Abstract
Gout is a self-limiting, auto-inflammatory arthritis induced by the deposition of monosodium urate crystals in the synovial fluid and periarticular tissues. The aim of this study was to investigate the associations between genetic variants in the interleukin (IL) and interleukin receptor (ILR) genes IL-33, IL-1RL1, IL-23R and signal transducer and activator of transcription 4 (STAT4) and susceptibility to gout in Chinese Han male individuals. The genetic distributions of rs3939286 in IL-33, rs13015714 in IL-1RL1, rs10889677 in IL-23R and rs7574865 in STAT4 were detected in 1100 men with gout and 1227 ethnically matched controls, using Taqman allelic discrimination real-time polymerase chain reaction (PCR). Differences in these polymorphisms between the groups were investigated using χ2 tests. The genotype-phenotype relationship among gout patients was tested by analysis of variance. There was a significant difference in genotypic frequencies of IL-23R rs10889677 between gout patients and controls (χ2 = 81.386, P < 0.001). However, there were no significant differences in distributions of the other polymorphisms between the groups. Our results revealed that the rs10889677 variant in IL-23R may be involved in the development of gout in Chinese Han male individuals. However, further studies in other ethnic groups are needed to confirm these results.
Similar content being viewed by others
Introduction
Gout is the most common inflammatory arthritis and is characterized by elevated serum urate levels and precipitation of monosodium urate crystals (MSU) in the joints, resulting in recurrent episodes of acute pain. The incidence of gout is increasing globally in line with an aging population according to the Global Burden of Disease studies1. However, the prevalence of gout varies among geographical areas and racial groups2, with a prevalence of 2.49% in the UK in 2012, compared with 6.24% in Taiwan in 20103,4. Gout is a polygenic hereditary disease and previous studies have focused on genetic variants related to key regulators of uric acid homeostasis, such as URAT1, ABCG2 and SLC2A95,6,7. However, in a previous epidemiological study, we found that only about 10% of hyperuricemia patients finally developed gout8, indicating that candidate genes related to uric acid metabolism are unable to provide a full explanation for the pathogenesis of gout. Increasing numbers of studies have therefore concentrated on variations in genes related to inflammatory mediators in gout.
The deposition of MSU caused by increased uric acid levels in the blood, together with several risk factors, can active the NLRP3 (NACHT, LRR and PYD domain-containing protein 3) inflammasome, resulting in caspase-1 activation and secretion of functional interleukin (IL)-1β and IL-18, accompanied by the release of other pro-inflammatory cytokines such as IL-6, IL-8 and tumor necrosis factor (TNF)-α9,10. These cytokines can finally lead to amplification of the inflammatory response, with neutrophil infiltration into the joints and periarticular tissues. IL-33 is an important member of the IL-1 family and a pro-inflammatory cytokine that enhances T-helper cell immune responses by binding to its receptor, IL-1 receptor-like 1 (IL-1RL1) and co-receptor, IL-1 receptor accessory protein (IL-1RAcP)11,12. The IL-33/IL-1RL1 axis plays a critical role in several autoimmune and inflammatory disorders. IL-23 is a member of the IL-12 family and also a pro-inflammatory cytokine, comprising a p19 subunit and a p40 subunit of IL-12B. After binding to the IL-23 receptor (IL-23R), IL-23 stimulates the secretion of a variety of inflammatory factors such as IL-1, IL-6, IL-8 and TNF-α by activated CD4 + Th17 cells, which in turn drive gouty inflammation13. In addition, signal transducer and activator of transcription 4 (STAT4), encoded by STAT4 mapped to chromosome 2q33, regulates the IL-23-related inflammatory response by transmitting signals in response to several cytokines such as IL-23 and IL-1214. STAT4 is also essential for the expansion of Th17 cells activated by IL-23, which contributes to the development of many autoimmune diseases15. The production and function of cytokines may be affected by polymorphisms in the functional regions of their genes, suggesting that IL-33, IL-1RL1, IL-23R and STAT4 may be candidate genes for the inflammatory pathogenesis of gout. The current study therefore aimed to investigate the associations between the IL-33 rs3939286 A/G, IL-1RL1 rs13015714 G/T, IL-23R rs10889677 A/C and STAT4 rs7574865 G/T single nucleotide polymorphisms (SNPs) and the risk of gout.
Results
Demographic and clinical characteristics
The clinical characteristics of all subjects are summarized in Table 1. The mean ages ( ± SD) of the two groups were 50.98 ± 13.92 and 59.72 ± 14.19 years, respectively. Compared with controls, gout patients had higher diastolic pressure (85.01 ± 11.71 vs. 88.07 ± 12.46 mmHg, P < 0.001), higher uric serum acid levels (313.87 ± 59.31 vs. 473.06 ± 122.44 μmol/L, P < 0.001), higher serum triglyceride (TG) levels (1.68 ± 4.44 vs. 2.36 ± 1.75 mmol/L, P < 0.001), higher total cholesterol (TC) levels (5.33 ± 1.07 vs. 5.47 ± 1.35 mmol/L, P = 0.006) and higher serum creatinine levels (80.80 ± 17.52 vs. 88.80 ± 33.66 μmol/L, P < 0.001). However, other parameters including body mass index (BMI), systolic pressure and blood glucose levels were similar in both groups (P = 0.967, P = 0.098 and P = 0.964, respectively; Table 1).
Genetic analysis
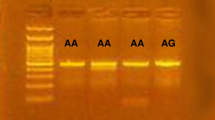
The distributions of the four SNPs among the controls were in Hardy–Weinberg equilibrium (for rs3939286, χ2 = 2.16, P = 0.142; for rs13015714, χ2 = 2.28, P = 0.131; for rs10889677, χ2 = 0.973, P = 0.324; for rs7574865, χ2 = 0.310, P = 0.578). The results for the whole study population are shown in Tables 2 and 3. We detected a significant difference in genotype frequency of IL-23R rs10889677 (χ2 = 81.39, P < 0.001; Table 3) between gout patients and controls. The frequency of the C allele was higher in gout patients compared with controls, but the difference was not significant (P = 0.059, OR = 1.137, 95% CI 0.995–1.298). When the samples were subdivided into AA + AC/CC groups or AA/AC + CC groups, there was a significant difference between cases and controls in terms of AA + AC and CC groups (P < 0.001, OR = 17.589, 95% CI 7.124–43.431). However, no significant differences in genotype or allele frequencies between the groups were identified for the remaining SNPs (for rs3939286, P = 0.478 by genotype, χ2 = 1.46, P = 0.309 by allele; for rs13015714, χ2 = 2.35, P = 0.770 by genotype, χ2 = 0.01, P = 0.228 by allele; for rs7574865, χ2 = 0.52, P = 0.905 by genotype, χ2 = 0.001, P = 0.972 by allele; Table 2). To confirm the accuracy of the genotyping results, 50 samples including cases and controls were selected randomly for sequencing validation and the results were consistent.
Genotype-phenotype analysis of the rs10889677 polymorphism
As shown in Table 4, a detailed genotype-phenotype analysis of IL-23R rs10889677 was conducted among gout patients in relation to clinical factors including demographic characteristics and serum biochemical parameters, as well as tophi and previous medical history (hypertension, diabetes mellitus and obesity). When gout patients were divided into AA, AC and CC groups, there were significant differences in hypertension history between the three groups (P = 0.020) and between the AA and AC + CC groups (P = 0.009). However, there were no significant differences between the three groups in any other parameters, including BMI, systolic pressure, diastolic pressure, other serum biochemical indexes, tophi, diabetes mellitus, or obesity. The results were as the same when the patients were subdivided into AA + AC/CC groups or AA/AC + CC groups (all P > 0.05; Table 4).
Discussion
Gouty inflammation is a paradigm of innate immunity and the IL-1β/IL-1R pathway plays a key role in acute gout attack16. Phagocytosis of MSU by macrophages can activate the NLRP3 inflammasome, which is considered to play a critical role in gouty arthritis. Caspase-1 is in turn activated by the NLRP3 inflammasome and can lead to the conversion of pre-IL-1β and pre-IL-18 to mature IL-1β and IL-18 and secretion of the latter. Binding of IL-1β to the IL-1β receptor in endothelial cells and resident macrophages, together with other pro-inflammatory cytokines and chemokines such as IL-6, IL-8 and TNF-α, can finally lead to gouty inflammation17.
Genetic factors are known to play key roles in the pathogenesis of gout and genetic variants in genes for cytokines involved in gouty inflammatory pathways, such as IL-1β, IL-1R, IL-6, IL-8 and IL-18, have been associated with susceptibility to gout17. We previously suggested that the IL-8 251T/A and IL-12B 1188A/C polymorphisms may be relevant host susceptibility factors for the development of gout18. In addition, a case-control study in 400 gout patients and 582 gout-free controls revealed that the IL-23R rs7517847 G/T polymorphism might be associated with gout in Chinese Han males19. Moreover, the analysis of linkage disequilibrium between rs7517847 and rs10889677 in IL-23R was done further and the D’ value was 0.653, while the r2 value was 0.157, which indicated that there was no linkage disequilibrium between these two SNPs. Sites rs10889677 and rs7517847 of IL-23R were independent to each other and this may demonstrated IL-23R as an important candidate gene for gout susceptibility. However, Tsai et al.20 found no significant association between four polymorphisms in the IL-6 gene promoter, including -597G/A, -572C/G, -373A/T and -174G/C and gout.
As an IL-1 family member, IL-33 is synthesized as a 30-kDa peptide and then cleaved by caspase-1 to form an active 18-kDa mature peptide, which is widely expressed in tissues, specifically in epithelial linings and smooth muscle cells21. Through binding to the IL-33R encoded by IL-1RL1, IL-33 can activate mast cells and Th2 cells as well as Th17 cells, leading to the production of pro-inflammatory cytokines and chemokines21. IL-23/IL-23R represents a novel pathway involved in inflammatory responses. The effect of IL-23 depends on its binding to IL-23R, a heterodimer of the IL-12RB1 subunit22, which was identified as an additional candidate gene in some autoimmune and inflammatory diseases such as inflammatory bowel disease, psoriasis, Graves’ ophthalmopathy, ankylosing spondylitis and multiple sclerosis23,24. A genome-wide association study in Japanese subjects reported that the rs12119179 variant in IL-23R was associated with Behcet’s disease25,26. As noted above, IL-23 may be associated with the production of IL-17A, IL-17F, IL-6 and TNF-α by active Th17 cells, which are important effector cytokines during gouty inflammation. In return, the engagement of IL-17 and/or IL-17F with endothelial cells promotes the expression of IL-1, IL-6, IL-8, TNF-α and ICAM-1, which are critical factors driving the inflammatory response13. Moreover, the ability of IL-23 to induce IL-17A, IL-17F and other pro-inflammatory factors appears important for neutrophil recruitment27. STAT4 transmits signals induced by several cytokines such as IL-12 and IL-23, which are key cytokines in the development of autoimmune diseases14. Furthermore, STAT4 plays a key role in IL-12-induced T-cell differentiation into Th1 cells14 and in IL-23-mediated production of IL-17 by activated Th17 cells15,28. The functions of these inflammatory cytokines suggest that they may play important roles in the inflammatory pathogenesis of gout. We therefore propose IL-33, IL-1RL1, IL-23R and STAT4 as potential candidate susceptibility genes for gout.
To the best of our knowledge, this is the first study to investigate the association between these SNPs in IL-1-related cytokine genes and the risk of gout. We found that IL-23R rs10889677 was significantly associated with susceptibility to gout in Chinese Han male individuals. The genotypic frequency differed significantly between gout patients and controls and patients with the AC or CC genotype were more likely to develop gout than patients with the AA genotype. It found that significant differences exist in patients with hypertension history between the AA and AC + CC groups and between the three groups when doing an analysis on the genotype-phenotype of IL-23R rs10889677, which showed C allele may be a risk allele for gout patients to develop hypertension, however, it may need further research as C allele frequency of rs10889677 is smaller than A allele. rs10889677 is located in the 3′-UTR of IL-23R, suggesting that it might regulate the expression and function of IL23R mRNA. The other three tested SNPs showed no significant association with gout risk. However, Carriere et al. and Oboki et al. reported that IL-33 rs3939286 and IL-1RL1 rs13015714 were associated with other autoimmune diseases such as Crohn’s disease, rheumatoid arthritis (RA), asthma and Alzheimer’s disease29,30. Hayakawa et al. also found that blocking IL-1RL1 receptor signaling could prevent arthritis development and airway inflammation31,32. A relevant study in western Algeria indicated that the STAT4 rs7574865 polymorphism was clearly associated with the risk of RA in an Algerian population33, while a meta-analysis by Jiang et al. also indicated a significant association between the rs7574865 polymorphism and a decreased RA risk in an Asian population34. In addition, Liu et al. found that rs7574865 in STAT4 was significantly associated with increased susceptibility to and severity of ankylosing spondylitis in a Chinese Han Population.
There were some limitations to this study. First, all the subjects were Chinese Han individuals from Shandong Province and further studies including participants from other nationalities and regions are therefore needed to determine the general applicability of the results, given the important roles of both genetic and environmental factors in the development of gout. Furthermore, other important SNPs exist on these genes and the contributions of these to gout susceptibility should also be investigated. However, despite these limitations, our study demonstrated an association between IL-23R and susceptibility to gout in Chinese Han male individuals.
Methods
Populations and clinical data
A total of 1100 men with gout who visited the Department of Gout, the Affiliated Hospital of Qingdao University, China between January 2009 and December 2014 were recruited to this study. Gout diagnosis followed the criteria published by the American College of Rheumatology in 199735. No patients had any history of cancer, hematopathy, nephropathy, or other autoimmune diseases. Some metabolic diseases such as hypertension, obesity, diabetes and so on were not excluded from gout patients because there were no researches have indicated relationships between the four SNPs and these diseases. A further 1227 gout-free male controls were enrolled. All the participants were Chinese Han men. Blood samples were collected into anticoagulant-coated tubes and transferred immediately to the laboratory for DNA extraction and genetic analysis. Demographic and laboratory parameters and medical histories were recorded by experienced endocrinologist physicians, including serum uric acid, glucose, TG, TC and creatinine levels, blood pressure and BMI, as well as age of onset, tophi and medical history among gout patients. This study was approved by the Ethics Committee of the Affiliated Hospital, Qingdao University and conducted in accordance with the ethical guidelines of the 1975 Declaration of Helsinki.
DNA extraction and genetic analysis
After obtaining informed consent, genomic DNA was extracted from peripheral blood samples from gout patients and controls using genomic DNA Extraction kits (Qiagen, Hilden, Germany) in accordance with the manufacturer’s instructions. All four SNPs (rs3939286, rs13015714, rs10889677, rs7574865) were genotyped by CFX96TM real-time polymerase chain reaction (PCR) (Bio-Rad, California, USA) using the TaqMan probe method. The TaqMan probes and primers were designed and synthesized by Applied Biosystems of Life Technologies (New York, USA). The sequences of the forward and reverse primers were: rs3939286, 5′-TCCACATCCCCATGGTTTGTTGTTG-3′ and 5′-TGCTTGTAGTGGGTTGTTGTTATCT-3′, respectively; rs13015714, 5′-CGGCTATGGGTTTCCCTTTTCCTTT-3′ and 5′-GTTAAATAACAGTTCTGCCACAAAA-3′, respectively; rs10889677, 5′-TTTAATTTTAGCCATTCTTCTGCCT-3′ and 5′-ATTTCTTAAAATTAGAGAATTAAGG-3′, respectively; rs7574865, 5′-TATGAAAAGTTGGTGACCAAAATGT-3′ and 5′-AATAGTGGTTATCTTATTTCAGTGG-3′, respectively. The amplifications were carried out by C1000TM thermal cycler in a 25-μL total reaction volume containing 12.5 μL 2× PCR Master Mix, 1.25 μL 20× SNP Genotyping Assay, 11.25 μL DNA sample and DNase-free water. The PCR conditions were as follows: initial denaturation at 95 °C for 3 min, followed by 45 cycles of denaturation at 95 °C for 15 s and 60 ˚C 1 min. Genotypes of all the samples were finally analyzed using Bio-Rad CFX manager 3.0 software.
Statistical analysis
Statistical analyses were carried out using the Statistical Package for Social Sciences version 22.0 (SPSS Inc., Chicago, IL, USA). The Hardy–Weinberg equilibrium of the control group was tested using the goodness-of-fit χ2 test. Differences in genotypes and alleles between cases and controls were analyzed using the Pearson’s χ2 test (or Fisher’s exact test if the expected values were < 5) and P-values < 0.013 were considered significant when Bonferroni’s correction was made. Differences in demographic and clinical indexes between the two groups were compared using the Student’s t-test. Differences between genotyping and clinical characteristics in gout patients, including clinical and biochemical parameters, tophi and past medical history were assessed by analysis of variance. The strength of the relationships in allelic distributions between gout patients and controls was assessed by OR and 95% CI. All statistical tests were two-sided with a significance level of 0.05 and differences were deemed to be significant at P < 0.01.
Additional Information
How to cite this article: Liu, S. et al. Associations between interleukin and interleukin receptor gene polymorphisms and risk of gout. Sci. Rep. 5, 13887; doi: 10.1038/srep13887 (2015).
References
Smith, E. et al. The global burden of gout: estimates from the Global Burden of Disease 2010 study. Annals of the rheumatic diseases 73, 1470–1476, 10.1136/annrheumdis-2013-204647 (2014).
Roddy, E., Zhang, W. & Doherty, M. The changing epidemiology of gout. Nature clinical practice. Rheumatology 3, 443–449, 10.1038/ncprheum0556 (2007).
Kuo, C. F., Grainge, M. J., Mallen, C., Zhang, W. & Doherty, M. Rising burden of gout in the UK but continuing suboptimal management: a nationwide population study. Annals of the rheumatic diseases. 10.1136/annrheumdis-2013-204463 (2014).
Kuo, C. F. et al. Epidemiology and management of gout in Taiwan: a nationwide population study. Arthritis research & therapy 17, 13, 10.1186/s13075-015-0522-8 (2015).
Takada, T. et al. ABCG2 dysfunction increases serum uric acid by decreased intestinal urate excretion. Nucleosides, nucleotides & nucleic acids 33, 275–281, 10.1080/15257770.2013.854902 (2014).
George, R. L. & Keenan, R. T. Genetics of hyperuricemia and gout: implications for the present and future. Current rheumatology reports 15, 309, 10.1007/s11926-012-0309-8 (2013).
Kolz, M. et al. Meta-analysis of 28,141 individuals identifies common variants within five new loci that influence uric acid concentrations. PLoS genetics 5, e1000504, 10.1371/journal.pgen.1000504 (2009).
Miao, Z. M. et al. Epidemiological survey of hyperuricemia and gout in coastal areas of Shandong province. Chin J Endocrinol Metab 22, 421–425, 10.3760/j.issn:1000-6699.2006.05.005 (2006).
Malawista, S. E., de Boisfleury, A. C. & Naccache, P. H. Inflammatory gout: observations over a half-century. FASEB journal: official publication of the Federation of American Societies for Experimental Biology 25, 4073–4078, 10.1096/fj.11-1201ufm (2011).
Popa-Nita, O. & Naccache, P. H. Crystal-induced neutrophil activation. Immunology and cell biology 88, 32–40, 10.1038/icb.2009.98 (2010).
Xu, D. et al. IL-33 exacerbates antigen-induced arthritis by activating mast cells. Proceedings of the National Academy of Sciences of the United States of America 105, 10913–10918, 10.1073/pnas.0801898105 (2008).
Palmer, G. et al. Inhibition of interleukin-33 signaling attenuates the severity of experimental arthritis. Arthritis and rheumatism 60, 738–749, 10.1002/art.24305 (2009).
Aggarwal, S. & Gurney, A. L. IL-17: prototype member of an emerging cytokine family. Journal of leukocyte biology 71, 1–8 (2002).
Watford, W. T. et al. Signaling by IL-12 and IL-23 and the immunoregulatory roles of STAT4. Immunological reviews 202, 139–156, 10.1111/j.0105-2896.2004.00211.x (2004).
Mathur, A. N. et al. Stat3 and Stat4 direct development of IL-17-secreting Th cells. Journal of immunology 178, 4901–4907 (2007).
Ghaemi-Oskouie, F. & Shi, Y. The role of uric acid as an endogenous danger signal in immunity and inflammation. Current rheumatology reports 13, 160–166, 10.1007/s11926-011-0162-1 (2011).
Kingsbury, S. R., Conaghan, P. G. & McDermott, M. F. The role of the NLRP3 inflammasome in gout. Journal of inflammation research 4, 39–49, 10.2147/JIR.S11330 (2011).
Liu, S., Yin, C., Chu, N., Han, L. & Li, C. IL-8 -251T/A and IL-12B 1188A/C polymorphisms are associated with gout in a Chinese male population. Scandinavian journal of rheumatology 42, 150–158, 10.3109/03009742.2012.726372 (2013).
Liu, S. et al. The rs7517847 polymorphism in the IL-23R gene is associated with gout in a Chinese Han male population. Modern rheumatology/the Japan Rheumatism Association, 1–4, 10.3109/14397595.2014.964823 (2015).
Tsai, P. C., Chen, C. J., Lai, H. M. & Chang, S. J. Analysis of polymorphisms in the promoter region and protein levels of interleukin-6 gene among gout patients. Clinical and experimental rheumatology 26, 841–847 (2008).
Schmitz, J. et al. IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity 23, 479–490, 10.1016/j.immuni.2005.09.015 (2005).
Ekinci, N. S., Alpsoy, E., Karakas, A. A., Yilmaz, S. B. & Yegin, O. IL-17A has an important role in the acute attacks of Behcet’s disease. The Journal of investigative dermatology 130, 2136–2138, 10.1038/jid.2010.114 (2010).
Huber, A. K., Jacobson, E. M., Jazdzewski, K., Concepcion, E. S. & Tomer, Y. Interleukin (IL)-23 receptor is a major susceptibility gene for Graves’ ophthalmopathy: the IL-23/T-helper 17 axis extends to thyroid autoimmunity. The Journal of clinical endocrinology and metabolism 93, 1077–1081, 10.1210/jc.2007-2190 (2008).
Sung, I. H. et al. IL-23R polymorphisms in patients with ankylosing spondylitis in Korea. The Journal of rheumatology 36, 1003–1005, 10.3899/jrheum.081121 (2009).
Mizuki, N. et al. Genome-wide association studies identify IL23R-IL12RB2 and IL10 as Behcet’s disease susceptibility loci. Nature genetics 42, 703–706, 10.1038/ng.624 (2010).
Remmers, E. F. et al. Genome-wide association study identifies variants in the MHC class I, IL10 and IL23R-IL12RB2 regions associated with Behcet’s disease. Nature genetics 42, 698–702, 10.1038/ng.625 (2010).
Happel, K. I. et al. Cutting edge: roles of Toll-like receptor 4 and IL-23 in IL-17 expression in response to Klebsiella pneumoniae infection. Journal of immunology 170, 4432–4436 (2003).
Miossec, P. Interleukin-17 in fashion, at last: ten years after its description, its cellular source has been identified. Arthritis and rheumatism 56, 2111–2115, 10.1002/art.22733 (2007).
Carriere, V. et al. IL-33, the IL-1-like cytokine ligand for ST2 receptor, is a chromatin-associated nuclear factor in vivo. Proceedings of the National Academy of Sciences of the United States of America 104, 282–287, 10.1073/pnas.0606854104 (2007).
Oboki, K., Ohno, T., Kajiwara, N., Saito, H. & Nakae, S. IL-33 and IL-33 receptors in host defense and diseases. Allergology international: official journal of the Japanese Society of Allergology 59, 143–160, 10.2332/allergolint.10-RAI-0186 (2010).
Hayakawa, H., Hayakawa, M., Kume, A. & Tominaga, S. Soluble ST2 blocks interleukin-33 signaling in allergic airway inflammation. The Journal of biological chemistry 282, 26369–26380, 10.1074/jbc.M704916200 (2007).
Iikura, M. et al. IL-33 can promote survival, adhesion and cytokine production in human mast cells. Laboratory investigation; a journal of technical methods and pathology 87, 971–978, 10.1038/labinvest.3700663 (2007).
Fodil, M. et al. Association of PTPN22 (rs2476601) and STAT4 (rs7574865) polymorphisms with Rheumatoid Arthritis in the Western Algerian population. Acta reumatologica portuguesa (2014).
Jiang, X., Zhou, Z., Zhang, Y., Yang, H. & Ren, K. An updated meta-analysis of the signal transducer and activator of transcription 4 (STAT4) rs7574865 G/T polymorphism and rheumatoid arthritis risk in an Asian population. Scandinavian journal of rheumatology 43, 477–480, 10.3109/03009742.2014.918174 (2014).
Wallace, S. L. et al. Preliminary criteria for the classification of the acute arthritis of primary gout. Arthritis and rheumatism 20, 895–900 (1977).
Acknowledgements
We thank all of the probands for their participation. This work was supported by the National Science Foundation of China (31371272 and 81371499).
Author information
Authors and Affiliations
Contributions
S.L. conceived and designed the experiments. Z.Z. performed the data analyses and drafted the manuscript. S.L. and Z.Z. contributed equally to this work. C.W. and M.G. collected the blood samples and extracted genomic DNA. N.C. coordinated and supervised data collection. C.L. conceptualized and designed the study. All authors read and approved the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Liu, S., Zhou, Z., Wang, C. et al. Associations between interleukin and interleukin receptor gene polymorphisms and risk of gout. Sci Rep 5, 13887 (2015). https://doi.org/10.1038/srep13887
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep13887
This article is cited by
-
Regulatory T cell frequency, but not plasma IL-33 levels, represents potential immunological biomarker to predict clinical response to intravenous immunoglobulin therapy
Journal of Neuroinflammation (2017)
-
Polymorphisms in key bone modulator cytokines genes influence bisphosphonates therapy in postmenopausal women
Inflammopharmacology (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.