Abstract
Mitogen-activated protein kinases (MAPKs) are key signal transducers involved in various cellular events such as growth, proliferation and differentiation. Previous studies have reported that H2O2 leads to phosphorylation of extracellular signal-regulated kinase (ERK), one of the MAPKs in endothelial cells. The current study shows that H2O2 suppressed ERK1/2 activation and phosphorylation at specific concentrations and times in human umbilical vein endothelial cells but not in immortalized mouse aortic endothelial cells or human astrocytoma cell line CRT-MG. Phosphorylation of other MAPK family members (i.e., p38 and JNK) was not suppressed by H2O2. The decrease in ERK1/2 phosphorylation induced by H2O2 was inversely correlated with the level of phosphorylation of Src tyrosine 530. Using siRNA, it was found that H2O2-induced suppression of ERK1/2 was dependent on Csk. Physiological laminar flow abrogated, but oscillatory flow did not affect, the H2O2-induced suppression of ERK1/2 phosphorylation. In conclusion, H2O2-induced Csk translocation to the plasma membrane leads to phosphorylation of Src at the tyrosine 530 residue resulting in a reduction of ERK1/2 phosphorylation. Physiological laminar flow abrogates this effect of H2O2 by inducing phosphorylation of Src tyrosine 419. These findings broaden our understanding of signal transduction mechanisms in the endothelial cells against oxidative stress.
Similar content being viewed by others
Introduction
Reactive oxygen species (ROS) such as superoxide, hydrogen peroxide (H2O2) and the hydroxyl radical are generated by all aerobic cells. Low-dose ROS act as signaling molecules and regulate both gene expression and signal transduction1,2,3,4. However, when excess ROS overwhelm endogenous antioxidant systems, oxidative stress occurs, leading to harmful effects such as aging, senescence and apoptosis5,6. In endothelial cells, excessive ROS mediate vasodilation, endothelial barrier dysfunction, actin reorganization and leukocyte extravasation7,8,9,10,11. ROS are abundant, especially in the endothelium at sites of inflammation and infection and are generated by various sources including vasoactive peptides, cyclic stretch, hypoxia-reoxygenation and infiltration of activated leukocytes12. The pathogenesis of diverse vascular diseases, including atherosclerosis, diabetes and ischemia-reperfusion injury, is associated with the overproduction of ROS8.
Endothelial cells that line the inner surface of blood vessels are continually exposed to various stresses, including oxidative stress and mechanical force induced by blood flow and pressure. Laminar shear stress, one of the primary forces that endothelial cells experience, is particularly important because it is critical to endothelial cell survival and proliferation and the regulation of various genes such as heme oxygenase, connexin 37 and growth arrest and DNA damage-inducible protein 15313,14. However, when unidirectional shear flow is disrupted, the gene expression profile is changed and pro-atherogenic signaling is activated15,16. When endothelial cells are exposed to oxidative stress, signaling molecules such as extracellular signal-regulated kinases (ERKs), protein kinase C and tyrosine kinase are activated8. Increasing ERK1/2 activation in endothelial cells by ROS influences several endothelial activities such as pro-survival effects against apoptosis, increased vascular permeability and MMP-9-mediated angiogenesis7,17,18.
The mitogen-activated protein kinases (MAPKs) are a family of serine-threonine kinases that act as key signaling molecules regulating numerous cellular functions19. Responding to both extracellular and intracellular stimuli such as growth factors, cytokines and oxidative stress, MAPKs phosphorylate specific serine and threonine residues on target proteins and control cellular activities including gene expression, metabolism, proliferation and apoptosis20. ERKs, c-Jun NH2-terminal kinases (JNKs) and p38 enzymes are major subfamilies of MAPKs. However, these subfamilies have somewhat different effects on various eukaryotic cells. In general, ERKs regulate cell survival and proliferation in differentiated cells, while JNK controls the apoptotic response to cellular stress and p38 plays a critical role in normal immune and inflammatory responses21,22,23. Dysregulation of ERK1 and ERK2 (ERK1/2) signaling pathways is related to various human diseases including neurodegenerative diseases, cancers and inflammation24. Previous studies have reported that extracellular H2O2 leads to phosphorylation of ERK1/2 and activation of the ERK signaling pathway in various cell types, including fibroblast, smooth muscle and endothelial cells8,25,26. ERK1/2 regulates cell functions by phosphorylating many cytosolic proteins such as p90 ribosomal s6 kinase, MAPK-interacting serine/threonine kinase and the transcription factor Elk27. MAPK kinase (MAPKK), MEK1/2 and MAPKK kinase (MAPKKK; A-Raf and B-Raf) are upstream activators of ERK1/2. Their activation is triggered by G protein-coupled receptors, receptor tyrosine kinases and non-receptor tyrosine kinases such as Src28,29.
Src is an oncogene that is overexpressed in many types of tumors30. In endothelial cells, Src activates several molecules that are involved in endothelial permeability and induce vascular leakage31. Src is regulated at two major tyrosine phosphorylation sites (419 and 530). When phosphorylation occurs at tyrosine 419, Src undergoes a conformational change so that substrates are able to access the Src kinase domain. However, when Src is phosphorylated at tyrosine 530, kinase activity is blocked32. Csk is a negative regulator of Src that phosphorylates Src at tyrosine 530 and maintains the inactive state33,34.
The present study was designed to determine the molecular mechanisms by which H2O2 suppresses p-ERK1/2 phosphorylation, focusing on the Csk-Src-ERK signaling pathway. The results demonstrate that phosphorylation of ERK1/2 is suppressed over a specific concentration range of H2O2 in human umbilical vein endothelial cells (HUVECs). The study also found that physiological laminar shear stress could modulate this suppression.
Results
H2O2 reduces the endogenous level of ERK1/2 phosphorylation
HUVECs were treated with various concentrations of H2O2 for 15 min and examined for changes in ERK1/2 phosphorylation by immunoblotting. In the presence of 0.25–1 mM H2O2, ERK1/2 phosphorylation was significantly reduced compared to the untreated control group, with no change in the total amount of ERK1/2 (Fig. 1a). However, when 2 mM concentrations of H2O2 were tested, ERK1/2 phosphorylation was slightly increased. Treating HUVECs with a mid-range concentration of H2O2 (0.5 mM) for varied time periods showed that p-ERK1/2 was slightly increased at 5 min but significantly reduced at 15 min (Fig. 1b). Interestingly, ERK1/2 phosphorylation was increased at 30 min after H2O2, compared to the 15 min-treated and control groups. After 1 h, ERK1/2 phosphorylation was decreased to levels comparable to the control.
Phosphorylation of ERK1/2 in HUVECs is suppressed by treatment with H2O2.
(a) HUVECs were treated with 0–2 mM H2O2 for 15 min. Cells were washed once with PBS. Whole cell lysates were then extracted and analyzed by immunoblotting. Anti-p-ERK1/2, anti-ERK1/2 and anti-tubulin antibodies were used. Tubulin was used as the loading control. Bottom panel: p-ERK1/2 levels were quantified densitometrically and normalized to the level of ERK1/2. The control level of p-ERK1/2 expression was assigned a value of 1. Data are expressed as mean values ± SEM of three independent samples. *p < 0.05 vs. untreated control. (b) HUVECs were treated with 0.5 mM H2O2 for the indicated times (5 min to 8 h). Whole cell lysates were extracted and analyzed by immunoblotting. Data are expressed as mean values ± SEM of three independent samples. *p < 0.05 vs. untreated control; §p < 0.05 between 15 min- and 30 min-treated cells. Ctrl, untreated control; p, phospho.
H2O2 reduces ERK1/2 phosphorylation but has little effect on the phosphorylation of p38 and JNK
ERK1/2, p38 and JNK influence each other in response to various stresses35. To test whether these MAPK subfamilies are affected by H2O2, HUVECs were treated with varied concentrations of H2O2 for 15 min and the phosphorylation of p38 and JNK was examined by immunoblotting. Treating HUVECs with 0.5 or 1 mM H2O2 decreased p-ERK1/2 but had no effect on p-p38 (Fig. 2a). Interestingly, phosphorylation of p38 was slightly increased with 0.05 mM and 1 mM H2O2, compared to control. There was also a non-significant tendency toward a decrease with 0.5 mM H2O2 compared to 0.05 mM H2O2-treated cells. p-JNK was not changed by treatment of HUVECs with 0.01–1 mM H2O2. To determine whether H2O2-induced suppression of ERK1/2 is a common occurrence in different cell types, immortalized murine aortic endothelial cells (iMAECs) and human brain astroglioma CRT-MG cells were tested. Phosphorylation of ERK1/2 was significantly increased in response to H2O2 in iMAECs (Fig. 2b), consistent with previous reports showing that H2O2 induces ERK1/2 phosphorylation. In CRT-MG cells, p-ERK1/2 was also induced by H2O2 in a concentration-dependent manner (Fig. 2c).
H2O2-mediated suppression of ERK1/2 is specific to HUVECs.
(a) HUVECs were treated with 0–1 mM H2O2 for 15 min. Whole cell lysates were extracted and analyzed by immunoblotting. Antibodies against p-p38, p38, p-JNK, JNK, p-ERK1/2, ERK1/2 and tubulin were used. Tubulin was used as the loading control. Right panel: p-p38 and p-JNK levels were measured densitometrically and normalized to the levels of p38 and JNK, respectively. The control level was assigned a value of 1. Data are expressed as mean values ± SEM of three independent samples. Varied concentrations of H2O2 were used to treat (b) iMAEC and (c) CRT-MG cells for 15 min. Whole cell lysates were extracted and analyzed by immunoblotting. Data are expressed as mean values ± SEM of three independent samples. *p < 0.05 vs. untreated control. p, phospho.
H2O2 influences HUVEC apoptosis and monolayer permeability
To test the effect of H2O2 on HUVEC apoptosis, cells were treated with 0.5 mM H2O2 for 15 min, stained with annexin V-conjugated FITC and propidium iodine (PI) and then analyzed by fluorescence activated cell sorting (FACS). As shown in Fig. 3a, H2O2 increased the number of early and late apoptotic cells from 2.16% and 1.82% to 4.43% and 6.48%, respectively. Treating monolayer HUVECs with 0.5 mM H2O2 for 15 min and measuring permeability using a FITC-dextran solution showed that permeability was significantly increased about 14% above control (Fig. 3b).
H2O2 influences HUVEC apoptosis and monolayer permeability.
(a) HUVECs were treated with 0.5 mM H2O2 for 15 min, stained with annexin V-FITC and propidium iodide (PI) and then analyzed by fluorescence activated cell sorting (FACS). The proportion of cells in each quadrant was presented. Data are representative of at least three independent experiments. (b) HUVECs cultured on collagen-coated inserts were exposed to 0.5 mM H2O2 for 15 min and then FITC-dextran solution was added to the inserts. After 20-min incubation at room temperature, the amount of FITC-dextran that passed through the HUVEC monolayer was measured using a fluorescence plate reader. Relative fluorescence intensity is shown. Data are expressed as mean values of three independent samples. *p < 0.05 vs. untreated control. HUVECs cultured on inserts were then lysed in lysis buffer and p-ERK level was analyzed by immunoblotting (right panel). p, phospho.
H2O2 induces Src phosphorylation at tyrosine 530 in HUVECs
The phosphorylation of ERK1/2 by H2O2 is dependent on the activation of Src29. To investigate the effect of H2O2 on Src activation, HUVECs were treated with varied concentrations of H2O2 for 15 min and immunoblotted with anti-p-Src tyrosine 419 and anti-p-Src tyrosine 530 antibodies. Phosphorylation of Src tyrosine 419 was decreased following treatment with 0.5–1 mM H2O2 compared with the control (Fig. 4a, first panel, lanes 5–6 vs. 1). In contrast, the phosphorylation of Src tyrosine 530 was significantly increased by these H2O2 treatments compared to the control (Fig. 4a, second panel, lanes 5–6 vs. 1). To determine whether H2O2 was directly involved in the phosphorylation of Src tyrosine 530, HUVECs were incubated for 15 min with 25, 50, or 100 units/ml catalase in the presence of 0.5 mM H2O2. As little as 25 units/ml catalase abrogated H2O2-induced changes in p-ERK1/2 (Fig. 4b, third panel, lanes 3 and 2), as well as p-Src tyrosine 419 (Fig. 4b, first panel). In contrast to the suppression and recovery patterns of p-ERK1/2 and p-Src tyrosine 419 induced by treatment with H2O2, p-Src tyrosine 530 was reduced to the control level by co-treatment with 25, 50 and 100 units/ml catalase (Fig. 4b, second panel, lanes 3–6 vs. 2).
H2O2 induces Src phosphorylation at tyrosine 530 in HUVECs.
(a) HUVECs were treated with the indicated concentrations of H2O2 for 15 min. Whole cell lysates were then extracted and analyzed by immunoblotting. Anti-p-Src Y419, anti-p-Src Y530, anti-p-ERK1/2, anti-ERK1/2 and anti-tubulin antibodies were used. p-ERK1/2 levels were quantified densitometrically and normalized to that of ERK1/2. p-Src Y419 and p-Src Y530 levels were quantified densitometrically and normalized to the level of tubulin. The control level was assigned a value of 1. Data are expressed as mean values ± SEM of three independent samples. *p < 0.05 vs. untreated control. (b) Different amounts of catalase were added to HUVECs in the presence or absence of 0.5 mM H2O2 for 15 min. Whole cell lysates were extracted and analyzed by immunoblotting. p-Src and p-ERK1/2 blots from three independent samples were quantified densitometrically. *p < 0.05 vs. untreated control; §p < 0.05 vs. H2O2 only-treated cells. p, phospho; Y, tyrosine; CAT, catalase.
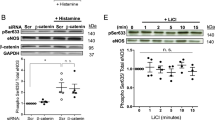
siRNA targeting Csk abrogates the suppression of p-ERK1/2 by H2O2 by controlling the phosphorylation status of Src tyrosine 530
To investigate whether Src was directly involved in the suppression of p-ERK1/2 by H2O2, HUVECs were transfected with siRNA targeting Src for 48 h, treated with or without H2O2 for 15 min and immunoblotted with anti-p-ERK1/2, anti-ERK1/2 and anti-Src antibodies. In the absence of H2O2, the phosphorylation of ERK1/2 in Src siRNA-transfected cells was significantly decreased compared to GFP siRNA-transfected cells. Interestingly, in the presence of H2O2, the phosphorylation of ERK1/2 was reduced in both GFP-siRNA transfected and Src-siRNA transfected H2O2-treated groups, with no significant difference in the phosphorylation level between the two groups (Fig. 5a). From a proportionate perspective, the amount of p-ERK1/2 suppressed by H2O2 was reduced from 65.4% to 46.9%. Because Csk is the key regulatory molecule that phosphorylates Src tyrosine 530 and inhibits Src kinase activity34, HUVECs were transfected with Csk siRNA and the effect of silencing Csk on Src and ERK1/2 was examined in H2O2-treated HUVECs. After exposure to 0.5 mM H2O2 for 15 min, the level of p-Src tyrosine 530 was reduced and p-ERK1/2 suppression was significantly abrogated in Csk siRNA-transfected cells, compared to GFP siRNA-transfected cells (Fig. 5b). In contrast to p-Src tyrosine 530, p-Src tyrosine 419 was not affected by Csk siRNA knockdown.
H2O2-induced suppression of ERK1/2 is dependent on Csk.
(a) HUVECs were transfected with 100 nM of GFP or Src siRNA using LipofectamineTM RNAiMAX (1.5 μl/ml). After 48 h, cells were treated with 0.5 mM H2O2 for 15 min. Whole cell lysates were extracted and analyzed by immunoblotting. Tubulin was used as the loading control. Right panel: p-ERK1/2 levels were quantified densitometrically and normalized to the level of ERK1/2. The control level of p-ERK1/2 expression was assigned a value of 1. Data are expressed as mean values ± SEM of three independent samples. *p < 0.05 between GFP siRNA-transfected control and H2O2-treated cells; **p < 0.05 between Src siRNA-transfected control and H2O2-treated cells; §p < 0.05 between GFP siRNA-transfected H2O2-treated cells and Src siRNA-transfected H2O2-treated cells. (b) HUVECs were transfected with 50 nM of GFP or Csk siRNA using LipofectamineTM RNAiMAX (1.5 μl/ml). After 24 h, cells were treated with 0.5 mM H2O2 for 15 min. Whole cell lysates were analyzed by immunoblotting. Data are expressed as mean values ± SEM of three independent samples. *p < 0.05 between GFP siRNA-transfected control and H2O2-treated cells; §p < 0.05 between GFP siRNA-transfected H2O2-treated cells and Csk siRNA-transfected H2O2-treated cells. p, phospho.
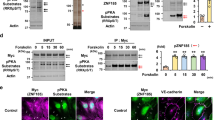
H2O2-induced Csk translocation is critical to suppressing p-ERK1/2
Csk exhibits its inhibitory function on Src through effects on membrane translocation34. To confirm the membrane translocation of Csk by treatment with 0.5 mM H2O2, HUVECs transfected with Csk siRNA were treated with 0.5 mM H2O2 for 15 min, followed by separation of whole cell lysates into detergent soluble and insoluble fractions using the nonionic detergent Brij 58. Immunoblotting analysis revealed that recruitment of Csk to the insoluble fraction was increased by H2O2 (Fig. 6a, first panel, lanes 6 and 5). When Csk was suppressed by siRNA knockdown, the amount of Csk and p-Src tyrosine 530 in the insoluble fraction was reduced, while the reduction in p-ERK1/2 in the insoluble fraction caused by H2O2 was abrogated (Fig. 6a, lanes 5–6 vs. 7–8). Immunofluorescence assay (IFA) data showed that Csk was diffusely distributed throughout the cytoplasm in untreated HUVECs and that a significant quantity of Csk was found at the cell periphery following H2O2 treatment, although a large quantity of Csk was still present in the cytoplasm (Fig. 6b).
Membrane translocation of Csk is induced by treatment with H2O2 in HUVECs.
(a) HUVECs transfected with GFP or Csk siRNA were exposed to 0.5 mM H2O2, then separated into soluble and insoluble fractions using Brij 58. Each fraction was analyzed by immunoblotting using anti-Csk, anti-p-Src Y530, anti-p-ERK1/2, anti-ERK1/2, anti-caveolin-1 and anti-tubulin antibodies. (b) HUVECs were incubated with or without 0.5 mM H2O2 for 15 min and then fixed with 4% paraformaldehyde. Fixed cells were probed with anti-Csk antibody followed by an Alexa fluor 568-conjugated secondary antibody. Data are representative of at least three independent experiments. (Original magnification × 400). p, phospho; Y, tyrosine; CON, control.
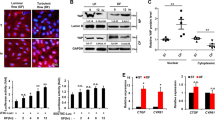
H2O2-induced suppression of ERK1/2 phosphorylation is abrogated by physiological laminar flow, but not by oscillatory flow
In a normal physiological state, endothelial cells lining the inner surface of blood vessels are exposed to mechanical shear stress from the circulating blood flow14. To determine whether H2O2-induced suppression of p-ERK1/2 occurs in the presence of shear stress, HUVECs were treated with or without 0.5 mM H2O2 for 15 min under conditions of laminar shear stress (16 and 25 dyne/cm2) or oscillatory shear stress. Interestingly, H2O2-induced suppression of p-ERK1/2 was abolished in the presence of laminar shear stress (Fig. 7a, first panel, lanes 4 and 6 vs. 2). In contrast, suppression of ERK1/2 phosphorylation induced by H2O2 was unaffected by oscillatory shear stress. HUVECs were also treated with various concentrations of H2O2 for 15 min in the presence or absence of 16 dyne/cm2 laminar shear stress. Under these conditions, ERK1/2 phosphorylation was increased and the suppression of p-ERK1/2 by H2O2 was significantly diminished, especially with 1 mM H2O2 (Fig. 7b, third panel, lanes 10 and 5). Interestingly, phosphorylation of Src tyrosine 530 was not changed by laminar shear stress but only by H2O2, while the phosphorylation of Src tyrosine 419 was increased by laminar shear stress and significantly reduced by H2O2.
Physiological laminar flow, not oscillatory flow, prevents the H2O2-induced suppression of ERK1/2 phosphorylation.
(a) Different strengths of laminar shear or oscillatory shear stress were used on HUVECs for 15 min in the presence or absence of 0.5 mM H2O2. After treatment, whole cell lysates from each group were extracted and analyzed by immunoblotting. (b) HUVECs were exposed to the indicated concentrations of H2O2 for 15 min with or without 16 dyne/cm2 laminar shear stress. Whole cell lysates were extracted and analyzed by immunoblotting. Bottom panel: p-ERK1/2 levels were quantified densitometrically and normalized to the level of ERK1/2. The control level of p-ERK1/2 expression was assigned a value of 1. Data are expressed as mean values ± SEM of three independent samples. *p < 0.05 vs. H2O2-untreated static control; §p < 0.05 between static cells and LSS-treated cells at each concentration of H2O2. LSS, laminar shear stress; OSS, oscillatory shear stress; p, phospho.
Discussion
The results of the current study showed that phosphorylation of ERK1/2 in HUVECs are suppressed by H2O2 at particular concentrations and times and that this suppression is dependent on Csk translocation and subsequent phosphorylation of Src tyrosine 530. Physiological laminar flow, but not oscillatory shear stress, abrogates the suppression of p-ERK1/2 by H2O2. The suppression of p-ERK1/2 by H2O2 seems to conflict with other reports showing that H2O2 induces the phosphorylation of ERK1/27,25,36,37,38,39. However, our findings differ from previous reports in that the suppression of p-ERK1/2 is specific to HUVECs. When either murine endothelial or astroglioma cells were treated with 0.5 mM H2O2 for 15 min, p-ERK1/2 was increased (Fig. 2b,c). This can explain the finding of Yang et al. that ERK1/2 phosphorylation is increased in bovine aortic endothelial cells treated in a similar manner37. The reason for the different response in HUVECs may be related to the high basal phosphorylation of ERK1/2 in these cells compared to other cell types. As a result, H2O2-induced suppression of p-ERK1/2 may be easier to detect in HUVECs. In addition to cell type, time and concentration are important factors in the effects of H2O2. Suppression of ERK1/2 phosphorylation by 0.5 mM H2O2 occurs in a relatively short time range (within 15 min) and rebounds at 30 min when there is a high level of p-ERK1/2 compared to untreated controls (Fig. 1b). The increased p-ERK1/2 observed at 30 min is consistent with the report of Kevil et al. showing that ERK1/2 is activated by 1 mM H2O2 after 30 min in HUVECs7. In addition, this fact suggests that transient ERK1/2 suppression provides a window during which cells can prepare for the later activation of ERK1/2. Suppression of p-ERK1/2 occurred only at H2O2 concentrations between 0.25 and 1 mM, while ERK1/2 phosphorylation was slightly increased at 0.1 mM H2O2 (Fig. 1a). This is consistent with other reports showing ERK1/2 activation at relatively low concentrations of H2O236,38. This result suggests that both concentration and time are important factors of suppression of p-ERK1/2 by H2O2.
ERK1/2 is a well-studied signaling molecule in various cell types, including endothelial cells. The phosphorylation of ERK1/2 activates many downstream signaling molecules and induces cell proliferation and protection against oxidative stress25. However, the role of ERK1/2 and the effect of ERK1/2 phosphorylation, has been controversial in endothelial cell biology. ERK1/2 mediates the loss of cell-to-cell junctional integrity and the increased cell permeability induced by H2O27. When ERK1/2 activity is suppressed by the pharmacological inhibitor PD98059, H2O2-evoked increases in permeability are prevented. ERK1/2 activation also activates pro-survival signaling and prevents the apoptosis induced by H2O2. Thus, apoptosis is increased when ERK1/2 function is blocked17,26,37. On the other hand, activation of ERK1/2 contributes to cell death induced by cerebral ischemia. Phosphorylation level of ERK1/2 increases by oxygen-glucose deprivation (OGD), in vitro ischemic model and a specific inhibitor of MEK protects cell injury against OGD40,41. In our results, treatment with 0.5 mM H2O2 increased apoptotic cell death and increased monolayer permeability in HUVECs. This result is consistent with previous reports that H2O2 increases endothelial cell permeability but conflicts with the finding that the activation of ERK1/2 contributes to the disruption of endothelial cell integrity7,42. This discrepancy might due to different time and concentration of H2O2.
Because MAPK subfamilies interact with each other, the levels of p-p38 and p-JNK were measured. Although ERK1/2 phosphorylation was decreased by 0.5 mM H2O2, p38 and JNK showed no significant change in phosphorylation (Fig. 2a). Interestingly, although the p-p38 level after treatment with 0.5 mM H2O2 was slightly decreased compared to the group treated with 0.05 mM H2O2, the much larger suppression seen with p-ERK1/2 was not observed. Generally, the function of ERK1/2 in eukaryotic cells differs from the function of p38 and JNK. While ERK1/2 primarily participates in cell growth and proliferation, p38 and JNK have a role in stress and inflammatory responses rather than mitogen responses43. Wang et al. reported that ERK1/2 had an anti-apoptotic effect while p38 did not affect H2O2-mediated apoptosis and JNK participated in pro-apoptotic signaling17.
Significant suppression of p-ERK1/2 by H2O2 was related with the inactivation of Src through the phosphorylation of Src tyrosine 530. When phosphorylation of ERK1/2 was suppressed by 0.5 mM H2O2, phosphorylation of Src tyrosine 530 was increased while phosphorylation of Src tyrosine 419 was reduced (Fig. 4a,b). Src activates various molecules involved in endothelial cell permeability and induces vascular leakage31. Src is regulated by two major phosphorylation sites, tyrosine 419 and tyrosine 530. When Src tyrosine 419 is phosphorylated, Src undergoes a conformational change resulting in activation; however, when Src tyrosine 530 is phosphorylated, Src is inactive and maintains a “closed” conformation32. Dephosphorylating Src at tyrosine 530 is as important to Src activation as is Src tyrosine 419 phosphorylation. In human colon cancer cells, increased Src activation is mediated by the reduction of Src tyrosine 530 phosphorylation rather than Src tyrosine 419 phosphorylation44,45. When Src expression was silenced by siRNA, the percentage of reduction in H2O2-mediated suppression of ERK1/2 phosphorylation was decreased from 65.4% to 46.9% (Fig. 5a). Although the difference in the percentage of reduction between the GFP siRNA- and Src siRNA-transfected H2O2-treated groups was relatively small, Src knockdown reduced the suppression of p-ERK1/2 by H2O2, perhaps because of changes in the Src expression level. The quantity of Src is reduced in Src knockdown cells, resulting in a decrease in the quantity of p-Src tyrosine 530 induced by H2O2. Thus, both Src activity and the suppression of ERK1/2 in the presence of H2O2 were decreased by Src knockdown. The observed difference in the efficacy of Src and Csk knockdown in reversing H2O2-mediated suppression of ERK1/2 phosphorylation might be due to the complicated mechanism of action of Src. The knockdown of Src induces depletion of two major phosphorylation sites, tyrosine 419 and tyrosine 530, which may result in a loss of counteraction.
Csk is a negative regulator of Src that phosphorylates Src tyrosine 530 and maintains Src in the inactive state. Membrane translocation and co-localization with Src are required for Csk functioning33,34. Suppression of ERK1/2 phosphorylation was abrogated by Csk siRNA knockdown (Fig. 5b). This indicates the dependency of Csk and Src tyrosine 530 for the suppression of p-ERK1/2 by H2O2. IFA and immunoblotting data showed that translocation of Csk to the cell membrane occurred in response to H2O2 (Fig. 6a,b). In addition, when the movement of Csk to the detergent-insoluble fraction was blocked by silencing Csk, the phosphorylation of Src tyrosine 530 was decreased and suppression of p-ERK1/2 by H2O2 was abolished (Fig. 6a). This finding suggests that the H2O2-induced suppression of pERK1/2 is dependent on the membrane translocation of Csk and subsequent phosphorylation of Src tyrosine 530. Interestingly, Cao et al. reported that both Src and Csk are activated by oxidative stress and this paradoxical activation of Csk with Src may be important to stabilize Src in its basal state46. Activation of Csk and subsequent inactivation of Src in this study may largely affect the H2O2-mediated ERK1/2 signaling pathway.
Endothelial cells lining the vascular wall are continuously exposed to mechanical force because of blood pressure and flow. Laminar shear stress, the tangential force created by blood flow, plays an important role in endothelial function and helps prevent atherosclerosis13,47. ERK1/2 has an important role in the various events mediated by shear stress, which activates both Src and ERK1/213. When different strengths of laminar shear stress were applied together with 0.5 mM H2O2 for 15 min, suppression of ERK1/2 was significantly abrogated (Fig. 7a). However, there was no change in H2O2-induced ERK1/2 suppression in either the static or oscillatory shear stress groups treated with H2O2. Interestingly, laminar shear stress increased the phosphorylation levels of Src tyrosine 419 as well as ERK1/2 (Fig. 7b). It has been reported that Src tyrosine 419 phosphorylation, Src tyrosine 530 dephosphorylation and Src activation occur with laminar shear stress as opposed to oscillatory shear stress48. In addition, Tian et al. reported that H2O2 primes the shear stress-induced activation of eNOS49. Thus, nitric oxide production is increased in response to treatment of cells with shear stress and H2O2 compared to H2O2 alone-treated cells. These results suggest that, in a normal endothelial cell environment where shear stress exists continuously, physiological laminar flow could abolish the H2O2-induced suppression of p-ERK1/2.
In conclusion, this is the first report describing that, in normal environments with physiological shear stress, laminar flow masks Csk and Src tyrosine 530-mediated suppression of ERK1/2 phosphorylation, which is induced by H2O2. As a result, p-ERK1/2 is not significantly altered by H2O2 in the presence of laminar shear stress. However, when physiological laminar flow is disrupted because of regional features such as a bifurcation point, atherosclerosis, or an obstruction, the H2O2-induced suppression of ERK1/2 could occur through Csk translocation and subsequent Src tyrosine 530 phosphorylation (Fig. 8). Further studies are needed to determine how HUVECs recognize concentration differences of H2O2 and activate different signaling pathways. This study, however, contributes to broaden our understanding of signaling mechanisms in endothelial cells against oxidative stress and shear stress.
Potential mechanism of ERK1/2 suppression by H2O2 in HUVECs.
During normal physiological shear stress, laminar flow induces phosphorylation of Src tyrosine 419, which masks Csk- and Src tyrosine 530-mediated suppression of p-ERK1/2 in the presence of H2O2. As a result, phosphorylation of ERK1/2 is not suppressed by H2O2. However, when physiological laminar flow is disrupted, such as during a static status or oscillatory flow, H2O2 induces Csk translocation and Src tyrosine 530 phosphorylation. This leads to the inactivation of Src and subsequent suppression of ERK1/2 phosphorylation in HUVECs.
Methods
Cell culture
Human umbilical vein endothelial cells (HUVECs) were kindly provided by Dr. Kihwan Kwon (Ewha Womans University, Seoul, Korea). HUVECs were maintained in Medium 200 (Life Technologies, Carlsbad, CA, USA) with 5% fetal bovine serum (FBS), 1% penicillin-streptomycin and low-serum growth supplement (LSGS; Life Technologies) and all dishes and flasks for HUVEC are pre-coated by 2% gelatin (Wako Pure Chemical Industries, Ltd., Osaka, Japan) solution. HUVECs in passage 5 to 8 were used in all experiments. Immortalized murine aortic endothelial cells (iMAECs) were grown in Dulbecco’s modified essential media (DMEM; GE Healthcare Life Science, Buckinghamshire, UK.) containing 100 μg/ml endothelial cell growth supplement (Sigma, St. Louis, MO, USA), 1x non-essential Eagle’s amino acid, 10% fetal bovine serum and 1% penicillin-streptomycin. Human astroglioma cell line CRT-MG were grown in DMEM (WelGENE, Daegu, Korea) with 10% fetal bovine serum and 1% penicillin-streptomycin.
Reagents and antibodies
H2O2 and catalase were purchased from Sigma. Antibodies against p-ERK1/2, ERK1/2, p-Src Y419, p-Src Y530 and Src were obtained from Cell Signaling (Beverly, MA, USA). Anti-Csk, anti-p-p38, anti-p38, anti-p-JNK, anti-JNK antibodies were obtained from Santa Cruz Technology (Santa Cruz, CA, USA). Antibody against caveolin-1 was obtained from BD Biosciences (San Diego, CA, USA). Tubulin antibody was obtained from Sigma. All Horseradish peroxidase (HRP)-conjugated secondary antibodies for immunoblotting were obtained from Santa Cruz Technology. Alexa flour 568-conjugated rabbit secondary antibody was obtained from Life Technologies.
Shear stress exposure
Cone and plate viscometer was used to apply shear stress to HUVEC in vitro. Confluent cells in 60-mm dishes were exposed to fluid shear stress. Unidirectional steady flow (shear stress of 16 dyne/cm2 or 25 dyne/cm2) for laminar shear stress or a bidirectional disturbed flow (shear stress of ± 5 dyne/cm2) for oscillatory shear stress was exposed to confluent HUVECs.
Small interfering RNA (siRNA) transfection
Cells were transfected with green fluorescence protein (GFP) siRNA from Samchully Pharm Co. Ltd. (Seoul, Korea), 50 nM of human Csk siRNA from Bioneer (Daejeon, Korea) or 100 nM of SMART pools of human Src siRNA from GE Healthcare Life Science using LipofectamineTM RNAiMAX reagent (Life Technologies) according to the manufacturer’s protocol. After transfection, HUVECs were incubated in media containing 5% FBS, 1% penicillin-streptomycin and LSGS for 24 h (in the case of Csk) or 48 h (in the case of Src) and then exposed to 0.5 mM H2O2 for 15 min.
Western blot analysis
Cells were washed once with phosphate-buffered saline (PBS) and lysed by ice-cold radioimmunoprecipitation assay (RIPA) buffer containing Xpert Protease Inhibitor Cocktail (GenDEPOT, Inc., Barker, TX, USA) and 0.5 mM sodium orthovanadate (Na3VO4). Lysates were centrifuged (12,000 g) at 4 °C for 30 min and then supernatants were used for immunoblot analysis. Proteins were separated by 10% or 12% SDS-PAGE and transferred to polyvinylidene difluoride (PVDF) membrane. The membranes were blocked by 5% skim milk in Tris-buffered saline containing 0.1% Tween-20 (TBS-T) and probed with primary antibodies followed by HRP-conjugated specific secondary antibodies. Immunoreactive blots were developed using ECL chemiluminescence system (Amersham, Buckinghamshire, UK) and exposed to blue X-ray film (AGFA, Mortsel, Belgium).
Vascular permeability assay
Endothelial cell permeability was measured using 24-well In vitro Vascular Permeability Assay (Millipore, Darmstadt, Germany) according to manufacturer’s protocol. 2 × 105 of HUVECs were seeded onto collagen-coated inserts and incubated for 72 h monolayer formation time. After incubation, 0.5 mM H2O2 were treated to HUVEC monolayer for 15 min and then FITC-dextran solution was added to each inserts. Inserts and receivers protected from light were further incubated for 20 min. At the end of the incubation time, 100 μl of media in each receiver tray was harvested and the amount of FITC-dextran which crossed the monolayer was measured using Synergy H1 Hybrid Multi-Mode Microplate Reader (BioTek Instruments, Inc., Winooski, VT, USA). After measurement, cell monolayer integrity was checked using Cell Stain (Millipore).
Cell apoptosis assay
The HUVEC apoptosis was examined using FITC Annexin V Apoptosis Detection Kit II (BD Biosciences) according to the manufacturer’s protocol. Confluent HUVECs were treated with 0.5 mM H2O2 for 15 min and then harvested cells were stained with annexin V-FITC and propidium iodine (PI) in the dark at room temperature for 15 min. Cells were analyzed by FACSCaliburTM flow cytometry (BD Biosciences).
Isolation of detergent-insoluble fraction
A detergent-insoluble fraction was isolated according to a protocol previously described50. Cells were washed once with PBS and lysed with HEPES buffer (10 mM sodium HEPES, pH 7.5, 150 mM NaCl, 5 mM EDTA, protease inhibitor mixture and 0.5 mM Na3VO4) containing 0.5% Brij 58. Cell lysates were centrifuged at 12,000 g for 30 min at 4 °C and then supernatants were obtained and used as soluble fractions in following experiments. The pellets were washed once with cold HEPES buffer without detergent, solubilized with lysis buffer (50 mM Tris–HCl, pH 7.4, 0.1% SDS, 0.5% sodium deoxycholate, 1% Nonidet P-40, 1 mM EDTA, 1 mM EGTA, protease inhibitor mixture and 0.5 mM Na3VO4) and centrifuged at 12,000 g for 30 min at 4 °C. These supernatants were used as insoluble fractions. Each fraction was analyzed by immunoblotting.
Immunofluorescence assay
HUVECs cultured on poly-D-lysine-coated coverslips were washed once with ice-cold PBS and fixed with 4% paraformaldehyde for 1 h at room temperature. Fixed cells were incubated in PBS-T (PBS with 0.25% Triton X-100) for 15 min and permeable cells were blocked by 1% BSA in PBS-T for 1 h. Cells were probed with anti-Csk antibody overnight, washed with ice-cold PBS twice and then probed by Alexa flour 568-conjugated secondary antibody for 90 min. Cells were mounted in mounting solution (Life Technologies) containing DAPI and observed using confocal microscope (Carl Zeiss, Oberkochen, Germeny).
Statistical analysis
Mean ± standard error of the mean (SEM) were showed in all bar graphs from at least three independent experiments and statistical significance was analyzed by student t-test using the SPSS software version 20.0 (SPSS Inc., Chicago, IL, USA). A value of p < 0.05 was regarded statistically significant.
Additional Information
How to cite this article: Jeon, B. K. et al. Csk-Induced Phosphorylation of Src at Tyrosine 530 is Essential for H2O2-Mediated Suppression of ERK1/2 in Human Umbilical Vein Endothelial Cells. Sci. Rep. 5, 12725; doi: 10.1038/srep12725 (2015).
References
Wagner, A. M. A role for active oxygen species as second messengers in the induction of alternative oxidase gene expression in Petunia hybrida cells. FEBS Letters. 368, 339–342 (1995).
Sen, C. K. & Packer, L. Antioxidant and redox regulation of gene transcription. FASEB J. 10, 709–720 (1996).
Sun, Y. & Oberley, L. W. Redox regulation of transcriptional activators. Free Radic Biol Med. 21, 335–348 (1996).
Allen, R. G. Oxidative stress and superoxide dismutase in development, aging and gene regulation. Age. 21, 47–76 (1998).
Taniyama, Y. & Griendling, K. K. Reactive oxygen species in the vasculature molecular and cellular mechanisms. Hypertension. 42, 1075–1081 (2003).
Kao, C. L. et al. Resveratrol protects human endothelium from H(2)O(2)-induced oxidative stress and senescence via SirT1 activation. J Atheroscler Thromb. 17, 970–979 (2010).
Kevil, C. G., Oshima, T., Alexander, B., Coe, L. L. & Alexander, J. S. H(2)O(2)-mediated permeability: role of MAPK and occludin. Am J Physiol Cell Physiol. 279, C21–30 (2000).
Lum, H. & Roebuck, K. A. Oxidant stress and endothelial cell dysfunction. Am J Physiol Cell Physiol. 280, C719–741 (2001).
Cai, H. et al. Akt-dependent phosphorylation of serine 1179 and mitogen-activated protein kinase kinase/extracellular signal-regulated kinase 1/2 cooperatively mediate activation of the endothelial nitric-oxide synthase by hydrogen peroxide. Mol Pharmacol. 63, 325–331 (2003).
Nguyen, A., Chen, P. & Cai, H. Role of CaMKII in hydrogen peroxide activation of ERK1/2, p38 MAPK, HSP27 and actin reorganization in endothelial cells. FEBS Letters. 572, 307–313 (2004).
Peshavariya, H. et al. NADPH oxidase isoform selective regulation of endothelial cell proliferation and survival. Naunyn Schmiedebergs Arch Pharmacol. 380, 193–204 (2009).
Boueiz, A. & Hassoun, P. M. Regulation of endothelial barrier function by reactive oxygen and nitrogen species. Microvasc Res. 77, 26–34 (2009).
Traub, O. & Berk, B. C. Laminar shear stress: mechanisms by which endothelial cells transduce an atheroprotective force. Arterioscler Thromb Vasc Biol. 18, 677–685 (1998).
Resnick, N. et al. Fluid shear stress and the vascular endothelium: for better and for worse. Prog Biophys Mol Biol. 81, 177–199 (2003).
Chien, S. Effects of disturbed flow on endothelial cells. Ann Biomed Eng. 36, 554–562 (2008).
Noguchi, N. & Jo, H. Redox going with vascular shear stress. Antioxid Redox Signal. 15, 1367–1368 (2011).
Wang, X., Martindale, J. L., Liu, Y. & Holbrook, N. J. The cellular response to oxidative stress: influences of mitogen-activated protein kinase signalling pathways on cell survival. Biochem J. 333, 291–300 (1998).
Genersch, E., Hayess, K., Neuenfeld, Y. & Haller, H. Sustained ERK phosphorylation is necessary but not sufficient for MMP-9 regulation in endothelial cells: involvement of Ras-dependent and -independent pathways. J Cell Sci. 113, 4319–4330 (2000).
Widmann, C., Gibson, S., Jarpe, M. B. & Johnson, G. L. Mitogen-activated protein kinase: conservation of a three-kinase module from yeast to human. Physiol Rev. 79, 143–180 (1999).
Johnson, G. L. & Lapadat, R. Mitogen-activated protein kinase pathways mediated by ERK, JNK and p38 protein kinases. Science. 298, 1911–1912, (2002).
Meloche, S. & Pouyssegur, J. The ERK1/2 mitogen-activated protein kinase pathway as a master regulator of the G1- to S-phase transition. Oncogene. 26, 3227–3239, (2007).
Cuadrado, A. & Nebreda, A. R. Mechanisms and functions of p38 MAPK signalling. Biochem J. 429, 403–417, (2010).
Dhanasekaran, D. N. & Reddy, E. P. JNK signaling in apoptosis. Oncogene. 27, 6245–6251 (2008).
Kim, E. K. & Choi, E. J. Pathological roles of MAPK signaling pathways in human diseases. Biochim Biophys Acta. 1802, 396–405 (2010).
Guyton, K. Z., Liu, Y., Gorospe, M., Xu, Q. & Holbrook, N. J. Activation of mitogen-activated protein kinase by H2O2. Role in cell survival following oxidant injury. J Biol Chem. 271, 4138–4142 (1996).
Lee, Y. J. et al. Oxidative stress-induced apoptosis is mediated by ERK1/2 phosphorylation. Exp Cell Res. 291, 251–266 (2003).
De Luca, A., Maiello, M. R., D’Alessio, A., Pergameno, M. & Normanno, N. The RAS/RAF/MEK/ERK and the PI3K/AKT signalling pathways: role in cancer pathogenesis and implications for therapeutic approaches. Expert Opin Ther Targets. 16, S17–27 (2012).
Cargnello, M. & Roux, P. P. Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol Mol Biol Rev. 75, 50–83 (2011).
Aikawa, R. et al. Oxidative stress activates extracellular signal-regulated kinases through Src and Ras in cultured cardiac myocytes of neonatal rats. J Clin Invest. 100, 1813–1821 (1997).
Bolen, J. B., Veillette, A., Schwartz, A. M., DeSeau, V. & Rosen, N. Activation of pp60c-src protein kinase activity in human colon carcinoma. Proc Natl Acad Sci USA. 84, 2251–2255 (1987).
Hu, G., Place, A. T. & Minshall, R. D. Regulation of endothelial permeability by Src kinase signaling: vascular leakage versus transcellular transport of drugs and macromolecules. Chem Biol Interact. 171, 177–189 (2008).
Bjorge, J. D., Jakymiw, A. & Fujita, D. J. Selected glimpses into the activation and function of Src kinase. Oncogene. 19, 5620–5635 (2000).
Okada, M. Regulation of the SRC family kinases by Csk. Int J Biol Sci. 8, 1385–1397 (2012).
Howell, B. W. & Cooper, J. A. Csk suppression of Src involves movement of Csk to sites of Src activity. Mol Cell Biol. 14, 5402–5411 (1994).
Hoefen, R. J. & Berk, B. C. The role of MAP kinases in endothelial activation. Vascul Pharmacol. 38, 271–273 (2002).
Chen, D. B., Li, S. M., Qian, X. X., Moon, C. & Zheng, J. Tyrosine phosphorylation of caveolin 1 by oxidative stress is reversible and dependent on the c-src tyrosine kinase but not mitogen-activated protein kinase pathways in placental artery endothelial cells. Biol Reprod. 73, 761–772 (2005).
Yang, B., Oo, T. N. & Rizzo, V. Lipid rafts mediate H2O2 prosurvival effects in cultured endothelial cells. FASEB J. 20, 1501–1503 (2006).
Ulrich-Merzenich, G., Zeitler, H., Panek, D., Bokemeyer, D. & Vetter, H. Vitamin C promotes human endothelial cell growth via the ERK-signaling pathway. Eur J Nutr. 46, 87–94 (2007).
Polidoro, L. et al. Vitamin D protects human endothelial cells from H(2)O(2) oxidant injury through the Mek/Erk-Sirt1 axis activation. J Cardiovasc Transl Res. 6, 221–231 (2013).
Narasimhan, P., Liu, J., Song, Y. S., Massengale, J. L. & Chan, P. H. VEGF Stimulates the ERK 1/2 signaling pathway and apoptosis in cerebral endothelial cells after ischemic conditions. Stroke. 40, 1467–1473 (2009).
Irving, E. A. & Bamford, M. Role of mitogen-and stress-activated kinases in ischemic injury. J Cereb Blood Flow Metab. 22, 631–647 (2002).
Kevil, C. G., Okayama, N. & Alexander, J. S. H(2)O(2)-mediated permeability II: importance of tyrosine phosphatase and kinase activity. Am J Physiol Cell Physiol. 281, C1940–1947 (2001).
Kyriakis, J. M. & Avruch, J. Mammalian mitogen-activated protein kinase signal transduction pathways activated by stress and inflammation. Physiol Rev. 81, 807–869 (2001).
Zhu, S., Bjorge, J. D. & Fujita, D. J. PTP1B contributes to the oncogenic properties of colon cancer cells through Src activation. Cancer Res. 67, 10129–10137 (2007).
Zhu, S., Bjorge, J. D. & Fujita, D. J. SRC is dephosphorylated at tyrosine 530 in human colon carcinomas. Chin J Cancer Res. 23, 229–231 (2011).
Cao, H., Sanguinetti, A. R. & Mastick, C. C. Oxidative stress activates both Src-kinases and their negative regulator Csk and induces phosphorylation of two targeting proteins for Csk: caveolin-1 and paxillin. Exp Cell Res. 294, 159–171 (2004).
Malek, A. M., Alper, S. L. & Izumo, S. Hemodynamic shear stress and its role in atherosclerosis. JAMA. 282, 2035–2042 (1999).
Spindel, O. N., Burke, R. M., Yan, C. & Berk, B. C. Thioredoxin-interacting protein is a biomechanical regulator of Src activity: key role in endothelial cell stress fiber formation. Circ Res. 114, 1125–1132 (2014).
Tian, J. et al. A novel role for caveolin-1 in regulating endothelial nitric oxide synthase activation in response to H2O2 and shear stress. Free Radic Biol Med. 49, 159–170 (2010).
Park, S. J. et al. Oxidative stress induces lipid-raft-mediated activation of Src homology 2 domain-containing protein-tyrosine phosphatase 2 in astrocytes. Free Radical Biol Med. 46, 1694–1702 (2009).
Acknowledgements
This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIP) Grant 2012R1A5A2A32671866 and by NRF-2013R1A1A3009978.
Author information
Authors and Affiliations
Contributions
B.K.J. and Y.H.C. designed research; B.K.J., K.K. and Y.H.C. conducted research; B.K.J., K.K., J.L.K. and Y.H.C. analysed data; B.K.J. and Y.H.C. wrote the paper; B.K.J. and Y.H.C. had primary responsibility for final content. All authors read and approved the final manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Jeon, B., Kwon, K., Kang, J. et al. Csk-Induced Phosphorylation of Src at Tyrosine 530 is Essential for H2O2-Mediated Suppression of ERK1/2 in Human Umbilical Vein Endothelial Cells. Sci Rep 5, 12725 (2015). https://doi.org/10.1038/srep12725
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep12725
This article is cited by
-
Csk regulates angiotensin II-induced podocyte apoptosis
Apoptosis (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.