG-XVI (P501) (2008).
Gregis, V. et al. Identification of pathways directly regulated by SHORT VEGETATIVE PHASE during vegetative and reproductive development in Arabidopsis . Genome Biol. 14, R56 (2013).
Du, Z., Zhou, X., Ling, Y., Zhang, Z. & Su, Z. agriGO: a GO analysis toolkit for the agricultural community. Nucl. Acids Res. 38, W64–W70 (2010).
Finn, R. D. et al. The Pfam protein families database. Nucleic Acids Res. 38, D211–D222 (2009).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Wang, Y. et al. MCScanX: A toolkit for detection and evolutionary analysis of gene synteny and collinearity. Nucleic Acids Res. 40, e49 (2012).
Tamura, K. et al. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 28, 2731–2739 (2011).
Brandao M. M., Dantas L. L. & Silva-Filho M. C. AtPIN: Arabidopsis thaliana protein interaction network. BMC Bioinformatics. 10, 454 (2009).
Muoki, R. C., Paul, A., Kumari, A., Singh, K. & Kumar, S. An improved protocol for the isolation of RNA from roots of tea (Camellia sinensis (L.) O. Kuntze). Mol. Biotechnol. 52, 82–88 (2012).
Patel, R. K. & Jain, M. NGS QC toolkit: A toolkit for quality control of next generation sequencing data. PLoS One 7, e30619 (2012).
Arya, P., Kumar, G., Acharya, V. & Singh, A. K. Genome-wide identification and expression analysis of NBS-encoding genes in Malus x domestica and expansion of NBS genes family in rosaceae. PLoS One 9, e107987 (2014).
Kumar, G. & Singh, A. K. Reference gene validation for qRT-PCR based gene expression studies in different developmental stages and under biotic stress in apple. Sci Hort. 10.1016/j.scienta.2015.10.025 (2015).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408 (2001).
Acknowledgements
This work was supported by grants from Council of Scientific and Industrial Research, New Delhi under Network Project PlaGen (BSC0107) to the CSIR-IHBT Palampur. Authors thank Director, CSIR-IHBT for constant encouragement and support. GK and PA gratefully acknowledge University Grant Commission (UGC), Government of India for providing fellowship. VR acknowledges fellowship from the CSIR-IHBT project MLP0071.
Author information
Authors and Affiliations
Contributions
A.K.S. conceived and designed the study. G.K. collected samples and conducted the experiments. G.K. and P.A. performed in silico analysis of MADS-box gene family. G.K. and K.B. performed RNA-seq data analysis. V.R. performed network analysis. G.K., P.A., V.A. and A.K.S. analysed the results. G.K., P.A., V.R. and A.K.S. wrote the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Kumar, G., Arya, P., Gupta, K. et al. Comparative phylogenetic analysis and transcriptional profiling of MADS-box gene family identified DAM and FLC-like genes in apple (Malusx domestica). Sci Rep 6, 20695 (2016). https://doi.org/10.1038/srep20695
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep20695
This article is cited by
-
Integrated transcriptome and proteome analysis provides insights into CpFPA1 for floral induction in Chimonanthus praecox (Magnoliidae) without FLC in genome
Plant Cell Reports (2024)
-
A comprehensive analysis of copy number variations in diverse apple populations
BMC Genomics (2023)
-
Systematic analysis of CCCH zinc finger family in Brassica napus showed that BnRR-TZFs are involved in stress resistance
BMC Plant Biology (2021)
-
A MADS-box gene, EgMADS21, negatively regulates EgDGAT2 expression and decreases polyunsaturated fatty acid accumulation in oil palm (Elaeis guineensis Jacq.)
Plant Cell Reports (2020)
-
Differential gene expression in non-transgenic and transgenic “M.26” apple overexpressing a peach CBF gene during the transition from eco-dormancy to bud break
Horticulture Research (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.